Abstract
Background:
There is no clear consensus on second-line chemotherapy for patients with advanced pancreatic ductal adenocarcinoma (PDAC) after failure of first-line gemcitabine plus nab-paclitaxel (GnP).
Objectives:
This study aimed to compare the efficacy and safety of oxaliplatin plus S-1 (SOX) versus modified FOLFIRINOX (mFFX) as second-line chemotherapy in this patient population.
Design:
A retrospective cohort study was conducted.
Methods:
Patients with advanced PDAC who received second-line SOX or mFFX after GnP failure at Peking Union Medical College Hospital were reviewed. Efficacy (disease control rate (DCR), overall survival (OS), progression-free survival (PFS)), and safety were analyzed. Molecular features were explored in a subgroup using targeted next-generation sequencing (NGS).
Results:
In total, 113 patients were included (65 SOX, 48 mFFX). The mFFX group had a significantly higher DCR than the SOX group (68.8% vs 40.0%, p = 0.005). Median PFS (4.8 vs 2.4 months, p = 0.001) and OS (10.4 vs 6.1 months, p = 0.001) were significantly longer with mFFX, even after propensity score matching adjustment. However, grade ⩾3 adverse events, particularly severe neutropenia (42.9% vs 13.5%, p = 0.004) and diarrhea (17.1% vs 1.9%, p = 0.031), were more frequent with mFFX. Multivariate analysis confirmed mFFX as an independent predictor for improved PFS (hazard ratio (HR) = 0.52, p = 0.004) and OS (HR = 0.46, p = 0.002). Exploratory NGS analysis in 45 patients suggested ARID1A, INPP4A, NTRK2, and PTPRS alterations may predict poor survival, but did not influence the relative efficacy of either regimen.
Conclusion:
The mFFX regimen demonstrated superior efficacy over SOX as second-line chemotherapy after GnP failure in advanced PDAC, significantly prolonging PFS and OS. However, this benefit was accompanied by a higher incidence of severe toxicities. Molecular alterations may hold prognostic value but did not guide regimen selection in this study.
Plain language summary
When first-line chemotherapy stops working for advanced pancreatic cancer, doctors need evidence to choose the best next treatment. This study compared two chemotherapy combinations (SOX: S-1 pill + oxaliplatin infusion vs. mFFX: modified four-drug cocktail) in 113 patients whose cancer worsened after standard gemcitabine/nab-paclitaxel treatment. Key findings: Patients receiving mFFX lived nearly twice as long (10.4 vs. 6.1 months average survival) with better short-term disease control (68.8% vs. 40% saw tumor shrinkage/stabilization). However, mFFX caused more severe side effects - 69% vs. 44% experienced significant blood cell drops or diarrhea requiring medical attention. Younger patients (average age 57) tended to receive mFFX more often. Genetic testing revealed four biological markers (ARID1A, INPP4A, NTRK2, PTPRS) that might help predict which patients benefit most from platinum-based therapies like SOX. While mFFX showed stronger anti-cancer effects, its higher toxicity suggests it may be better suited for younger, fitter patients. SOX remains a viable option for those needing gentler treatment. The discovery of potential genetic predictors opens doors for personalized therapy selection in future care. This real-world analysis provides practical guidance for oncologists balancing treatment intensity with patient tolerance in this challenging clinical scenario. Patients should discuss both options’ risks/benefits with their care team based on individual health status and treatment goals.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most aggressive and deadliest diseases, with a 5-year overall survival (OS) rate of only 12%. 1 Only 20% of PDAC patients can be treated with initial surgical resection. 2 Therefore, systemic chemotherapy plays a crucial role in the treatment of patients with advanced PDAC. Despite modest progress in chemotherapy based on gemcitabine, the 5-year OS for patients with metastatic PDAC is still as low as 3%. 1
In the PRODIGE 4/ACCORD 11 trial, a chemotherapy regimen consisting of 5-fluorouracil (5-FU)/leucovorin (LV), irinotecan, and oxaliplatin (FOLFIRINOX) significantly improved survival compared with gemcitabine monotherapy for metastatic PDAC patients with a good performance status. 3 In 2013, the MPACT trial comparing gemcitabine plus nab-paclitaxel (GnP) and gemcitabine alone in patients with metastatic pancreatic cancer showed significantly superior OS and progression-free survival (PFS). 4 Recently, the JCOG1611/GENERATE trial suggested that compared with modified FOLFIRINOX (mFFX), GnP had a longer (but not statistically significant) OS for first-line treatment of metastatic PADAC, and its safety may be better. 5 This suggests that higher chemotherapy intensity, more chemotherapy drugs, and adverse events (AEs) may not necessarily contribute to better survival outcomes in patients with PDAC.
