Abstract
Background:
Few data are available on programmed cell-death-protein-1–ligand-1 (PD-L1) expression on large-cell neuroendocrine carcinomas of the lung (LCNECs). We analyzed PD-L1 expression on tumor (TCs) and inflammatory cells (ICs) from LCNEC patients to assess relationships between this expression, clinical characteristics, and disease outcomes.
Methods:
PD-L1 expression was determined by immunohistochemistry with monoclonal antibody 22C3 in consecutive LCNEC patients managed in 17 French centers between January 2014 and December 2016.
Results:
After centralized review, only 68 out of 105 (64%) patients had confirmed LCNEC diagnoses. Median overall survival (OS) (95% CI) was 11 (7–16) months for all patients, 7 (5–10), 21 (10–not reached) and not reached months for metastatic, stage III and localized forms (p = 0.0001). Respectively, 11% and 75% of the tumor samples were TC+ and IC+, and 66% had a TC–/IC+ profile. Comparing IC+ versus IC– metastatic LCNEC, the former had significantly longer progression-free survival [9 (4–13) versus 4 (1–8) months; p = 0.03], with a trend towards better median OS [12 (7–18) versus 9.5 (4–14) months; p = 0.21]. Compared to patients with TC– tumors, those with TC+ LCNECs tended to have non-significantly shorter median OS [4 (1–6.2) versus 11 (8–18) months, respectively]. Median OS was significantly shorter for patients with TC+/IC– metastatic LCNECs than those with TC–IC+ lesions (2 versus 8 months, respectively; p = 0.04).
Conclusion:
TC–/IC+ was the most frequent PD-L1–expression profile for LCNECs, a pattern quite specific compared with non-small-cell lung cancer and small-cell lung cancer. IC PD-L1 expression seems to have a prognostic role.
Highlights
After centralized review, only 64% of initial diagnoses of large-cell neuroendocrine carcinoma of the lung (LCNEC) were confirmed.
Respectively, 11% and 75% of the tumor samples expressed PD-L1 on tumor (TCs) and immune cells (ICs).
IC+TC– was the most frequent co-expression pattern.
Median overall survival of metastatic LCNEC patients with the TC+/IC– profile was shorter than for those with the TC–/IC+ pattern.
Introduction
Large-cell neuroendocrine carcinomas of the lung (LCNECs) are rare tumors representing 1–3% of all primary lung cancers. 1 They are classed in the subgroup of lung neuroendocrine tumors in the World Health Organization 2015 classification. 2 Histologically, these tumors are characterized by a neuroendocrine morphology, high mitotic index >11 mitoses/mm2, frequent necrotic plaques and neuroendocrine immunohistochemistry (IHC)-detected markers, for example, chromogranin A, synaptophysin and CD56. 2 Despite being well-individualized since 1991,3,4 the histological diagnosis of these tumors remains difficult, particularly for small-sized tissue samples, with high inter-observer variability interpreting distinction from small-cell lung cancers (SCLCs). 5 Clinically, the prognosis of metastatic LCNECs remains dismal, with median overall survival (OS) of ~9 months. First-line therapeutic options are limited, based on phase-II studies with small sample sizes.6–8 There are no second-or-more-line standard treatments. Immune-checkpoint inhibitors have revolutionized the management of non-small-cell lung cancers (NSCLCs), with efficacy against metastatic disease as first- or second-line therapy.9,10 The percentage of IHC-detected programmed cell-death-protein-1–ligand-1 (PD-L1) expression on tumor cells (TCs) is a clearly identified predictive marker of improved efficacy. 10 Used less frequently, the percentage of PD-L1–expressing tumor-infiltrating inflammatory cells (ICs) may be also a marker predictive of efficacy, 9 as is a high tumor-mutation burden (TMB), >16 mutations/Mb, and independently of PD-L1 status. 11 In light of the rarity of LCNECs, little is known about their frequency of PD-L1 expression.
This multicenter retrospective study was undertaken to study PD-L1 expression on TCs and ICs in the tumors of consecutive patients managed for LCNEC, and to look for relationships between those expression rates and patients’ clinical characteristics and disease evolution.
