Abstract
Pancreatic cancer is a lethal disease characterized by highly dense stroma fibrosis. Only 15–20% of patients with pancreatic cancer have resectable tumors, and only around 20% of them survive to 5 years. Traditional cancer treatments have little effect on their prognosis, and successful surgical resection combined with effective perioperative therapy is the main method for maximizing long-term survival. For this reason, chemotherapy is an adjunct treatment for resectable cancer and is the main therapy for incurable pancreatic cancer, including metastatic pancreatic adenocarcinoma. However, there are various side effects of chemotherapeutic medicine and low drug penetration because the complex tumor microenvironment limits the application of chemotherapy. As a novel strategy, polymer nanoparticles make it possible to target the tumor microenvironment, release cytotoxic agents through various responsive reactions, and thus overcome the treatment barrier. As drug carriers, polymer nanoparticles show marked advantages, such as increased drug delivery and efficiency, controlled drug release, decreased side effects, prolonged half-life, and evasion of immunogenic blockade. In this review, we discuss the factors that cause chemotherapy obstacles in pancreatic cancer, and introduce the application of polymer nanoparticles to treat pancreatic cancer.
Introduction
Pancreatic cancer is the fourth leading cause of cancer death in the United States and can lead to an estimated of 22,7000 deaths per year worldwide. 1 Pancreatic cancer is an aggressive disease, with a 5-year survival rate of less than 5%, which is associated with late presentation.1,2 This disease is more common in older people than in younger people. 2 Although the cause of pancreatic cancer is complex and changeable, smoking and family history are concerns. 2 Unfortunately, less than 20% of patients present with localized, potentially curable tumors; thus, most of the patients who present with symptoms attributable to pancreatic cancer are already in the advanced stages of the disease. For patients with resectable disease, surgery is still the main method of treatment. 3 However, adjuvant treatment is recommended for individuals who undergo pancreatic resection with curative intent. 4 Chemotherapy is also the main treatment for individuals with advanced disease. For patients with locally advanced pancreatic cancer, chemoradiotherapy can downstage about 30% of them to a resectable state, and the median survival of these individuals is similar to the patients whose tumors are initially resectable without the need for any preoperative treatment. 5 Gemcitabine-based combination treatments have been assessed for advanced pancreatic cancer. Single-agent gemcitabine has been the standard regimen and is widely adopted in clinical practice. In addition, the novel regimens FOLFIRINOX [oxaliplatin, irinotecan, fluorouracil (5FU), and leucovorin] and gemcitabine/paclitaxel have shown better curative effects. No standard second-line treatment exists for pancreatic cancer, and many patients with advanced disease progress too rapidly to tolerate such regimens. Second-line fluoropyrimidine-based therapy is sometimes used if gemcitabine has been given as the first-line treatment. 2
Pancreatic cancer tissues comprise several distinct elements, including pancreatic cancer cells (PCCs), pancreatic cancer stem cells, and the tumor stroma. 6 Pancreatic cancer is characterized by the formation of a dense stroma, termed the desmoplastic reaction, whose microenvironment contains a mixture of interacting cellular and noncellular elements. The stromal cells comprise fibroblasts, pancreatic stellate cells (PSCs), endothelial cells, immune and inflammatory cells, and adipocytes.6,7 Cancer-associated fibroblasts (CAFs) are cellular components of the desmoplastic stroma that contribute to chemotherapy resistance. 8 PSCs are key cellular elements in the stroma and they are the most important source of CAFs. 9 Multiple types of immune and inflammatory cells can promote an immunosuppressive microenvironment. 10 Other components of the stroma, including the poor density of the vascular architecture and epithelial mesenchymal transition (EMT), also play significant roles in therapeutic resistance.8,11–13 In addition, a mature pancreatic cancer cell carries an average of 63 genetic alterations, which can be grouped into 12 core signaling pathways. 14 Each site of the signaling pathway can be a potential target for the chemotherapy of pancreatic cancer.
To overcome the biological barriers of pancreatic cancer for chemotherapy, advanced polymer nanoparticles are often used as excipients to improve the penetration and decrease the side effects of anticancer therapeutic agents. Compared with normal tissue, the tumor microenvironment (TME) has some unique properties, such as vascular abnormalities, hypoxia, and an acidic microenvironment. 15 In addition, various types of cells are fed by disordered blood vessels in pancreatic cancer. 16 Therefore, nanoparticles can exploit the TME to enhance therapeutic effectiveness. They have been used in vivo to protect drug entities in the systemic circulation, restrict the access of drugs to the chosen sites, and deliver drugs at a controlled and sustained rate to the site of action. 17 A nanoparticle drug delivery system (DDS) has many significant advantages over conventional molecular agents,18,19 such as the protection of incorporated agents from degradation, targeting the tumor site, controlled release of incorporated agents, multimodality for diagnostics and therapeutics, and easier elimination from the body. 20 Notably, the natural biocompatibility and biodegradability of nanoparticles make them extremely promising in drug delivery applications. Therefore, polymer nanoparticles represent a novel and promising delivery system for pancreatic cancer to overcome the natural chemotherapy obstacles (Figure 1). 21

Nanoparticles used as a drug carrier pass through complex tumor extracellular stroma to treat pancreatic cancer.
This review illustrates the mechanism of chemotherapy obstacles toward pancreatic cancer, including the components of the stroma, the changes in the TME, and the interaction of various elements. We also summarize pancreatic cancer models and discuss their advantages and disadvantages. Conclusions regarding the advantages, current challenges, and perspectives of nanoparticles for the chemotherapy of pancreatic cancer are provided.
Chemotherapy obstacles toward pancreatic cancer
The stromal component of pancreatic cancer and the interaction of different components
Pancreatic cancer is a very malignant disease. The tumor tissues of pancreatic cancer consist of cancer cells and stromal elements, which create a special TME for cancer cells. The exclusive TME is characterized by dense desmoplasia and profound infiltrations of immunosuppressive cells. 22 Notably, the desmoplastic stroma accounts for more than 80% of pancreatic cancer tissue. The cellular components of the stroma include PSCs, CAFs, tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs), neutrophils, adipocytes, epithelial cells, pericytes, mast cells, and lymphocytes. 23 Noncellular components include the extracellular matrix (ECM), a variety of enzymes, cytokines, and growth factors. The ECM consists of multiple materials, such as collagen, laminin, integrin, fibronectin, glycosaminoglycan, matrix metalloproteinase (MMP), and secreted protein acidic and rich in cysteine (SPARC). 24 These materials come into being an organic whole in which they interact with each other and accelerate the process of tumorigenesis, pancreatic cancer growth, migration, invasion, metastasis, and resistance to chemotherapy.