Although GnP is recommended as the first-line treatment for patients with advanced PDAC, almost all patients experience disease progression. According to the NCCN guidelines, 6 for patients who have failed GnP and are without actionable genomic alterations, second-line treatment typically involves fluorouracil-based regimens, including fluorouracil or capecitabine combined with oxaliplatin (FOLFOX or CapeOx) or mFFX. mFFX has also been reported to be a promising second-line therapy for PDAC patients after GnP therapy.7,8 On the other hand, mFFX is often associated with an incidence of grade 3 or higher adverse reactions exceeding 50%.7,8 S-1 is an oral fluoropyrimidine derivative that consists of tegafur, gimeracil, and oteracil potassium at a molar ratio of 1.0:0.4:1.0. 9 Notably, meta-analyses have proposed that patients with resected pancreatic cancer who choose oral S-1 as adjuvant chemotherapy can achieve survival outcomes similar to or even better than those of patients receiving mFFX, and S-1 is associated with better tolerance.10,11 Several studies have also demonstrated that oxaliplatin plus S-1 (SOX) is an advisable second-line regimen for treating gemcitabine-refractory PDAC with acceptable treatment-related toxicity.12,13 However, there is no clear consensus on the optimal second-line treatment for patients with advanced PDAC after GnP failure. In the present study, we compared the efficacy and safety of SOX and mFFX as second-line chemotherapies for advanced PDAC patients after GnP failure.
Patients and methods
Patients
This retrospective study included patients who received SOX or mFFX as second-line chemotherapy after GnP therapy from March 1, 2018 to October 1, 2023 at Peking Union Medical College Hospital (Beijing, China). Patient data were retrieved from the Electronic Medical Record Analytical Database (PUMCH-EMERALD). Ethics approval for this study was obtained from the Medical Ethics Committee of Peking Union Medical College Hospital (Approval Identifier: S-K2099). This study conformed to the principles of the Helsinki Declaration and ethical requirements involving human subjects. Informed consent was waived by the Medical Ethics Committee of Peking Union Medical College Hospital because of the retrospective design and anonymization of the study.
The inclusion criteria were as follows: (1) patients who had histologically or cytologically confirmed PDAC; (2) were older than 18 years; (3) experienced disease progression after first-line GnP treatment; (4) received at least 1 cycle of SOX or mFFX as second-line chemotherapy; and (5) had adequate hematologic, renal, and liver function. The exclusion criteria were as follows: (1) patients who died or were lost to follow-up before the first assessment of treatment efficacy and (2) had other primary tumors.
Treatment and assessment
In the SOX group, patients received oxaliplatin (100 mg/m2) intravenously on day 1 plus oral S-1 (40 mg for body surface area (BSA) <1.25 m2, 50 mg for BSA ⩾1.25 m2 but <1.5 m2, or 60 mg for BSA ⩾1.5 m2) twice per day for 14 days, followed by a 7-day pause in each cycle. Patients in the mFFX group received oxaliplatin at a dose of 85 mg/m2 intravenously on day 1, irinotecan at a dose of 150 mg/m2 intravenously on day 1, LV at 200 mg/m2 on day 1, and a continuous infusion of 5-FU at 2400 mg/m2 for 46 h without bolus infusion every 2 weeks. Compared to the standard FOLFIRINOX, mFFX omits the 5-FU 400 mg/m2 bolus and reduces the dose of irinotecan from 180 to 150 mg/m2. Treatments were continued until intolerable toxicity, disease progression, or discontinuation at the discretion of investigators or patients.