Patients and methods
The principal inclusion criteria were: age >18 years, LCNEC diagnosis and managed in participating centers between 1 January 2014 and 31 December 2016. Clinical information (age, sex, smoker status, etc.) was collected retrospectively. Each patient’s general condition was evaluated with the Eastern Cooperative Oncology Group Performance Status (ECOG PS), 12 the TNM stage at diagnosis according to the 7th edition. 13 The treatments received were also recorded (surgery, radiotherapy, chemotherapy, first, second and third-line regimens and immunotherapy), the objective response rate (ORR) to first-line chemotherapy according to Response Evaluation Criteria In Solid Tumors RECIST 1.1 criteria, 14 progression-free survival (PFS) and OS.
Centralized review
A panel of 6 pathologists, specialized in thoracic diseases, reviewed all tumor samples centrally and anonymously. The histological diagnosis of LCNEC was retained when the 6 experts reached consensus based on the neuroendocrine morphology, mitotic index, presence of necrotic tumor tissue and IHC-positivity for standard neuroendocrine markers (CD56, chromogranin A, synaptophysin). 2 For endobronchial ultrasonography (EBUS) cytological samples, ⩾100 TCs were required for analysis. In the case of discordant interpretations among the experts and the impossibility of retaining an alternative diagnosis, an unclassifiable endocrine tumor was retained.
IHC
PD-L1–immunolabeling of TCs and ICs used the anti-PD-L1 22C3 antibody (kit and automat Dako, Dako, Agilent, USA). The TC score was defined as the percentage of TCs with membrane PD-L1 labeling, regardless of intensity, with ⩾1% considered TC+. IHC-determined PD-L1 expression, that is, the percentage of the IC surface labeled, was quantified, as reported previously: 9 IC–: <1%; IC1: 1–5%; IC2: 5–10%; and IC3: >10%. Alveolar macrophages, when present, were not included in determining the IC score. For cytological samples from lymph nodes, IHC-determined PD-L1 expression is performed only if more than 100 cancer cells are present. For each patient’s specimen, immunostaining was performed as recommended by Dako, with the 22C3 monoclonal antibody on the Dako autostainer. For each bench of the experiment, negative control was included (control slide without antibody) and pathologists considered that almost one immune cell should be positive for PD-L1 on each slide. When all cells were negative, a new immunostaining on another slide was performed. Two totally negative immunostaining was considered as truly negative.
Statistical analyses
Qualitative variables are expressed as n (%) [95% confidence interval (95% CI)]. Quantitative parameters are expressed as mean ± standard deviation or median [interquartile range (IQR)/95% CI]. OS was defined from the date of diagnosis until the date of death or last update. PFS was defined as the date of treatment onset until the date of first progression or last update. Chi-squared tests were used to determine the relationships between PD-L1 expression and the LCNEC stage. The Kaplan–Meier method was used to analyze FPS and OS, and the log-rank test to analyze survival differences. A p < 0.05 defined significance.
Patients alive at the time of inclusion in the trial were given an information sheet and their written consent for the use of his/her personal information was included in the medical chart. For patients deceased at the time of inclusion, the information was obtained from their case-report forms, unless he/she had provided formal written opposition during his/her lifetime. Île-de-France Ethics Committee (Comité de Protection des Personnes-8) approved this research (ID RCB 2017-A00418-45), which is registered at ClinicalTrials.gov [ClinicalTrials.gov identifier: NCT03305133].
