Abstract
Background:
The programmed death-1/programmed death-ligand-1 (PD-1/PD-L1) axis may represent a target for cervical cancer; however, it is poorly understood in human immunodeficiency virus (HIV)-infected patients.
Methods:
We evaluated HIV-positive (n = 42) and HIV-negative (n = 110) women with locally advanced cervical cancer regarding their PD-L1 expression, determined by combined positive score (CPS) ⩾ 1 and tumor proportion score (TPS) ⩾ 25%, and PD-L1 copy number alterations, assessed by fluorescence in situ hybridization.
Results:
Regardless of HIV status, 84.9% and 44.8% of cases were PD-L1-positive according to CPS ⩾ 1 and TPS ⩾ 25%. Per CPS ⩾ 1, PD-L1 positive rate was similar between HIV-positive and HIV-negative women, whereas a significant difference was seen per TPS ⩾ 25%. Tumor size and parametrial invasion were correlated with PD-L1 positivity in HIV-negative women, whereas anti-retroviral therapy (ART) was correlated with TPS < 25%. Low CD4-positive cell counts were associated with CPS < 1 in HIV-positive women. No significant difference was observed in PD-L1 copy number status between HIV-positive and HIV-negative women. PD-L1 amplification and polysomy were independently associated with TPS ⩾ 25%, whereas the presence of parametrial invasion was independently associated with CPS ⩾ 1. Cancer stage and PD-L1 amplification were identified as independent predictors of recurrence-free survival [hazard ratio (HR) = 2.40 (1.32–4.36) and HR = 5.33 (1.94–14.61)] and cancer-specific survival [HR = 13.62 (5.1–36.38) and HR = 3.53 (1.43–8.69)]. PD-L1 polysomy was an independent predictor of locoregional recurrence-free survival [HR = 3.27 (1.27–8.41)]. HIV status and PD-L1 expression (CPS ⩾ 1 or TPS ⩾ 25%) were not associated with poor patient outcomes.
Conclusion:
PD-L1 amplification and polysomy are the strongest drivers of PD-L1 expression in cervical cancer, and could represent prognostic biomarkers for anti-PD-1/PD-L1 therapy. Cervical cancer biology may be modulated by HIV infection, CD4-positive cells, and HIV treatments.
Keywords
Introduction
Cervical cancer is one of the most common cancers in women with human immunodeficiency virus (HIV). 1
A previous study demonstrated that HIV-positive patients have greater tendency to develop precursor lesions in the cervix and cancer. HIV-positive individuals had DNA microsatellite alterations that could result in genetic instability, and, ultimately, cervical cancer. 2 Other reports also stated that low CD4-positive T cell counts were not associated with the incidence of cervical cancer. 3 Only a few studies have examined the development of such tumors at the molecular level. Therefore, the molecular pathogenesis of cervical cancer in HIV patients remains poorly understood.
Overexpression of PD-L1 has been observed in several cancers, such as melanoma, lung cancer, and colorectal cancer.4–7 Moreover, PD-L1 overexpression has been associated with poorer patient prognosis and worse overall survival in gastric carcinoma, transitional cell carcinoma, hepatocellular carcinoma, and oesophageal carcinoma. 4
Immune checkpoint blockade therapy using monoclonal antibodies targeting immune co-inhibitory molecules is an emerging technology of cancer treatment. The therapeutic antibody inhibits the interaction between PD-1 on immune effector cells and PD-L1 on tumor cells, preventing anti-tumor immunity suppression by cancer cells.
Currently, few immune checkpoint blockade agents are available for treating cervical cancer. Pembrolizumab (marketed as Keytruda) was approved by the United States Food and Drug Administration (FDA) for the treatment of PD-L1-positive cervical cancer and nivolumab (marketed as Opdivo) is still under development for clinical use in cervical cancer.
Clinical studies have shown that expression of PD-L1 could reflect a response of tumors to anti-PD-1/PD-L1 therapy in patients with cervical cancer. The recent KEYNOTE-158 and KEYNOTE-358 trials demonstrated the clinical activity of PD-1 targeted therapies in cervical cancer patients.8,9 The response to therapy was directly associated with PD-L1 expression, with a higher response rate in tumors with combined positive score (CPS) ⩾ 1 compared with tumors with CPS < 1.
A large number of companies and academic laboratories have been attempting to develop an efficient primary antibody to detect PD-L1 protein on the surface of tumor cells by immunohistochemistry. Although many products are commercially available, each uses different staining protocols, scoring algorithms, and threshold criteria. Hence, the best PD-L1 antibody and the appropriate cut-off expression level for determining PD-L1 expression remain controversial as it is based on different detection systems and testing platforms.
