Abstract
New insight on the interaction between the immune system and tumor has identified the programmed death-1/programmed death-1 ligand pathway to be a key player in evading host immune response. The immune checkpoint modulator, nivolumab (BMS-936558/ONO-4538), is the first PD-1 inhibitor to gain regulatory approval, for the treatment of patients with unresectable melanoma. This review will discuss results from early phase studies of nivolumab in solid tumors including non-small cell lung cancer (NSCLC) as well as studies of nivolumab in combination with chemotherapy, other immune modulators and molecular targeted therapy in patients with NSCLC.
Keywords
Introduction
A total of 1.6 million new cases of lung cancer are diagnosed each year, with 1.4 million deaths annually [Jemal et al. 2011]. Whilst advances in chemotherapy and molecular targeted therapy have improved outcomes in the advanced setting, survival remains poor with a 5-year overall survival (OS) of 4–5% [Howlader et al. 2014]. The advent of immunotherapy in non-small cell lung cancer (NSCLC) has begun to change the landscape of the management of this disease, offering a potential for prolonged responses and survival. For many years, lung cancer was believed to be nonimmunogenic with the failure of many agents such as Bacillus Calmette-Guerin (BCG), interleukin (IL)-2 and interferon [Einhorn et al. 1978; Jansen et al. 1992; Schiller et al. 1995]. However, new models of immunology and a better understanding of the interaction between the immune system and tumor has enabled the development of a new generation of cancer vaccines and immune modulators for NSCLC.
Immune checkpoint inhibition
Genetic and epigenetic variations in transformed cells lead to variation in antigen expression, but there is much controversy on whether antigenic signals on cancer cells are due to genomic instability of the cancer cells or associated with specific antigens related to the transformation [Schreiber et al. 2011; Wrangle et al. 2013]. Tumor recognition followed by tumor antigen presentation to T cells and T-cell activation leads to tumor cell kill. T-cell-mediated immune response is modulated by stimulatory and inhibitory signals. Checkpoint molecules (co-inhibitory molecules) include cytotoxic T-lymphocyte antigen-4 (CTLA-4), programmed death-1 (PD-1), T-cell immunoglobulin- and mucin domain-3-containing molecule 3 (TIM3), lymphocyte-activation gene 3 (LAG3) and killer cell immunoglobulin-like receptor (KIR) [Pardoll, 2012]. These immune checkpoints exist in a normal physiological state to protect against autoimmunity and inflammation. In a neoplastic state, dysfunction of these immune checkpoint proteins can lead to tumor tolerance and eventually allow for tumor ‘escape’ from the immune system. Targeting the molecules that regulate the immune response using antibodies has been the subject of much research and has yielded some promising and exciting results. This review will focus on a promising immune checkpoint inhibitor, an anti-PD-1 antibody, nivolumab. The role of other immune checkpoint inhibitors has been reviewed elsewhere [Brahmer, 2013, 2014; Brahmer and Pardoll, 2013; Sundar et al. 2014].
PD-1 and PD-L1
PD-1 receptor is expressed on several cells including CD4 and CD8 lymphocytes, B lymphocytes, natural killer (NK) cells and T regulatory cells (Tregs) [Keir et al. 2008]. Ligands of PD-1 include PD-L1 (or CD274, B7-H1) and PD-L2 (CD 273, B7-DC) [Dong et al. 2002]. PD-L1 is expressed in T and B cells, dendritic cells and macrophages (Figure 1). It is upregulated in a range of solid tumors including NSCLC. Upon induction by cytokines such as IL-4, IL-10, interferon (IFN) α, β or γ, PD-L1 activates PD-1 on T cells, and downregulates T-cell effector function. Through this mechanism the PD-1 pathway plays a role in the control of T-cell activity in peripheral tissue during an inflammatory response. In a similar manner, tumor cells can exploit the PD-1/PD-L1 pathway as a means of evading T-cell-regulated immune surveillance. PD-L1 is upregulated by IFNγ produced by T cells or through constitutive oncogenic signaling via the mitogen-activated protein kinase (MAPK) pathway or the phosphoinositide 3-kinase (PIK3) pathways [Pardoll, 2012]. Constitutive signaling through the EGFR pathway can also promote tumor escape via activation the PD-1/PD-L1 pathway [Akbay et al. 2013].

