Abstract
Background:
Aromatase inhibitors (AIs) influence blood lipid profiles. However, relatively few studies have directly compared the treatment effects of steroidal and nonsteroidal AIs.
Methods:
A prospective single-center cohort study was conducted to investigate the effects of steroidal and nonsteroidal AIs on lipid profiles during the first 24 months of endocrine therapy in hormone receptor-positive postmenopausal patients with early breast cancer. The primary endpoint was the cumulative incidence of lipid events, while the secondary endpoints were changes in lipid profiles and lipid event-free survival.
Results:
Comparison of the lipid profiles of the two groups showed that triglycerides (TGs) and total cholesterol (TC) levels were significantly higher in the nonsteroidal AI group over 24 months (p < 0.05), whereas low-density lipoprotein cholesterol (LDL-C) was significantly higher only at 3 months (p = 0.017) and 6 months (p = 0.026). High-density lipoprotein cholesterol (HDL-C) was significantly lower in the steroidal group at all time points (p < 0.05), except at 18 months (p = 0.085). The cumulative incidence of lipid events in the steroidal and nonsteroidal groups at 24 months was 25.3% and 37.0%, respectively. Multivariate analysis results indicated that TG, LDL-C, and steroidal AIs were independently associated with blood lipid events.
Conclusion:
This trial showed that a significantly higher cumulative incidence of lipid events occurred in the nonsteroidal AI group than in the steroidal AI group, which indicated that steroidal AIs exerted a protective effect against blood lipid events in postmenopausal women receiving an AI as adjuvant therapy for breast cancer.
ClinicalTrials.gov identifier:
NCT02765373
Keywords
Introduction
Aromatase inhibitors (AIs) suppress aromatase activity, thereby reducing estrogen concentrations and leading to improved survival in patients with hormone receptor-positive breast cancer. However, this improved survival concomitantly increases the relative risks of age-associated diseases, including cardiovascular events, in patients with early breast cancer. 1 Altered lipid profiles are considered as the likely means through which the relative risks for cardiovascular events increase in these patients. 2
Third-generation AIs such as exemestane, letrozole, and anastrozole have proven to be more active than tamoxifen for postmenopausal women diagnosed with hormone receptor-positive breast cancer. 3 Consequently, AIs are frequently used as adjuvant therapy in patients with hormone receptor-positive breast cancer as per National Comprehensive Cancer Network (NCCN) and European Society for Medical Oncology (ESMO) guidelines. However, long-term estrogen deprivation by AI treatment also affects the physiological functions of estrogen and leads to changes in lipid profiles as well as bone loss. 4
Several studies involving patients with early breast cancer have recognized that cardiovascular disease, related to altered lipid profiles, is the leading cause of death among elderly survivors.5,6 Studies estimating the effect of AIs in early breast cancer patients reported contradictory results in terms of net changes in the composition of specific lipid fractions. Furthermore, diverse endpoints and time points were used for analysis in these studies, further confounding the results.7–10
Although both steroidal and nonsteroidal AIs substantially decrease whole-body aromatization and inhibit the synthesis of estrogen in postmenopausal women, their chemical structure and mechanism of action differ which may lead to differential effects on blood lipids. 11 Nonsteroidal AIs such as letrozole (C17H11N5) and anastrozole (C17H19N5) interact noncovalently and reversibly with the heme moiety of aromatase. In contrast, the steroidal AI, exemestane (C20H24O2), forms an irreversible covalent bond with aromatase. The active site of aromatase recognizes exemestane as an alternative substrate as it is an analog of the natural aromatase substrate androstenedione. 12 However, to date, data comparing the effects of the two types of AIs on lipid profiles are scarce. The present prospective study was designed to compare changes in the lipid profiles of Chinese postmenopausal patients with hormone receptor-positive breast cancer receiving steroidal or nonsteroidal AIs as adjuvant endocrine therapy (ET). The results may provide evidence to aid in selecting the optimal strategy for ET.
Methods
Study design and patients
A prospective, single-center, cohort study was conducted at the Cancer Hospital of the Chinese Academy of Medical Sciences.
