Abstract
Background:
The aim of this study was to prospectively analyse, for the first time worldwide by
Methods:
Each enrolled patient underwent full ophthalmologic examination including
Results:
A total of 10 patients were consecutively enrolled. Median follow-up was 5 months. No Common Terminology Criteria for Adverse Events Version 4.0 grade 4 toxicity was documented. Two (20%) grade 3 toxicities were documented at week 8. CCM examination detected in all eyes multiple and diffuse hyperreflective white round spots in the corneal basal epithelial layers (100%), progressive subbasal nerve plexus layer fibres fragmentation followed by full disappearance (100%) and appearance of round cystic structures in the corneal epithelium (100%). All CCM documented side effects reached the peak of prevalence and severity after a median of 3 infusions. After treatment discontinuation, the reversibility of corneal side effects was documented at CCM after a median of 4 weeks.
Conclusion:
ABT-414 toxicity is not only directed to the corneal epithelium, but also to corneal nerves. Side effects are detectable in all treated patients and CCM documents early corneal epithelium and subbasal nerve plexus toxicity, with subsequent progressive restoration after treatment discontinuation. Ocular side effects due to ABT-414 can be manageable.
Keywords
Introduction
Epidermal growth factor receptor (EGFR) is commonly over expressed, amplified or mutated in many human cancer cells. 1 Therefore, EGFR-inhibitors have been introduced for the treatment of different solid tumors.1,2 Monoclonal antibodies against EGFR have been proven to improve progression-free survival and overall survival of patients affected by tumours with EGFR overexpression, while preserving a reasonable quality of life for the patient. 2 Antibody-drug conjugates (ADC) are a promising class of anticancer drugs characterized by the targeting properties of monoclonal antibodies and the antitumour effects of cytotoxic agents. Depatuxizumab mafodotin (formerly ABT-414) is an ADC consisting in a monoclonal antibody (depatuxizumab) binding a specific epitope of the EGFR and a chemotherapeutic agent, monomethyl-auristatin-F, a strong antimitotic agent that inhibits cell division by blocking the polymerization of tubulin.3,4
Glioblastoma is the most common primary central nervous system malignant tumour in adults, with a 5-year survival rate around 5%.1,5,6 Frequent abnormalities have been observed in the expression of the EGFR in glioblastoma, and approximately 50% of glioblastomas harbour EGFR gene amplification, with consequent overexpression of the EGFR protein.1,7 Therefore, ABT-414 is considered a promising drug for patients affected by this tumor.
1
In the eye, EGFR is constitutively localized in the basal epithelial cells of the cornea and conjunctiva and corneal toxicity has been previously reported in patients treated by monoclonal antibodies against EGFR.1,8–13 Unfortunately, the analysis of ocular side effects secondary to ABT-414 was performed using only standard ophthalmological procedures, mainly slit lamp examination with fluorescein corneal staining. Therefore, morphological analysis, at microscopic level, of corneal and ocular surface side effects related to the use of ABT-414 was never performed
The aim of this study was to prospectively analyse, for the first time worldwide, corneal side effects by clinical confocal microscopy (CCM) in a cohort of patients affected by glioblastoma and treated with the EGFR-inhibitor ABT-414 as compassionate use in combination with temozolomide.
Materials and methods
This was an institutional, observational, descriptive cohort study with prospective enrolment, compliant with the tenets of the Declaration of Helsinki and approved by the Veneto Institute of Oncology IOV-IRCCS institutional review board (No. 08.2019). Patients treated with ABT-414 plus temozolomide for EGFR-amplified, recurrent glioblastoma, were consecutively recruited. Informed consent covering patient inclusion in the present study was obtained from each patient. Treatment with ABT-414 (1.5 mg/kg every 2 weeks) plus temozolomide 150–200 mg/m2/day for five consecutive days every 28 days was continued until either intolerable toxicity or disease progression was reached, as assessed using the response assessment in neuro-oncology criteria.1,14 Brain magnetic resonance imaging was performed at baseline, every 2 months or when clinically indicated. Patients were treated at Veneto Institute of Oncology (Padua, Italy) and started the treatment from November 2018 to April 2019, as compassionate use. Each enrolled patient underwent full ophthalmologic examination, including best corrected visual acuity (BCVA) using the Early Treatment Diabetic Retinopathy Study (ETDRS) protocol, and slit lamp examination with fluorescein corneal staining.
15
Visual acuity reduction of at least five letters was defined as clinically significant.
Results
Population, treatment and baseline ophthalmologic characteristics
A total of 10 patients affected by EGFR-amplified, recurrent glioblastoma and treated with ABT-414 were consecutively recruited. Patient characteristics are reported in Table 1.