Interaction between PD-1/PD-L1 and T cell immune response. T cells are primed and activated through the interaction with antigen-presenting cells. T cells recognizes tumor via MHC–antigen interaction. Tumor PD-L1 and PD-L2 is upregulated by interferon γ released from activated CD8 T cells. PD-L1 in turn inhibits T cells via PD-1.
PD-L1 is localized in the cell membrane and/or the cytoplasm and is expressed in about 20–65% of NSCLC [Konishi et al. 2004; Mu et al. 2011; Chen et al. 2012, 2013; Sundar et al. 2014]. The overexpression of PD-L1 in resected NSCLC is associated with a poorer prognosis [Chen et al. 2012; Azuma et al. 2014] whilst other studies have reported either improved outcomes [Velcheti et al. 2014] or no association with survival [Yang et al. 2014; Boland et al. 2013; Konishi et al. 2004]. In advanced stage NSCLC, no association between PD-L1 expression and survival was reported [Sorenson et al. 2014].
PD-L2 interacts with PD-1 to inhibit T-cell effector function [Tseng et al. 2001]. Unlike PD-L1, which is expressed more broadly, the expression of PD-L2 is more limited, with expression mainly in macrophages and dendritic cells [Rozali et al. 2012]. This suggests PD-L2 is less effective in regulating peripheral T-cell response [Chen et al. 2012]. The expression of PD-L2 in tumor tissue is associated with a poorer survival in patients with resected NSCLC [Zhang et al. 2014].
Several antibodies directed against PD-1 have been developed including nivolumab (BMS936558, Opdivo®), pembrolizumab (MK-3475, lambrolizumab, Keytruda®), and pidilizumab (CT-011). AMP-224 is a fusion protein targeting PD-1. Nivolumab is the first PD-1 inhibitor to gain regulatory approval and is now approved for use in patients with unresectable melanoma in Japan [ONO Pharmaceutical, 2014]. Nivolumab has been granted Fast Track designation by the US FDA in NSCLC, melanoma and renal cell carcinoma. A rolling submission for nivolumab in the third-line treatment of squamous NSCLC was initiated in April 2014 based on the CheckMate063 study (to be discussed later) and submission is expected to be completed by the end of 2014 [Bristol-Myers Squibb, 2014]. In this review we present the latest data on nivolumab in NSCLC.
Nivolumab
Nivolumab/MDX-1106/BMS-936558/ONO-4538 is a genetically engineered, fully human immunoglobulinG4 (IgG4) monoclonal antibody specific for human PD-1. The IgG4 isotype was engineered to obviate antibody-dependent cellular cytotoxicity (ADCC). Most monoclonal antibodies in therapeutic oncology contain the IgG1 subtype, which have the most significant ADCC whereas IgG4 subtype possesses minimal ADCC activity. An intact ADCC has the potential to deplete activated T cells and tumor-infiltrating lymphocytes and diminish activity as PD-1 is expressed on T effector cells and other immune cells [Chen et al. 2012]. Nivolumab binds PD-1 with high affinity (KD 2.6 nmol/l by Scatchard analysis to polyclonally activated human T cells) and blocks its interactions with both B7-H1 and B7-DC [Brahmer et al. 2012].
Early phase trials of nivolumab in lung cancer
In a first in human phase I study, patients with advanced stage solid tumors were treated with a single dose of nivolumab at 0.3, 1, 3 or 10 mg/kg [Brahmer et al. 2010]. Patients with clinical benefit were eligible for repeat dosing at 3 months. Nivolumab was well tolerated, dose-limiting toxicities (DLTs) were not reached and the maximum tolerable dose (MTD) was not defined. Of the six patients with NSCLC, one had significant lesional response not amounting to a partial response (PR). Response was seen in 12 out of the 39 patients. Whilst the measured half-life of nivolumab was 12–20 days, the pharmacodynamic effects of PD-1 receptor occupancy was even more prolonged at 85 days, indicating the biological durability of this high-affinity mAb. Based on the safe toxicity profile and signs of clinical activity, a larger multidose trial was subsequently conducted.