Participants were eligible if they were postmenopausal women, diagnosed with hormone receptor-positive, ductal carcinoma in situ (DCIS; stage 0) or stages I to III invasive breast cancer, for whom AI therapy was considered for initial adjuvant ET. Patients were included irrespective of having received previous postoperative adjuvant chemotherapy and/or radiotherapy. Steroidal or nonsteroidal AIs were prescribed based on the patient’s preference and the physician’s clinical judgment. The patients in the steroidal AI group received exemestane at a dose of 25 mg orally once daily for 5 years, while those in the nonsteroidal AI group received letrozole at a dose of 2.5 mg or anastrozole at a dose of 1 mg orally once daily for 5 years.
Patients diagnosed with low-density lipoprotein cholesterol (LDL-C) levels ⩾4.14 mmol/L at baseline, those with a previous history of ET, those prescribed lipid-lowering medications, and those with severe cardiovascular/cerebrovascular disease or other malignant tumor diagnosed within 6 months were excluded from the study.
Endpoints
The primary endpoint of the study was the cumulative incidence of lipid events and factors associated with lipid events during 24 months of follow-up. The lipid events included an LDL-C level ⩾4.14 mmol/L, which was determined according to the 2007 Chinese guidelines on the prevention and treatment of dyslipidemia in adults, 13 or initiation of lipid-lowering medication. The secondary endpoints were the changes in lipid profiles during 2 years of treatment and lipid event-free survival (LEFS), which was defined as the time from receiving adjuvant ET to occurrence of lipid events.
Sample collection, assessment time points, and measurement of lipid profiles
Physical examination and collection of laboratory samples were scheduled at baseline and 3, 6, 9, 12, 18, and 24 months after the initiation of ET. Fasting (at least 12 h) blood samples were collected for the estimation of serum lipid parameters, including total cholesterol (TC), LDL-C, high-density lipoprotein cholesterol (HDL-C), and triglyceride (TG) concentrations, in clinical laboratories at the Cancer Hospital of the Chinese Academy of Medical Sciences.
Statistical analysis
The full analysis set consisted of patients with data from a minimum of two assessment time points (patients with no sequential time point assessment were also included in the analysis) who complied with the study protocol and were free of disease recurrence or metastasis. In the case of patient withdrawal, data collected prior to withdrawal were used for the analysis. Data for continuous variables were expressed as means ± standard deviation (SD) or medians (minimum, maximum), as appropriate, based on the Kolmogorov–Smirnov test. Normally distributed variables were analyzed using Student’s t-test (intergroup comparisons) or paired t-test (intragroup comparisons over treatment), while the Mann–Whitney U test was used for non-normally distributed variables. Categorical variables were presented as frequencies (percentage) and were analyzed using the chi-square test or Fisher’s exact test if >20% of the cells had an expected frequency <5. Time-to-event analysis was performed using the Kaplan–Meier method and the log-rank test. Cox analysis was used for univariate and multivariate regression analyses of the primary outcome. SPSS 22.0 (IBM, Armonk, NY, USA) and GraphPad Prism 6 (GraphPad Software Inc., San Diego, CA, USA) were used for analysis. All statistical tests were two-tailed, with p-values < 0.05 suggesting statistical significance.
Highlights
What is the current knowledge of the topic?
AIs are used as adjuvant therapy in hormone receptor-positive postmenopausal patients with early breast cancer.
To date, data comparing the effects of the two types of AIs on lipid profiles are scarce.
What question did this study address?
Head-to-head comparison of the effects of steroidal and nonsteroidal AIs on lipid profiles.
What does this study add to our knowledge?
A higher incidence of cumulative lipid events was observed with nonsteroidal AIs.
Patients on steroidal AIs showed favorable changes in lipid profiles.
How might this change clinical pharmacology or translational science?
The study will help physicians in developing treatment strategies for adjuvant therapy.
Results
Study population
A total of 647 patients who had undergone surgery received postoperative ET with AIs from November 2014 to December 2016 at the Cancer Hospital of the Chinese Academy of Medical Sciences. The demographic characteristics of the patients are presented in Table 1. Overall, 415 patients were included for lipid profile analysis. Of these, 192 were treated with steroidal AIs (exemestane) and 223 with nonsteroidal AIs (anastrozole or letrozole) (Figure 1).
Characteristics of the patients.

