Abstract
Background:
Cetuximab (CTX) has been approved to be administered concurrently with radiotherapy (RT) to treat locally advanced head and neck squamous cell carcinoma (HNSCC). The aim of this study was to assess the efficacy and safety of concurrent CTX with RT (ExRT).
Method:
The
Results:
In total, 32 articles with 4556 patients were included. The pooled HRs indicated that ExRT achieved an unfavorable overall survival (HR: 1.86,
Conclusion:
ChRT should remain the standard treatment for locally advanced HNSCC patients. ExRT was recognized as an effective alternative treatment for locally advanced HNSCC patients who experienced unbearable toxicities caused by non-CTX treatments.
Introduction
Head and neck cancer encompasses malignancies of the oral cavity, oropharynx, hypopharynx, and larynx, which remains a significant cause of morbidity and mortality and ranks the seventh most common cancer worldwide in 2018. 1 Over 90% patients are classified as head and neck squamous cell carcinoma (HNSCC), and the landscape of HNSCC treatment has evolved over the past decade. Approximately 30–40% of HNSCC patients present with early stage (stage I/II), which is commonly curable with surgery alone or in combination with radiotherapy (RT). 2 More than 60% of HNSCC patients are diagnosed with locally advanced stage (stage III/IV), which has a high risk of local recurrence with a poor prognosis.3,4 For locally advanced stage HNSCC, the established standard treatment is concurrent platinum-based chemotherapy with RT (ChRT), which further improves the survival rate and life quality of patients than RT alone.5,6 However, the usage of platinum-based chemotherapy especially with high-dose cisplatin could be accompanied by severe acute toxicities such as nephrotoxicity, hematologic toxicity, and nausea/vomiting during the early stage, and late toxicities in the long term such as severe difficulty in swallowing or feeding tube dependency. 7 Therefore, there is an urgent need for discovering new treatments to further improve the prognosis and tolerance for locally advanced HNSCC.
Epidermal growth factor receptor (EGFR), a 170-kDa polypeptide, belongs to a member of the Her family of tyrosine kinase receptors and regulates critical cell functions such as proliferation, migration, differentiation, and survival of cancer cells. 8 As a biological marker abnormally elevated in some epithelial malignancies such as esophagus cancer, colorectal cancer, and non-small cell lung cancer, EGFR emerges as an essential target for new therapeutic strategies.9,10 Cetuximab (CTX) is a monoclonal antibody derived from the murine antibody M225, and it specifically binds to EGFR, then inhibits its downstream signaling pathway. The potential mechanisms of CTX used in malignancies involve inhibiting cell cycle at G1 phase and enhancing radiation-induced apoptosis. 9 Currently, CTX has been approved by the United States Food and Drug Administration (FDA) to improve patient outcomes in many cancers, such as metastatic colorectal cancer. 11
In HNSCC, EGFR expression is confirmed to be considerably increased, compared with the adjacent normal tissue, suggesting EGFR as a potentially important target.
12
In 2006, Bonner
Because of lacking adequate rationale for the superiority of ExRT, the number of studies comparing ExRT with ChRT in locally advanced HNSCC has been growing, but different conclusions have been drawn, especially comparing ExRT with ChRT. By assessing the efficacy outcomes, some trials approved the superiority of ChRT compared with ExRT in HNSCC. Koutcher
Therefore, it is necessary to conduct an updated systematic review by retrieving more recent publications to clarify the pooled effects of ExRT for locally advanced HNSCC. In the present study, we summarize published data to evaluate the therapeutic effects and safety of ExRT in contrast to ChRT.
Materials and methods
Study identification and searching strategy
This study complies with the Preferred Reporting Item for Systematic Reviews and Meta-Analyses guidelines.
21
The
Inclusion and exclusion criteria
The following inclusion criteria were adopted based on the following PICOS criteria: (P) Patients: patients with locally advanced stage III/IV HNSCC. (I) Intervention: patients received ExRT. (C) Comparator: patients receiving ChRT or RT (only for analyzing the safety of ExRT). (O) Outcomes: the existing literature provided with sufficient data for estimating the following outcomes: OS, DSS, LRC, PFS, metastasis-free survival (MFS), and grade 3/4/5 toxicities. (S) Study design: RCT, prospective cohort study, retrospective cohort study.