In a phase Ib study of patients with advanced stage solid tumors including melanoma, NSCLC, renal cell cancer, prostate cancer and colorectal cancer, escalating doses of nivolumab biweekly were administered [Topalian et al. 2012]. Of the NSCLC cohort (n = 129) many were heavily pretreated, with 54% receiving at least 3 prior lines of therapy. The overall response rate (ORR) was 17% with a median duration of response of 74 weeks (range 6.1–133.9 weeks). Amongst the responders, 50% showed reduction in tumor size within the first 2 months of treatment. 33% of the squamous tumors (6 out of 18) and 12% of the nonsquamous tumors (7 out of 56) responded. Sustained response of >24 weeks was seen in 57% (8/14) of patients and 2 patients had a response of more than 1 year. Stable disease of more than 24 weeks was seen in 5 of the nonsquamous tumors. Updated results presented in abstract form reported prolonged survival with a median of 9.2–14.9 months across the cohorts with a 1-year survival of 32–56% and 12–45%, respectively. For the 3 mg/kg cohort, the median OS was 14.9 months, 1-year OS of 56% and 2-year OS of 45% [Brahmer et al. 2014].
Toxicity of nivolumab
In the phase Ib study discussed previously, nivolumab was generally well tolerated, with 41% developing adverse events, but only 6% developing grade 3 or 4 toxicities. Skin toxicities were the most common toxicity occurring in 31% of patients, with the most common being that of rash (12%), pruritus (9%) and vitiligo (3%). Gastrointestinal toxicities such as diarrhea occurred in about 11%, while pneumonitis occurred only in 3% of patients, with only 1% having grade 3 or 4 pneumonitis. Other toxicities included abnormalities in transaminases (7%), thyroid dysfunction (3%) and infusion-related reactions (3%).
Pneumonitis is a serious adverse event and is of major concern in lung cancer patients who may already have poor lung reserve due to prior smoking or metastatic disease. In the lung cancer cohort, pneumonitis was reported in 6% (8/129), 2% was grade 3/4 (3 patients) and two patient deaths were attributed to pneumonitis [Brahmer et al. 2013]. These toxicities have been similar to other PD-1 agents and other nivolumab trials [Brahmer et al. 2012; Horn et al. 2013; Garon et al. 2014b]. Pneumonitis rates for nivolumab are similar to or lower than rates of other commonly used drugs in NSCLC such as docetaxel (4.6%) [Grande et al. 2007] and gefitinib (3.5%) [Konishi et al. 2005]. However, as nivolumab is currently being tested in combination with several other agents, cumulative toxicity of pneumonitis would be of concern. This is of particular concern since several patients with advanced NSCLC are likely to have poor lung reserve function and prior thoracic radiation might also worsen pulmonary inflammation and pneumonitis.
PD-L1 is expressed in the small intestine [Keir et al. 2008] whereas PD-L2 is important in regulating TH2-mediated inflammation in the lung [Akbari et al. 2010]. PD-1 inhibitors are intended to block PD-1 from interacting with any of its ligands, hence preventing engagement between PD-1 with PD-L1 and PD-1 with PD-L2. In comparison PD-L1 inhibitors prevent binding between PD-1 with PD-L1 and PD-L1 with CD80 (B7.1) but not necessarily PD-1 with PD-L2 binding. The different mechanism of action in agents targeting PD-1 and PD-L1 may potentially result in a different side-effect profile. PD-L1 inhibitors may potentially have a lower risk of pneumonitis whereas targeting PD-1 may have a lower risk of gastrointestinal side effects. As the studies conducted to date differ greatly in terms of patient population, sample size, and duration of follow-up, direct comparisons between the PD-1 and PD-L1 inhibitors should be made with great caution. Further studies are required to determine differences in side effects between PD-1 and PD-L1 inhibitors.
Current ongoing trials of nivolumab in lung cancer
There are currently multiple trials examining the efficacy of nivolumab in patients with advanced stage NSCLC (Table 1). Of interest is the combination of PD-1 blockade with CTLA-4 inhibition (nivolumab plus ipilimumab) and combination with other immune checkpoint modulators. In preclinical studies multiple immune checkpoint blockades with combination PD-1 and CTLA4 Ab can allow for increased T-cell responsiveness and decreased T-cell anergy, in preclinical models [Curran et al. 2010]. This approach has been supported by results of a phase I study of nivolumab and ipilimumab in patients with advanced-stage melanoma where an ORR of 53% was observed in patients receiving concurrent nivolumab and ipilimumab [Wolchok et al. 2013]. However, a cautious approach is warranted given the potential to exacerbate autoimmunity.
Clinical studies of nivolumab in non-small cell lung cancer.