Continuous data are presented as median (range).
BMI, body mass index; ER, estrogen receptor; HDL-C, high-density lipoprotein cholesterol; HER2, human erbB-2 receptor; LDL-C, low-density lipoprotein cholesterol; PR, progesterone receptor; TC, total cholesterol; TG, triglyceride.

Cohort flowchart.
Primary endpoints
Lipid events
The cumulative incidence of serum lipid events at 24 months in the steroidal and nonsteroidal groups was 25.3% and 37.0%, respectively. Time-to-lipid event analysis revealed that steroidal AIs were associated with a 36% lower incidence of lipid events when compared with non-steroidal AIs, with a hazard ratio (HR) of 0.64 [95% confidence interval (CI), 0.44–0.93; p = 0.018; Figure 2].
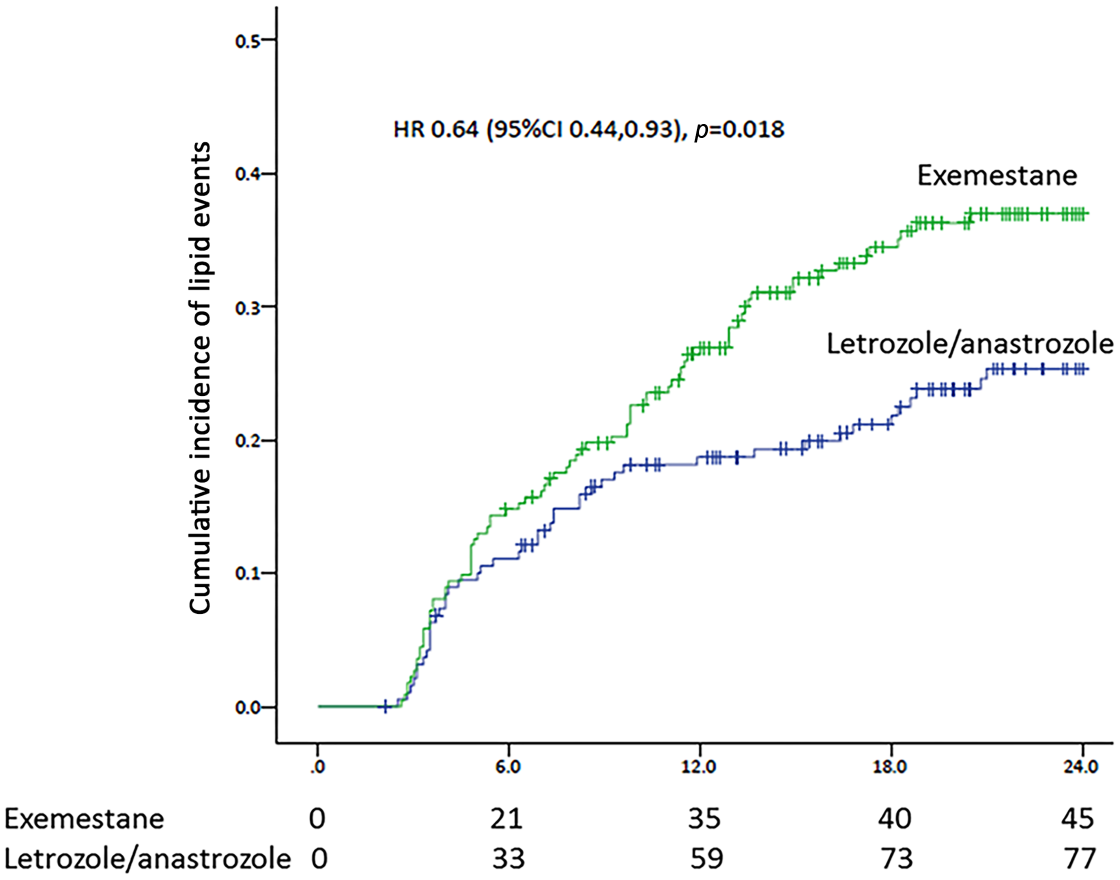
Kaplan–Meier curve for cumulative lipid events in patients with breast cancer treated with exemestane or letrozole/anastrozole.