Among these factors, CAFs are critical components of the TME. CAFs can originate from PSCs, resident fibroblasts, the differentiation of bone-marrow-derived mesenchymal stem cells, and EMT. 25 CAFs can be activated via various cytokines and other types of cells in pancreatic cancer tissue, such as transforming growth factor beta (TGF-β) and chemokine (C-X-C motif) ligand 2 (CXCL2). 23 CAFs play an important role in desmoplasia by secreting various ECM components, such as collagen, fibronectin, proteoglycans, and glycosaminoglycans. Hyaluronic acid is one of the most important glycosaminoglycans, which will be discussed later in this article. By contrast, the secretion of ECM components increases the mechanical pressure, leading to cancer-cell migration, and increases the tumor vascularization. 23 The ECM components are biophysical barriers that hinder the pharmacokinetics and pharmacodynamics of drugs, reducing their therapeutic efficacy. In addition, CAFs produce various molecules and cytokines to promote tumor proliferation, accelerate tumor invasion and metastasis, and induce angiogenesis and chemoresistance. For example, Begum and colleagues cocultured CAFs from patients’ tumor tissues with pancreatic ductal adenocarcinoma (PDAC) cells. They found that CAFs enhanced PDAC growth and self-renewal and increased the frequency of cancer stem cells through type I collagen production. 26
PSCs, another important component of the pancreatic stroma, comprise about 4–7% of normal pancreas. 27 PSCs are normally quiescent and are termed qPSCs. The qPSCs can transit into an activated myofibroblast-like phenotype, termed as aPSCs. The aPSCs express fibroblast activation proteins, such as α-smooth muscle actin (α-SMA). 28 It is now commonly accepted that aPSCs are essential for the desmoplastic reaction in pancreatic cancer. 29 Amrutkar and coworkers cocultured PSCs with seven different PCC lines, respectively, by both direct and indirect means, and various degrees of chemoresistance were detected among all PCC lines. By examining PSC-conditioned medium, they found several proteins that participated in the construction of ECM, such as fibronectin and collagen. 30 Koikawa and colleagues found that PSCs and PCCs frequently invaded the collagen matrix successively to establish three-dimensional matrix remodeling. PCCs invade after the PSCs because PSCs remodeled the ECM and increased parallel fibers along the direction of invading PSCs. 31 In addition to CAFs, PSCs are another main source of fibronectin and collagen in pancreatic cancer and they contribute to the chemotherapy obstacles.30,32
In TME, TAMs and MDSCs are critical drivers of immune escape and resistance to cytotoxic therapies. 33 PCCs and fibroblasts in the TME can polarize M2-like macrophages and MDSCs via immunosuppressive cytokines. 34 MDSCs are a mixture of different types of immune cells that accumulate in cancer tissues. Myeloid cells are recruited from the bone marrow through chemokines secreted by cancer cells and stromal cells. MDSCs are blocked at different stages in the progression of maturation via tumor-derived factors. Currently, MDSCs in human cancer tissues can be identified by the expression of CD 14 or CD15. The main function of MDSCs is suppressing host immunity by reactive oxygen and arginase produced by various mechanisms. 35 There are abundant immunosuppressive TAMs in the TME. 36 However, the origin of TAMs remains unclear. Research has shown that they are not only derived from hematopoietic precursors, but also from embryonic precursors with self-renewal capability. 37 TAMs and other inflammatory cells, such as mast cells, lymphocytes, and neutrophils, are key players connecting inflammation and cancer by secreting pro-inflammatory cytokines and chemokines. 37 TAMs affect inflammation, metastasis, angiogenesis, and hypoxia of tumors by regulating the levels of interleukin (IL)-10, IL-12, C-C motif chemokine ligand (CCL)13, CCL18, 38 vascular endothelial growth factor alpha (VEGF-A), CCL2, 39 TGF-β, IL-23, 40 and bone morphogenic protein (BMP). 41 TAMs induce EMT and coordinate with other stromal components (e.g. MDSCs) to promote therapeutic resistance. 42 Halbrook and coworkers found that TAMs could release pyrimidines (e.g. deoxycytidine), which inhibit gemcitabine via molecular competition in the process of drug uptake and metabolism. 43
There are rare CD8+ T cells that recognize and clear cancer cells, whereas abundant regulatory T cells induce immune tolerance. 9 High levels of neutrophils are always associated with poor prognosis in patients with pancreatic cancer. 44 Solid epidemiological evidence proves that obesity is associated with high morbidity and low overall survival. 44 However, the function of adipocytes in pancreatic cancer remains ambiguous. 45 Endothelial cells are located in the innermost layer of tumor blood vessels and are one of the targets of antitumor vascular therapy.
The supplement changes of oxygen and blood
Tissue hypoxia and vascular abnormalities are notable characteristics of the microenvironment in pancreatic cancer. Through immunofluorescence and phosphorescence lifetime imaging of oxygen-sensitive nanoparticles, Conway and coworkers demonstrated the existence of hypoxic regions. 46 In addition, tumor blood vessels are immature and leaky, with low expression of the vascular tight junction marker CD31. 47 Furthermore, the immature vasculatures of cancer tissue are disordered, with small pore sizes. 48
Many factors can lead to changes of the oxygen level in the TME. First, the deposition of a large amount of desmoplastic stroma increases the pressure of the TME, leading to vascular collapse and hypoxia.49,50 Second, the composition of the blood is also related to the supplemental changes of oxygen. Li and colleagues established both in vivo and in vitro models and found that a hyperglycemic environment was associated with tissue hypoxia and poor prognosis. 51 Third, the stromal components can influence the supplements of oxygen through various signaling pathways. For example, Xu and coworkers suggested that hyaluronan could influence angiogenesis by regulating small guanosine triphosphatases and receptors of tyrosine kinase. 52 Finally, PCCs have an effect on angiogenesis. Maity and colleagues showed that PCCs activate Sonic Hedgehog (SHH) via the CCN1 (formerly known as Cyr61, a matricellular protein of the CCN-family) signaling pathway to promote tumor angiogenesis. 53
Hypoxia can affect the prognosis of pancreatic cancer by affecting angiogenesis, EMT, the immunosuppressive phenotype, tumor migration, invasion, and metastasis. 54 There is a close relationship between hypoxia and angiogenesis. Among various pro-angiogenic factors, such as VEGF, fibroblast growth factor-2, platelet-derived growth factor, angiopoietin-1, and tyrosine kinase with immunoglobulin and epidermal growth factor homology domains-2 (Tie-2),55,56 VEGF has attracted extensive research attention. In vitro, all pancreatic cancer-cell lines that are exposed to low oxygen show a high level of VEGF production. 57 Tsuzuki and coworkers established abdominal-wall windows for intravital microscopy and found that the TME promotes VEGF expression. 58 VEGF can be activated by the binding of hypoxia-inducible factor-1 (HIF-1), an oxygen-sensitive transcriptional activator, to the VEGF promoter. 59 In normal tissues, the degradation of HIFs can be regulated by prolyl hydroxylase domain 3 (PHD3), a rate-limiting enzyme that might be a novel target in the therapy of pancreatic cancer. 60 In addition, hypoxia can facilitate EMT via the NOTCH or c-MET pathways and inhibit the immune response by transiting immune cells into an immunosuppressive phenotype. 61 Hypoxia played a synergistic role in AKT-signaling-mediated gemcitabine resistance of pancreatic cancer. 62 Wu and colleagues demonstrated that hypoxia might facilitate the migration of PCCs by upregulating quiescin sulfhydryl oxidase 1 (QSOX1), which extends the EMT of PCCs. 63 Shi and coworkers indicated that hypoxia extended the expression of QSOX1 in vitro, which promoted the invasion of PCCs. 64 In addition, Sun and colleagues indicated that metastasis-associated 1 (MTA1) could promote the metastasis of PCCs via the HIF-α/VEGF pathway, which was associated with hypoxia. 65
Since the article about angiostatin properties was published by a Children’s hospital in Boston about 40 years ago, the research on antiangiogenesis therapy has never stopped. 66 Antivascular therapy has produced objective effects in certain tumors, such as kidney cancer,67,68 ovarian cancer, 69 colorectal cancer, 70 and breast cancer.71,72 Human PCCs also overexpress various angiogenic factors, as mentioned above. Although antiangiogenic agents are able to reduce the microvessel density (MVD) and the tumor volume, and improve the survival time in some animal models, 73 the results of clinical trials using antivascular drugs have been disappointing, which might because therapy-induced hypoxia can increase the process of EMT. 74 Based on unsuccessful clinical trials, some scholars advanced new strategies to treat pancreatic cancer to improve prognosis by improving hypoxia rather than inhibiting angiogenesis. These strategies might improve the antitumor efficacy via better delivery of antitumor immune cells and antineoplastic drugs through stable vessels. 75 Hyaluronic acid, a component of the stroma, oppresses blood vessels, leading to hypoxia in pancreatic cancer. Li and colleagues targeted hyaluronic acid using polyethylene glycol (PEG)ylated recombinant human hyaluronidase (PEGPH20), which dramatically reduced the stromal component. Moreover, PEGPH20 downregulated the level of VEGF-A165 and diminished the angiogenic potential of the TME in vivo. 50 Banerjee and coworkers applied Minnelide (a water-soluble prodrug of triptolide, currently in a phase I clinical trial) in tumor-bearing mice and patient tumor xenografts. Minnelide could reduce the components of the ECM, such as hyaluronic acid and collagen, while increasing the functional vasculature to four times that of the control group. 49 However, attempts to eliminate stromal components might result in the formation of more malignant phenotypes of PCCs. Signal transducer and activator of transcription 3 (STAT3) inhibition combined with gemcitabine increases the MVD and promotes vascular normalization, which could enhance the efficacy of drug delivery without depleting stromal components like collagen or hyaluronan. 76 Shin and coworkers applied Fuco-MnO2-nanoparticles to efficiently generate oxygen in the presence of H2O2 in vitro. By increasing the oxygen levels in tumor tissues, HIF-1 expression and downstream pathways were suppressed. 77 Sasajima and colleagues attempted to rebuild an effective vascular system that could meet the metabolic needs and maintain homeostasis using bone marrow mononuclear cells (BMMNC). 78
The TME is the reason for chemotherapy obstacles in the treatment of pancreatic cancer
Medical researchers have performed intensive research into chemotherapeutic drugs and schemes for pancreatic cancer. In recent years, FOLFILINOX and nano-albumin-bound (Nab)-paclitaxel plus gemcitabine have been reported to work better than gemcitabine monotherapy and can increase overall survival for several months for patients with advanced pancreatic cancer. 79 However, for patients with pancreatic cancer, further studies are needed to achieve longer survival time. Limited chemotherapy effect and poor prognosis might be attributed to the characteristics of the TME in pancreatic cancer. 80 Smith and coworkers proposed that the solid tissues of a tumor could be divided into two types: the tumor vessel phenotype, which responds to antiangiogenic drugs, and the stromal vessel phenotype, with vessels distributed in the stroma around the tumor, which is poorly responsive to antiangiogenic drugs. 81 In fact, many solid tumors belong to the second type. Notably, the stromal component accounts for more than 80% of the pancreatic tumor tissues. In this review, we discuss the effects of the TME on the chemotherapy obstacles due to two main aspects.