The primary endpoint was OS. OS was defined as the interval between second-line chemotherapy initiation and death due to any cause. The secondary endpoints included PFS, disease control rate (DCR), and safety. PFS was defined as the time from the initiation of second-line chemotherapy to disease progression or death due to any cause. Contrast-enhanced computed tomography or magnetic resonance imaging was performed at baseline and every 2–3 months after second-line chemotherapy initiation to assess tumor responses according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 14 Carbohydrate antigen 19-9 (CA19-9) was measured before second-line chemotherapy and every 1–2 months thereafter. AEs were evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. All patients were followed up until death or loss to follow-up, and the last day of follow-up was June 1, 2024. The reporting of this study conforms to ESMO Guidance for Reporting Oncology real-World evidence (GROW). 15
Targeted next-generation sequencing
Hybrid capture-based next-generation sequencing (NGS) of targeted genes was conducted as previously described. 16 Briefly, DNA was extracted from formalin-fixed paraffin-embedded cancer tissue. Sequencing was performed on a Nextseq500 sequencer (Illumina, Inc., San Diego, California, USA), for 520 genes containing whole exons of 312 genes, hotspot mutation regions (exons, introns, and promoter regions) of 208 genes, and fusions of 16 genes (Table S1). The output data were analyzed for quality control, sequence alignment, and variant calling using a vendor-supplied bioinformatics pipeline.
Statistical analyses
All the statistical analyses were performed with R version 3.6.1 (https://www.r-project.org/). The Mann–Whitney U test or Pearson’s Chi-square test was used to compare continuous or categorical variables. A Cox proportional hazard model was used to calculate the hazard ratios (HRs) with 95% confidence intervals (CIs) of variables associated with OS and PFS in patients. Survival curves were generated with Kaplan–Meier analyses and compared using the log-rank test. Since clinicians may tend to recommend the mFFX regimen over the SOX regimen for younger and physically robust patients, and tumor stage and first-line treatment efficacy can influence patient prognosis, propensity score matching (PSM) was used to minimize the impact of confounding factors. Propensity scores were estimated using a logistic regression model, where the binary treatment variable (regimen: mFFX vs SOX) was regressed on the following baseline covariates: age, performance status, body mass index (BMI), baseline serum albumin level, tumor stage, baseline CA19-9, and first-line efficacy. We employed nearest-neighbor matching with a 1:1 ratio to create balanced treatment and control groups. A caliper of 0.02 standard deviations of the propensity score was used to ensure adequate similarity between matched pairs, reducing the likelihood of poor matches. For balance diagnostics, we assessed the standardized mean differences (SMDs) of all covariates before and after matching. Covariates were considered balanced if the post-matching SMD was <0.1, indicating minimal residual confounding. A two-tailed probability value of p < 0.05 was considered to indicate statistical significance.
Results
Patient characteristics
A total of 113 patients with advanced PDAC refractory to first-line GnP therapy were included according to the selection criteria. The baseline characteristics of the study cohort are listed in Table 1. Among these patients, 65 received SOX, and 48 received mFFX as second-line chemotherapy. The median age was 59 years (range, 37–76), and 70.8% (n = 80) of the patients were male. Most patients had an Eastern Cooperative Oncology Group (ECOG) performance status score of 0 (43.4%) or 1 (54.0%). Notably, patients treated with SOX were generally older (median age: 63 vs 57 years; p < 0.001) and had lower BMIs (median: 21.9 vs 22.9 kg/m2; p = 0.018) and baseline albumin levels (median: 39 vs 41 g/dL; p = 0.004) than patients treated with mFFX. A cohort including 58 matched patients was generated by propensity score matching (PSM), with 29 patients each receiving mFFX and SOX regimens. The balance of baseline covariates after PSM is confirmed in Figure S1, which plots the SMDs of key variables before and after matching—all SMDs were reduced to <0.1, indicating minimal residual confounding between the two groups. Table S2 details the clinical profiles of the 58 matched patients (29 per group), confirming balanced distribution of key variables such as ECOG performance status (all 0–1) and tumor stage, which supports the validity of subsequent survival analyses.