The following studies were excluded: (1) non-human study; (2) the essential information was not sufficiently complete to extract the data for meta-analysis; (3) review, letter, and case report. The flow chart of searches is shown in Figure 1.
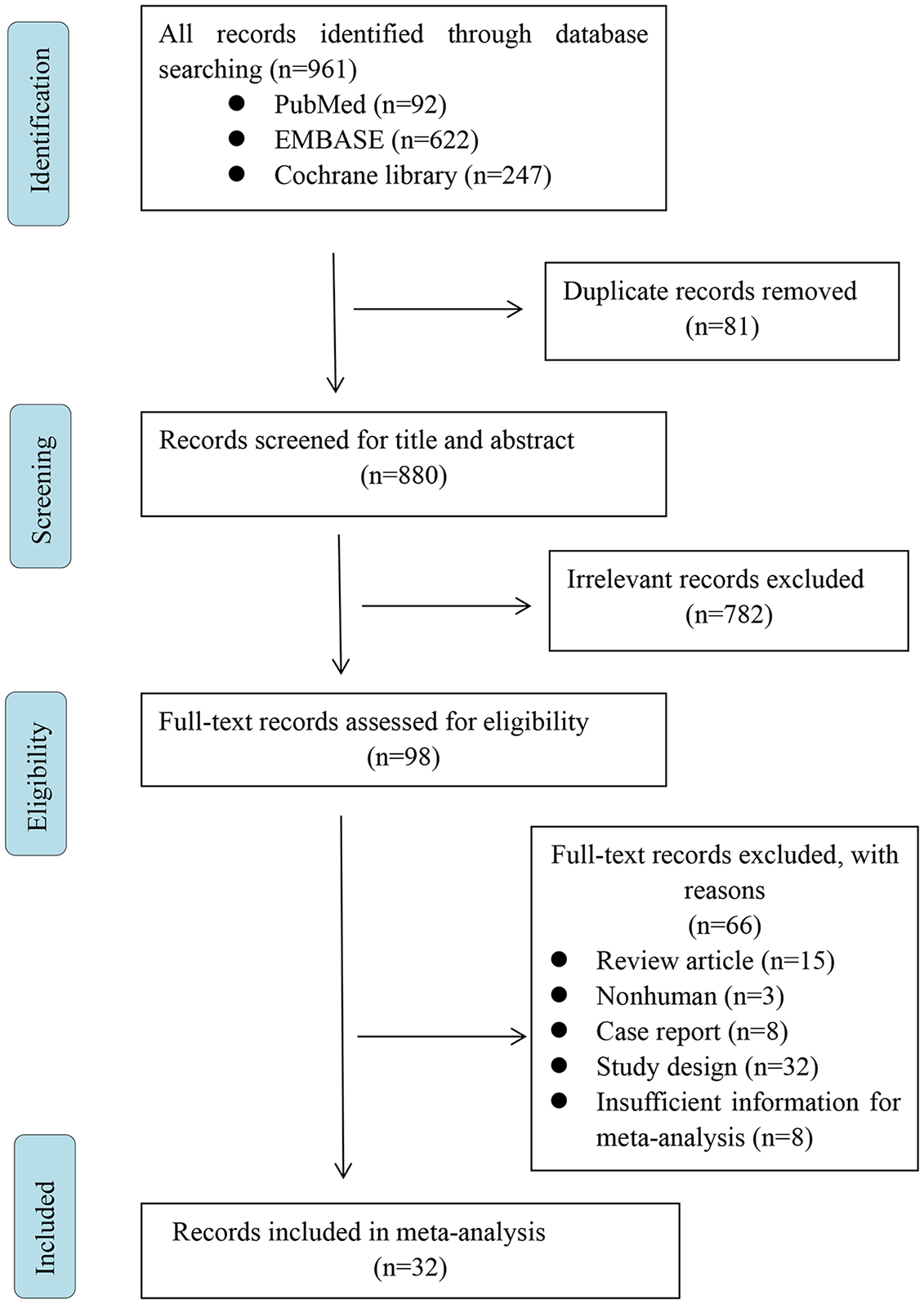
Flowchart of study selection.