Apart from the above-mentioned problems, the optimal criteria to select patients who may benefit the most from immune checkpoint inhibitors also represent a problem because the expression of PD-L1 could be heterogeneous and inconsistent. Therefore, a process to overcome these limitations is necessary. For example, some studies have reported that 9p24.1 gene locus amplification is a vital mechanism for PD-L1 overexpression in many cancers, including nodular sclerosing Hodgkin lymphoma, primary mediastinal large B-cell lymphoma, subsets of colorectal adenocarcinoma, gastric cancer, triple-negative breast cancer, and glioblastoma.10–12 Using fluorescence in situ hybridization (FISH), researchers haves found that 67% of cervical and 43% of vulvar squamous cell carcinomas positive for PD-L1 and PD-L2 had multiple copies of CD274 and PDCD1LG2 in the same chromosome. 13 Therefore, it could be assumed that 9p24.1 gene amplification is an underlying mechanism of enhanced PD-L1 expression in cervical squamous cell carcinomas.
Most studies assessing PD-L1 expression have included only healthy subjects, and excluded HIV-positive patients from their analysis. Data on PD-L1 expression in HIV-infected cervical cancer patients and on the efficacy of immune checkpoint blockade therapy with anti-PD-1 monoclonal antibodies against cervical cancer in HIV-infected patients are limited.
Our previous study showed that tumors of HIV-infected cervical cancer patients who had undergone antiretroviral therapy (ART) had lower prevalence of PD-L1 immunopositivity, PD-L1 amplification, and polysomy compared with patients who did not receive ART and those that were HIV-negative. 14 Having additional expression data on PD-L1 and its encoding gene in HIV-positive patients would be useful for clinical management.
This study aimed to retrospectively evaluate the relationship between pre-treatment PD-L1 expression and copy number gains of CD274, as well as its association with clinical outcomes, in HIV-positive and HIV-negative cervical cancer patients.
Materials and methods
Patients
The study cohort consisted of 152 patients diagnosed with cervical cancer (42 HIV-infected subjects and 110 HIV-uninfected controls) between December 2008 and December 2016 at the Faculty of Medicine of Navamindradhiraj University, the Rajavithi Hospital, and the National Cancer Institute of Thailand.
These patients were selected and considered eligible for our study based on the following inclusion criteria: (i) diagnosis of IB2-IVA cervical cancer according to the International Federation of Gynecology and Obstetrics (FIGO) staging system, and (ii) histologically confirmed diagnosis of squamous cell carcinoma. Patients who had non-squamous malignancies of the cervix had been exposed to chemoradiation therapy, or had synchronous or metachronous malignancy, were excluded from the study.
This study complied with the Declaration of Helsinki and was approved by the Institutional Review Boards of Navamindradhiraj University (No. 137-2017), Rajavithi Hospital (No. 157-2017), and the National Cancer Institute (No. 33-2017). All patients provided informed consent, and their clinical information was recorded and coded anonymously. Hematoxylin and eosin (HE) stained tissue sections were reviewed by two clinical pathologists (KL and NP). Complete clinicopathologic data were reviewed and available for all patients.
Immunohistochemistry
All immunohistochemical studies were prepared from formalin-fixed paraffin-embedded tissues (FFPE). Immunohistochemistry was performed using 4 µm whole-tissue sections that were cut and stained with monoclonal antibodies recognising PD-L1 (clone SP263, Ventana Medical Systems, Tucson, AZ, USA) and performed on an automated staining platform (BenchMark ULTRA, Ventana Medical Systems). The OptiView DAB IHC Detection Kit (Ventana Medical Systems) was used according to the manufacturer’s guidelines for the visualization of the primary anti-PD-L1 antibody. For all immunohistochemical staining reactions, human placental tissue was used as a positive control.
PD-L1 immunohistochemical expression in cancer cells was assessed using CPS and tumor proportion score (TPS) methods. All slides were independently assessed by two experienced clinical pathologists (KL and NP). In case of disagreement, the slides were reviewed together to achieve consensus.
Scoring methods
The CPS is given by summing the number of PD-L1 positive cells, including tumor cells, lymphocytes, and macrophages, divided by the total number of viable tumor cells × 100. Specimens with CPS ⩾ 1 were considered to be ‘positive’ (Figure 1a–b). This cut-off value was used according to the KEYNOTE-158 and CHECKMATE-358 studies.