Physicochemical conditions of the microenvironment
Physical barrier. Abundant accumulation of cellular components and acellular components, such as fibrosis, collagen, SPARC, and hyaluronan, enhance the stiffness and reduce the elasticity of the TME. 82 Chemotherapy agents must extravasate and pass through the stiff and thick physical barrier to target the cancer cells, which will lead to inefficient permeability to cancer nests and limit the efficacy of chemotherapy.80,83,84
Physical pressure. Increased interstitial fluid pressure (IFP), attributed to abundant hyaluronan and the tough desmoplastic stroma, leads to the collapse of blood vessels. Abnormal vascular structures result in low flow perfusion, which restricts the penetration of chemotherapy drugs. Some researchers have focused on improving perfusion by increasing the vasculature density, which improved the efficacy of chemotherapy in a variety of models.76,78,85 In addition, compared with the hypoperfusion in pancreatic cancer tissues, healthy tissues have a normal vascular system and normal perfusion, which unfortunately results in highly nonspecific toxicity. The dosage of chemotherapeutic agents is limited because of certain serious side effects, such as neuropathy suppression, bone marrow suppression, and gastrointestinal reaction. This limitation could affect the efficacy of chemotherapy. 79
Hypoxia condition. The induction of reactive oxygen species (ROS) generation plays an important role in current clinical treatments using chemotherapeutic agents, molecular-targeted drugs, photodynamic therapy (PDT), and radiotherapy using ionizing radiation. 86 The changes in supplemental oxygen caused by hypoperfusion are one of the key factors of chemotherapy obstacles. For example, doxorubicin, a widely used antineoplastic agent, cannot produce sufficient ROS when reacting with insufficient oxygen, which suppresses the chemotherapy efficacy of doxorubicin. 87 Nomura and coworkers found that hypoxia could lead to altered metabolic profiles of cells. Cells obtain energy by aerobic glycolysis instead of oxidative phosphorylation, which limits the production of ROS induced by drugs like paclitaxel, 5FU, and gemcitabine. 88 Raykov and coworkers used myo-inositol trispyrophosphate (ITPP) to improve vessel structure and increase the supplement of oxygen. This reversed hypoxia and improved the cells’ susceptibility to gemcitabine in pancreatic cancer tissues. 89
Low pH value. The hypoxic characteristics of tumors often coincide with low PH values. 90 Changes in metabolism significantly increase lactate dehydrogenase A and tightly regulate carbonic anhydrases (Cas), which are key players in the acidification of the TME.91,92 The change of pH value reduces the absorption of drugs, and contributes to increased drug efflux. 13 Tumor initial cells (TICs) resist damage by chemotherapeutic compounds through the ABC transporter family which can be activated by special TME to maintain a low intracellular drug concentration. 13 Moreover, tumor cells remove extra protons through extracellular vesicles (Evs), which are involved in the development of tumor biological processes and chemotherapy resistance. 93
Biological function of cells and networks of cells in the microenvironment
PSCs. PSCs are neglected components of the TME. As we described earlier, a large amount of the ECM is produced by PSCs, such as collagens and hyaluronic acid, playing an important role in the physical barrier, physical pressure, hypoperfusion, and hypoxia. Besides, PSCs interact with cancer cells at all stages of cancer development. 52 For example, through the NOTCH signaling pathway, PSCs promote the proliferation, migration, differentiation, and chemotherapy resistance of cancer cells.82,94,95 Fibronectin secreted by PSCs promotes the chemoresistance of PCCs to gemcitabine via increasing phosphorylation of the ERK1/2. 30 As aforementioned, PSCs are important source of CAFs, which also interact with cancer cells at all stages of cancer development. Complex connections between cells in the TME increase the difficulty of treatment. Furthermore, several signaling pathways induced by PSCs related to chemotherapeutic resistance were summarized by Fu and coworkers, including IL-6/Janus kinase (JAK)/ STAT signaling, paracrine SHH signaling, and the stromal-derived factor 1 (CXCL12)/SDF-1 receptor (CXCR4) signaling axis. 28 Moreover, hyaluronic acid secreted by PSCs can bind with the hyaluronic acid receptor expressed by PCCs and activate many signaling pathways, such as RAS, Ras-related C3 botulinum toxin substrate (Rac), mitogen-activated protein kinase (MAPK), and phosphatidylinositol-3-kinase (PI3K). These signaling pathways also accelerate chemotherapeutic resistance in pancreatic cancer. 82 Last but not least, SPARC is another vital component of the TME secreted by PSCs. The inverse correlation of SPARC with the overall survival of patients further demonstrates the role of PSCs in chemotherapeutic resistance. 96
CAFs. CAFs can originate from PSCs and they are the main source of the desmoplastic stroma. CAFs are not only closely related with the physicochemical conditions in the TME, but also are involved in chemotherapy obstacles through the release of Evs and a variety of signaling pathways, such as the SDF1A/CXCR4 axis, the FAK/AKT, mTOR/4E-BP1 axis, TGF-β-activin receptor-like kinase (ALK) 5-Smad2/3 signaling, insulin-like growth factors (IGF) 1 and 2, IL-6, and CYR61.8,25,97 In addition, Lakiotaki and colleagues observed that CAFs were densely arranged in a ring around the cancer nest. 16 Interestingly, Hessmann and coworkers found that the lack of key metabolic enzymes involved in gemcitabine inactivation contributed to the high concentration of cytotoxicity agents in CAFs. 98 Combining these two findings, it seems that CAFs are ‘selfless shields’ against chemotherapeutic agents that act as drug scavenging for PCCs.
TAMs. The apoptosis of PCCs induced by chemotherapeutic agents can induce the infiltration of TAMs. Deshmukh and colleagues observed more macrophages in tumor-bearing mice treated with gemcitabine than in those treated with vehicle. An increased number of macrophages might correlate with the regulation of IL-8. 99 TAMs release deoxycytidine to inhibit the uptake and metabolism of gemcitabine via molecular competition. 43 Moreover, TAMs release IGF to bind with the IGF receptors in PCCs. The interaction between TAMs and PCCs improves the chemoresistance of pancreatic cancer. 100
MDSCs. When treated with gemcitabine or 5FU, MDSCs secret cathepsin B, which activates NLRP3 inflammasomes. The activation of NLRP3 inflammasomes reduces the antineoplastic power of chemotherapeutic drugs because of the secretion of IL-1β. 101 Besides, the immunosuppression function of MDSCs plays a synergistic role in chemotherapeutic resistance.
Although the TME is the main reason for chemotherapy obstacles in the treatment of pancreatic cancer, it also provides promising avenues for better prognosis.
The establishment of pancreatic cancer model in mice
To obtain a comprehensive understanding of biological characteristics and therapeutic response of pancreatic cancer, the establishment of pancreatic cancer models is essential. It is difficult to set up pancreatic cancer models because of its genetic heterogeneity and complex TME. 102 However, we already possess a series of PDAC models with their respective advantages and weaknesses, and the development of new methods to establish models is ongoing. Pancreatic cancer models can be divided into two main categories: in vitro models and in vivo models.
In vitro models
It is convenient for researchers to observe and manipulate experimental conditions using in vitro models. 103 According to the cancer-cell source, in vitro models can be divided into two types: commercially available human PDAC cell lines and patient-derived tumor models.
Human PDAC cell lines are homogenous and easy to culture. Moreover, this method is cheaper and easier to perform than other experiments. 102 PDAC cell lines have played a vital role in chemotherapy development. However, genetic drift might occur during long-term culture and there are only a few commercial PDAC cell lines to choose compared with other tumor cell lines. 104 Among these commercial PDAC cell lines, some classical ones have been acquired from metastasis-derived cells, such as Capan-1, Aspc-1, and CFPAC.105,106 To date, there is no unified proposal whether metastatic derivatives can replace primary tumors. 105 Additionally, commercial cell lines are preferably propagated with monolayer cultures without the TME, which makes it difficult to recapitulate in vivo conditions using these cell lines. 102
Patient-derived in vitro models and patient-derived primary cell models can represent the heterogeneous nature with a variety of genetic mutations. They are established based on the surgical specimens from individual patients; therefore, patient-derived primary cell models can closely reflect the original tumors, which are useful for studying disease-related biology, as well as guiding future precision medicine. Beyond PDAC, patient-derived primary cell models can be established in other diverse pathological types of pancreatic cancer, such as adenosquamous carcinoma and ductal adenocarcinoma with oncocytic intraductal papillary mucinous neoplasm. Patient-derived primary cell models allow the assessment of novel therapeutic plans for these low-frequency pathological types of pancreatic cancer. 107 Nevertheless, the generation of primary cells from patient-derived tumor tissues is difficult because of its time-intensive nature and the use of huge amounts of patient-derived tumor tissue.107,108 Patients with overall survival less than 6 months would hardly get any benefit from experiments based on patient-derived primary cell models. Patients with resectable pancreatic cancer account for only a small proportion of all patients with pancreatic cancer. To solve the problem of primary tumor material, some researchers use patient-derived xenograft mice models to expand the volume of clinical samples. However, the rate of success is low because of the overgrowth of mouse stromal cells and bacterial contamination. 105
These two models cannot recapitulate the complex TME of pancreatic cancer very well. To obtain more reliable experimental results for therapeutic regimens, coculture methods (PSCs and PCCs) were developed. PSCs can be harvested by density-gradient centrifugation and outgrowth methods. Cell culture of PSCs and coculture of PSCs and PCCs help researchers to further explore the role of PSCs in the TME as well as the therapeutic response of PCCs in a complex microenvironment. 103 To date, there is no standard for experimental manipulation of culture conditions. According to different experimental purposes, investigators can adjust the cell line of PSCs, species of PSCs (human or mouse), proportion of PSCs and PCCs, and pattern of coculture (indirect coculture method or direct coculture method). Coculture of PSCs and PCCs partly help us understand the interaction between PCCs and the TME.