Baseline characteristics of all 113 included patients.

BMI, body mass index; CA19-9, carbohydrate antigen 19-9; ECOG, Eastern Cooperative Oncology Group; mFFX, modified 5-fluorouracil/leucovorin, irinotecan, and oxaliplatin; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; SOX, oxaliplatin plus S-1. The bold values indicate they are statistically significant (p < 0.05).
Efficacy
The treatment outcomes of all included patients are shown in Table 2. At the time of the last follow-up, 100 out of the 113 patients had stopped second-line chemotherapy, 70.8% (n = 80) terminated treatment due to disease progression, and 3 patients receiving mFFX were still continuing second-line treatment. Among all patients, 5 (4.4%) had a partial response (PR), 54 (47.8%) had stable disease (SD), and 54 (47.8%) had progressive disease according to RECIST1.1. No patient in either group achieved a complete response (CR). The DCR in the mFFX group was significantly greater than that in the SOX group (68.8% vs 40.0%, p = 0.005). The objective response rate (ORR) was similar for both groups (4.2% vs 4.6%).
Response to chemotherapy of all 113 included patients.

Treatment discontinuation directly attributed to AEs, regardless of the type or grade of the AE, and irrespective of whether discontinuation was initiated by the physician (based on clinical judgment) or by the patient (due to inability to tolerate symptoms).
Tumor progression confirmed by radiological assessments (CT and/or MRI) in accordance with the Response Evaluation Criteria in Solid Tumors version 1.1.
(1) Patients who remained on treatment at the time of data cutoff. (2) Patients lost to follow-up before treatment completion, where medical records lacked documentation of a clear reason for potential discontinuation, and attempts to confirm via follow-up communications were unsuccessful.
AE, adverse event; CA19-9, carbohydrate antigen 19-9; CT, computed tomography; DCR, disease control rate; mFFX, modified 5-fluorouracil/leucovorin, irinotecan, and oxaliplatin; MRI, magnetic resonance imaging; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease; SOX, oxaliplatin plus S-1.
Of the 113 enrolled patients, 21 failed to monitor CA19-9 per the study plan (both 1 month before and during second-line treatment), and 17 had no CA19-9 expression. The remaining 75 patients were included in the CA19-9 response analysis, among whom 33.3% (11/33) in the mFFX group and 31.0% (13/42) in the SOX group experienced the greatest decrease of more than 30%. However, the levels of CA19-9 decreased to within the normal range after SOX or mFFX treatment in only 4 (5.3%) patients. Table S3 shows efficacy outcomes in the matched cohort: ORR was similar between mFFX and SOX groups (3.4% vs 6.9%, p = 1.000), and CA19-9 reduction ⩾30% was comparable (27.6% vs 24.1%, p = 0.976). However, mFFX still had a significantly higher DCR (75.9% vs 41.4%, p = 0.016), consistent with the total cohort.