Data extraction and analysis
Two authors used pre-designed tables to extract data from the studies, which included the following items: first author, publication year, the population (country), study design, age, gender, grouping, the total number of patients with HNSCC in ExRT group and control group, follow-up years, OS [hazard ratio (HR), 95% confidence interval (CI)], DSS (HR, 95% CI), LRC (HR, 95% CI), PFS (HR, 95% CI), MFS (HR, 95% CI) as well as a range of grade 3/4/5 toxicities [risk ratio (RR), 95% CI]. Any divergences between authors concerning the eligibility of a study were resolved by consulting a third author or by consensus.
Statistical methods
Most studies provided log-rank curves of OS, DSS, LRC, PFS, and MFS, but did not directly offer HR data. We calculated HR by using log-rank curve point-taking method. For OS, we also combined the results of multivariate Cox regression analysis with HR to analyze the risk factors for it. The RR of adverse events from included studies was analyzed between groups. All extracted data were entered into RevMan 5.3 (Cochrane Collaboration’s Information Management System) for statistical analysis. The heterogeneity test between studies was evaluated by
Quality assessment
We used the Newcastle–Ottawa Scale (NOS) for assessing the quality of prospective cohort studies. NOS is suitable for evaluating case–control studies and cohort studies. It evaluates cohort studies and case–control studies with three blocks and eight items, including population selection, comparability, exposure evaluation or outcome evaluation. All investigators independently assessed the quality and bias of the study. The differences were resolved by consensus or by arbitration by the third investigator.
Assessment of risk of bias
Publication bias was detected by funnel plots, and Begg’s and Egger’s tests using Stata software version 14.0, with a
Results
Eligible studies and description of studies