Results
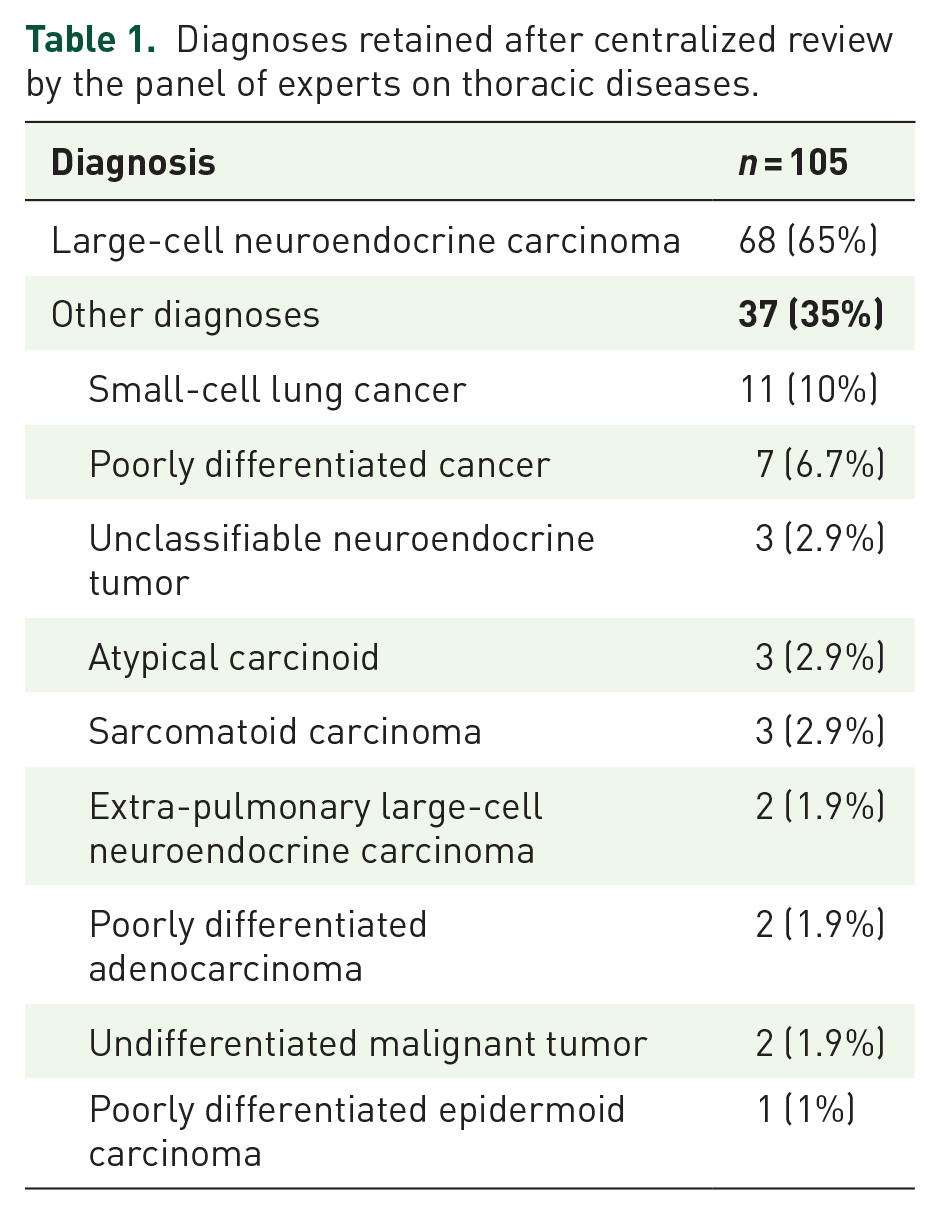
LCNECs were initially diagnosed in 105 patients who attended 17 centers. Their tumor samples were reevaluated during 4 centralized review meetings: LCNEC was not retained as the final diagnosis for 32% (Table 1); SCLC was the most frequently advanced alternative diagnosis. For three patients, no tumor tissue remained or was uninterpretable for further analysis. Only 68 out of 105 (65%) of the patients with a final consensus diagnosis of LCNEC were included in this analysis. They were predominantly men (85%), mean age 64 (range, 38–87) years. Of all the patients for whom the information was available, 92% were smokers or ex-smokers, with a mean of 40 pack-years (Table 2). At diagnosis, 59% of the patients had metastatic LCNECs, 30% locally advanced tumors and 11% localized disease. Most were in good general condition, with ECOG PS 0/1 for 50 (79%) of the 63 patients with available values.
Diagnoses retained after centralized review by the panel of experts on thoracic diseases.
Characteristics of the 68 LCNEC patients.

ECOG PS, Eastern Cooperative Oncology Group Performance Status; LCNEC, large-cell neuroendocrine carcinoma of the lung.
Among the 40 patients with metastatic LCNECs at diagnosis, 29 (72%) had received first-line platinum–etoposide. The ORR to that regimen, evaluated for 31/40 patients, was partial (45%), stable (35%) or progressive disease (20%). During their management, 10 patients had received anti-PD1 (nivolumab) immunotherapy, always as third-line treatment; it controlled the disease for 2 months for half of them (3 stable and 2 partial), sometimes with prolonged durations of control [median, 9 (5–22) months].