Factors associated with lipid events
The factors associated with the occurrence of lipid events were analyzed by univariate and multivariate analyses. In univariate analysis (p < 0.05), TGs, TC, LDL-C, and steroidal AIs were associated with blood lipid events, whereas in multivariate analysis TGs (HR = 1.26, 95% CI = 1.05–1.51; p = 0.013), LDL-C (HR = 2.73, 95% CI = 1.51–4.98; p = 0.001), and steroidal AIs (HR = 0.602, 95% CI = 0.42–0.87; p = 0.008) were independently associated with blood lipid events (Table 2).
Multivariate analysis of factors associated with LEFS.
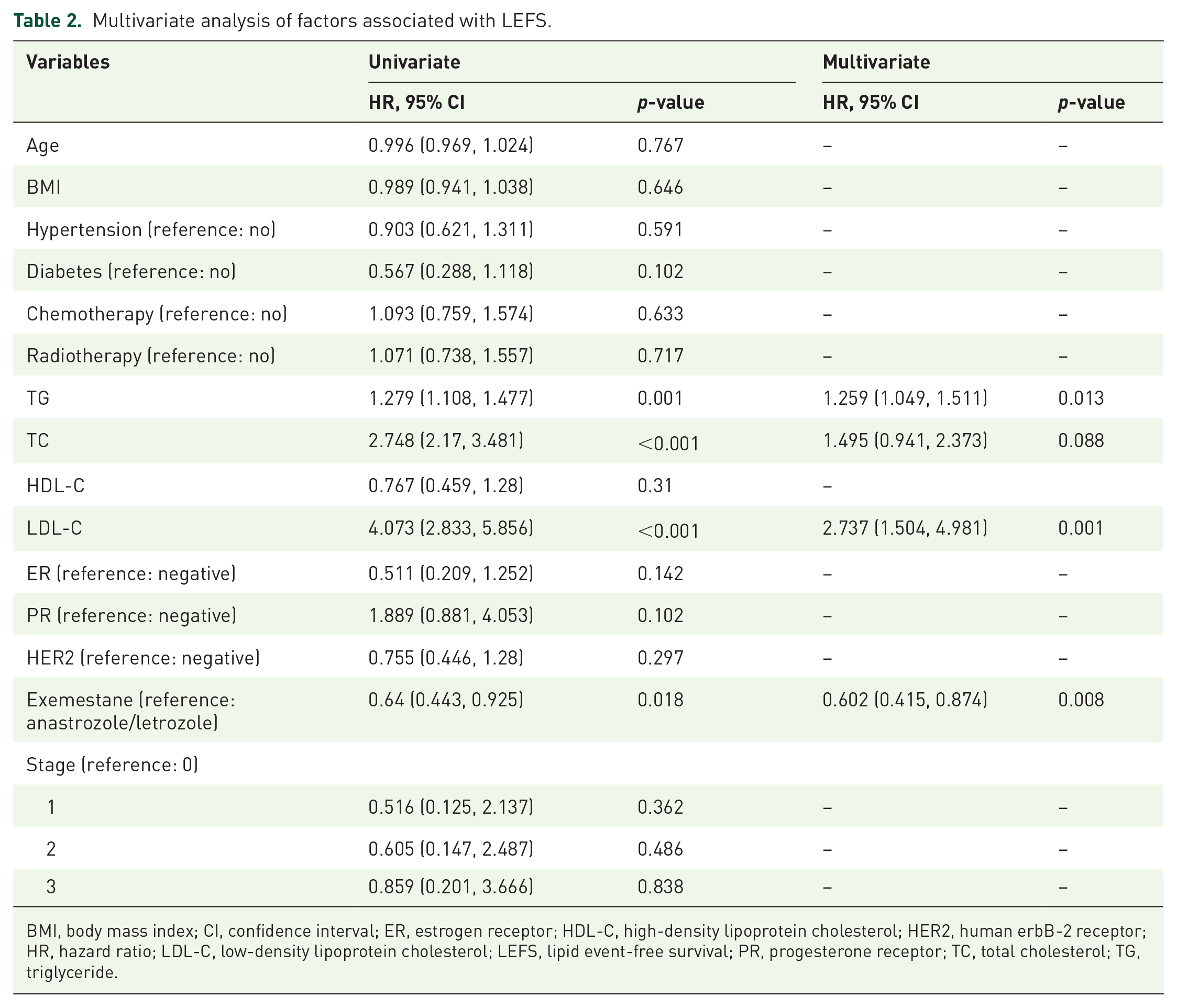
BMI, body mass index; CI, confidence interval; ER, estrogen receptor; HDL-C, high-density lipoprotein cholesterol; HER2, human erbB-2 receptor; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; LEFS, lipid event-free survival; PR, progesterone receptor; TC, total cholesterol; TG, triglyceride.