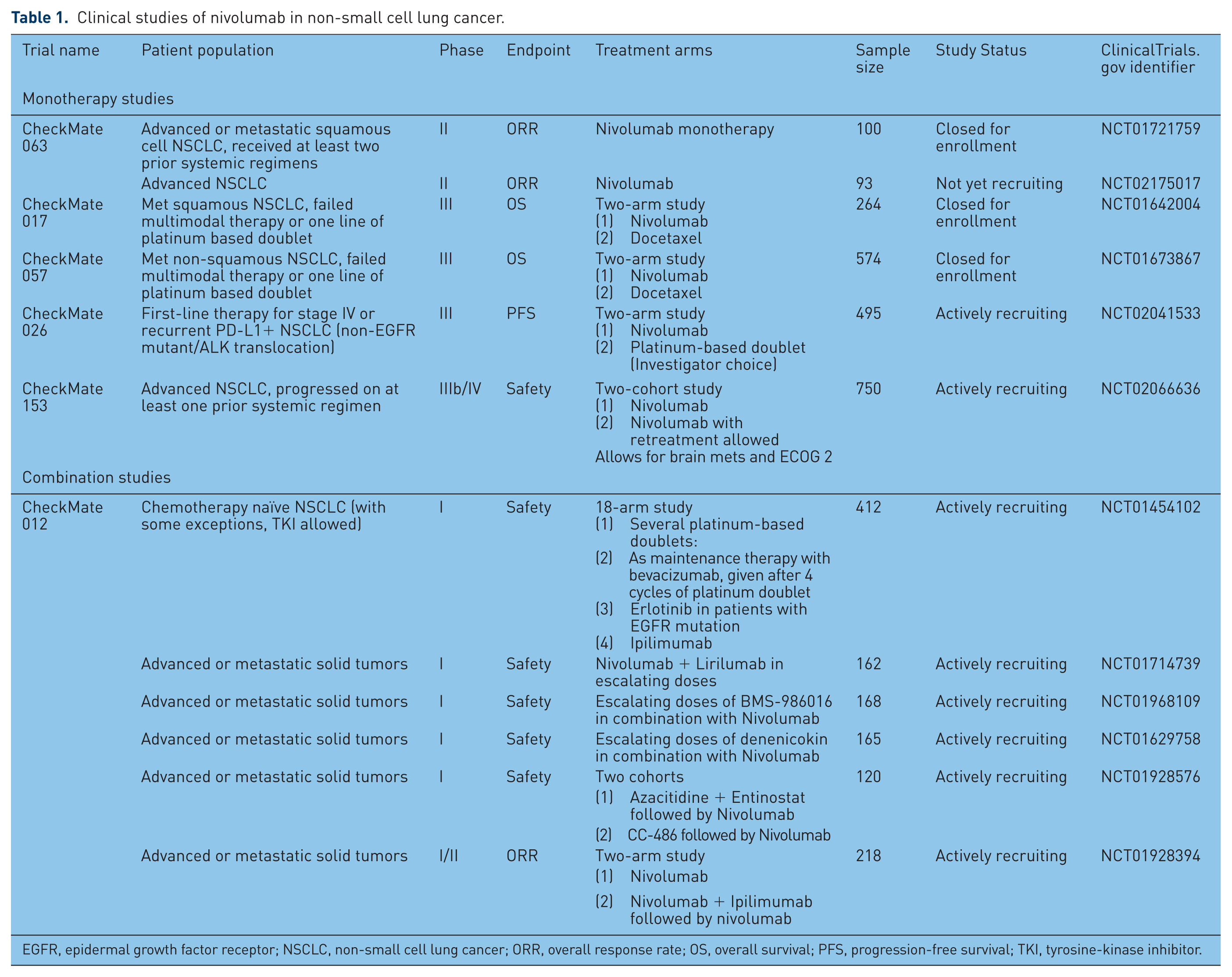
EGFR, epidermal growth factor receptor; NSCLC, non-small cell lung cancer; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; TKI, tyrosine-kinase inhibitor.