Cervical cancer specimens stained with a monoclonal anti-PD-L1 antibody. (a) Specimen with CPS < 1, (b) Specimen with CPS > 1, (c) Specimen with TPS < 25%, and (d) Specimen with TPS > 25% (original magnification × 20).
The TPS is the percentage of viable tumor cells showing partial or complete membrane staining at any intensity above background, relative to all viable tumor cells present in the sample. PD-L1 status was considered ‘positive’ when TPS ⩾ 25% of tumor cells exhibiting membrane staining (Figure 1c–d), according to data from the same antibody clone used in clinical studies of durvalumab in urothelial carcinoma, non-small-cell lung cancer, and head and neck squamous cell carcinoma (HNSCC).15–19
PD-L1 fluorescence in situ hybridization
Tissue microarrays (TMAs) with 3 mm core diameter were obtained from representative cervical cancer tissue samples. A minimum of two, and up to four, tumor cores from the tumor invasion front and/or tumor center were taken in areas previously marked by two clinical pathologists (KL and NP). All TMA cores were validated as containing sufficient number of tumor cells through HE staining. FFPE samples were deparaffinized, dehydrated in 100% ethanol, and air-dried. The ZytoLight SPEC CD274, PDCD1LG2/CEN9 dual-color probe, containing a mixture of fluorochrome-labelled probes specific for CD274/PDCD1LG2 cluster, and an orange fluorochrome-labelled CEN 9 probe specific for the classical satellite III region of chromosome 9 (D9Z3) at 9q12 (ZytoVision, Bremerhaven, Germany), was used according to the manufacturer’s guidelines.
After screening the entire area of individual cores, the probe signals for a monolayer of at least 50 tumor cell nuclei were counted at ×100 magnification in at least five representative images per patient. As previously described, 20 PD-L1 amplification was expressed as PD-L1/CEP9 ratio ⩾ 2.0 (Figure 2a), while polysomy was expressed as the median copy number of PD-L1 indications ⩾ 3.0, and the proportion to CEP9 signals was < 2.0 (Figure 2b). All other tumors were deemed to display disomy (Figure 2c).

Representative pictures of PD‑L1 FISH, showing (a) PD-L1 amplification, (b) PD-L1 polysomy, and (c) PD-L1 disomy (original magnification × 100). The PD-L1 gene is labelled in green, centromere 9 in red.
Statistical analysis
Statistical analysis was performed using Stata software (version 13, StataCorp LP, College Station, TX, USA). The distribution of qualitative data was compared between groups using the χ 2 -test or Fisher’s exact test, depending on the cell counts of the corresponding contingency table. Logistic regression was used to compute the relationship between PD‑L1 expression and its copy number gains with adjustment for potential confounding factors, including HIV status (HIV versus non-HIV), ART use, age, tumor size, FIGO stage, the presence of parametrial invasion, metastatic lymph node, and type of treatment.
From clinical data, we evaluated recurrence-free survival (RFS), which was calculated as the period from the first day of treatment to the date of confirmed recurrence or disease progression, or to the last noted date of disease-free status on the medical records. Cancer-specific survival (CSS) was also calculated as the time of the initial diagnosis of cervical cancer until the date of disease-related death or last follow up, as well as locoregional recurrence-free survival (LRR) as the interval after achieving complete response to the first biopsy proven, clinically, or radiologically detected relapse within the pelvis. The Kaplan–Meier method was used to compute RFS, CSS, and LRR. Univariate and multivariate analyses were performed through the Cox proportional hazards model, and the differences between groups were computed with the log-rank test. For all statistical analyses, p < 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of the patients
HIV status and different clinical cut-offs for PD-L1 expression for the patients included in the study are shown in Table 1. For the entire cohort (n = 152), the median follow-up time was 40 months (range: 1–120 months) for the HIV cohort (n = 42) and 28 months (2–82 months) for the non-HIV cohort (n = 110). HIV-positive patients were more likely to be younger (median age 41.52 ± 10.05 versus 55.34 ± 12.61 years, p < 0.001). Overall, 58 patients (38%) had FIGO stage III–IV tumors, and 40 patients (26%) had node-positive disease. For the entire cohort, the FIGO stage was as follows: I, 16 (11%); II, 78 (51%); III, 45 (29%); and IV, 13 (9%).
Clinicopathological characteristics of HIV and non-HIV patients with cervical cancer related to different clinical cutoffs for PD-L1 expression.