Secondary endpoints
Changes in lipid profiles
The median values of lipid parameters at each time point are presented in Figure 3. Comparison of the lipid profiles of the two groups showed that TGs and TC levels were significantly higher in the nonsteroidal AI group over 24 months (p < 0.05), whereas LDL-C was significantly higher only at 3 months (p = 0.017) and 6 months (p = 0.026). HDL-C was significantly lower in the steroidal AI group at all time points (p < 0.05), except at 18 months (p = 0.085). Figure 4 depicts the changes in lipid profiles from baseline over the study period and the distribution of lipid values are provided in Figure 5.

(a) Triglyceride (TG), (b) total cholesterol (TC), (c) high-density lipoprotein cholesterol (HDL-C), and (d) low-density lipoprotein cholesterol (LDL-C) levels at different time points over the course of exemestane or anastrozole/letrozole treatment in women with breast cancer. Data are presented as median and range.
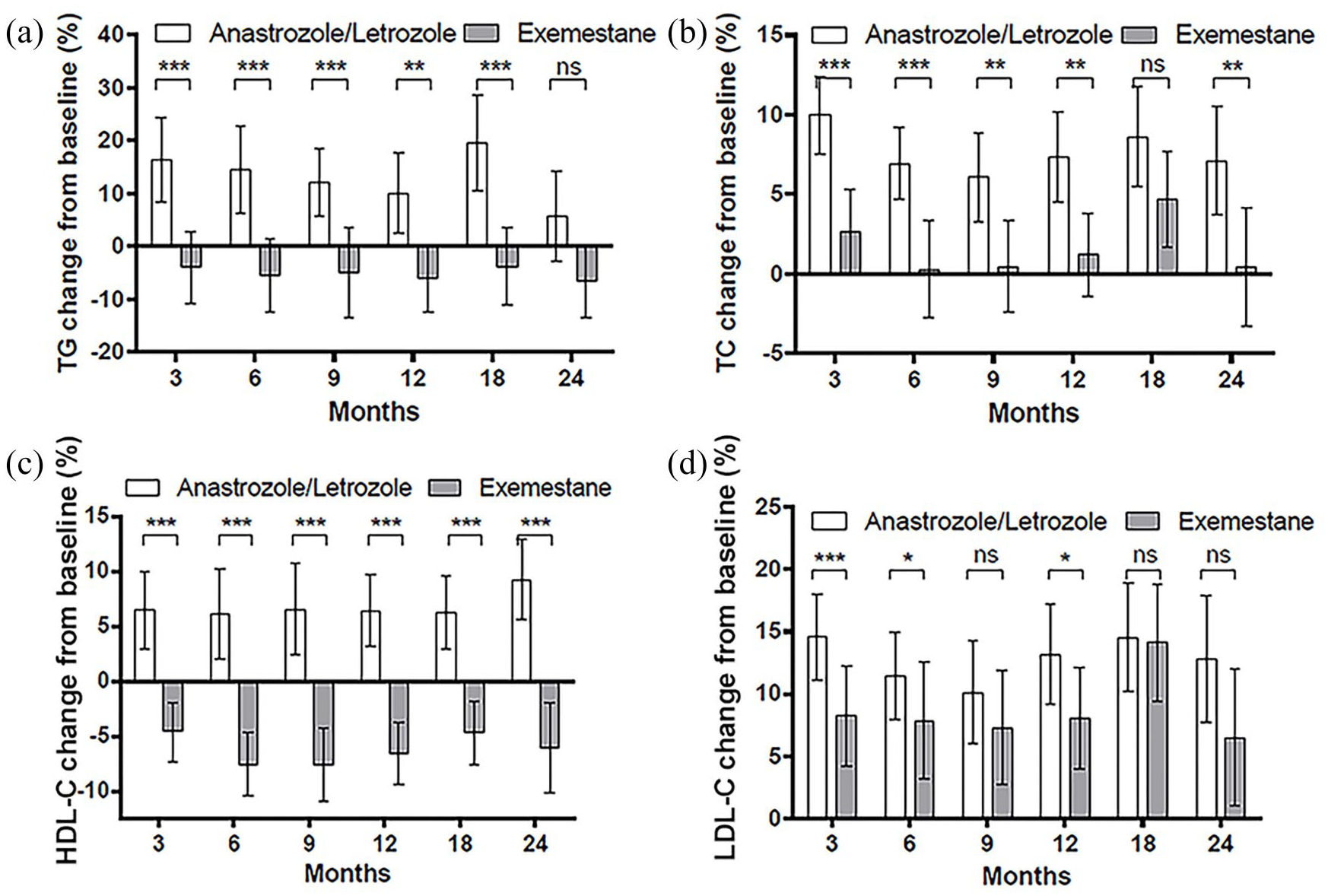
Changes from baseline (%) of (a) triglyceride (TG), (b) total cholesterol (TC), (c) high-density lipoprotein cholesterol (HDL-C), and (d) low-density lipoprotein cholesterol (LDL-C) levels at different time points.

Distribution of lipid values. (a) triglyceride (TG), (b) total cholesterol (TC), (c) high-density lipoprotein cholesterol (HDL-C), and (d) low-density lipoprotein cholesterol (LDL-C) levels at different time points.
Lipid event-free survival
The median LEFS could not be estimated for either group.
Discussion
In the present study, we focused on changes in lipid profiles, which are considered to be surrogate markers for the risk of cardiovascular events in patients with early breast cancer. Steroidal AI treatment elicited better outcomes in terms of a lower incidence of increases in all the lipid parameters, including HDL-C levels. In multivariate analysis, steroidal AIs exerted a protective effect against blood lipid events, which is in accordance with a previous study. 2 Earlier studies on the effects of AIs on lipid profiles have reported contradictory and ambiguous results. Reduced TC and LDL-C levels were reported for exemestane treatment, whereas higher doses of anastrozole and letrozole led to increases in TC and LDL-C levels.14–17
Trials with exemestane and letrozole have previously compared their lipid-altering effects with placebo. The results of the MAP.2 trial and the MA.17 subgroup study reported no difference in blood lipid levels with exemestane and letrozole treatment when compared with placebo.18,19 Similarly, Hozumi et al showed that exemestane and anastrozole treatment exerted no clinically significant effect on serum lipid levels. 20 In the MA.27 study, the incidence of hypertriglyceridemia and hypercholesterolemia in the anastrozole treatment group was higher than in the exemestane group. 21 This disparity was likely due to ethnicity-specific differences in AI metabolism.22,23