At the final analysis carried out in November 2018, with a mean follow-up of 41 (range, 23–57) months, 75% of the patients had died. The median PFS for the entire population was 6 (95% CI, 4–9) months, 4 months for patients with metastatic LCNECs and 11 months for those with stage III disease (p < 0.0001). Median OS for the entire population was 11 (95% CI, 7–16) months, 7 (95% CI: 5–10) months for those with metastatic LCNECs, 21 (95% CI, 10–not reached) months for stage III disease and not reached for localized tumors (p = 0.0001).
PD-L1 immunolabeling of TCs and ICs
Among the 68 tumor samples examined, seven (10.3%) were large surgical resection, 50 (73.5%) bronchial biopsies and 11 (16.2%) lymph node biopsies. Seven (11%) samples were 1–80% TC+. Among the 65 specimens assessable for the IC score (3 EBUS samples could not be evaluated), 25% were IC–, 75% IC+ (25%, 20%, and 31%, respectively: IC1, IC2, and IC3) (Table 3). TC–/IC+ was the most frequent PD-L1-expression profile, found on 66% of the tumor samples (Figure 1).
PD-L1 expression on tumor (TCs) or inflammatory cells (ICs) in 65 LCNECs.
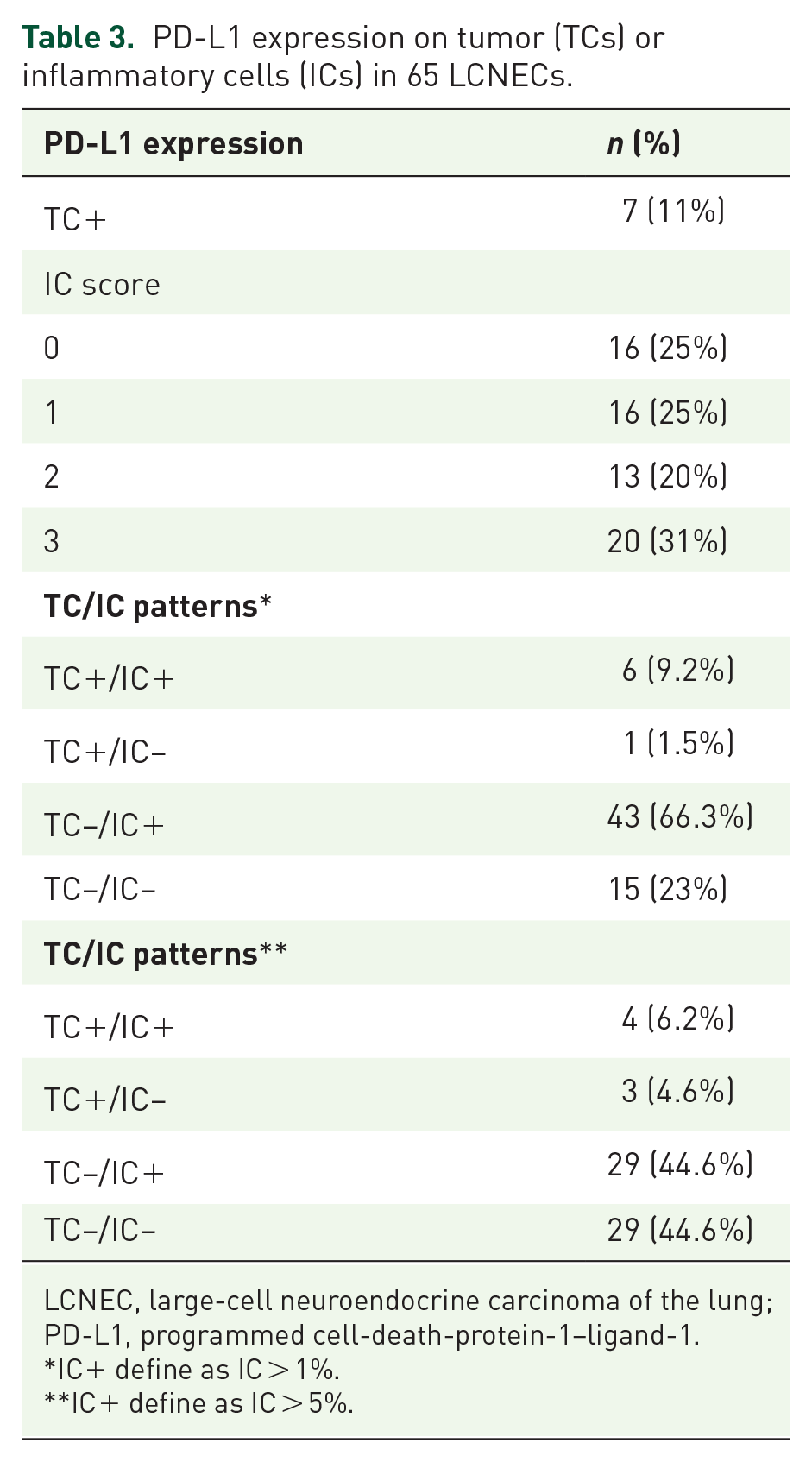
LCNEC, large-cell neuroendocrine carcinoma of the lung; PD-L1, programmed cell-death-protein-1–ligand-1.