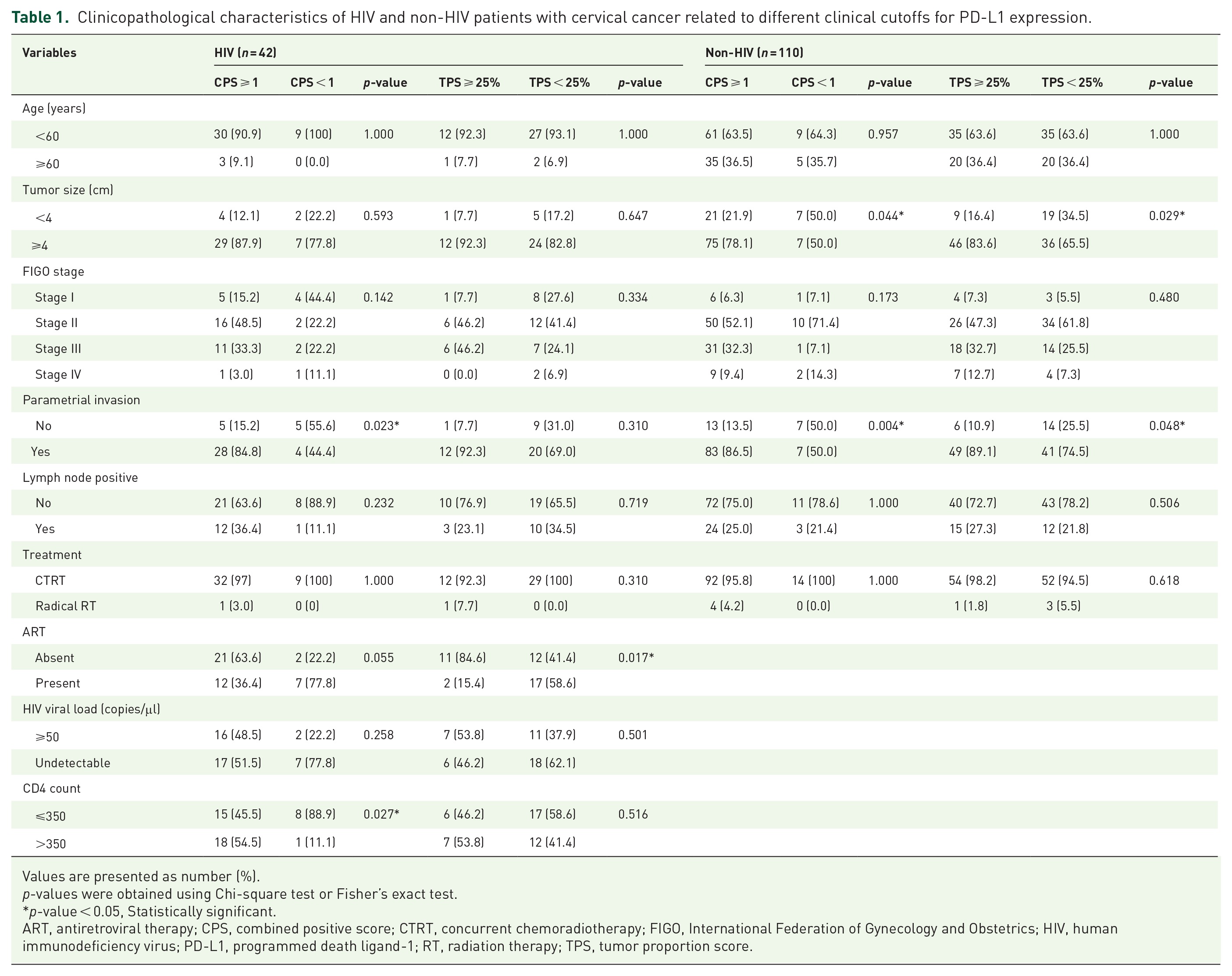
Values are presented as number (%).
p-values were obtained using Chi-square test or Fisher’s exact test.
p-value < 0.05, Statistically significant.
ART, antiretroviral therapy; CPS, combined positive score; CTRT, concurrent chemoradiotherapy; FIGO, International Federation of Gynecology and Obstetrics; HIV, human immunodeficiency virus; PD-L1, programmed death ligand-1; RT, radiation therapy; TPS, tumor proportion score.
Among HIV-positive patients, 19 (45%) were on ART at the time of treatment (defined as the reported use of three or more anti-retroviral medications, a protease inhibitor, a non-nucleoside reverse transcriptase inhibitor, or a nucleoside reverse transcriptase inhibitor). In total, 57% of patients had an undetectable HIV viral load, with a mean CD4-positive count of 316.83 ± 181.5 cells/μl.
