Abstract
Immune checkpoint inhibitors targeting PD-1 and PD-L1 have demonstrated anti-tumor activity in several advanced solid malignancies. In previously treated metastatic castration resistant prostate cancer (mCRPC), a small subset of patients have a therapeutic response to checkpoint inhibition. Those who do respond to anti-PD-1/PD-L1 therapy have a marked, durable response to treatment, suggesting some derive long-term benefit from immune checkpoint blockade. In other cancers, one strategy to increase the efficacy of immune checkpoint blockade is to combine it with a pro-immune stimulatory agent, such as radiation. Here we present a case of a patient with heavily treated mCRPC who had a significant tumor response to concurrent pembrolizumab and radiation therapy to the primary prostatic mass. We review the growing evidence supporting the use of this combination therapy in other cancers and its potential benefit and safety in mCRPC. Our report highlights a potential therapeutic approach that should be further investigated in previously treated mCRPC.
Introduction
While there have been significant advances in the treatment of metastatic castration resistant prostate cancer (mCRPC), this advanced malignancy remains incurable despite successive lines of therapy. Immune checkpoint inhibitors (ICPi) targeting regulatory T-cell molecules such as cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), and programmed cell death protein ligand 1 (PD-L1) have emerged as paradigm-shifting therapies with durable responses observed across solid tumors. Recent studies have shown that a subset of pretreated mCRPC patients have a marked durable responses to anti-PD-1 therapy with a median duration of >15 month, suggesting that some mCRPC patients derive long-term benefit from these novel treatments.1–5 However, only a small minority of this population (4–20%) respond to anti-PD-1/PD-L1 therapy. Current work has focused on increasing the proportion of mCRPC patients who benefit from ICPi. One approach is to identify biomarkers that better predict responsiveness to PD-1/PD-L1 inhibition to better identify those patients who will benefit from these agents. A parallel approach is to identify safe, combinatorial treatments that synergize with ICPi to enhance its therapeutic effect and durability. In particular, combining ICPi with radiation has been previously shown to positively augment anti-PD-1 treatment in other cancers. However, its tolerability and efficacy in mCRPC remain underexplored.
Here, we present a patient with previously treated high-volume mCRPC with a large primary prostatic mass invading the rectum that dramatically responded to combination pembrolizumab, an anti-PD-1 therapy, and radiation therapy (RT). We discuss the potential therapeutic synergy and safety of radiation and pembrolizumab in our patient’s significant response and its role in mCRPC treatment. We also highlight the potential implications of genomic biomarkers in our patient’s clinical response.
Case presentation
A 75-year-old gentleman with a history of remote para-aortic radiation for testicular seminoma when he was 30 years old presented in April 2017 with obstructive uropathy and hematuria requiring chronic Foley catheter placement. Computer tomography (CT) demonstrated an exophytic prostatic mass and biopsy confirmed de novo metastatic Gleason 5+5 prostate adenocarcinoma. Prostate-specific membrane antigen (PSMA)-positron emission tomography (PET)/magnetic resonance imaging (MRI) revealed a prostatic tumor abutting the rectal wall with bladder lumen and bilateral seminal vesicles invasion. He had a right acetabular and bilateral pelvic sidewall nodal metastases. Prostate serum antigen (PSA) at diagnosis was 20.4 ng/ml.
He was initially treated with leuprolide, abiraterone, and prednisone from June to November 2017, but PSA rose and the primary tumor size increased within four months of treatment initiation, consistent with development of castration resistance (Figure 1). Systemic carboplatin and docetaxel was trialed for 2–3 months with minimal PSA response and interval imaging in January 2018 revealed worsening disease (Figures 1 and 2(a) and (b)). The patient also had recurrent hospital presentations for obstructive uropathy and symptomatic anemia from hematuria. In February 2018, he was switched to systemic carboplatin and cabazitaxel. During cycles 1 and 2 of carboplatin/cabazitaxel, the patient received stereotactic body radiotherapy (SBRT) of 3800 cGy to the primary tumor over four treatments to provide symptomatic relief from uropathy and hematuria. Chemoradiation decreased primary tumor size and PSA by >90% after four cycles (Figure 2(c) and (d)). He continued with carboplatin/cabazitaxel for 10 total cycles.