In vivo models
Compared with in vitro models, in vivo models provide a pathophysiological environment which is closer to that of the human body. Xenograft models can be established by injecting PCCs or transplanting freshly resected tumor pieces orthotopically or ectopically into nude mice. 109 The cells used to build xenograft models can originate from commercial human PDAC cell lines, pancreatic ascites/pleural effusion cancer cells, or diverse types of cocultured PCCs.
Xenograft models based on cell lines share some shortcomings with commercial human PDAC cell lines. The National Cancer Institute suggests a moderate predictive value for the models after more than 10 years of extensive screening. 110 The clinical efficacy of numerous agents that work relatively well in preclinical models is disappointing. The disconnection between the outcomes of in vivo testing and clinical utility suggest that further evolution of tumor models is required.
Patient-derived xenografts maintain the tumor morphology, genetic stability, and metastatic potential. 111 Moreover, patient-derived xenografts have consistent biological properties with pancreatic cancer, such that they can mirror patients’ responses to chemotherapeutic agents. However, just like patient-derived primary cell models, patient-derived xenografts need a long time and more financial support to reach clinical utility. 102
About 20% of patients with advanced PDAC have ascites effusion or pleural effusion. Golan and colleagues utilized PCCs from ascites and pleural effusion to establish tumor models without surgery. This kind of model has complimentary value in investigating advanced PDAC. 112
Xenograft models originating from diverse types of cocultured cells have potential in the study of stromal components as well as novel therapeutic regimens. Three-dimensional (3D) pancreatic cancer spheroids, based on cocultured PCCs and PSCS, improve the production of ECM, comprising fibronectin, collagen, laminin, and hyaluronic acid, with a highly reproducible and uniform architecture. Interactions between PCCs and stromal components increase exponentially because of the added third dimension. 102 Thus, 3D cocultured pancreatic cancer spheroids improved the efficacy of the establishment of subcutaneous xenograft models significantly. The 3D cocultured pancreatic cancer spheroids have almost met the requirements for chemotherapeutic drug studies, such as optimal reproducibility, high throughput, simple assay readouts, and simulation of the TME. 113 Nowadays, 3D coculture of pancreatic cancer spheroids has become the most widely used animal model to test the efficiency of novel therapeutic approaches. 114
Cells or resected tumor pieces can be inoculated subcutaneously, intraperitoneally, or orthotopically. All routes of administration can produce xenograft models. Among them, subcutaneous implantation is used most frequently, whereas orthotopic and intraperitoneal implantation are more suited to study the cachexia induced by pancreatic cancer. 115 Although orthotopic xenograft models work better in recapitulating the clinical, pathological, genetic, and molecular aspects of pancreatic cancer, subcutaneous xenograft models are more popular with pharmaceutical companies. Subcutaneous transplantation is convenient and intuitive for the assessment of tumor data. Orthotopic transplantation is more costly because there is no easy way to discern the therapeutic efficacy.102,116 Yu and colleagues developed a method for building an orthotopic implantation model. They established a red fluorescent protein (RFP) orthotopic implantation model utilizing stable high RFP-expressing cells, SW1990-RFP. As a result, they could obtain tumor data in a noninvasive way with appropriate sensitivity and specificity. 117 Delitto and coworkers established an orthotopic implantation model via injecting a low-temperature matrigel suspension that mimics the pathological features of pancreatic cancer. Matrigel could form a gel drip that avoided leakage from the injection site and dispersion in the abdominal cavity. 111 All the xenograft mice models we have mentioned should be established in immunodeficient nude mice in which immunotherapy studies cannot be conducted. Without the effect of the immune system, the TME is incomplete in these models. This may be one of the reasons xenograft mice models cannot be used to assess therapeutic approaches perfectly. 105
Genetically engineered mouse models (GEMMs) avoid the common problems of xenograft models. The intact immune system of GEMMs is useful to test the immune response and immunotherapeutic approaches. 102 Compared with xenograft models, GEMMs fully replicate vital morphology features of human pancreatic cancer, such as the desmoplastic stroma and abnormal vasculature. In addition, GEMMs exhibit pathophysiological characteristics defined in human pancreatic cancer, such as pain and cachexia. Besides, GEMMs could be used to discover biomarkers. However, they are not widely applied because of their extremely high cost. Another disadvantage is that there is no close relationship between GEMMs with specific gene mutations and patients with various gene-mutation profiles.
Beyond xenograft models and genetically engineered mouse models, innovative cancer models are evolving. For instance, organoid models have been developed to study biological questions associated with pancreatic cancer since 2015. 102 Further tests are needed to innovate and assess the value of new models.
Advanced polymer nanoparticles as excipients of pancreatic cancer chemotherapy
Polymeric organic nanoparticles as a carrier for chemotherapy
Since Birrenbach and Speiser utilized nanoparticles in the pharmaceutical field in the 1970s, polymeric nanoparticles have received growing interest as DDSs.118,119 Polymeric nanoparticles have been investigated in a variety of diseases, such as atherosclerosis,120,121 Alzheimer’s disease, 122 diabetes, 123 psoriasis, 124 alopecia, 125 human immunodeficiency virus, 126 and cancer. Among these diseases, nanoparticles have been mostly studied in the application of cancer chemotherapy.
Chemotherapy is one of the main treatment methods against cancer. However, conventional chemotherapeutic agents have narrow therapeutic indices because of critical issues, such as low aqueous solubility, rapid drug metabolism and excretion, lack of selectivity, and dose-limiting toxicity.127,128 Some patients cannot benefit from chemotherapy because anticancer agents have poor bioavailability and severe side effects. 48 Furthermore, some chemotherapeutic agents may induce the occurrence of multidrug resistance and metastasis (MDR). 129 The application of nanoparticles as drug carriers can address these concerns. 129 The pharmacokinetics and pharmacological properties of chemotherapeutic agents can be altered by improving their solubility. Protected by polymeric nanoparticles, anticancer drugs can avoid rapid elimination or premature degradation. 130 The prolonging circulation time reduces the required drug dosage, as well as enhances the antitumor efficacy, with minimal side effects.118,129 Polymeric nanoparticles make it possible for controlled and targeted drug delivery at sites of action.118,128 In addition, nanoparticle DDSs have excellent biodegradability, good biocompatibility, and low toxicity. 131 Another important feature of polymeric nanoparticles is their high surface-to-volume ratio, which provides polymeric nanoparticles with high drug-loading ability and more interactions with tumor environment. 127 The properties and functions of nanoparticles can be regulated in various ways to satisfy different therapeutic or diagnostic needs. For instance, to address the issue of real-time observation of biodistribution or therapeutic effectiveness, we can incorporate imaging probes, such as fluorescent dyes, into polymeric nanoparticles.
Polymeric nanoparticles are solid colloidal systems. There are several dominant methods for synthesizing polymeric nanoparticles: (a) emulsification and solvent evaporation/extraction; (b) nanoprecipitation; (c) supercritical antisolvent method; and (d) salting out. 132 According to their morphological structure, polymeric nanoparticles can be divided into nanospheres and nanocapsules. When drugs are encapsulated within a polymeric matrix, the nanoparticles are termed nanospheres. Meanwhile, nanocapsules utilize a polymeric membrane to entrap drugs in a cavity. 133 The raw materials of nanoparticles range from natural sources, including gelatin, chitosan, alginate, dextran, heparin, collagen, albumin, and polyhydroxyalkanoates (PHAs); to synthetic sources, including poly(lactic–coglycolic acid; PLGA), poly-n(cyanoacrylate), poly(caprolactone; PCL), PEG, and cyclodextrins.118,132,133 In general, natural materials have limited sources and relatively complicated extraction processes, but have better bioavailability and biodegradability. For example, chitosan is a kind of natural polymer produced from chitin, derived from marine organisms.134,135 Compared with natural materials, the preparation of synthetic materials is simple, but the biodegradability is relatively poor. Albumin and PLGA are both approved by the US Food and Drug Administration for the usage in biomaterial pharmaceuticals. Human serum albumin is the most abundant serum protein naturally occurring in the human circulatory system and has been proven a safe carrier for drugs. 136 Moreover, human serum albumin is a catabolic source of glutamine for tumor, so albumin drug carriers could be taken up and scavenged by solid tumors.137,138 Meanwhile, PLGA can also be used for tissue engineering repair, with the retention time of at least 1 month in vivo.139,140 The degradation rate of PLGA is relatively slower in vivo than that of natural materials.