Cytotoxic chemotherapy, targeted therapies and radiotherapy can modulate immune response to tumors. An understanding of these immunomodulatory effects may enable the design of rational combinations of chemotherapy, targeted therapy and radiotherapy with immunotherapy. Cytotoxic chemotherapy can modulate the immune system by several mechanisms such as: (1) inducing immunogenic cell death, a form of cell death that induces dendritic cells to stimulate tumor antigen presentation to T cells [Kroemer et al. 2013]; (2) stimulating T-cell activation via increasing the expression of MHC-1 molecules (such as oxaliplatin, gemcitabine, paclitaxel) [Liu et al. 2010]; (3) stimulating dendritic cell maturation (gemcitabine, oxaliplatin) [Liu et al. 2010]; and (4) reducing the immunosuppressive function of regulatory T cells (cyclophosphamide, paclitaxel) [Ghiringhelli et al. 2007; Zhang et al. 2008] and myeloid derived suppressor cells (MDSCs; gemcitabine, docetaxel) [Suzuki et al. 2005; Kodumudi et al. 2010]. Vascular endothelial growth factor (VEGF) may have immunosuppressive effects. VEGF stimulate MDSC in peripheral immune organs, promote regulatory T cells and inhibit dendritic cell maturation [Goel et al. 2011]. As such VEGF inhibition in combination with a checkpoint inhibitor may be synergistic [Huang et al. 2013] and a study using this combination in advanced stage NSCLC is ongoing [ClinicalTrials.gov identifier: NCT01454102]. Ionizing radiation can increase tumor susceptibility to T-cell killing through increased expression of death receptors, MHC class 1 molecules, costimulatory molecules, adhesion molecules and stress-induced ligands [Formenti and Demaria, 2013, Frey et al. 2014]. As these anticancer treatments can potentially modulate antitumor immune response, there is good rationale to combine it with immune checkpoint inhibitors.
As immune checkpoint inhibitors induce T-cell response, patients treated with supraphysiological doses of corticosteroids are inappropriate candidates for immune checkpoint inhibitors as systemic corticosteroids may attenuate the potential beneficial effects of these drugs [Harvey, 2014]. As such, patients on systemic doses of corticosteroids have been excluded from clinical trials of nivolumab and other immune checkpoint inhibitors. [Topalian et al. 2012; Brahmer et al. 2012; Wolchok et al. 2013; Hamid et al. 2013]. Low-dose steroids as an anti-emetic or premedication for paclitaxel [Lynch et al. 2012] can be used, as it is unlikely that the therapeutic effect of immune checkpoint inhibitors will be affected at low doses. In fact, continued antitumor activity has been observed in patients with immune-related adverse events treated with high-dose steroids [Downey et al. 2007; Harmankaya et al. 2011]. Generally steroids may be used if needed, but should be avoided if not required [Fecher et al. 2013].
CheckMate 012 [ClinicalTrials.gov identifier: NCT01454102] is a multi-arm phase I study of nivolumab with various anticancer agents or as monotherapy (Table 1). Preliminary results from four treatment arms from this study have been presented recently at the 2014 ASCO and are summarized below (Table 2).
Preliminary results from the multi-arm phase I study of nivolumab (Checkmate 012).
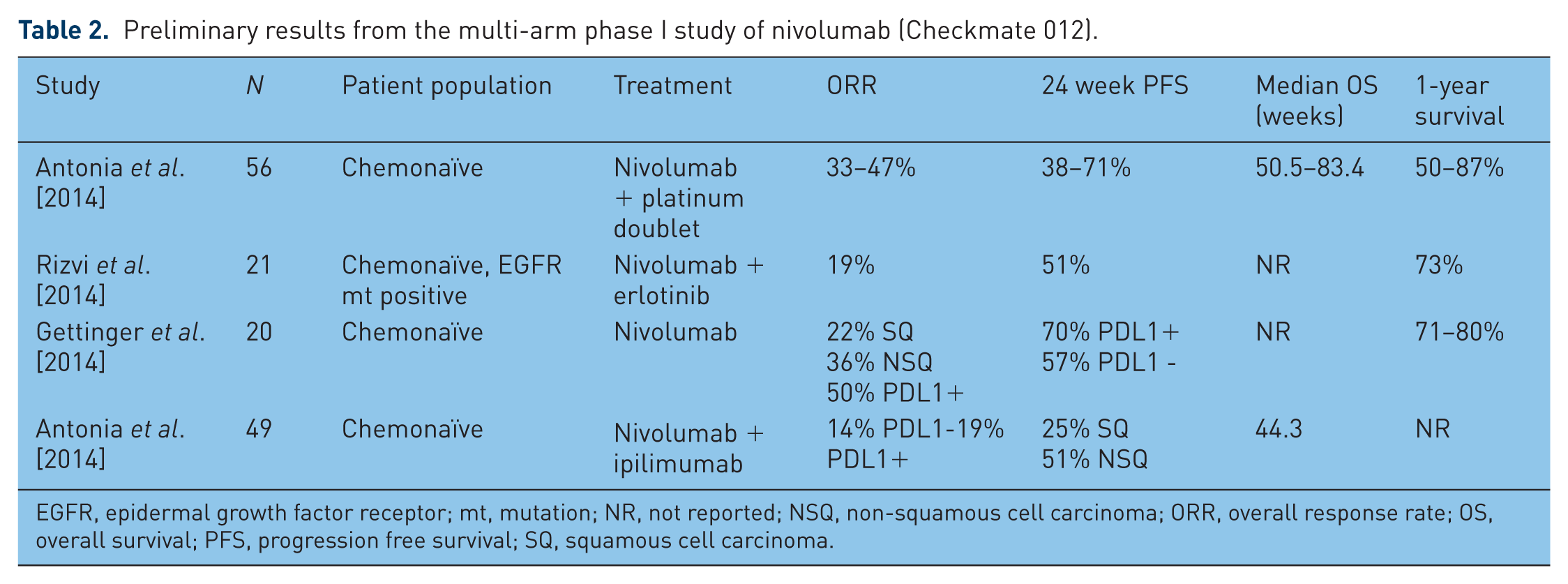
EGFR, epidermal growth factor receptor; mt, mutation; NR, not reported; NSQ, non-squamous cell carcinoma; ORR, overall response rate; OS, overall survival; PFS, progression free survival; SQ, squamous cell carcinoma.