Prostate serum antigen (PSA) and corresponding prostatic tumor size across patient’s treatment course. Measured PSA (boxes) with trend line (solid black line) across the patient’s treatment course with corresponding prostate tumor size by magnetic resonance imaging at the time of treatment switch or at PSA nadir. Above the graph displays the systemic treatments and specific radiation treatment the patient received. The duration of each systemic therapy is the time interval between dotted grey lines. Note, the patient has continued to receive leuprolide every 3 months since June 2017.
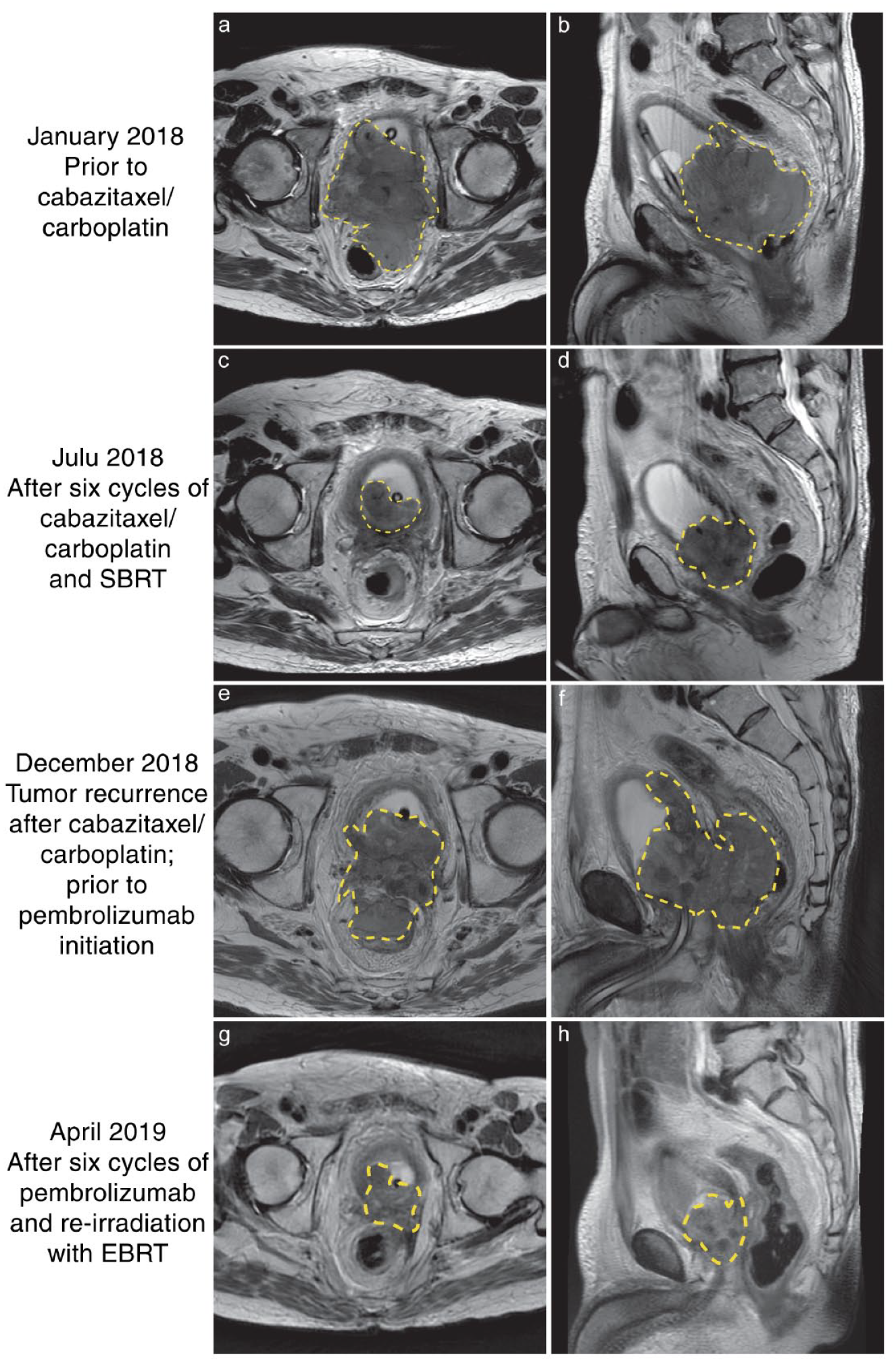
Serial magnetic resonance imaging pelvis imaging of the patient’s tumor after sequential treatments.