IC+ define as IC > 1%.
IC+ define as IC > 5%.
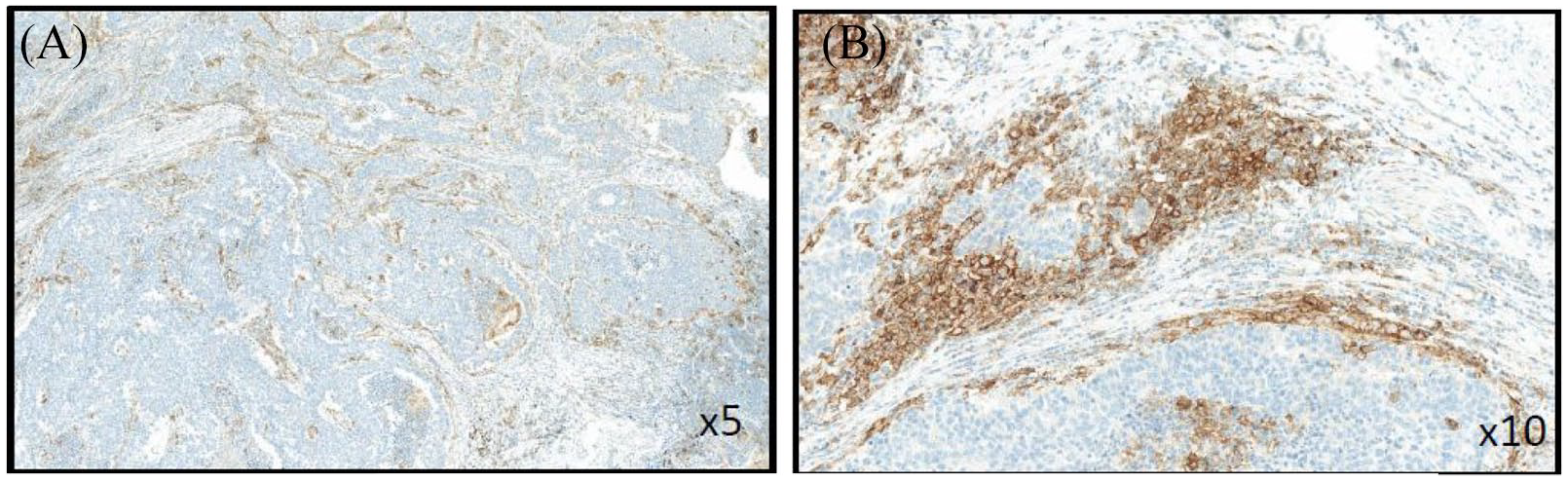
Example of negative programmed cell-death-protein-1–ligand-1 labeling on tumor cells (TC–) and strong positive staining on inflammatory cells (IC+) in the same sample at low (A) and high (B) magnification.
No relationship could be established between the TC score, IC score and the LCNEC stage (data not shown). When the analysis was limited to metastatic disease, no relationship could be found between the TC score, IC score and the OR (R to first-line treatment (data not shown). Moreover, no relationship was found between the ORR to third-line immunotherapy and the TC-score and IC scores (data not shown).