Clinical and demographic characteristics of enrolled patients.

ABT-414, depatuxizumab mafodotin; CI, confidence interval; SD, standard deviation.
At the time of our analysis, median follow up time was 5 months [95% confidence interval (CI) 3.2–6.7]. The median treatment time was 2 months (95% CI 1.5–2.5). The median number of ABT-414 infusion was 4 (range 2–6). No patient discontinued the treatment because of ocular side effects. Two patients (20%) reduced ABT dose to 1 mg/kg due to grade 3 ocular side effects. In total, six patients (60%) were still in treatment 2 months after starting the therapy. Six patients were followed at least 1 month after treatment discontinuation and three patients up to 2 months. The mean BCVA at baseline was 86 ± 3 letters. No significant corneal or anterior segment clinically relevant abnormalities were documented at baseline (by biomicroscopy and CCM), excluding cortical cataract in two patients (3 eyes) (Table 2).
Clinical and clinical laser scanning

ETDRS, Early Treatment Diabetic Retinopathy Study; NA, not applicable; N, number; SD, standard deviation.
Corneal and ocular surface side effects
The prevalence and severity (median and maximal severity) of ocular side effects in our cohort of patients is reported in Table 2. No CTCAE grade 4 toxicities were documented. Two grade 3 toxicities (20%) were documented at week 8. Symptoms were experienced by all patients starting at 10 ± 3 days after the first drug infusion and included: blurred vision (eight patients), eye pain (nine patients) and photophobia (five patients) (Table 2). At the first follow-up examination, the mean BCVA was 84 ± 4 letters and no patient experienced more than five ETDRS letter losses from baseline. Slit lamp examination with fluorescein corneal staining examination documented the appearance of mild conjunctival hyperaemia (10 eyes), mild blepharitis (12 eyes), superficial punctate epitheliopathy (8 eyes) (Figure 1) and multiple corneal epithelial cysts (10 eyes) (Figure 2). When present, superficial punctate epitheliopathy was graded as moderate in four cases and severe in another four cases. The corneal epithelial cysts were characterized by round shape and optically empty content, with preferential distribution in the central cornea (Figure 2). CCM examination revealed the presence of multiple and diffuse epithelial hyperreflective white round spots in all examined eyes, mainly distributed in the basal epithelial layers (Figure 3). The presence of these spots was graded as mild in 8 eyes, moderate in 10 eyes and severe in 2 eyes. The subbasal nerve plexus layer was characterized by evident fibre fragmentations and a reduction in the number of fibres in all eyes, mainly when compared with baseline images (Figure 4). The documentation of the round epithelial cystic structures, characterized by a hyperreflective and well-defined wall, was documented in 12 eyes and graded as mild in 9 and moderate in 3 eyes. An increase in ocular symptoms and signs was documented in the first 2 months, with a subsequent stabilization over time during the treatment period (Table 2). The superficial punctate epitheliopathy increased in incidence, affecting 16 eyes at 4 weeks and 20 eyes at 8 weeks, and became severe in 6 eyes at 4 weeks and in 12 eyes at 6 weeks. A single eye presented two severe episodes of corneal abrasion-like epithelial defect (corneal ulceration) with positive fluorescein staining associated with mild anterior chamber inflammation. This side effect regressed using topical antibiotics and steroids. Corneal stroma oedema was documented in two eyes at a 4-week examination and its incidence increased at 4 weeks (nine eyes). Clinically significant BCVA reduction started at 4 weeks (68 ± 9 ETDRS score), with stabilization at 8 weeks (64 ± 11 ETDRS score). By CCM, the evidence of multiple and diffuse epithelial hyperreflective white round spots, documented in all patients since week 2, persisted during the treatment and increased in severity, becoming severe in 7 eyes at 4 weeks and in 11 eyes at 6 weeks. Conversely, the presence of the round cystic structures at CCM were documented in all patients at week 4, persisting during the treatment (Table 2). Round cystic structures also increased in severity, becoming severe in five eyes at 4 weeks and in nine eyes at 6 weeks. The subbasal nerve plexus layer, characterized by fibre fragmentation and reduction at week 2, was then characterized by a complete disappearance of the plexus fibres in 14 eyes (70%) at week 4 and in 20 eyes (100%) at week 8. Keratocytes activation in the corneal stroma was also documented in two eyes at a 4-week examination and its incidence increased at 4 weeks (nine eyes). All documented side effects reached the peak of prevalence and severity in a range of 4–8 weeks (2–4 ABT 414 infusions; median 3 infusions) from starting the treatment, maintaining the same prevalence and severity during the subsequent treatment period.