In the platinum doublet arm, where three platinum-based chemotherapy regimens (cisplatin/gemcitabine; cisplatin/pemetrexed; and carboplatin/paclitaxel) were combined with nivolumab (n = 56), the ORR was 33–50%, 24-week progression-free survival (PFS) 36–71% and 1-year OS was 59–87% [Antonia et al. 2014a]. No DLTs were seen in the first 6 weeks of treatment, but 45% of patients had grade 3 or grade 4 toxicities and 7% (n = 4) had pneumonitis. Based on these preliminary results, the antitumor activity of first-line nivolumab in combination with platinum doublets is highly promising.
In the erlotinib and nivolumab arm, chemotherapy-naïve patients (n = 21) with EGFR mutations were enrolled. A total of 20 patients had prior treatment with first-line erlotinib. The ORR was 19% and the 24-week PFS was 51% [Rizvi et al. 2014]. Interestingly, of those with acquired erlotinib resistance, 3 (15%) had a PR, 9 (45%) achieved SD, and 1 had an unconventional immune related response. Grade 3 toxicities were reported in 19% of patients (there were no grade 4 toxicities). Common side effects included skin rash, fatigue, paronychia, diarrhea and skin fissures. Whilst this combination show encouraging activity, the data is preliminary and validation and comparison with the third-generation EGFR tyrosine-kinase inhibitors (TKIs) in tumors harboring T790M mutations would be needed. Further work on the interaction between tumors harboring T790M mutations and the tumor microenvironment and immune checkpoints would also be of interest.
In the cohort of chemotherapy-naïve patients with advanced NSCLC treated with single-agent nivolumab (n = 20), the ORR was 30%. The 1-year OS and median PFS was 75% and 36 weeks, respectively [Gettinger et al. 2014]. Grade 3/4 treatment related toxicities were reported in four (20%) patients. In exploratory analysis, the ORR, PFS rate at 24 weeks, median PFS and 1-year OS were numerically higher in patients with tumors expressing PD-L1 compared with PD-L1 negative tumors. A phase III study of single-agent nivolumab versus investigator choice of a platinum doublet in the first-line treatment of advanced stage NSCLC is ongoing [ClinicalTrials.gov identifier: NCT02041533].
In the nivolumab and ipilimumab combination arm, the ORR was 22%, stable disease was 33% and the PFS rate at 24 weeks was 20–51%. No association was found between PD-L1 status and clinical activity. Treatment-related adverse events were reported in 88% of patients, most commonly fatigue (45%). Pneumonitis was reported in six (12%) patients (all grades) and three (6%) were grade 3/4. All cases were reversible upon discontinuation of study drugs and use of corticosteroids. Grade 3-4 treatment-related toxicity was reported in 24 patients (49%). Diarrhea was the most common toxicity (10%) [Antonia et al. 2014b] Treatment-related deaths occurred in three patients (two from pulmonary toxicities and one from toxic epidermal necrolysis). The data, although preliminary, is promising as this combination is active in both PD-L1-positive and PD-L1-negative NSCLC. This combination may be warranted in selected patients, such as patients with PD-L1-negative tumors where nivolumab alone may have lower clinical activity. Additional studies are required to further define safety and clinical activity of this combination.