Overall, 129/152 patients (84.9%) had tumors with CPS ⩾ 1, of whom 33/42 (78.6%) were HIV-positive and 96/110 (87.3%) were HIV-negative (p = 0.181). On the other hand, 68/152 (44.8%) had tumors with TPS ⩾ 25%, of whom 13/42 (31%) were HIV-positive and 55/110 (50%) were HIV-negative (p = 0.035). The distribution of PD-L1 protein expression with CPS ⩾ 1 and TPS ⩾ 25% in each group of patients is shown in Figure 3.

PD-L1 immunoreactivity and PD-L1 copy number alterations in HIV-positive and HIV-negative cervical cancer patients. (a) Percentage of PD-L1 immunoreactivity with CPS ⩾ 1 versus CPS < 1 across patient groups. (b) Percentage of PD-L1 immunoreactivity with TPS ⩾ 25% versus TPS < 25% across patient groups. (c) Percentage of PD-L1 copy number alterations (disomy, polysomy, and amplification) across patient groups. The analysis was conducted using the χ 2 -test and the data are presented as mean ± standard error of the mean.
Among HIV-positive patients (n = 42), those with CPS ⩾ 1 had significantly higher CD4 counts (54.5% versus 11.1%; p = 0.027) and higher risk for parametrial invasion (84.8% versus 44.4%; p = 0.023) than those with CPS < 1. No significant correlations were observed between CPS ⩾ 1 and <1 groups regarding patient age, tumor size, FIGO stage, nodal metastasis, radio/chemotherapy, ART use, or undetectable viral loads.
HIV-negative patients with CPS ⩾ 1 had bigger tumors (78.1% versus 50%; p = 0.044) and had a significantly higher risk for parametrial invasion (86.5% versus 50.0%; p = 0.004) compared with patients with CPS < 1. No significant correlations were found between CPS ⩾ 1 and <1 groups regarding patient age, FIGO stage, nodal metastasis, or radio/chemotherapy.
In the HIV-positive cohort with TPS ⩾ 25%, a significant difference in PD-L1 expression was observed between ART-treated (n = 19) and untreated (n = 23) patients (15.4% versus 84.6%; p = 0.017). However, no significant correlation was seen between patients with TPS ⩾ 25% or less regarding patient age, tumor size, FIGO stage, the presence of parametrial invasion, nodal metastasis, radio/chemotherapy, increased CD4 counts, or undetectable viral loads.
HIV-negative patients with TPS ⩾ 25% were found to have significantly bigger tumors (83.6% versus 65.5%; p = 0.029) and parametrial invasion at diagnosis (89.1% versus 74.5%; p = 0.048) compared with patients with TPS < 25%. No significant correlation was observed between TPS level and patient age, FIGO stage, nodal metastasis, or radio/chemotherapy.
Clinicopathological and molecular factors predicting PD-L1 positivity with CPS ⩾ 1
Based on CPS ⩾ 1, univariate analyses revealed that the features that associated with PD-L1 positivity were bigger tumor size [odds ratio (OR) = 2.67 (95% CI: 1.04–6.88), p = 0.041], parametrial invasion [OR = 6.73 (2.58–17.53), p < 0.001], and ART non-exposure or HIV negative [OR = 0.16 (0.03–0.92), p = 0.039 and OR = 0.25 (0.08–0.74), p = 0.012, respectively] (Table 2). However, only parametrial invasion was associated with CPS ⩾ 1 in the multivariate analyses [OR = 4.99 (1.78–13.95), p = 0.002].
Univariate and multivariate analyses for factors predict CPS ⩾ 1 (n = 152).

ART, antiretroviral therapy; CI, confidence interval; CPS, combined positive score; CTRT, concurrent chemoradiotherapy; FIGO, International Federation of Gynecology and Obstetrics; HIV, human immunodeficiency virus; OR, odds ratio; ORadj, adjusted odds ratio; PD‑L1, programmed cell death ligand-1; RT, radiation therapy.
p-value < 0.05, Statistically significant.