Targeted drug delivery is the most important characteristic of polymeric nanoparticles. Mechanisms of targeted DDS include passive targeting and active targeting. 141 Nanoparticles with acid-sensitive spacers are stable at the bloodstream pH and facilitate the release of anticancer components when pH value falls below 5.5 in the TME. 142 Enhanced permeation and retention (EPR) caused by immature vasculature and poor lymphatic drainage play an important role in passive targeted delivery. Compared with normal tissues, it is easier for small nanoparticles to reach and remain in cancer tissues because of the special TME. 143 Nanoparticles with specific ligands, such as antibodies, small molecules, peptides, proteins, transferrin, and folate can be employed to achieve active targeted drug delivery. 48 Stimuli-responsive nanoparticles have been created as smart carriers to release drugs under specific stimuli, including pH, temperature, and specific enzymes. 144 Li and coworkers showed that ultrasound upregulated the drug release rate from the nanoparticles into cancer tissues and accelerated the treatment effectiveness of antineoplastic components. 120 Molecularly imprinted nanoparticles have emerged as a novel platform for a ligand-free targeted DDS. 145 Esfandyari-Manesh and colleagues synthesized molecularly imprinted polymer–polyethylene-glycol–folic acid (MIP-PEG-FA) nanoparticles by mini-emulsion polymerization technology to transport paclitaxel (PTX) to cancer cells. The MIP-PEG-FA nanoparticles were taken up into cells more effectively, with lower half-maximal inhibitory concentrations (Figure 2). Molecularly imprinted polymer is a kind of polymeric material which is able to mimic natural recognition entities, such as biological receptors and antibodies. Therefore, molecularly imprinted polymer could be adapted to a variety of demands by adjusting the morphological features. These polymeric materials have broad application in various fields, especially in medical therapy. 146

Preparation of MIP-PEG-FA nanoparticles and characteristics.
Nanoparticles can be used as a carrier for chemotherapy to treat a variety of cancers, such as pancreatic cancer, breast cancer,133,147,148 non-small cell lung cancer (NSCLC), gastrointestinal cancer, 149 hepatic cancer, 150 colorectal cancer,151,152 ovarian carcinoma,147,153,154 and glioblastoma multiforme. 155 Abraxane, based on albumin, received ribonucleic acid approval for clinical application in treating pancreatic cancer, breast cancer, and NSCLC. 156 Faramarzi and coworkers showed that metformin-encapsulated PLGA–PEG nanoparticles had enhanced antiproliferative and pro-apoptotic effects on human ovarian carcinoma cells. 153
Multifunctional polymeric nanoparticles for pancreatic cancer
Gemcitabine plus Nab-paclitaxel (NAB-P + GEM) may be the most suitable regimen for treating pancreatic cancer. 157 Nab-paclitaxel, also termed Abraxane®, is the only nanoparticle to receive FDA approval (in 2013) for clinical application. 156 Abraxane® could improve overall survival in a multicenter international, open-label randomized trial. 118 In consideration of the role of the highly dense tumor stroma in chemotherapy resistance, Nab-paclitaxel was utilized to deplete the tumor stroma through the interaction between albumin and SPARC. SPARC is a kind of albumin-binding glycoprotein overexpressed both in PCCs and stromal cells in pancreatic cancer. 158 Stromal depletion and increased microvasculature caused by Nab-paclitaxel led to a higher gemcitabine concentration within the tumor.159,160 The phase III Metastatic Pancreatic Adenocarcinoma Clinical Trial (MPACT) showed notable tumor shrinkage benefit of Nab-paclitaxel plus gemcitabine compared with gemcitabine alone. 161 Although the combination of gemcitabine and Nab-paclitaxel increased the median survival by about 1.8 months, 159 the regimen still needs to be improved. Nitric oxide (NO), an intrinsic vascular modulator, enhances the permeability and retention of nanoparticles. Kinoshita and colleagues evaluated the effect of S-nitrosated human serum albumin dimers (SNO-HSA Dimer) as a pretreatment agent in Nab-paclitaxel therapy. SNO-HSA Dimers enhanced tumor selectivity of Nab-paclitaxel, the combination of SNO-HSA Dimer and Nab-paclitaxel showed high antitumor activity in a SUIT2 human pancreatic cancer orthotopic model. 162
In recent years, nanoparticles have emerged as a promising approach for delivering drugs to tumor sites in pancreatic cancer treatment. In addition to Nab-paclitaxel, we have made a brief summary and classified the multifunctional organic nanoparticles into the following categories: facile nanoparticles; ligand-based active-targeted nanoparticles; pH-sensitivity nanoparticles; redox-responsive targeted nanoparticle; traceable nanoparticles; multidrug codelivery nanoparticles; and other nanoparticles for new drugs or composed of new materials. Polymer nanoparticles engineered by researchers to treat pancreatic cancer in recent years are summarized in Table 1.
Polymer nanoparticles as a carrier for treatment of pancreatic cancer.

17AAG, 17- N-allylamino- 17-demethoxygeldanamycin; α-SMA, alpha smooth muscle actin; AA, acrylic acid; ASO, antisense oligonucleotides; bPEI, branched PEI; BPTES, bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl sulfide; BSANPs, bovine serum albumin nanoparticles; cRGD, cyclic arginine–glycine–aspartic acid; DBCO, dibenzylcyclooctyne; DNA, deoxyribonucleic acid; dsRNA, double-stranded RNA; DTX, docetaxel; DOX, doxorubicin; DZNep, 3-deazaneplanocin A; ECs, endothelial cell lines; EGFR, epidermal growth factor receptor; ENPs, cationic ε-polylysine copolymer; EPR, enhanced permeation and retention; GEM, gemcitabine; Glu, glutamine; hENT, human equilibrative nucleoside transporter; HIF1α, hypoxia-inducible factor 1 alpha; HSA, human serum albumin; ICG, indocyanine green; LENP, cationic ε-polylysine copolymer coating with PEGylated lipid bilayer; mPEG, methoxy polyethylene glycol; NanoCurc, nanoparticle encapsulated curcumin; NDAT, nano-diamino-tetrac; NF, nanoformulation; NIPAAM, N-isopropylacrylamide; NPs, nanoparticles; ORM, ormeloxifene; P@-GEM-HSA-NPs, pheophorbide-a- gemcitabine-human serum albumin-nanoparticles; PDMAEMA, poly(dimethylaminoethyl methacrylate); pDNA, plasmid DNA; PDX, patient-derived xenograft; PEG, polyethylene glycol; PEI, polyethylenimine; PLA, polylactic acid; PLA-COONa, polylactic acid monovalent salt; PLGA, poly(lactic–coglycolic acid); PNP, polymeric nanoparticle; POEGMA, poly[oligo(ethylene glycol) methyl ether methacrylate]; PTX, paclitaxel; RAFT, reversible addition–fragmentation chain transfer polymerization; RGD, arginine–glycine–aspartic acid; RIG-I, retinoic-acid-inducible gene I; RNA, ribonucleic acid; scFv, single-chain variable fragment; SCID, severe combined immunodeficient; siRNA, small interfering RNA; SQdFdC, squalenoyl prodrug of gemcitabine; TF, transferrin; TPP, triphenyl phosphonium.