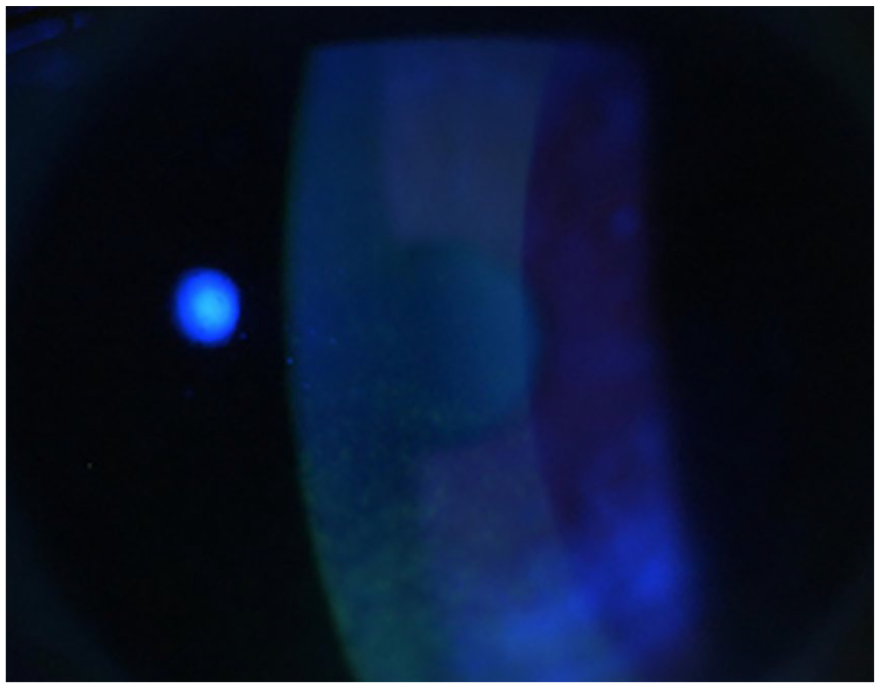
Standard slit lamp examination with fluorescein staining in a patient affected by superficial punctate epitheliopathy secondary to ABT-414 systemic treatment.

Standard slit lamp examination in a patient showing the presence of diffuse intraepithelial cysts secondary to ABT-414 systemic treatment. Please note the round shape and an optically empty content.

Clinical

Clinical
Effect of treatment discontinuation
In the three patients followed for at least 2 months after treatment discontinuation, corneal side effects regressed progressively. After 8 weeks post treatment discontinuation, corneal side effects were almost completely resolved, with partial subbasal nerve plexus restoration and the disappearance of most epithelial cysts (Figures 3 and 4). Punctate keratopathy also became mild, progressively reducing in severity. All these patients reached the baseline ETDRS score. In six patients, followed for at least 1 month after treatment discontinuation, there was an evident trend toward reversibility of ocular side effects.
Discussion
Epidermal grow factor (EGF) is a small polypeptide characterized by a potent stimulatory effect on epidermal cell proliferation and differentiation. 8 It plays a key role in the maintenance of ocular surface homeostasis. 8 In physiological situations, EGF binds the EGFR, which is localized in the basal cell layer of the conjunctiva and cornea, mainly in peripheral and limbal epithelial corneal cells.8,18 ABT-414 is a promising new drug, and a recent randomized comparative phase II trial (INTELLANCE 2) showed that ABT-414, in combination with temozolomide, could improve survival in recurrent glioblastoma patients compared with the standard treatment (temozolomide alone or lomustine). 19 This drug binds an epitope present only when EGFR is in an extended (activated) conformation.1,7,10,11 Therefore, it seems to be associated with less systemic adverse events, avoiding toxicity typically associated with other systemic EGFR drugs inhibitors. 1 Other authors have reported ocular adverse events during systemic therapy with EGFR inhibitors. Ahn and colleagues reported corneal toxicity secondary to systemic vandetanib, an inhibitor of both EGFR and VEGFR2, clinically describing using the standard slit lamp examination with fluorescein corneal staining, the development of vortex keratopathy and diffuse subepithelial haze in both corneas of one treated patient. 13 In addition, Tullo and colleagues, using the same standard approach, reported the development of mild conjunctival hyperaemia associated with mild and reversible superficial punctate keratopathy, blepharitis, trichiasis, keratoconjunctivitis sicca and meibomitis during treatment with Gefitinib, an EGFR tyrosine kinase inhibitor. 12 Other authors reported ocular toxicity specifically due to ABT-414, mainly analysing ocular side effects from a medical oncologist’s point of view, reporting the common appearance of dry eyes, blurred vision, eye pain and photophobia, secondary to microcystic corneal degeneration.1,9 –11 These authors reported that most patients experienced at least one ocular adverse effect, including blurred vision (65%) and dry eye (29%). 1 A recent study analysed the safety and efficacy of ABT + temozolomide in 60 recurrent glioblastoma patients with EGFR amplified; the authors showed that 87% of patients reported any grade of ocular adverse events. Similarly to our results, 22% of cases reported severe (grade 3–4) adverse events. Among these, the most frequent severe adverse events were keratitis (22%), blurred vision (5%) and photophobia (3%). Unfortunately, these side effects were not fully characterized, and just one case report (one patient) was described by the standard ophthalmological point of view. 19
Confocal microscopy was introduced in the ophthalmological clinical practice as a noninvasive tool to observe at high magnification the structure of the human cornea and conjunctiva