PFS for patients with metastatic IC+ LCNECs was significantly longer than for those with IC– tumors (Figure 2A). The same tendency could be observed for patients with IC2/3 tumors compared with those IC–/1 (Figure 2B). OS was longer, but not significantly so, for patients with IC+ metastatic LCNECs, 9.5 (95% CI: 4–14) versus 12 (95% CI: 7–18) months, p = 0.21. That trend was also found when comparing patients with IC2/3 versus IC–/1 tumors, 9.5 (95% CI: 5–14) versus 14 (95% CI: 7–25) months, p = 0.08. Median OS for patients with TC– LCNECs compared with those with TC+ tumors was non-significantly shorter [4 (95% CI, 1–6.2) versus 11 (95% CI: 8–18) months].
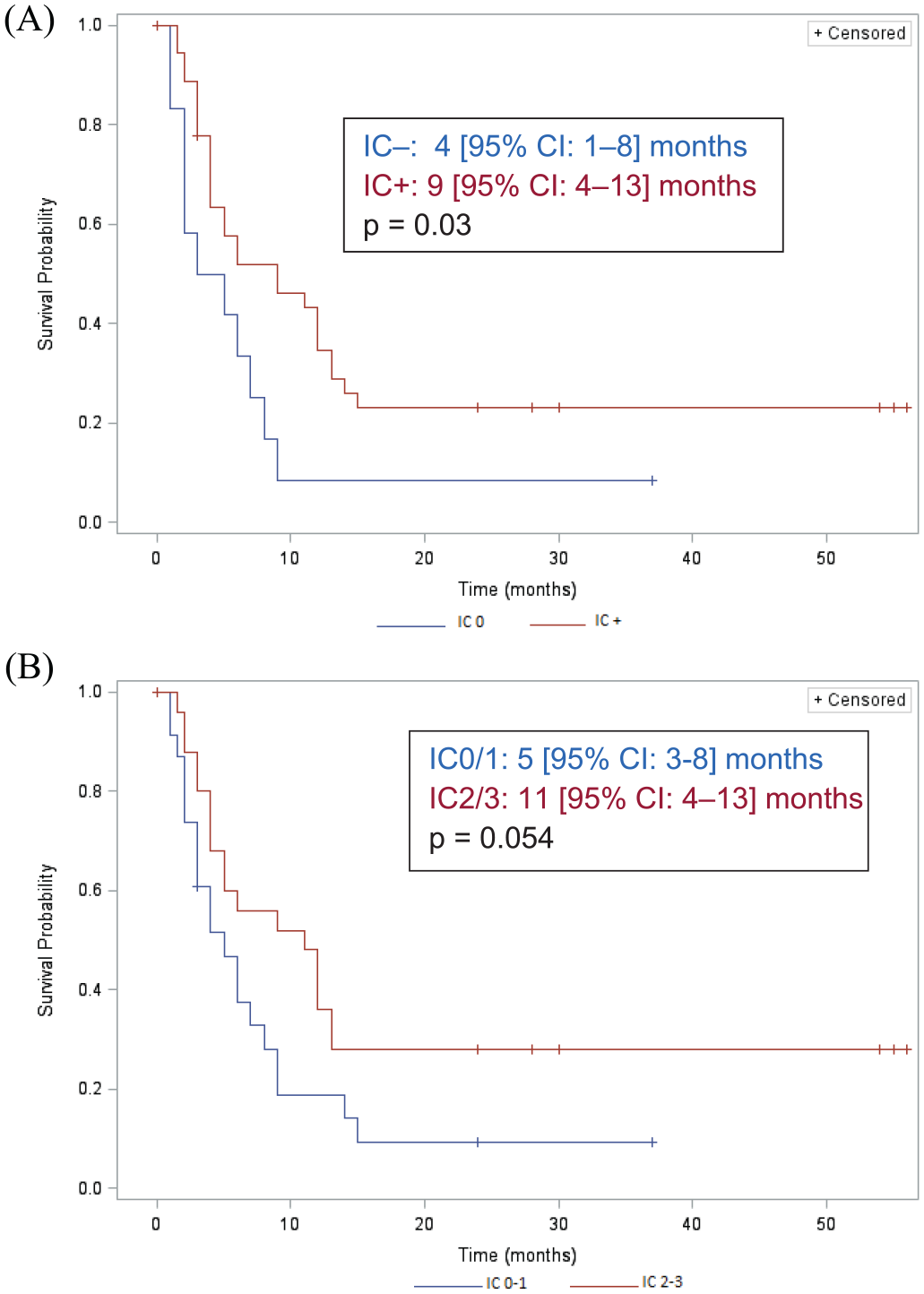
Median progression-free survival (95% confidence interval) of patients with metastatic large-cell neuroendocrine carcinomas of the lung, according to the intensity of programmed cell-death-1–ligand-1 expression on inflammatory cells (ICs): (A): negative IC–: <1%+; or positive (IC+) or (B) negative or IC1: 1–5%+ (IC0–1) or more intense IC2: 5–10%+; and IC3: >10%+ (IC2/3).