Facile nanoparticles composed of frequently used materials are used to deliver antipancreatic cancer drugs. PLGA, PEG, or the combination of these two materials are the most frequently used synthetic materials. Meanwhile, albumin is one of the most popular natural sources materials. As a result of the EPR effect, facile nanoparticles passively accumulate at the tumor site. Ernsting and coworkers investigated a Cellax-docetaxel (DTX) polymer to reduce the stroma components in pancreatic cancer. Cellax-DTX polymer, a conjugate of PEGylated carboxymethylcellulose and DTX, can condense into nanoparticles in saline by a nanoprecipitation technique. Cellax-DTX was suitable for intravenous injection because the nanoparticles were stable in serum and long circulating compared with Nab-PTX. Moreover, Cellax-DTX could be stored at 4°C due to the structural stability of nanoparticles. The effect of Cellax-DTX in depleting stroma components was better than that of Nab-paclitaxel (Figure 3). 163 Elgogary and colleagues developed a kind of nanoparticle composed of block copolymers of PLGA and PEG to deliver glutaminase inhibitors to regulate glutamine metabolism in tumor tissue while protecting normal liver and kidney function. Sucrose esters were used as nano-emulsion stabilizers to increase the density of PEG on the nanoparticle surface, which assisted in increasing the pharmacological inhibition of glutaminase. Dense PEG coatings protected nanoparticles from aggregation, opsonization and phagocytosis, thereby prolonging the systemic circulation time of nanoparticles and improving drug delivery to the tumor. 192 The results from a patient-derived orthotopic pancreatic tumor model proved that glutaminase inhibition by PEGylated PLGA nanoparticles was promised as a novel therapy for pancreatic cancer. 164 Other therapeutic components, like ormeloxifene, 165 PH-427, 166 gemcitabine,167,168 docetaxel, 169 and RIG-I agonists, 170 were successfully delivered through facile nanoparticles and made sense in the therapy of pancreatic cancer. Genetic material can also be delivered to tumor sites via polymer nanoparticles for gene therapy. Arora and coworkers enscapsulated miR-150 to target the MUC4 gene in nanoparticles composed of PLGA and polyethylenimine (PEI). Ribonucleic acid (RNA) is characterized by negative charge, sensitivity to nuclease in plasma and poor uptake efficiency at the tumor site. Nanoparticles prevented the deletion of RNA in plasma and promoted RNA uptake by tumor cells. In particular, nanoparticles composed of PEI had a high encapsulation efficiency of RNA, because PEI is a cationic polymer. 171

Tissue microenvironment effects and tumor prevention efficacy of Cellax-DTX polymer in a PAN02 pancreatic tumor model.
Ligand-based active-targeted nanoparticles are advantageous and promising drug carriers because of their higher penetrability and higher affinity for cancer cells, such that they are more easily taken up by targeted cells. Yu and colleagues prepared cyclic arginine–glycine–aspartic-conjugated (cRGD), gemcitabine-loaded albumin nanoparticles that had a more robust tumor targeting ability than free drugs or ordinary nanoparticles. 172 Similarly, Ge and coworkers constructed polyplex micelles with an elevated PEG coat and used cRGD as a ligand. PEG was applied to confer long circulation properties to nanoparticles by decreasing nonspecific interactions with biological components. The cRGD at the end of PEG broke PEG shielding at the tumor site by interacting with integrins that were overexpressed in malignant tumor cells and tumor angiogenic endothelial cells. So the polyplex micelles succeeded in prolonging the systemic circulation time by PEG coat and facilitating targeted delivery by using cRGD as a ligand. 173 Min and colleagues conjugated antihuman tissue factor Fab’ on the surface of polymeric micelles to improve the targetability of nanoparticles. 174 The results from a pancreatic cancer xenograft model exhibited more than 15-fold increased cellular binding and rapid cellular internalization of the antibody (tissue factor- targeting Fab’) fragment-installed nanoparticles compared with that of ordinary nanoparticles. 175 As the expression level of the folate receptor (FR) on PCCs is relatively high, FR is another popular ligand. Zhou and coworkers developed FR-mediated gemcitabine nanoparticles with a core-shell structure. From the studies based on COLO357, SW1990, MIA PaCa-2, Capan1, L3.6pl, and BxPC3 cell lines and human pancreatic cancer orthotopic xenografts tumor models, they found that tumor cells could take up FR-based nanoparticles in multiple ways and this kind of nanoparticle could enhance the retention hysteresis effect in pancreatic cancer. 176 Tetrac targets the integrin αvβ3 receptor on the plasma membrane of cancer cells. Nano-diamino-tetrac (NDAT) could assist paclitaxel to reduce the tumor volume. Moreover, the reduction of cell viability confirmed that NDAT could increase the therapeutic effect of paclitaxel. 177 Valetti and colleagues investigated a new ligand, the CKAAKN peptide, conjugated with squalene (SQCKAAKN). They obtained a unique targeted nanoparticle to deliver gemcitabine with remarkable selectivity. 178
Beyond ligand-based active-targeting DDSs, utilization of special materials to form nanoparticles is another new strategy for achieving targeted delivery. Their increased expression of fucosyltransferase implies that PCCs have a crucial requirement for fucose. Therefore, Yoshida and coworkers developed L-fucose-bound nanoparticles containing cisplatin to inhibit tumor growth as well as prolong overall survival in mouse xenograft models. 179
As described previously, hypoxia and acidic pH are significant characteristics of pancreatic cancer. Low pH contributes to chemotherapeutic resistance. However, if we adjust the properties of nanoparticles, low pH can also be used as a breakthrough to increase therapeutic efficacy. Lucero-Acuna and Guzman developed smart nanoparticles with ultrahigh pH sensitivity that switched size instantaneously in the acidic TME of pancreatic cancer. The essential and unique material properties of this nanoparticle improved the penetration of encapsulated antitumor drugs. 180 Colby and colleagues explored the application of expansile nanoparticles (eNPs) to leverage the intraperitoneal route of administration to treat pancreatic cancer. 193 The eNP monomer unit comprises a hydrophobic, pH-responsive protecting group, a hydrophilic triol-linker, and a methyl methacrylate end group that enables polymerization. The diameter of eNPs expanded 2–10 times in water which was triggered by a mildly acidic environment at the tumor site, and drugs loaded in the eNPs could be released. Therefore, eNPs had comparable efficacy and reduced toxicity compared with the standard clinical formulation of paclitaxel, Taxol®. 193
Redox-responsive targeted nanoparticles efficiently deliver their payload because of the fracture of disulfide bonds in the presence of excess intracellular glutathione (GSH). 181 The dual-responsive polymer micelle, which showed ultrasensitive pH response when the nanoparticle accumulates at the tumor site via the EPR effect, and showed a redox response after cellular internalization, could deliver the antitumor agent to the nucleus. Wang and coworkers synthesized the dual-responsive polymer micelle by the typical solvent evaporation method and tested the therapeutic effect both in vitro and in vivo. The dual-responsive nanoparticle ensured high cytotoxicity against the target cells. 182