Our data shows, for the first time, that corneal damage by ABT-414 is not limited to the corneal epithelium, but also affects the corneal nerves and anterior corneal stroma. We hypothesize that the corneal subbasal nerve plexus involvement is explained by its location immediately under the basal epithelial membrane. Indeed, in physiological conditions EGFR is constitutively localized in the corneal basal epithelial cells, and ABT-414 binding to it could deliver its direct cytotoxic effect into the corneal basal epithelial layer, in the subbasal nerve plexus and also in the anterior stroma. 8 Moreover, an ADC could hypothetically contribute to ocular toxicity not only through an on-target mechanism (antibody-mediated homing and delivery), but also by an off-target mechanism (uptake of the unconjugated cytotoxin by tissue having a specific tropism for it). Therefore, a second pathogenetic hypothesis is the possible existence of a direct uptake of ABT-414 into rapid proliferating cells of the corneal limbus (corneal stem cells) that differentiate and give rise to the corneal epithelium, moving from the limbus toward the central cornea.20 –22 This migration may also explain the preferential location of the epithelial cysts in the central cornea. Eaton and colleagues suggest that this intracellular uptake can be simply related to the general mechanisms of endocytosis in cells characterized by high replication and differentiation rate, rather than specific targeting of ABT-414 to activated EGFR in the cornea. 21 Although this mechanism is suggested by preclinical observations reporting that corneal side effects are present regardless of the antibody epitope of the ADC, this second hypothesis may also suggest that human stem cells, in tissues characterized by high replication and differentiation rate, may be affected by similar toxicity. 21 Conversely, nonocular side effects are less common and severe, 22 suggesting that corneal homing may be present. The most frequently reported nonocular side effect is fatigue sensation (41%). 22 Considering that our CCM analysis demonstrated that corneal innervation is directly affected by the ABT-414 toxicity, we cannot exclude a similar mechanism of toxicity at the level of the neuromuscular junctions or other peripheral nervous structures responsible for this side effect.
A third pathogenetic hypothesis of corneal ABT-414 toxicity is that the antibody-drug conjugate may be taken up from the blood flow by the lacrimal gland and secreted with tears, delivering the drug to the cornea.20 –22
Our study confirms that ocular side effects can improve upon treatment discontinuation. However, a precise definition of a median time to resolution was not established because of the limited number of patients followed after treatment discontinuation. This limitation of the study is related to the patient’s systemic conditions: most of our patients discontinued follow-up for progressive disease. A second limitation of this study is the relative low number of patients enrolled. However, this is the first study on ABT-414 ocular toxicity analysed
In conclusion, this is the first study analysing ocular side effects by CCM in patients with recurrent glioblastoma treated with ABT-414. ABT-414 toxicity is not only directed to the corneal epithelium, but also to the subbasal nerve plexus. Corneal side effects are detectable in all treated patients, and all documented side effects reached the peak of prevalence and severity after a range of 1–2 months (2–4 ABT-414 infusions; median 3 infusion) from starting the treatment, maintaining the same prevalence and severity during the subsequent treatment period. After treatment discontinuation there is a significant evident trend toward reversibility, and CCM documented a progressive corneal epithelium and subbasal nerve plexus regeneration. The added value of using CCM when quantifying corneal side effects of affected patients is proven by our data. However, these data, obtained
Footnotes
Acknowledgements
RP, GL, LF and EM contributed to the study conceptualization, design, interpretation of data, drafting and revising and agreed on the final approval and to be accountable for all aspects of the work.
FL, DL, MP, MC, GM, SB and VZ contributed to data acquisition/analysis/interpretation, drafting and revising of work, formal analysis, final approval and agree to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The contribution of the Fondazione Bietti in this paper was supported by the Ministry of Health and Fondazione Roma.
Conflict of interest statement
The authors declare that there is no conflict of interest.