A study by Ma et al on 60 Han Chinese American, African American, Caucasian, and Mexican American patients found that Thr364, Cys264, and double-variant Arg39Cys264 aromatase allozymes showed significantly decreased activity when compared with the wild-type enzyme. The Arg39Cys264 allozyme also exhibited a significantly increased inhibitor constant for letrozole. Compared with Caucasian Americans (2.5%) and Mexican Americans (5%), Cys264 was found at a higher frequency in Han Chinese Americans (11.7%) and African Americans (22.5%). In Han Chinese Americans, the Arg39 variant was present at a frequency of 6.7% but was rare in other ethnic groups. The results of this study suggest that patients with decreased aromatase activity may not receive the full benefit of AI treatment. 24
The lipid event we defined in this trial could, to a certain extent, predict the risk of cardiovascular events such as coronary heart disease. The differences in the cumulative incidence of lipid events between the two types of AI may have been due to their chemical structures and mechanisms of action. AIs reduce estrogen levels by suppressing the cytochrome P450-dependent enzyme, aromatase, which is responsible for the conversion of androgens to estrogens. In the case of exemestane, 17-hydroxy exemestane, a metabolite of exemestane, also possesses AI activity, which may contribute to its enhanced efficacy. This is a likely reason for the observed decrease in HDL-C levels in patients treated with exemestane. 11 The precise effect of this decrease in HDL-C concentrations will be difficult to ascertain as previous Mendelian randomization studies have reported an increased incidence of hormone receptor-positive breast cancer in patients with a genetic predisposition for increased levels of HDL-C. 25
In the current study, a substantial decrease in TG levels was observed in patients treated with steroidal AIs. TGs serve as an important source of oxidizable energy, which drives cellular and tumor growth. 26 Therefore, the TG lowering effect of steroidal AIs may indirectly contribute to tumor suppression. Other studies have reported similar results. 2
Heterogeneity in genes involved in estrogen signaling has been suggested to affect the lipid profiles of women undergoing AI treatment. 27 For example, in postmenopausal women on adjuvant AI therapy, specific polymorphisms in the estrogen receptor alpha (ESR1) gene were shown to be associated with increased LDL-C and TG concentrations, 28 while changes in CYP19A1 were associated with a decrease in TG levels and changes in HDL-C concentrations. 29 Environmental causes, such as an unhealthy lifestyle, can also lead to elevated levels of blood lipids. As it was difficult to follow up on the eating habits of patients, the lifestyle such as eating habits was not considered in the final statistical analysis. In the multivariate analysis, body mass index, hypertension, and diabetes, which are commonly considered as potential risk factors for changes in blood lipid levels, had no effect on lipid events in our study.
Several studies have suggested that changes in lipid profiles induced by letrozole are reversible and manageable with drug-lowering therapy, while others have indicated that these changes are resistant to lipid-lowering medication.30–32 A recent study on hormone receptor-positive early stage breast cancer patients on adjuvant ET reported that cholesterol-lowering medications may have a role in preventing breast cancer recurrence. 33 In the present study, patients taking a lipid-lowering medication at baseline were excluded. These factors should be taken into consideration in future studies.