To observe the distribution of nanoparticles noninvasively, traceable nanoparticles are emerging as a means of diagnosis and treatment. Magnetic iron oxide nanoparticles are one of the most used traceable nanoparticles by magnetic resonance imaging (magnetic resonance imaging), which will be discussed later. Nanoparticles for photothermal therapy are multifunctional drug carriers. Like facile nanoparticles, they are targeted DDSs for chemotherapy. Their tracer ability is triggered by near-infrared irradiation and helps to detect the drug distribution. Meanwhile, photothermal damage in coordination with chemotherapy improves the therapeutic efficacy for malignant tumors with a high mortality rate. For instance, Yu and colleagues designed and synthesized such a nanoparticle compound, termed ‘pheophorbide-a (P@)-Gem-human serum albumin (HSA)-nanoparticles.’ Modified by P@, a second-generation photodynamic therapy agent, P@-Gem-HSA-nanoparticles could be used to track drug delivery because P@ had a high extinction coefficient in the near-infrared region. By combining chemotherapy and PDT, P@-Gem-HSA-nanoparticles had excellent therapeutic effects. 183 Roh and coworkers synthesized cationic photosensitizer-encapsulated polymeric nanoparticles (PS-pNPs), comprised with PEG, PEI, and chlorin e6 (Ce6), a chlorin-based photosensitizer via the carbodiimide reaction. This compound increased the solubility and stability of Ce6 in the aqueous-phase chlorin. Importantly, PS-pNPs broke the limitation of chlorin-based photosensitizers that these agents would be discharged by cells with high expression level of cell membrane transporters. Compared with Ce6, PS-pNPs increased the intracellular levels of the photosensitizer in both AsPC-1 and MIAPaCa-2 cell lines which had high and low cell membrane transporters, respectively, and both had better effects of PDT compared with free Ce6. 184 Zhao and colleagues developed indocyanine green-loaded PLGA–PEG core-shell nanoparticles via single-step nanoprecipitation, which offered a real-time imaging monitor in BxPC-3 xenograft tumor model. Indocyanine green is a dye with substantial absorption and fluorescence in the near-infrared region but is unstable in the living body. PLGA–PEG core-shell nanoparticles prevented indocyanine green from quick degradation and clearance and made it possible to apply indocyanine green in PDT. Near-infrared imaging demonstrated that PLGA–PEG core-shell nanoparticles between 20 nm and 50 nm in diameter were rapidly taken up by tumor cells and significantly suppressed tumor growth. 185
For pancreatic cancer, a highly malignant tumor, monotherapy often fails to achieve the desired therapeutic effect. Current first-line treatment regimens include more than one drug. A DDS should meet the need of codelivery of multiple drugs. To improve the chemosensitivity of pancreatic cancer to gemcitabine, Hung and coworkers developed some multidrug codelivery nanoparticles to chronologically release 3-deazaneplanocin A (DZNep), a histone methylation reversal agent, followed by gemcitabine to improve the chemosensitivity of PCCs to nucleoside analogs. 188 Pancreatic cancer is prone to chemotherapeutic resistance. To solve this problem, Zhao and colleagues developed a cationic ε-polylysine copolymer with a PEGylated lipid bilayer coat by the double-emulsion method. They encapsulated gemcitabine to the hydrophilic core and absorbed an HIF1α short interfering RNA (si-HIF1α) onto the surface of the cationic ε-polylysine copolymer due to the negative charge of si-HIF1α. The PEGylated lipid bi-layered coat maintained the stability of the nanoparticles in the bloodstream. The suppression of HIF1α expression enhanced the antitumor effect of gemcitabine. 189
Other nanoparticles composed of new materials for drug delivery are emerging. A polymeric nanoparticle encapsulated with curcumin (NanoCurc) was engineered by SignPath Pharmaceuticals, Inc. NanoCurc™ consists of N-isopropylacrylamide (NIPAAM), vinylpyrrolidone (VP), and acrylic acid (AA), which have remarkable systemic bioavailability. 191 Teo and colleagues developed a kind of star polymer using reversible addition–fragmentation chain transfer polymerization (RAFT), which contained different lengths of cationic poly(dimethylaminoethyl methacrylate; PDMAEMA) side arms and varied the amounts of poly[oligo(ethylene glycol) methyl ether methacrylate; POEGMA]. The star-POEGMA tended to self-assemble with siRNA to form nanoparticles. Importantly, these star-POEGMA polymers were nontoxic to normal cells. 190 Zeng and coworkers explored a new regimen, a combination of siRNA-based Kras-silencing therapy and arsenic therapy. The siRNAs were carried by poly (ethylene glycol)-block-poly (L-lysine) nanoparticles and arsenic was carried by poly(ethylene glycol)-block-poly(DL-lactide) nanoparticles. They demonstrated that co-administration of these two kinds of nanoparticles was a new potential therapeutic regimen to treat pancreatic cancer. 194
The combination of organic and inorganic composite nanoparticles
Metal nanoparticles have been applied in many fields, especially to overcome medicinal problems. 195 Decorating metal nanoparticles with a polymer coating diversifies their functions and widens their applications in the medical field. Biopolymer coating improves the colloidal stability, biocompatibility, and systemic circulation time of the nanoparticles. 196 Furthermore, with the help of functional coatings, these advanced metal nanoparticles can actively target tumor cells or release drugs in response to specific stimuli, which are hard to achieve using pure metal nanoparticles alone. The nanoparticles comprising organic and inorganic components were named as a metal organic framework (MOF) by some scholars. 127 Nanoparticles consisting of polydentate bridging ligands and metal ions or clusters are named nanoscale coordination polymers (NCPs). 197 Commonly used metal materials for nanoparticles are superparamagnetic iron oxide, gold, silver, and zinc. The combination of organic and inorganic composite nanoparticles as carriers for drugs to treat pancreatic cancer is summarized in Table 2.
The combination of organic and inorganic composite nanoparticles as a carrier for treatment of pancreatic cancer.
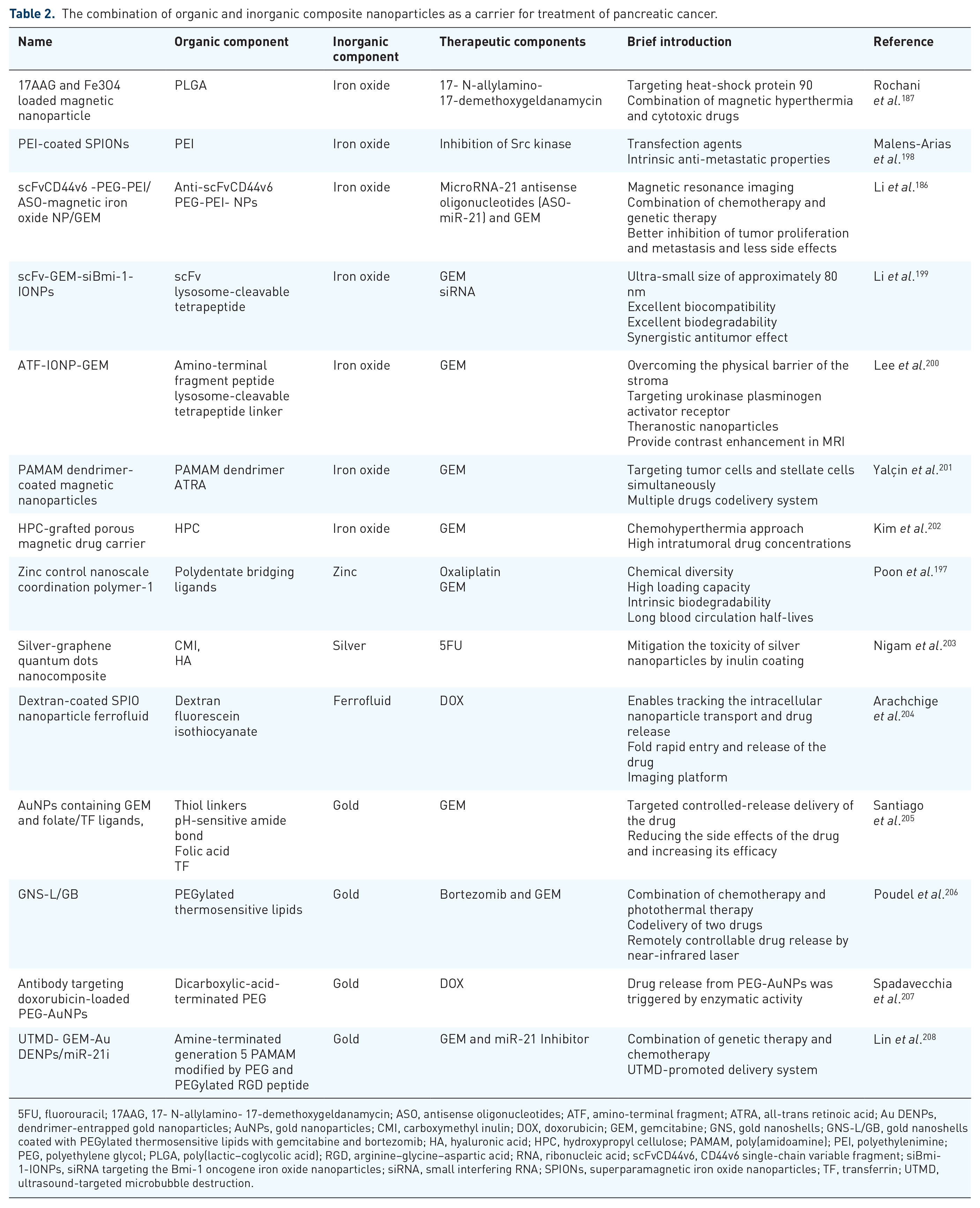
5FU, fluorouracil; 17AAG, 17- N-allylamino- 17-demethoxygeldanamycin; ASO, antisense oligonucleotides; ATF, amino-terminal fragment; ATRA, all-trans retinoic acid; Au DENPs, dendrimer-entrapped gold nanoparticles; AuNPs, gold nanoparticles; CMI, carboxymethyl inulin; DOX, doxorubicin; GEM, gemcitabine; GNS, gold nanoshells; GNS-L/GB, gold nanoshells coated with PEGylated thermosensitive lipids with gemcitabine and bortezomib; HA, hyaluronic acid; HPC, hydroxypropyl cellulose; PAMAM, poly(amidoamine); PEI, polyethylenimine; PEG, polyethylene glycol; PLGA, poly(lactic–coglycolic acid); RGD, arginine–glycine–aspartic acid; RNA, ribonucleic acid; scFvCD44v6, CD44v6 single-chain variable fragment; siBmi-1-IONPs, siRNA targeting the Bmi-1 oncogene iron oxide nanoparticles; siRNA, small interfering RNA; SPIONs, superparamagnetic iron oxide nanoparticles; TF, transferrin; UTMD, ultrasound-targeted microbubble destruction.