As seen in Figure 1, at the initial search stage, a total of 961 articles were identified from three databases. Following screening of the titles and abstracts, we excluded duplicates and irrelevant articles, leaving 98 articles. Later, through reading the full text, 66 articles were excluded with reasons: review article (
Characteristics of included studies.
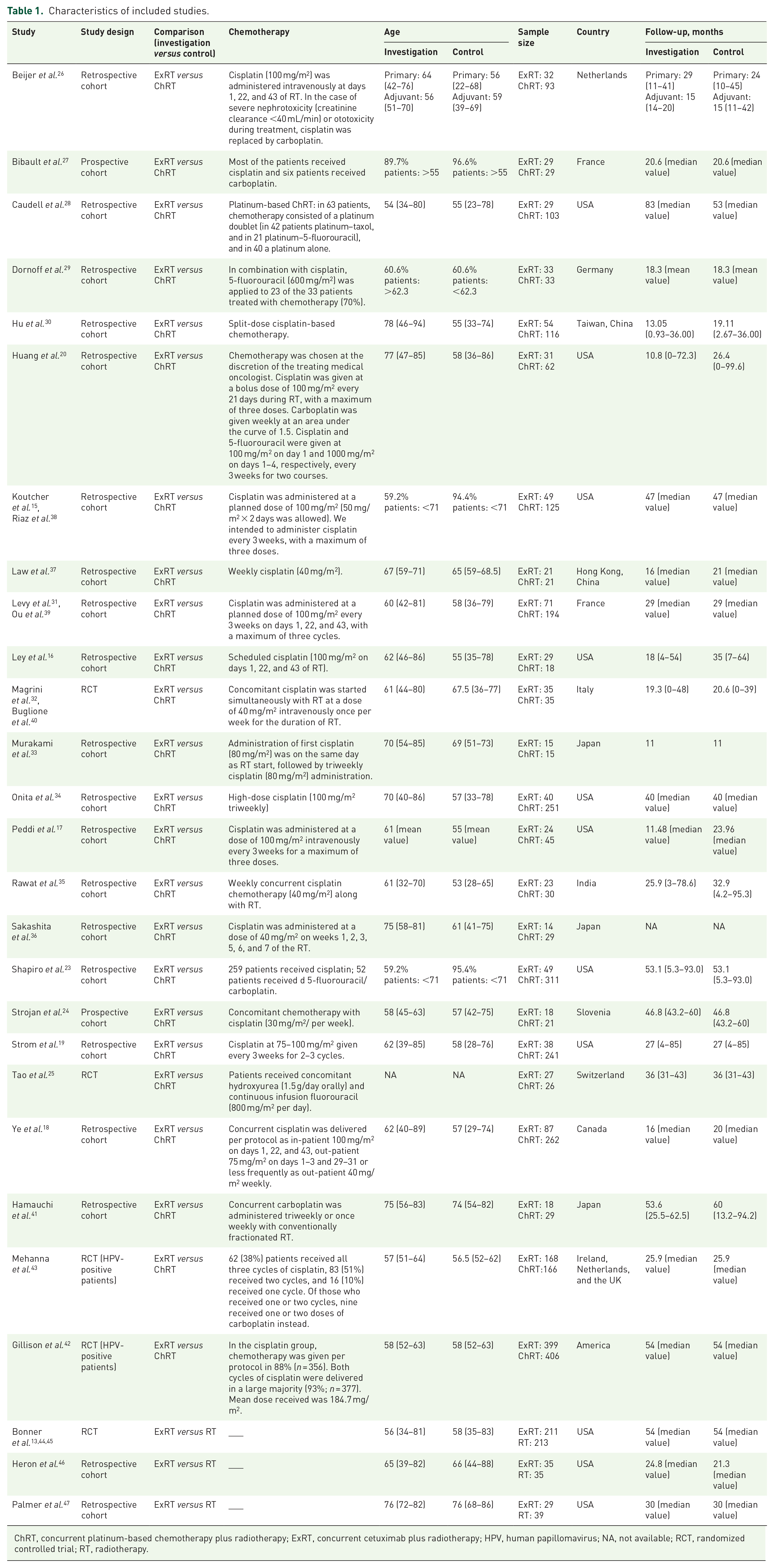
ChRT, concurrent platinum-based chemotherapy plus radiotherapy; ExRT, concurrent cetuximab plus radiotherapy; HPV, human papillomavirus; NA, not available; RCT, randomized controlled trial; RT, radiotherapy.
Survival outcomes of ExRT versus ChRT for locally advanced HNSCC patients
OS
Nineteen studies reported the OS of HNSCC patients receiving ExRT in contrast to ChRT. Using a random-effects model according to the test of heterogeneity (

Forest plots comparing cetuximab plus concomitant radiotherapy with concurrent platinum-based chemotherapy plus radiotherapy in locally advanced head and neck squamous cell carcinoma. (A) Forest plot of hazard ratio (HR) for overall survival (OS). (B) Forest plot of HR for disease-specific survival (DSS). (C) Forest plot of HR for locoregional control (LRC). (D) Forest plot of HR for progression-free survival (PFS). (E) Forest plot of HR for metastasis-free survival (MFS).
DSS
Four studies reported the DSS of HNSCC patients receiving ExRT compared with ChRT, and the data were analyzed with a fixed-effects model according to the test of heterogeneity (
LRC
Eight studies investigated the LRC of HNSCC patients receiving ExRT compared with ChRT. Using a fixed-effects model according to the test of heterogeneity (
PFS
Eight studies reported the PFS of HNSCC patients receiving ExRT in contrast to ChRT, and the data were analyzed with a random-effects model according to the test of heterogeneity (
MFS