The analyses of OS according to PD-L1 expression on TCs and ICs showed that the TC+/IC– group had the worst prognosis, with the shortest, but not significantly different, median compared with longest for the TC–/IC+ subgroup (Figure 3A). When the analysis was restricted to metastatic TC+/IC– and TC–/IC+ LCNECs, the difference reached significance (p = 0.04), while median OS was comparable for the TC+/IC+ and TC–/IC– subgroups (Figure 3B).

Median overall survival (95% confidence interval) for all patients with (A) large-cell neuroendocrine carcinomas of the lung or (B) those with metastatic disease according to tumor (TC) or inflammatory cell (IC) expression of programmed cell-death-1–ligand-1.
Discussion
The results of this multicenter analysis that included 105 patients initially diagnosed as having LCNECs showed that a centralized review by a panel of expert pathologists revised 32% of those diagnoses. That finding is consistent with the literature. In an earlier study by our group, 6 27% of the diagnoses were revised after review. In addition, Derks et al. 15 reported that only 148 out of 232 (63%) initial LCNEC diagnoses were confirmed after a centralized review. The most frequent alternative diagnosis retained by our panel was always SCLC, further substantiating the difficulties associated with differentiating between LCNEC and SCLC, especially based on small-sized tumor specimens. 16
Only 11% of the tumor specimens retained with a final LCNEC diagnosis had TCs expressing PD-L1. This observation is also consistent with recent publications of TC PD-L1 expression ranging from 9% to 25%.17–23 Those studies used diverse antibodies and different positivity thresholds. We used the monoclonal antibody 22C3 that is currently used extensively for routine IHC labeling and whose reproducibility has been validated for NSCLCs.10,24
In contrast, PD-L1 expression on ICs was very frequent, seen on 75% of the tumor samples. This finding was also reported by others examining PD-L1 co-expression on LCNEC TCs and ICs. PD-L1 expression was systematically found to be higher on ICs than TCs, respectively, by Kim et al. (34% and 15%), 17 Eichhorn et al. (36% and 22%) 19 and Kasajima et al. (44% and 9%); 18 the latter also used the 22C3 antibody. Test performances alone cannot explain the very high IC PD-L1 expression in our series compared with rates in the literature. 25 On the other hand, the cut-off levels differed from one study to another: semi-quantitative for Eichhorn et al. 19 and different thresholds for Kim et al. 17 and Kasajima et al. 18 Like others, 17 we found a non-significantly higher IC– level in metastatic LCNECs.
Notably, 66% of the LCNEC specimens had a TC–/IC+ PD-L1–expression pattern, different from that observed for NSCLCs, with ~60% TC+ samples with the 22C3 antibody, 26 or for SCLCs which had the same TC score but much fewer IC+ patients. 17
PFS was significantly longer for patients with IC+ LCNECs than those IC–, with a tendency towards non-significantly better OS, which was also found when IC–/1 and IC2/3 were compared. Similar findings were reported for a population comprised of LCNEC and SCLC patients 17 and surgically treated LCNEC patients.18,19 Even though the precise mechanisms involved in the relationship between IC+ infiltration of the tumor stroma and a better prognosis remains poorly understood, it has been shown that IC+ were predominantly macrophages, whose presence was significantly associated with CD8+ T-lymphocyte infiltration of the tumor, 18 attesting to a local immune reaction and, thus, tumor immunogenicity.