Clinicopathological and molecular factors predicting PD-L1 positivity with TPS ⩾ 25%
Based on TPS ⩾ 25%, features that could significantly predict PD-L1 positivity were HIV-positive [OR = 0.45 (0.21–0.95), p = 0.037], increased tumor size [OR = 2.67 (1.04–6.88), p = 0.041], parametrial invasion [OR = 6.73 (2.58–17.53), p<0.001], PD‑L1 amplification [OR = 16.84 (3.39–83.6), p = 0.001], and PD‑L1 polysomy [OR = 7.3 (3.42–15.57), p<0.001]. In subgroup analyses, ART exposure was inversely correlated with TPS ⩾ 25% compared with ART-untreated patients [OR = 0.13 (0.02–0.69); p = 0.016] and HIV-negative patients [OR = 0.12 (0.03–0.53), p = 0.006]. The analyses also revealed that only PD‑L1 amplification and PD‑L1 polysomy were significant predictors of TPS ⩾ 25% [OR = 13.09 (2.43–70.61), p = 0.003; and OR = 6.41 (2.87–14.33), p < 0.001; respectively]. The ability of each parameter to predict PD-L1 positivity is summarised in Table 3.
Univariate and multivariate analyses for factors predict TPS ⩾ 25% (n = 152).

ART, antiretroviral therapy; CI, confidence interval; CTRT, concurrent chemoradiotherapy; FIGO, International Federation of Gynecology and Obstetrics; HIV, human immunodeficiency virus; OR, odds ratio; ORadj, adjusted odds ratio; PD-L1, programmed cell death ligand-1; RT, radiation therapy; TPS, tumor proportion score.
p-value < 0.05, Statistically significant.
PD-L1 gene copy number alterations
Figure 3c shows the proportion of PD-L1 copy number alterations in each patient group. Among all patients included in the study 12 (7%) had tumor cells positive for gene amplification, of whom 4/42 (8%) were HIV-positive and 8/110 (7%) were HIV-negative patients. Polysomy was seen in 57 (38%) cases, among whom 14/42 (33%) were HIV-positive and 43/110 (39%) were HIV-negative patients, whereas PD-L1 disomy was seen in 83/152 (55%) cases, of whom 24/42 (57%) were HIV-positive patients and 59/110 (54%) were HIV-negative patients. Gene copy number gain was restricted to tumor cells and was not present in the inflammatory cell component. No significant differences in PD-L1 amplification, polysomy, and disomy between HIV-positive and HIV-negative patients were found.
Survival outcomes
Survival of HIV-positive and HIV-negative patients according to immunohistochemistry- and FISH-based expression status of PD-L1 are shown in Figures 4–5. For PD-L1 status determined by TPS ⩾ 25, only poor RFS was observed in HIV-negative patients positive for PD-L1. No significant survival differences between HIV-positive or negative patients with PD-L1 status determined by CPS ⩾ 1 criteria were observed. Of note, PD-L1 amplification was significantly correlated with unfavorable patient survival (RFS and CSS), regardless of HIV status.

Kaplan–Meier survival curves for HIV patients in relation to PD-L1 immunoreactivity and genetic category. (a) LRR, (b) RFS, and (c) CSS based on PD-L1 expression with CPS ⩾ 1 versus CPS < 1. (d) LRR, (e) RFS, and (f) CSS based on PD-L1 expression with TPS ⩾ 25% versus TPS < 25%. (g) LRR, (h) RFS, and (i) CSS based on PD-L1 amplification versus disomy and PD-L1 polysomy versus disomy.

Kaplan–Meier survival curves for non-HIV patients in relation to PD-L1 immunoreactivity and genetic category. (a) LRR, (b) RFS, and (c) CSS based on PD-L1 expression with CPS ⩾ 1 versus CPS < 1. (d) LRR, (e) RFS, and (f) CSS based on PD-L1 expression with TPS ⩾ 25% versus TPS < 25%. (g) LRR, (h) RFS, and (i) CSS based on PD-L1 amplification versus disomy and PD-L1 polysomy versus disomy.
Moreover, significant poor prognostic factors for patient survival were advanced FIGO stage (LRR, RFS, and CSS), large tumor size (RFS and CSS), parametrial invasion (RFS), positive nodal status (RFS and CSS), ART-untreated HIV-positive, PD-L1 positive (with TPS ⩾ 25%) (RFS), positive PD-L1 amplification (RFS and CSS), and polysomy (LRR and RFS). Multivariate analyses showed that only FIGO stage (RFS and CSS), polysomy (LRR), and PD-L1 amplification (RFS and CSS) remained significant poor prognostic factors. Of note, HIV status was not associated with worse survival outcomes. Univariate and multivariate analyses evaluating the impact of these prognostic factors are shown in Table 4.