Three studies reported the MFS of HNSCC patients receiving ExRT in contrast to ChRT. Using a fixed-effects model according to the test of heterogeneity (
Multivariate analysis by Cox proportional hazards model on OS
To confirm whether the OS of HNSCC patients was associated with ExRT treatment in contrast to ChRT, we made multivariate analysis by Cox proportional hazards model, which included the clinical, tumor, and therapeutic variables extracted from six studies. Using a fixed-effects model according to the test of heterogeneity (
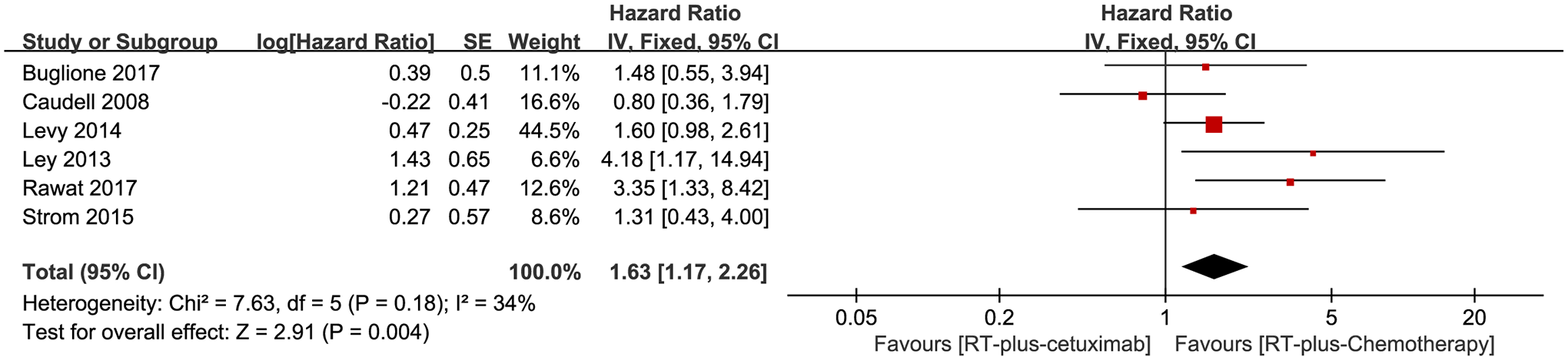
Multivariate analysis by Cox proportional hazards model on overall survival among patients with locally advanced head and neck squamous cell carcinoma receiving cetuximab plus concomitant radiotherapy
Survival outcomes of ExRT versus ChRT for HPV-positive locally advanced HNSCC patients
OS
HPV-positive oropharyngeal squamous cell carcinoma is considered to be a distinct disease entity from the HPV-negative ones. Three studies reported the OS of HPV-positive HNSCC patients receiving ExRT in contrast to ChRT, and the data were analyzed with a random-effects model according to the test of heterogeneity (

Forest plots comparing cetuximab plus concomitant radiotherapy with concurrent platinum-based chemotherapy plus radiotherapy in human papillomavirus-positive locally advanced head and neck squamous cell carcinoma patients. (A) Forest plot of hazard ratio (HR) for overall survival (OS). (B) Forest plot of HR for disease-specific survival (DSS). (C) Forest plot of HR for locoregional control (LRC).
DSS
Three studies reported the DSS of HPV-positive HNSCC patients receiving ExRT in contrast to ChRT, and the data were analyzed with a fixed-effects model according to the test of heterogeneity (
LRC
Three studies reported the LRC of HPV-positive HNSCC patients receiving ExRT in contrast to ChRT. Using a fixed-effects model according to the test of heterogeneity (
Adverse events of ExRT versus non-CTX therapy