Checkmate 063 (Table 1) is a study of single-agent nivolumab in patients who had failed two or more lines of systemic therapy. Preliminary results reported an ORR of 15% (95% confidence interval [CI] 8.7–22.2), with a median OS of 8.2 months and a 1-year OS of 41% (95% CI 31.6–49.7). Pneumonitis rate was reported as 3.4% [Ramalingam et al. 2014]. Results are highly promising given 65% had been treated with three or more lines of therapy.
KIR is a family of NK-cell regulators represents a novel class of immunotherapy and is beginning to garner interest in various tumor types. KIR is one of the negative regulators of NK-cell effector function. The inhibition of KIR results in NK-cell-mediated antitumor activity [Ruggeri et al. 2002]. Lirilumab is an antibody that binds to KIR2DL1, 2 and 3 receptors, causing NK-cell-mediated cell kill [Romagne et al. 2009]. A phase I study combining lirilumab and nivolumab in patients with advanced tumors is ongoing [ClinicalTrials.gov identifier: NCT01714739].
Lymphocyte-activation gene 3 (LAG3; CD223) is a co-inhibitory receptor expressed in activated T cells, Tregs, dendritic cells and NK cells. LAG3 is a CD4-related protein that binds to major histocompatibility complex (MHC) class II and reduces T-cell proliferation resulting in tumor evasion [Huang et al. 2004; Gandhi et al. 2006]. LAG3 gene expression is upregulated in a silica-mediated lung inflammation murine model [Freire et al. 2013]. Further work is needed to characterize LAG3 in NSCLC, with a current phase 1 study looking at the role of BMS-986016, a LAG3 monoclonal antibody with or without nivolumab in advanced solid tumors [ClinicalTrials.gov identifier: NCT01968109].
The role of epigenetic priming on subsequent treatment with a PD-1 checkpoint inhibitor is being investigated. It is hypothesized that epigenetic modulation with azacitidine, a cytidine analogue that causes DNA hypomethylation, and entinostat, a histone deacetylase inhibitor, affects inflammation within the tumor by affecting the interferon pathway, leading to PD-L1 upregulation. Azacitidine can also induce cancer testis antigens which are not expressed in normal tissues (except the testes in males and the placenta in pregnant females), while being expressed almost ‘tumor-specifically’ in several different tumors [Van Den Eynde and Boon, 1997]. This leads to an enhanced immune response against the tumor. A study of azacitidine and entinostat or azacitidine alone prior to nivolumab with the primary endpoint of ORR is ongoing [ClinicalTrials.gov identifier: NCT01928576].
Comparison with other PD-1/PD-L1 inhibitors
Although cross-trial comparisons are not possible, especially in this very early stage of development of various immune checkpoint inhibitors, PD-1/PD-L1 inhibitors appear to have similar efficacy in pretreated advanced stage NSCLC. The ORR of the anti-PD-1 antibody pembrolizumab (MK-3475) is 15–24% [Garon et al. 2013, 2014a]. In a pooled analysis of pembrolizumab in phase I studies, an ORR of 21% (16–27%) and a median PFS of 27 weeks (95% CI 14–45 weeks) was reported in treatment-naïve patients, with a 24-week PFS of 51%. In pretreated patients, the median PFS was 10 weeks [Garon et al. 2014a]. Anti-PD-L1 antibodies MPDL3280A and MEDI4736 have ORR of 23% and 15%, respectively [Soria et al. 2013, Lutzky et al. 2014], similar to that of nivolumab with an ORR of 24.3% (3mg/kg cohort) [Brahmer et al. 2014b].
Toxicities amongst various antibodies of the same class appear similar as well, with most drugs reporting a pneumonitis rate of less than 5%, while the most common adverse event is that of fatigue. Grade 3 or 4 toxicities of all adverse events appear to be less than 10% for all drugs, suggesting that this class of drugs is a very well tolerated.