Univariate and multivariate analyses for LRR, RFS, and CSS in cervical cancer patients. (n = 152).

CI, confidence interval; CPS, combined positive score; CSS, cancer-specific survival; CTRT, concurrent chemoradiotherapy; FIGO, International Federation of Gynecology and Obstetrics; HIV, human immunodeficiency virus; HR, hazard ratio; HRadj, adjusted hazard ratio; LRR, locoregional recurrence; PD-L1, programmed cell death ligand-1; RFS, recurrence-free survival; RT, radiation therapy; TPS, tumor proportion score.
p-value < 0.05, Statistically significant.
Discussion
Comparison of PD-L1 expression and copy number alterations in cervical cancer tissue specimens of patients with and without HIV infection, we found that HIV-positive women had significantly lower protein levels by using the clinical cut-off TPS ⩾ 25% but not by CPS ⩾ 1. However, there was no significant difference in PD-L1 copy number categories (amplification, polysomy, and disomy) between women positive and negative for HIV. Our findings suggest that HIV status was associated with cervical cancer cell biology; nevertheless, HIV effect was not associated with differences in genomic alterations between HIV-positive and HIV-negative women. In addition, we found that the CPS was related to the CD4-positive cell count in HIV-positive women, whereas TPS was associated with ART use, suggesting that the tumor microenvironment in cervical cancer may be influenced by CD4 cell count and HIV treatment.
In HIV-positive patients, the infection promotes PD-L1 expression in dendritic cells, B cells, and neutrophils, leading to an inhibitory environment for T cell-mediated anti-tumor immune activity.21,22 In addition, the use of anti-retroviral therapy may modulate the immune status of these patients, further promoting a tumor-promoting immune environment. 21 Nevertheless, while these explanations are theoretically meaningful, the underlying mechanism contributing for the poor outcomes of HIV patients with cervical cancer remains unclear.
An in vitro study in HNSCC cell lines revealed that PD-L1 expression is significantly upregulated in response to interferon gamma (IFN-γ), which is an important cytokine triggering de novo PD-L1 induction in tumor cells. 23 Multiple downstream signalling pathways, including NF-kB, MAPK, mTOR, PI3K, and JAK/STAT, are expressed and activated under extrinsic stimuli that act through the IFN-γ receptor. These pathways play an important role in cell proliferation, cell cycle progression, transcription factor regulation or activation, and regulation of the nuclear translocation of transcription factors to the PD-L1 promoter. 24
T helper cells (Th1), which can express CD4 receptors, act as regulators of the immune response. The CD4-positive Th1 subset produces cytokines, such as interleukin (IL-2) and IFN-γ, that stimulate macrophages to eliminate intracellular pathogens. 25 In the setting of HIV infection, the innate and adaptive immune responses are affected. Fewer CD4-positive cells may prevent IL-2 and IFN-γ secretion, 26 even after a lengthy period of antiretroviral therapy. 27 Hence, it is possible that lower levels of pro-inflammatory cytokines, particularly IFN-γ, can affect the tumor microenvironment and lead to a decrease in PD-L1 expression in tumor cells or immune cells of HIV patients. In addition, some studies have demonstrated that several signalling genes, such as NF-κB, MAPK, and JAK/STAT, are also down-regulated after ART.28,29 Nevertheless, the exact molecular mechanism for these findings remains unknown. Further studies are warranted on these novel observations regarding cervical cancer cell biology, as well as on the impact of ART and CD4-positive T cells on PD-L1 expression in HIV-positive patients.
Based on immunohistochemistry data and the cut-off value of CPS ⩾ 1, we found that 79% and 87% of cervical carcinomas were positive for PD-L1 in HIV-positive and negative patients, respectively, which was in accordance with previous reports.8,9 When using the cut-off value of TPS ⩾ 25%, only 31% and 50% of HIV-positive and HIV-negative cervical carcinoma, respectively, were determined as PD-L1 positive. Nevertheless, these frequencies were within the 30–70% range previously reported.30–33 Several factors may contribute for the different rates of PD-L1 expression seen, including different scoring techniques used between studies or the defined positive cut-off points. The heterogeneity of PD-L1 staining in tumors, along with the tissue fixation conditions and epitope stability, may further contribute to these differences.