Out of all adverse events reported in the included studies, we retrieved 14 adverse events (grade ⩾3) from studies comparing ExRT with non-CTX group (ChRT and RT). After combining the data from the included trials, we analyzed the RR for each event. The adverse events are listed in Table 2 according to the different organs involved, and the forest plot of each RR is shown in Figure 5. The results showed that the incidence of mucositis (RR: 1.17, 95% CI 1.05–1.31,
Toxicity grade ⩾3.


Forest plot of risk ratio of toxicities among patients with locally advanced head and neck squamous cell carcinoma receiving cetuximab plus concomitant radiotherapy
Publication bias
The publication bias of the individual studies for locally advanced HNSCC patients was evaluated by funnel plots. By comparing ExRT with ChRT, the results showed that no obvious publication bias was observed in the funnel plots for the HR of OS (Figure 6A), HR of LRC (Figure 6B), HR of PFS (Figure 6C), and HR of OS based on multivariate analysis by Cox proportional hazards model (Figure 6D), for all
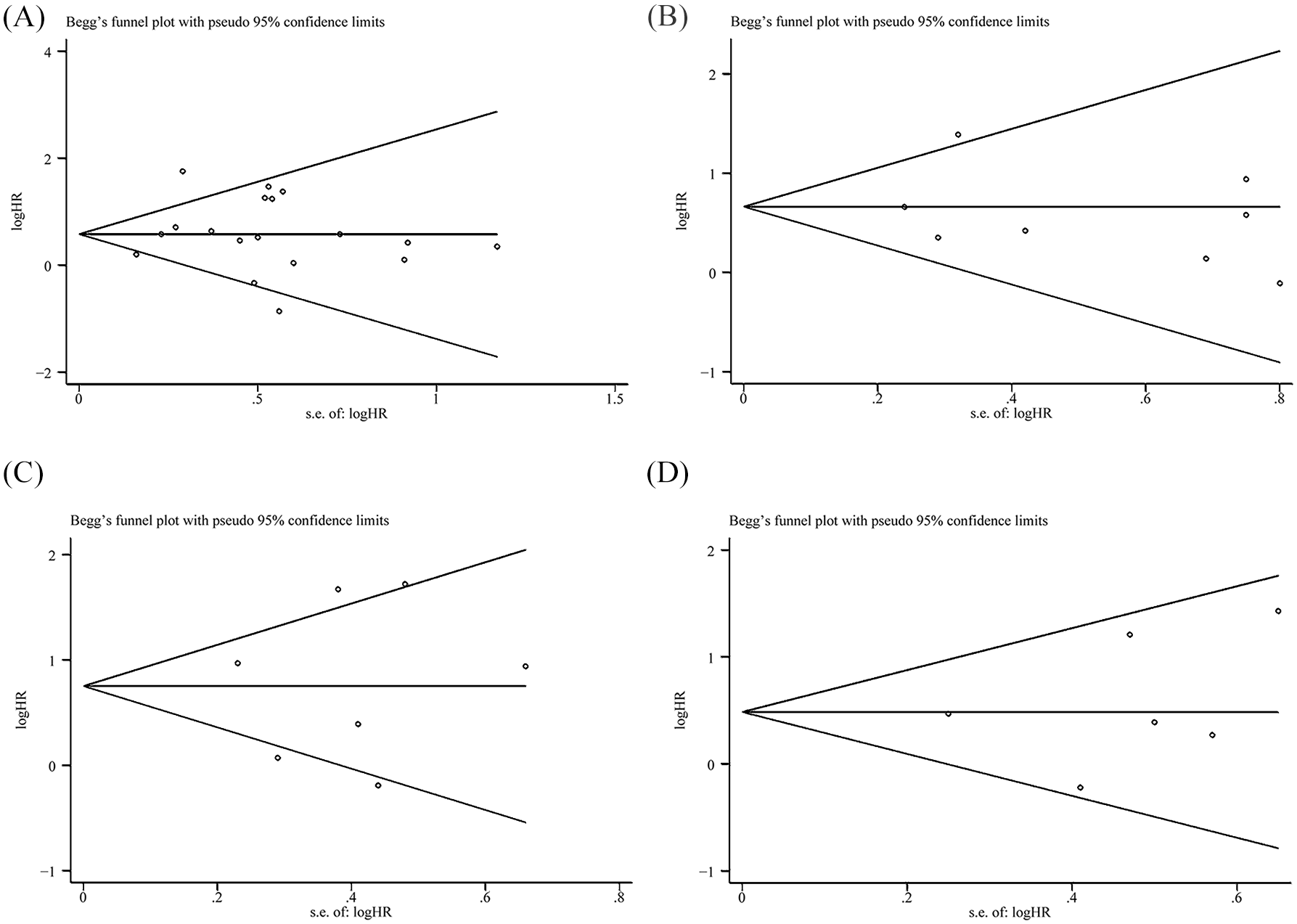
Begg’s funnel plots among patients with locally advanced head and neck squamous cell carcinoma receiving cetuximab plus concomitant radiotherapy
Discussion
This study was mainly focused on the comparison of clinical efficacy and safety between ExRT and ChRT in locally advanced HNSCC patients. Our results verified that in contrast to ChRT, ExRT was associated with worse survival outcomes including OS, DSS, LRC, and PFS for locally advanced HNSCC patients. Consistently, in HPV-positive HNSCC patient subgroups, ExRT showed inferior DSS and LRC to ChRT regimen. Furthermore, ExRT increased the incidence of mucositis, skin toxicity, and infection, and was associated with lower incidence of anemia, leukocytopenia, neutropenia, nausea/vomiting, and renal toxicity in contrast to non-CTX therapies.