Measurement of tumor response
One of the challenges faced in the development of nivolumab and other inhibitors of the PD-1/PD-L1 pathway is the assessment of tumor response. The use of RECIST1.1 for tumor assessment in patients receiving immunotherapy has limitations. For example, it does not capture (i) patients who initially progress as defined by RECIST 1.1 but subsequently respond or (ii) patients with a mixed response or new lesions, but the overall tumor burden is decreased. Based on this, an immune-related response criterion has been created [Wolchok et al. 2009]. Immune-related progression-free survival (irPFS) accounts for the apparent increase in tumor size followed by sustained tumor response, which has been documented with these agents in the past [Oxnard et al. 2012]. This phenomenon of ‘pseudo-progression’ may be due to peritumoral lymphocyte infiltration or delayed immune activity.
Biomarkers
The need for predictive biomarkers is important, as nivolumab is highly active in a very select group of patients. In the landmark phase I study, the association between ORR and PD-L1 expression in NSCLC was investigated [Topalian et al. 2012]. PD-L1-positive tumors, defined as expression in at least 5% of tumor cells on immunohistochemistry (IHC), were seen in 49% (31/63) of patients. The ORR in these heavily pretreated patients with PD-L1 positive and PD-L1 negative tumors was 16% and 13%, respectively [Antonia et al. 2013]. Using archival tumor tissue may not be ideal for assessing PD-L1 status especially in a heavily pretreated group of patients hence the disparate results between studies. In a more recent study of chemotherapy-naïve patients with advanced-stage NSCLC treated with nivolumab, the ORR was 50% in PD-L1-positive tumors whereas the ORR was 0% in PD-L1-negative tumors [Gettinger et al. 2014]. The use archival tumor samples from pretreated patients in the pivotal phase I study may explain the differences in ORR according to PD-L1 expression between the two studies. PD-L1 expression appeared to correlate with pembrolizumab ORR, with strong PD-L1 expression (>50%) having an ORR of 39%, while PD-L1 negative being only 9% [Garon et al. 2014a]
As the frequency of PD-L1 positive NSCLC tumors is about 20% [Sundar et al. 2014], potentially a large number of patients with advanced stage NSCLC may be suitable for nivolumab treatment. In comparison, the frequency of patients with EML-ALK translocation or ROS1 rearrangement is about 4% [Solomon et al. 2009] and 1–2% [Leow et al. 2012], respectively, depending on the population studied and detection methods used.
It is recognized PD-L1 expression varies with the tumor microenvironment [Taube et al. 2012] and thus PD-L1 expression at a single time point may not represent a dynamic immune response. Thus PD-L1 expression as a predictive biomarker may be difficult to implement. Further studies are needed to characterize immunologic correlates of response in the pretreatment and on-treatment setting.
Future directions
Several questions regarding the optimal duration of therapy and the role of retreatment with nivolumab are yet to be answered. A recent clinical trial [ClinicalTrials.gov identifier: NCT02066636] described aims to answer some of these questions. Issues of long-term toxicities and consequences of prolonged exposure to immunotherapy are to be addressed by this study.
It has been reported that tumor bulk can affect the ability to mount a successful tumor-specific immune response [Stewart and Abrams, 2007]. One potential approach would be to administer nivolumab and other PD-1, PD-L1 inhibitors after complete gross tumor resection to target minimal residual disease. As such studies of nivolumab in the adjuvant setting should be explored.
Another area of great interest is elucidating the interaction between oncogene-driven signaling pathways regulating immune escape and the regulation of tumor-infiltrating lymphocytes. It is likely that tumor genomic profiling will be integrated with profiling of the tumor microenvironment to provide a more comprehensive approach to treatment selection.
Conclusion
A deeper understanding of the immune system in tumor immunosurveillance has led to the development of a novel approach to the management of advanced stage NSCLC. Whilst results from early phase studies of immune checkpoint modulators such as nivolumab, in a range of solid tumors including NSCLC are highly promising, these trials require confirmation. Research is ongoing to combine immunotherapy agents with chemotherapy and molecular targeted therapy and to identify patients who are likely to respond to PD-1/PD-L1–inhibitors.
Footnotes
Acknowledgements
RAS is supported by the National Research Foundation, Singapore and the Singapore Ministry of Education under its Research Centres of Excellence initiative.
Conflict of interest statement
RS, BCC and RAS have no conflicts of interest to declare. JBR has received research funding from Merck and BMS, is a paid consultant for Merck and is a compensated advisory board member for BMS.