It is also important to note that the different scoring algorithms and cut-off values for PD-L1 staining are specific to tumor entities and certain clinical decisions. The TPS only considers membranous staining of tumor cells, while the CPS comprises PD-L1 expression in both tumor and immune cells into a single score, providing a broader view of the cancer immune status. However, these scoring methods, as well as cut-off values, might differ between HIV-positive and HIV-negative patients. Our results indicated that there are differences in cervical cancer cell biology between these patients. Moreover, HIV-associated factors, including CD4-positive cell counts and ART may also impact on PD-L1 expression and promote inconsistent results among studies. Altogether, we believe that use of PD-L1 immunohistochemistry should be reassessed and exercised with caution in patients with HIV.
Owing to the limitations of immunohistochemical studies, determination of aberrant amplification of the 9p24.1 locus by FISH may represent an alternative option to evaluate PD-L1 status. An increase in 9p24.1 locus copy number represents a somatic change that results in the gain of a fraction of the DNA region and amplification of a number of significant genes, a process that has been associated with tumor progression and carcinogenesis. These genes can act as triggers and/or supporting factors of the underlying mechanism involved in drug resistance. Therefore, they may serve as prognostic markers and potential therapeutic targets. 34 For example, in the case of 9p24.1 locus amplification, its encoded genes PDCD1LG2, CD274, and JAK2 can promote PD-L1 expression in many cancers, such as nodular sclerosing Hodgkin lymphoma, mediastinal large B-cell lymphoma, glioblastomas, and carcinomas of colon, gastric, and triple-negative breast cancers, as well as of cervix and vulva.10–13
Our study showed that copy number gains from the amplification of PD-L1 occurred in a small number of cases (8% in HIV patients and 6.5% in non-HIV patients), which was a much lower incidence than the 67% PD-L1 amplification among HIV-negative cervical cancer patients reported by Howitt et al. 13 This difference may be due to differences in sample sizes, stage of the disease, or health conditions of the participants in each study.
Our study also demonstrated that evaluation of copy number gains for PD-L1 (amplification and polysomy) was more efficient to predict survival outcomes than immunohistochemical PD-L1 expression assessment. PD-L1 copy gain could serve as a reliable prognostic marker in cervical carcinoma patients in both HIV and non-HIV patients. Therefore, treatment of cervical cancer with anti‑PD‑1/PD‑L1 therapy may be tailored according to PD‑L1 copy number alterations. Further studies are warranted to evaluate the relationship between anti‑PD‑1/PD‑L1 therapy and clinical outcomes of cervical cancer patients with CD274 amplification.
Although this study added useful information to the current knowledge of cervical cancer, it had some limitations. The main limitation was the small sample size of HIV patients who had or had not received ART, which may have led to low statistical power. Since this was a retrospective study, various treatments were administered to the patients; therefore, the prognostic factors influencing survival outcomes may have not been thoroughly assessed. Another limitation was the use of tissue microarray techniques for pathological procedures and assessment. The limited area of tumor sampling may not represent the genuine PD-L1 copy number status, which might be heterogeneous. Nevertheless, we endeavored to overcome these limitations by increasing the case:control ratio in order to improve the statistical power of our study and by assessing 3–4 core tissue punching for each case in the TMA procedure.
Locally advanced cervical cancer generally has poor prognosis and frequently fails after primary treatment. Therefore, the stability and prognostic significance of PD-L1 copy number status and protein expression in tumor tissue from metastatic or recurrent settings would be clinically useful and should be explored in future studies.
In summary, PD-L1 amplification and polysomy are the most potent drivers of PD-L1 expression in cervical cancer cells, whereas use of PD-L1 immunohistochemistry should be re-assessed and exercised with caution in patients with HIV due to differences in cancer cell biology and HIV-associated influential factors. An increase in the PD-L1 gene copy number is more reliable and could be an alternative prognostic and possible predictive biomarker for anti-PD-1/PD-L1 therapy, regardless of HIV status.
Footnotes
Acknowledgements
The authors would like to thank Somneuk Jesadapatarakul, Sujitra Tanvanich, Unaporn Sitthivilai, and Pornpimon Kongjan in the Department of Anatomical Pathology, Navamindradhiraj University for academic advice and support; Thawi Yingsa-nga (Division of Nuclear Medicine, Maharat Nakhon Ratchasima Hospital) and Wanniga Saengsuri (Division of Gynecologic Oncology, Taksin Hospital) for their advice in clinical practise; and Oraphan Wanacharoen (BCC MDx Co., Ltd., Bangkok, Thailand) for her kind technical support.
Author contributions
KL and NP designed the study and wrote the manuscript. JT and NiP performed the statistical analysis and revised the manuscript. ST provided clinical information and revised the manuscript. All authors have read and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Navamindradhiraj University Research Fund.