Survival
After a median follow-up of 15.3 months, 75 patients died—22 (45.83%) in the mFFX group and 53 (81.5%) in the SOX group. The median PFS of the mFFX group was significantly longer than that of the SOX group (4.8 months (95% CI, 3.9–7.2 months) vs 2.4 months (95% CI, 2.0–3.5 months), p = 0.001; Figure 1(a)). A significant improvement in OS in the mFFX group (median: 10.4 months (95% CI, 8.4–not reached months) vs 6.1 months (95% CI, 5.0–7.4 months), p = 0.001) was also observed (Figure 1(b)). Consistently, the median PFS (4.8 months (95% CI, 3.9–8.0 months) vs 2.1 months (95% CI, 5.8–15.1 months), p = 0.036) and OS were longer for patients treated with mFFX (11.3 months (95% CI, 8.4–not reached months) vs 6.6 months (95% CI, 5.8–15.1 months), p = 0.028) in the matched cohort (Figure 1(c) and (d)). According to the subgroup analysis based on baseline characteristics, PFS and OS were greater for patients who received mFFX than for patients who received SOX in most subgroups (Figures 2 and 3), including those with ECOG performance status scores of 1–2.

Kaplan–Meier curves of progression-free survival and overall survival for all 113 included patients (a, b) or 58 matched patients (c, d).

Forest plot for progression-free survival in selected subgroups.
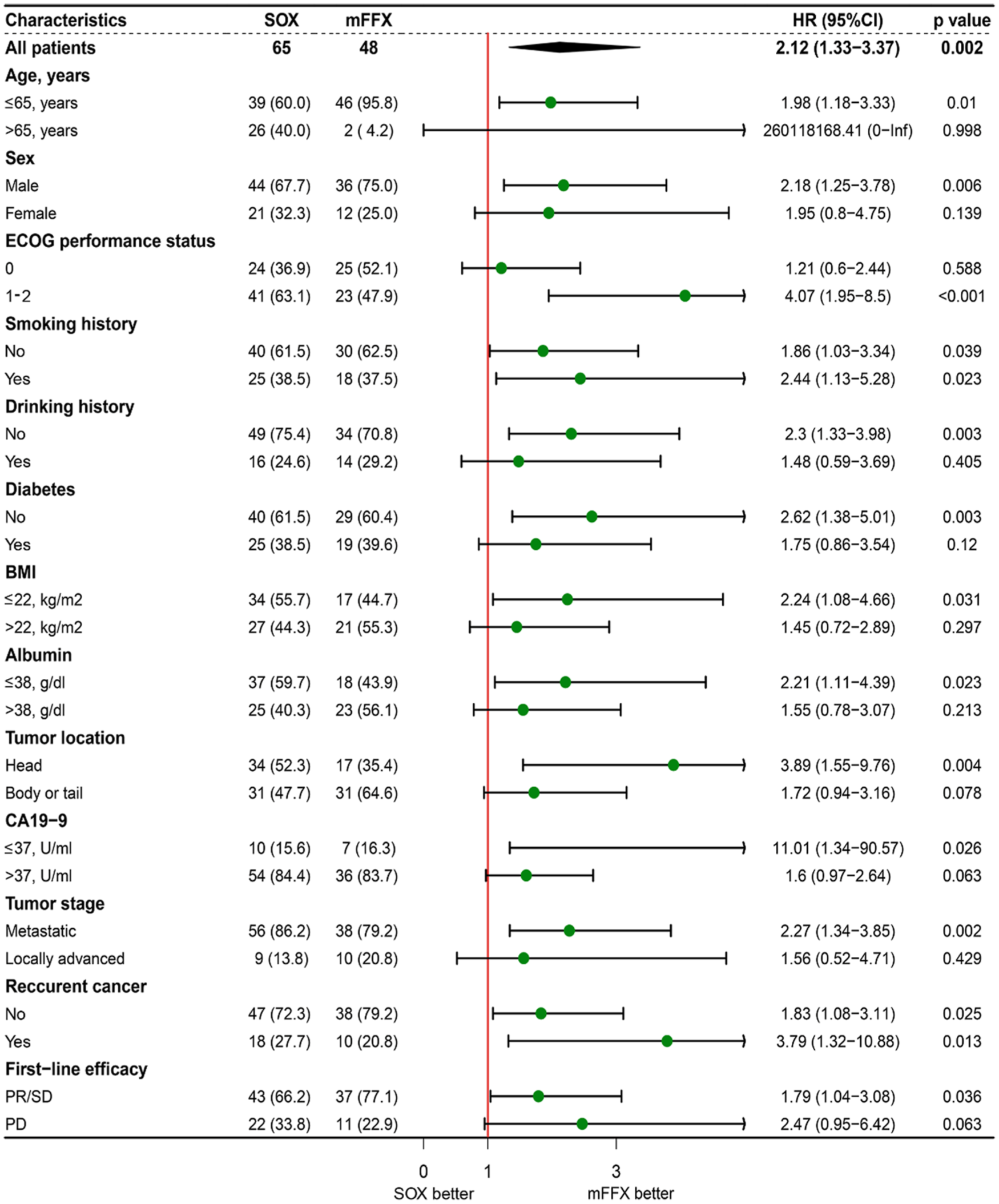
Forest plot for overall survival in selected subgroups.