By September 2018, the patient’s PSA began to rise. Restaging MRI pelvis in December 2018 revealed an enlarging primary tumor size, worsening seminal vesicle involvement, rectal invasion, and a right pubic bone lesion without other measurable soft tissue lesions by Response Evaluation Criteria in Solid Tumors (RECIST) criteria (Figures 1 and 2(e) and (f)). At that time, he was started on compassionate-use pembrolizumab. Given the rectal invasion, the patient received palliative radiation to the primary tumor and pelvic lymph nodes with conventionally fractionated external beam RT in January 2019 during pembrolizumab cycles 2 and 3. RT was initially prescribed to 4500 cGy for 25 fractions with a simultaneous integrated boost to 5000 cGy to the gross primary tumor volume. Analysis of serum circulating tumor DNA of 70 cancer-associated genes via Clinical Laboratory Improvement Amendments (CLIA)-approved assay through FoundationOne 6 was sent prior to pembrolizumab initiation. Results obtained during pembrolizumab cycle 2 demonstrated high levels of microsatellite instability (MSI-H) and multiple pathogenic mutations in AR, ATM, BRCA1, BRCA2, CDK12, PTEN, and TP53, with mean allelic frequencies ranging 0.23–7.2%. Remarkably, the patient demonstrated an exceptional treatment response after two cycles of pembrolizumab and 1920 cGy of RT with visible reduction of rectal involvement.
At the time of pembrolizumab initiation, the patient was undergoing evaluation for unilateral left lower extremity weakness and neuropathy. MRI neurogram obtained during cycle 1 of pembrolizumab revealed asymmetric left sciatic nerve and lumbar plexus enhancement and thickening consistent with treatment-related inflammation. Given the patient’s dramatic treatment response after two cycles and to minimize the risk of treatment-related toxicity, RT was held after 1920 cGy and the radiation field was adjusted to target only the gross residual tumor volume for an additional 2400 cGy over eight fractions followed by stereotaxic cone down boost to 800 cGy over two fractions.
In April 2019, after RT and six cycles of pembrolizumab, the patient’s PSA was undetectable and imaging revealed a decreased prostatic mass with no radiographic evidence of rectal wall involvement (Figure 2(g) and (h)). His right pubic bone lesion remained stable on post-treatment imaging. As of October 2019, the patient continues on pembrolizumab monotherapy. His neuropathy and leg weakness have remained stable. He underwent a trial of void and remains catheter free. He denies symptoms of bladder and colorectal toxicity related to RT.
Discussion and implications
We report a patient with heavily treated mCRPC with a large primary prostatic mass who had a striking response to concurrent pembrolizumab and focal radiation. ICPi has vastly improved outcomes in melanoma, non-small-cell lung cancer (NSCLC), and head and neck cancers, but its activity in mCRPC is modest. Combination RT and anti-PD-1 therapy is actively being explored as an approach to enhance the response rate of ICPi in other solid tumors, but studies investigating combination therapy in mCRPC treatment remain few. While the exact mechanism underlying our patient’s dramatic response remains uncertain, our case highlights the possible benefit and safety of combination RT/anti-PD-1 in treated mCPRC and suggests that dedicated investigation is needed to determine whether combination therapy is effective in this patient population.