The current study differed from earlier studies in terms of its strengths. To the best of our knowledge, this is the largest head-to-head prospective study comparing the effects of steroidal and nonsteroidal AI treatment as initial adjuvant therapy on lipid profiles in Asians. It was also the first study using cumulative incidence of lipid events as the primary endpoint, and the changes in lipid levels as the secondary endpoints, which provides a comprehensive overview of changes in lipid profile.
However, our study had several limitations. This was a prospective observational study and the patients were not randomized to receive the study drugs. Furthermore, all the patients were Chinese and from a single center, limiting the applicability of the results to patients worldwide. In addition, because of the relatively short follow-up period, the median LEFS in the two groups could not be estimated and the effect of the AIs on major cardiovascular outcomes was not evaluated. Additional studies are necessary to address these limitations and refine the results, as well as to verify whether lipid-lowering medications improve the prognosis of early breast cancer patients.
Conclusion
In conclusion, patients with early breast cancer treated with steroidal AIs presented with favorable changes in lipid profiles when compared with those treated with nonsteroidal AIs. Because a large number of women with chronic adjuvant ET are vulnerable to cardiovascular disease, physicians are encouraged to develop treatment strategies after evaluating the risk factors for disorders of lipid metabolism. Both physicians and patients should give more consideration to the results of laboratory tests, lifestyle counseling, and appropriate drug therapy options when using AIs.
Footnotes
Acknowledgements
We wish to thank all the patients who took part in this research, their families, and the investigators. A special thanks to Jing Liu for trial monitoring and data management and Pfizer Oncology Medical Affairs for their support.
Author’s contribution
PY designed the trial. XW collected the data. XW, AZ, JW, FM, JL, YF, YL, PZ, QL, BX, and PY analyzed the data. All the authors vouch for the accuracy and completeness of the analyses and data reported and also confirm adherence to the protocol. XW wrote the initial manuscript. All the authors contributed to subsequent drafts and gave their final approval for submission of the manuscript for publication.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The trial was conducted in accordance with the principles of Good Clinical Practice as specified in the Declaration of Helsinki. The study protocol was approved by the Institutional Ethics Committee (ID: CH-BC-030), and all the participants provided written informed consent. It will be made clear that patients may withdraw from the study at any time without providing a reason and without affecting their current or future care. Physicians in our team will be kept informed on the study’s progress.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