In contrast, patients with TC+ LCNECs had poorer prognoses than those TC– in our series, with a respective median OS of 4 versus 11 months. The impact of TC PD-L1 expression on the prognosis of LCNEC patients is an ongoing debate, associated with better OS in some studies20,22,23 but worse in others.19,27 TC PD-L1 expression has been associated with a poor outcome in various tumor models27–29 and NSCLCs (21). Unfortunately, the sizes and limited numbers of LCNECs tumor specimens examined herein do not enable us to conclude definitively.
However, the prognostic value of TC–IC co-expression seems to be more informative. As for Eichhorn et al.’s surgically treated series, patients with the TC+/IC– profile had more dismal prognoses that those TC–/IC+, with the difference being statistically significant herein for metastatic stages. In contrast, OS was comparable for patients with TC+/IC+ and TC–/IC– LCNECs. The poor prognosis for TC+/IC– patients needs to be validated in other tumor models, as it is not clearly explained by reported results. It might be a marker of an immune pattern carrying a poor prognosis or a tumor escape mechanism requiring the further characterization of immune subsets, notably CD8+ T lymphocytes and regulatory T cells.
Our results confirmed the poor prognosis for patients with metastatic LCNECs, with 4-month PFS and 7-month OS, in agreement with reported durations.6–8 Nivolumab-treated patients had a 50% tumor control rate, sometimes with prolonged durations. That control rate, obtained with third-line therapy is promising, in light of LCNEC aggressivity. Immunotherapy for LCNECs has not been extensively evaluated but several case reports and small series described efficacy, including against TC– tumors.27,30,31 LCNECs seem to have a higher frequency of elevated TMBs, a marker predictive of immunotherapy efficacy, and an elevated TMB was recently associated with higher IC PD-L1 expression for LCNEC patients. 17 Those findings led to undertaking prospective trials to evaluate immunotherapy safety and efficacy in LCNEC patients. 32
Our analysis has several limitations that support prudent interpretation of the results. The study was retrospective, with the investigator evaluating the ORR to treatment and PFS. Also, all of the patients were included, regardless of the quality of their tumor samples, and many specimens were small-sized. “Roche score” was published using SP142 PD-L1 clone, but for this analysis we apply the manufactural clone 22C3 from Dako as it was the best antibody for the Dako autostainer. Otherwise, the use of a centralized analysis limits in this analysis the heterogeneity of the interpretation. Finally, LCNEC is a rare disease and, despite this being a multicenter study, a relatively small number of patients could be included, thereby limiting the statistical power and, hence, significance of some findings. Nonetheless, the inclusion of all consecutive patients managed in participating centers and centralized review by a panel of thoracic disease experts reinforces the validity of the results obtained. Furthermore, while most of the series related to surgical specimens, this analysis included LCNEC patients diagnosed consecutively and unselected pathological sampless.
In conclusion, TC–/IC+ was the most frequent LCNEC PD-L1–expression pattern, a rather specific profile compared with those of NSCLCs and SCLCs. IC PD-L1 expression seems to have a prognostic impact. Prospective studies are now needed to confirm these findings.
Supplemental Material
EpnecFig1S_1 – Supplemental material for PD-L1-expression patterns in large-cell neuroendocrine carcinoma of the lung: potential implications for use of immunotherapy in these patients: the GFPC 03-2017 “EPNEC” study
Supplemental material, EpnecFig1S_1 for PD-L1-expression patterns in large-cell neuroendocrine carcinoma of the lung: potential implications for use of immunotherapy in these patients: the GFPC 03-2017 “EPNEC” study by Dominique Arpin, Marie-Christine Charpentier, Marie Bernardi, Isabelle Monnet, Aurelie Boni, Emmanuel Watkin, Isabelle Goubin-Versini, Régine Lamy, Laurence Gérinière, Margaux Geier, Fabien Forest, Radj Gervais, Anne Madrosyk, Florian Guisier, Cécile Serrand, Chrystèle Locher, Chantal Decroisette, Pierre Fournel, Jean-Bernard Auliac, Thierry Jeanfaivre, Jacques Letreut, Hélène Doubre, Geraldine Francois, Nicolas Piton, Christos Chouaïd and Diane Damotte in Therapeutic Advances in Medical Oncology
Footnotes
Authors’ note
This work was presented in part as posters at the 2018 ESMO and 2018 IASCL congresses.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Academic grant from Bristol-Myers Squibb, France.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