Superparamagnetic iron oxide nanoparticles (SPIONs) were used initially as contrast agents in MRI. With the help of polymer coatings, SPIONs could be used not only in diagnosis, but also in image-guided therapies. 209 Magnetic hyperthermia synergy with antitumor drugs increased the therapeutic effect on pancreatic cancer.187,198 Besides, polylactic acid (PLA), PEG, dextran and chitosan, could also act as the polymer organic components of advanced SPIONs. 209 Another two research teams conjugated iron oxide nanoparticles with single-chain variable fragment (scFv), which provided the nanoparticles with an active-targeting ability to deliver chemotherapy drugs and therapeutic genes. Chemotherapy drugs (gemcitabine) were linked through a lysosome-cleavable tetrapeptide linker. Their scFv-Gem-siRNA-nanoparticles displayed a great targeting effect, few side effects, and excellent inhibition of tumor proliferation and metastasis.186,199
Polymer-coated gold nanoparticles (P-AuNPs) have attracted a lot of attention in recent years. Gold nanoparticles (AuNPs) can be exploited for photothermal therapy because of their ability to convert absorbed light into heat. Meanwhile, their polymer coating, with special ligands or chemotherapeutic components, has the ability to assist nanoparticles in reaching their therapeutic sites and increases therapeutic effect. 210 Considering the low pH value in the pancreatic TME, Santiago and colleagues immobilized gemcitabine on AuNPs using a pH-sensitive amide bond. This kind of amide bond prevents gemcitabine from being metabolized under normal physiological conditions but breaks via the reduced pH in the TME. 205 Poudel and coworkers modified plasmonic hollow gold nanoshells (GNS) with PEGylated thermosensitive lipid coating to deliver gemcitabine and bortezomib. Bortezomib loaded in lipid bilayers was used to induce photothermal therapy, while gemcitabine loaded in the hydrophilic interior of the plasmonic hollow GNS was used for chemotherapy. These nanoparticles achieved remotely controllable drug release in the presence of a near-infrared laser. The authors demonstrated that the combination of photothermal therapy and chemotherapy was promising, even in resistant pancreatic cancer, using in vitro experiments. 206 Spadavecchia and colleagues synthesized dicarboxylic acid-terminated PEG-AuNPs to deliver doxorubicin. For more precise treatment, they further modified the advanced AuNPs with an anti-Kv11.1 polyclonal antibody to increase tumor targeting and cell internalization. The authors demonstrated that the application of nanomedicines to treat pancreatic cancer has considerable potential (Figure 4). 207 Lin and coworkers reported dendrimer-entrapped gold nanoparticles (Au DENPs) that formed an ultrasound-targeted microbubble destruction (UTMD) DDS to carry gemcitabine and an miR-21 inhibitor. Experiments in vitro and in vivo attested that this smart DDS might be a novel strategy for treating pancreatic cancer. 208

Schematic of potential cell internalization routes of anti-Kv11.1 polyclonal antibody-modified dicarboxylic-acid-terminated PEG-AuNPs. 207
Discussion
The TME of pancreatic cancer is complex, and the details of the interactions of different stromal components are unclear. As a distinct feature, the dense stroma of the pancreas represents a promising treatment target. The dense stroma plays an important role in chemoresistance. Stroma-depleting therapy, assisted by cytotoxic drugs targeting PCCs, worked well in improving chemotherapeutic efficiency. For example, the combination of gemcitabine and Nab-paclitaxel increased the 1.8 month median survival compared with that of gemcitabine monotherapy in the recent phase III multinational MPACT trial. 159 However, a complete stroma elimination not only reduces intercellular pressure, but also provides growth space for PCCs. Several studies in murine PDAC models indicated that the complete stroma elimination decreased overall survival. 102 Acidic pH and hypoxic conditions are also notable features of pancreatic cancer. The considerable changes in oxygen levels and blood contribute to invasiveness, metastasis, and chemotherapy resistance. Meanwhile, hypoxia facilitates EMT and immunosuppression. HIF-1 plays an important role in these processes through a variety of signaling pathways. In addition, antiangiogenesis therapy has yielded encouraging results in many tumors. Although pancreatic cancer tissues overexpress angiogenic factors, many clinical trials aimed at antiangiogenesis have failed. Interestingly, acidic pH and hypoxic conditions are used for targeted drug delivery in nanomedicine. Facile nanoparticles accumulate at the tumor site, mainly through the EPR effect. With their special design, nanoparticles are sensitive to low pH values. Adverse conditions can be transformed to treatment-friendly features via nanotechnology innovations.
The advent of smarter nanoparticles increased the likelihood of developing a treatment for pancreatic cancer. Nanoparticle DDSs help to overcome the shortcomings of traditional chemotherapeutic drugs. Furthermore, using antibody-based ligands, such as pH-sensitive chemical bonds, redox-responsive disulfide bonds, magnetic iron oxides, or other metals, nanoparticles can achieve targeted, controlled, and detectable drug delivery. At the same time, nanoparticles also facilitate the cotransportation of a variety of drugs. To date, gemcitabine-based combination treatments and FOLFIRINOX have been considered as the first-line chemotherapy for patients with locally advanced pancreatic cancer or metastatic pancreatic cancer. 159 Kim and colleagues compared the efficacy and safety of these two common first-line therapies via a retrospective cohort study that reviewed 654 medical records. Patients who received NAB-P + GEM chemotherapy had similar overall survival to the patients treated with FOLFIRINOX; however, a lower rate of side effects, such as diarrhea, fatigue, mucositis, nausea, and vomiting, was reported for the NAB-P + GEM treated group. 211 Gharaibeh and coworkers evaluated economic efficiency and found that NAB-P + GEM had an economic benefit over FOLFIRINOX. 212 NAB-P + GEM therapy might be the most suitable regimen to treat pancreatic cancer. 157 In addition to these two first-line therapies, other combination therapies have also been studied extensively in preclinical trials. Gene therapy, working in coordination with chemotherapeutic drugs is a potential novel treatment strategy. Nanoparticles stabilize free nucleic acids in circulation and decrease their recognition by the immune system. 213 Photosensitizer and chemotherapeutic drugs encapsulated within polymeric nanoparticles can improve the therapeutic efficiency. These nanoparticles reduce the volume of tumors by the combined effect of photothermal damage and cytotoxic drugs. Moreover, they are visible under near-infrared irradiation.
Although these novel nanoparticles are very promising for better diagnosis and therapy, few of them have entered clinical applications. 214 The big gap between exciting experimental data and a clinical application effect is partly caused by the imperfect models used in preclinical trials. To better understand the relationship between the TME and PCCs, animal models that can better mimic the human pancreatic TME need to be established. Some models, with advantages and disadvantages, are already in use. In one model, to stimulate a desmoplastic reaction in pancreatic cancer, researchers tried to cotransplant fibroblasts and tumor cells. Patient-derived xenograft models also partly address this problem. For xenograft models, the absence of the immune system is the most troublesome problem. GEMMs successfully avoid the problem of immune deficiency. However, the species differences and expenses plague researchers. The lack of genetic diversity and the larger tumor-to-body-weight ratio in mice models are also key differences. 133 As a result, further studies are needed to facilitate the clinical application of specific modified nanoparticles in the treatment of pancreatic cancer.
Conclusion and perspectives
Pancreatic cancer is a highly malignant tumor with a special TME that plays an important role in therapeutic resistance. However, tumor models to study the TME are not sufficiently mature. Therefore, there is still a need to build a perfect tumor model that can be used widely and simulates the special TME. To overcome the limitations of traditional therapeutic drugs, a large number of DDSs using nanomaterials have been studied. Nanoparticles have the natural advantage of easy penetration into the tumor site through immature tumor vessels. The diameter of nanoparticles is one of their most important properties. To date, the size of nanoparticles has been kept highly uniform and stable through a new technology termed flash nanocomplexation (FNC). 215 However, compared with inorganic nanoparticles, especially metal nanoparticles, the diameter of organic nanoparticles is relatively large. Organic nanoparticles with smaller diameters may be worthy of further investigation. Smart nanoparticles with exquisite designs have enhanced the ability to target tumors and controlled-release drug at the proper site. However, the more complex the structures and compositions of nanoparticles, the more difficult it is to study their detailed action in vivo. Large amounts of data indicate that smart nanoparticles work well in the short-term treatment of various diseases. However, the efficiency of many smart nanoparticles has only been demonstrated using in vitro experiments and most of the in vivo experiments only proved short-term therapeutic efficacy. The overall survival of patients with advanced pancreatic cancer is short; however, for patients with curable diseases, research into the safety and effectiveness of long-term mass application of nanoparticles, especially nanoparticles with complex structures and compositions, still requires a lot of effort. For instance, for frequent injections of ligand-based nanoparticles, the following aspects should be clarified: how long do they retain their ability to target the tumor site? How long do the nanomaterials take to degrade completely in vivo (especially in the presence of cachexia)? How to detect the content of undesirable harmful solvents in the production of nanoparticles, and what level of harmful solvents should be controlled.
There is still a long way to go before nanoparticles can meet clinical requirements. The ideal polymer nanoparticles should have the following basic conditions: (a) cause no harm to health; (b) the degradation time of nanomaterials should be consistent with the drug delivery frequency; (c) the nanoparticles should have good long-term storage and retain their physical and chemical properties; (d) they should show an ideal therapeutic effect with minimal side effects. The progress of nanotechnology is encouraging, and we look forward to innovations in nanoparticles for the treatment of refractory diseases such as pancreatic cancer.
Footnotes
Funding
This study was financially supported by the Financial Department of Jilin Province (Grant No. 2019SCZT045).
Conflict of interest statement
The authors declare that there is no conflict of interest.