Univariate and multivariate Cox analyses performed to evaluate potential prognostic factors associated with PFS and OS for all 113 patients are presented in Tables 3 and 4. In the univariate analysis for PFS, age, ECOG performance status, and second-line regimen were associated with PFS. Furthermore, multivariate analysis confirmed that second-line mFFX (HR, 0.52; 95% CI, 0.34–0.81; p = 0.004) was independently associated with improved PFS (Table 3). According to the univariate analysis for OS, ECOG performance status, baseline CA19-9 level, best response to first-line chemotherapy, and second-line chemotherapy regimen were potential prognostic factors for OS. Multivariate analysis further confirmed that the second-line mFFX regimen (HR, 0.46; 95% CI, 0.28–0.76; p = 0.002), better first-line efficacy, and better performance status were independent prognostic factors for favorable OS (Table 4).
Univariate and multivariate analyses of PFS.

BMI, body mass index; CA19-9, carbohydrate antigen 19-9; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; mFFX, modified 5-fluorouracil/leucovorin, irinotecan, and oxaliplatin; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; SOX-1, oxaliplatin plus S-1. The bold values indicate they are statistically significant (p < 0.05).
Univariate and multivariate analyses of overall survival.

BMI, body mass index; CA19-9, carbohydrate antigen 19-9; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; mFFX, modified 5-fluorouracil/leucovorin, irinotecan, and oxaliplatin; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; SOX-1, oxaliplatin plus S-1. The bold values indicate they are statistically significant (p < 0.05).
Adverse events
There were 35 patients receiving mFFX and 52 patients receiving SOX with evaluable safety data, with the median number of treatment cycles being 4 (range, 1–12) and 2 (range, 1–8) for the mFFX and SOX regimens, respectively. No patients died due to AEs, while 47 (54.0%) patients experienced grade 3 or 4 AEs: 24 (68.6%) in the mFFX group and 23 (44.2%) in the SOX group (p = 0.044; Table 5). The incidences of grade 3 or 4 neutropenia (42.9% vs 13.5%, p = 0.004) and diarrhea (17.1% vs 1.9%, p = 0.031) in the mFFX group were significantly greater than those in the SOX group. The most common grade 3 or 4 AEs (⩾5%) in the mFFX group were leukopenia, neutropenia, anemia, thrombocytopenia, fatigue, anorexia, nausea, diarrhea, and peripheral neuropathy. In the SOX group, neutropenia, anemia, thrombocytopenia, fatigue, and anorexia were frequently observed.
Grade 3 or 4 adverse events (safety population, n = 87).

mFFX, modified 5-fluorouracil/leucovorin, irinotecan, and oxaliplatin; SOX-1, oxaliplatin plus S-1. The bold values indicate they are statistically significant (p < 0.05).