Chemotherapy improves survival in HNSCC patients. Decades ago, cisplatin had been utilized as a radiosensitizing agent concomitantly with RT. 48 A meta-analysis in 2009, which included 16,485 HNSCC patients from 87 randomized trials conducted between 1965 and 2000, demonstrated that chemotherapy concurrently with locoregional treatment (surgery and/or RT) significantly increased OS and achieved an absolute benefit of 4.5% at 5 years in contrast to locoregional therapy alone. 49 And in 2019 another meta-analysis, which including 5000 patients from 24 trials conducted between 1965 and 2000, reported that concurrent chemotherapy added to surgical locoregional treatment significantly improved OS, while induction chemotherapy did not increase OS. 50 Nowadays, ChRT, in which concurrent platinum-based chemotherapy is performed with RT, remains the standard-of-care therapy for locally advanced HNSCC, and high-dose cisplatin (100 mg/m2) is recognized as the gold standard of radiosensitizer. 14 High-dose cisplatin was associated with increased severe toxicities such as nephrotoxicity, hematologic toxicity, difficulty in swallowing, and requirement for a feeding tube. A previous study which included 230 patients receiving ChRT reported that 43% of assessable patients had severe late toxicities. 7 Thus, the acute early and late toxic effects induced by platinum-based drugs, especially high-dose cisplatin, were limiting factors for its use and provided an impetus for seeking alternative strategies for radiation sensitization. Low-dose cisplatin was found to offer ease of administration and reduced toxicities, 51 but no definite low-dose regimen was approved to be as effective as high-dose cisplatin therapy, and ongoing studies are investigating different low-dose regimens. 52 Non-cisplatin regimens were proposed for vulnerable patients to avoid unbearable toxicities from cisplatin, and the regimens were intended to be associated with the administration of carboplatin, docetaxel or paclitaxel, 5-fluorouraci, or the anti-EGFR drugs, especially CTX. 53
EGFR belongs to the ErbB family of tyrosine kinase receptors. The EGFR signaling pathway is involved in the tumorigenesis and progression of HNSCC,
54
and about 90% of HNSCC patients are detected overexpressing EGFR.55,56 CTX is a chimeric IgG1-subclass monoclonal antibody, which binds to the extracellular domain of EGFR with higher affinity than the natural ligand. It is the only approved targeted strategy for HNSCC patients.13,57 The binding of CTX to EGFR blocks the binding of a series of natural ligands, resulting in downregulation of the EGFR signaling cascade. RT-associated CTX decreases the capacity of DNA repair and inhibits tumor angiogenesis; meanwhile it facilitates apoptosis, sensitizes G1-phase cells to radiation, and reduces radioresistance of S-phase cells.58–60 In 2006, the trial by Bonner
Increasing trials were performed to compare the efficacy of ExRT with the standard-of-care ChRT for locally advanced HNSCC patients. We searched the relevant trials and performed this study. As seen in Table 1, in most of the trials, ChRT involved the administration of cisplatin, and most of the trials included were non-RCT studies. The pooled analysis showed that in contrast to ChRT, ExRT was associated with worse survival outcomes including OS, DSS, LRC, and PFS. These data are consistent with a previous meta-analysis which showed that ChRT significantly improved OS, LRC, and PFS compared with ExRT for locally advanced HNSCC, and out of the 15 trials included in this meta-analysis, 13 trials utilized cisplatin as platinum-based chemotherapy drug. 64 Our results indicated that, so far, ExRT has failed to achieve better survival outcomes than ChRT for locally advanced HNSCC patients.
HPV-positive oropharyngeal cancer (OPC) is a unique entity among HNSCCs, 65 because HPV-positive OPC patients achieve better prognosis generally. 66 We then assessed the efficacy of ExRT compared with ChRT for HPV-positive HNSCC patients. The analysis included six trials which contained two RCTs, namely NRG Oncology RTOG 1016 and the European De-ESCALaTE.42,43 The ChRT in the six trials all involved the administration of cisplatin. Results showed that ExRT achieved inferior DSS and LRC in contrast to ChRT, and ExRT induced no significant difference in OS compared with ChRT. A previous meta-analysis in 2019 included eight studies and, out of the trials, three were the same included trials as with our study, and they reported that in patients with HPV-positive HNSCC, ExRT was associated with inferior OS and LRC compared with ChRT, which to some extent was consistent with our findings. 67 To sum up, for locally advanced HPV-positive HNSCC patients, our results demonstrated that ChRT, especially the cisplatin-based RT, should remain the cornerstone of treatments.