To our knowledge, there are no reported studies in mCRPC patients directly comparing anti-PD-1 therapy with or without radiotherapy and we are among the first to highlight the potential benefit of this combination in mCRPC. Radiotherapy stimulates tumor associated antigen release, T-cell activation, and an inflammatory microenvironment, resulting in a systemic anti-tumor host immune response. A prevailing hypothesis is that these pro-stimulatory effects synergize with ICPi to enhance its systemic efficacy. 7 These systemic effects also occur outside the localized radiation treatment field—prior studies have noted that tumors outside the treatment field decreased in size, a phenomenon known as the “abscopal effect.” In preclinical models of other solid tumors, ICPi and RT enhanced tumor regression with signs of the abscopal effect and improved survival compared with each modality individually.8–12 Early phase clinical studies and case reports have supported this synergy.8,13–18 Maity et al. report that two out of twelve patients with NSCLC or melanoma who developed disease progression on anti-PD-1 monotherapy had a partial response in the unirradiated lesion when focal radiation was added to continued anti-PD-1 inhibition, supporting the hypothesis that radiation may reinvigorate immunotherapy. 14 A recent phase II study examining SBRT with or without pembrolizumab in NSCLC found that combination therapy increased the objective response rate (ORR) compared with RT alone, although this increased ORR was not statistically significant nor was it associated with an increase in overall or progression free survival. 19 Collectively, these studies suggest that radiotherapy enhances ICPi efficacy and may increase the number of patients who respond to immunotherapy. However, more studies are needed to evaluate whether this response translates to improved overall or progression free survival.
In contrast, studies examining combination anti-PD-1/RT in mCRPC have been limited. One preclinical study demonstrated that combination therapy in a CRPC subcutaneous tumor graft mouse model enhanced tumor shrinkage and increased survival compared with single agent anti-PD-1 or PD-L1 therapy. 20 A retrospective case series found that 7/10 patients treated with pembrolizumab and SBRT had either a response or stable disease, although it is unclear whether this significant response is superior to single agent pembrolizumab alone. 21 There are ongoing prospective trials in mCRPC investigating anti-PD-1/PD-L1 therapy combined with different forms of radiation therapies, including SBRT (ClinicalTrials.gov identifier: NCT03477864), brachytherapy (ClinicalTrials.gov identifier: NCT03543189), alpha-radiation from Radium-223 preferentially absorbed by the bone (ClinicalTrials.gov identifiers: NCT04071236, NCT03093428, NCT04109729, NCT03795207) and the targeted radio-ligand to the PSMA lutetium-177-PSMA-617 (ClinicalTrials.gov identifier: NCT03805594).
mCRPC studies combining RT with another class of ICPi, the anti-CTLA-4 targeted therapies, have been mixed. A phase I/II dose-escalation trial examining ipilimumab with or without RT found that 4/34 patients treated with combination ICPi/RT had a >50% PSA decline and one had a partial response by RECIST and was well tolerated. 22 However, the randomized phase III study CA184-043 testing radiotherapy with or without ipilimumab in docetaxel-treated patients failed to show improved overall survival in an unselected mCRPC population, although did show improved progression free survival. 23 While these results may cast doubt on the potential efficacy of PD-1 immune checkpoint inhibitors, it is important to underscore that CTLA-4 and PD-1/PD-L1 pathways impact T-cell activity and function through distinct, non-redundant mechanisms. Clinically, melanoma patients had different response rates to these two classes of ICPi. 24 As such, these mixed results from earlier anti-CTLA-4 trials should not hamper exploration of combination anti-PD-1 therapy/RT in mCRPC.
The safety profile of combination anti-PD-1/RT is an important consideration. Prior studies found that the addition of RT to anti-PD-1 therapy was not associated with an increased rate of adverse toxicities compared with anti PD-1 monotherapy.25–27 Similarly, the addition of RT to the anti-CTLA-4 inhibitor ipilimumab in melanoma and mCRPC patients did not increase the number of patients with Grade 3/4 adverse events compared with ipilimumab monotherapy, suggesting that radiation can be safely added to ipilimumab without significant increase in toxicity.22,28 It is also important to note that our patient’s neurologic symptoms could be sequelae from his treatment. Radiation-induced lumbosacral plexopathies are rare, but can occur months to years after RT and present with motor predominant symptoms. 29 Peripheral neuropathy and plexopathies are uncommon side effects of anti-PD-1 therapies, having only been described in case reports.30,31 While our patient’s symptoms preceded pembrolizumab and remained stable after treatment initiation, we cannot exclude the possibility that his symptoms and abnormal imaging were exacerbated by pembrolizumab administration or concurrent pembrolizumab/RT.