For locally advanced HNSCC patients treated with the combination of CTX, adverse events (grade ⩾3) were comprehensively analyzed in our study. The results showed that ExRT was associated with an increased risk of mucositis (RR: 1.17, 95% CI: 1.05–1.31,
So far, there is no clear predictive marker which could determine that concurrent CTX is more useful and tolerable in some populations of locally advanced HNSCC. Bonner reported that for the patients treated with CTX, OS was significantly improved in those who suffered an acneiform rash of grade ⩾2 severity in contrast to the patients with no rash or grade 1 rash, but this adverse event could not be predicted prior to deciding the therapeutic regimen. 44 The potential markers such as EGFR mutations, HPV status, and performance status have not been validated to predict effectively or be adequately powered by significant differences in survival outcomes between the patient subsets.71,72 In 2019, a meta-analysis including 2653 HNSCC patients from five RCTs indicated that EGFR inhibition (three RCTs with CTX, one with gefitinib, and one with panitumumab) was associated with better OS in young patients than in older counterparts, and neither HPV status nor performance status had much effect on the relative OS benefit from EGFR inhibition. 72 To determine the difference of survival outcomes between subsets which divided according to potential influencing factors, more prospective and RCT trials should be conducted in future, which would further help to find the most suitable patients to use CTX.
There are several limitations in our study. First, out of our included trials comparing survival outcomes between ExRT and ChRT, most included articles were non-RCT studies, so there would be statistically differences in patients’ baseline. For instance, the patients in ExRT arms commonly had more pre-existing conditions, such as cardiac comorbidity, older age, and worse performance status, and obtained a higher Charlson Comorbidity Index,20,31 thus resulting in ExRT arms suffering from more severe systemic diseases. These could lead to heterogeneity between studies. More prospective trials and RCTs should be conducted in future. Second, since quality of life in locally advanced HNSCC patients has not been analyzed in our work, to identify the relation between CTX-induced toxicities with CTX-associated quality of life in future could help us in seeking methods to improve HNSCC patient compliance with CTX. Third, the cost-effectiveness of ExRT
Conclusion
In conclusion, this study illustrated that in locally advanced HNSCC patients, CTX was an effective radiosensitizer, while ChRT achieved better survival outcomes than ExRT. Moreover, in HPV-positive HNSCC patient subgroup, ExRT also obtained inferior survival outcomes in contrast to ChRT. ExRT is associated with a lower incidence of severe adverse events, including anemia, leukocytopenia, neutropenia, nausea/vomiting, and renal toxicity compared with non-CTX therapies, and the CTX-associated adverse events included mucositis, skin toxicity, and infection, which occurred more frequently in ExRT arms. It was demonstrated that ChRT should remain the first-line treatment for locally advanced HNSCC patients, and considering that some frail HNSCC patients were intolerable to severe toxicities induced by ChRT, ExRT could be an effective alternative treatment.
Supplemental Material
sj-docx-1-tam-10.1177_1758835920975355 – Supplemental material for Comparative efficacy and safety of radiotherapy/cetuximab versus radiotherapy/chemotherapy for locally advanced head and neck squamous cell carcinoma patients: a systematic review of published, primarily non-randomized, data
Supplemental material, sj-docx-1-tam-10.1177_1758835920975355 for Comparative efficacy and safety of radiotherapy/cetuximab versus radiotherapy/chemotherapy for locally advanced head and neck squamous cell carcinoma patients: a systematic review of published, primarily non-randomized, data by Mei Mei, Yu-Huan Chen, Tian Meng, Ling-Han Qu, Zhi-Yong Zhang and Xiao Zhang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-1-tam-10.1177_1758835920975355 – Supplemental material for Comparative efficacy and safety of radiotherapy/cetuximab versus radiotherapy/chemotherapy for locally advanced head and neck squamous cell carcinoma patients: a systematic review of published, primarily non-randomized, data
Supplemental material, sj-xlsx-1-tam-10.1177_1758835920975355 for Comparative efficacy and safety of radiotherapy/cetuximab versus radiotherapy/chemotherapy for locally advanced head and neck squamous cell carcinoma patients: a systematic review of published, primarily non-randomized, data by Mei Mei, Yu-Huan Chen, Tian Meng, Ling-Han Qu, Zhi-Yong Zhang and Xiao Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank Beijing Zhiyun Data Technology Co. LTD, for providing methodological advices.
Author contributions
Study concept and design: Mei Mei, Zhi-Yong Zhang, Xiao Zhang. Acquisition of data: Mei Mei, Yu-Huan Chen, Tian Meng. Analysis and interpretation of data: Mei Mei, Ling-Han Qu. Drafting of the manuscript: Mei Mei. Critical revision of the manuscript for important intellectual content: Mei Mei, Zhi-Yong Zhang, Xiao Zhang.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (grant number 81700993), and is available via the SAGE Open Access.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