Another consideration is the optimal dose and fractionation of RT used in combination with ICPi. During re-irradiation with combination pembrolizumab, our patient received two different radiation regimens. Preclinical studies have found varying effects on anti-tumor activity and immune system activation depending on the radiation dose (high versus low dose) and number of fractions.32–34 In clinical studies, both conventionally fractionated and hypo-fractionated regimens in combination with immunotherapy were safe and well tolerated.14,35,36 However, additional studies are needed to determine whether dosing and fractionation impacts outcomes. Interestingly, one retrospective single institution study found improved overall survival in patients who received hypo-fractionated radiotherapy. 37
We cannot exclude the possibility that our patient would have responded to repeat radiation or pembrolizumab monotherapy. Genomic analysis of our patient’s tumor revealed MSI-H and pathogenic mutations in BRCA1, BRCA2, ATM, and CDK12, genes associated with DNA damage response (DDR) and homologous recombination (HR). Although pembrolizumab is approved for solid tumors that harbor MSI-H or have deficient DNA mismatch repair (dMMR) agnostic of tumor location only one prostate cancer patient was enrolled in the two studies leading to Food and Drug Administration approval.38,39 Growing evidence from case reports and a recent retrospective cohort study supports the use of anti-PD-1/PD-L1 therapy in MSI-H mCRPC and several case series suggest that DDR mutations and HR deficiency predict ICPi responsiveness.1,5,40–43 Importantly, only half of dMMR/MSI-H mCRPC patients in the retrospective cohort study responded to anti-PD-1/PD-L1 therapy, suggesting that MSI-H is not sufficient to predict anti-PD-1/PD-L1 treatment response. 41 One of the five mCPRC responders in the retrospective cohort initially did not respond to pembrolizumab monotherapy, having a significant PSA rise 10 weeks after treatment initiation. He subsequently received focal prostate irradiation with pembrolizumab, resulting in an 81% PSA decline that remained stable 30–40 weeks post-iraddiation. 41 Similar to the findings from Maity et al., this second case raises the possibility that, RT can re-invigorate an initial response to anti-PD-1 therapy and enhances durability. Together, these two cases in mCRPC patients, ours using concurrent ICPi/RT and the case described in Abida et al. using an additive approach, argue for further exploration of combination ICPi and radiation in pretreated mCRPC.
In sum, our case report provides evidence supporting the hypothesis that combination radiation and anti-PD-1 therapy is safe and potentially efficacious in heavily treated mCRPC. Several accruing studies combining various forms of RT with anti-PD-1 therapies are underway, which will likely provide significant insight into the safety and efficacy of this combination. Nevertheless, dedicated randomized studies directly comparing single agent ICPi versus combination therapy are needed to best answer whether the addition of radiation to anti PD-1 therapy is both safe and beneficial to patients. In addition, further studies are needed to better define the optimal timing, dose, and fractions of radiation that would best synergize with ICPi. Regardless of the outcome, investigating RT in combination with anti-PD-1 therapy will likely uncover novel pathways or targets that provide better insight into biomarkers for anti-PD-1 responsiveness and how to best combine anti-PD-1 therapy with other pro-immunogenic agents.
Footnotes
Acknowledgements
We thank the patient for allowing us to share his medical history and clinical course. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The NIH had no involvement in the data collection and writing of this report.
Conflict of interest statement
RA receives research support from Merck for a research study unrelated to this case. This research support was received after initiating treatment for the patient in this case and did not influence the clinical decision-making for this patient. The other authors declare no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: YRL is a recipient of a NIH F32 individual postdoctoral fellowship [5F32CA232635].
Informed consent
This patient has provided written, informed consent for his medical history, treatment history, objective laboratory data, and images to be published in your journal, social media, news release, and/or website. The patient is aware that his medical information will be published without his name attached and understands that complete anonymity cannot be guaranteed. He also is aware that his right to revoke consent can occur at any time prior to publication, but once it has been committed to publication, it will not be possible to revoke consent. A copy of the signed consent form is available on request.