Molecular features
Forty-five patients had available NGS data (NGS cohort), with 24 and 21 patients receiving mFFX and SOX, respectively, as second-line chemotherapy. Forty-two (93.3%) samples used for NGS originated from the primary pancreas, while the remaining 2 and 1 samples originated from peritoneal and liver metastases, respectively. The baseline characteristics of the patients in the NGS cohort are listed in Table S4. A total of 279 genomic alterations were detected in 118 genes. KRAS (91.1%, n = 41) was the most frequently occurring somatic mutation in the NGS cohort, and other common mutations included mutations in TP53 (75.5%), SMAD4 (28.9%), and CDKN2A (17.8%; Figure 4).

Summary of the common prevalent genomic alterations and clinical characteristics of 45 patients with available NGS data.
Univariate Cox and survival analyses were performed to explore the correlation between molecular features and prognosis. After excluding genomic alterations that occurred in only one patient, Cox and survival analyses suggested that ARID1A, INPP4A, NTRK2 (two patients with missense mutations), and PTPRS alterations may be prognostic factors for poor PFS and OS, while ASXL1, BRCA2, RNF43, and SETD2 alterations may only be prognostic factors for PFS or OS (Table S5 and Figure S2). Moreover, alterations in homologous recombination (HR) pathway genes are considered to be associated with platinum-based chemotherapy response and the prognosis of patients with PDAC. 17 However, Cox analysis did not reveal a significant correlation between HR pathway alterations and prognosis in PDAC patients receiving second-line therapy with SOX or mFFX (Table S5). In the subgroup analysis based on the top 10 most common gene alterations and HR pathway alterations, the PFS and OS benefits of mFFX remained consistent across all subgroups (Figures S3 and S4). Furthermore, molecular features, including HR pathway alterations, were not associated with the tumor response to second-line chemotherapy (Figure S5).
Discussion
The prognosis of pancreatic cancer patients is extremely poor. We have been committed to optimizing the treatment strategy for pancreatic cancer, whether it is adjuvant or palliative chemotherapy.18–20 Nab-paclitaxel has been found to improve the distribution and penetration of gemcitabine in tumors, thereby exerting a synergistic antitumor effect with gemcitabine in murine models of pancreatic cancer.21,22 GnP is currently the most prevalent first-line chemotherapy regimen for advanced PDAC. 5 However, the optimal second-line treatment for PDAC progression after GnP treatment has not yet been determined, although oxaliplatin-based regimens are currently commonly used. 23 In this study, 113 patients with advanced PDAC refractory to GnP therapy were included. Our results demonstrated the superiority of mFFX in extending survival compared with that of SOX (median PFS: 4.8 vs 2.4 months, p = 0.001; median OS: 10.4 vs 6.1 months, p = 0.001). The median OS and PFS for advanced PDAC patients included in the present study are in line with the survival results reported in previous studies.8,12 However, some of the baseline characteristics in our study differed between the SOX and mFFX groups. The mFFX regimen tended to be administered to younger patients with higher BMIs and serum albumin level in our center. Notably, the median PFS and OS were still significantly longer in the mFFX group even after using the PSM method to control for confounding factors. Multivariate Cox analysis also suggested that the mFFX regimen was an independent predictor of better PFS and OS for PDAC patients after GnP therapy failure. However, the tumor response to first-line GnP was associated with OS, but not PFS, for PDAC patients receiving second-line chemotherapy. This suggests that the biological behavior of the tumor is directly related to the patient’s survival time (OS), but appropriate second-line treatment can prevent rapid tumor progression, contribute to longer PFS, and potentially prolong OS.
There is a concern that the toxicity may be intolerable for elderly patients with poor performance status. 24 Consistent with previous studies,8,25 grade 3 or 4 AEs were more frequent in the mFFX group (68.6% vs 44.2%, p = 0.044), especially neutropenia and diarrhea. No new safety signals were observed in this study, and no patients died due to AEs. Interestingly, the results of subgroup analyses suggested that young patients with a good performance status may have greater survival benefits when receiving second-line mFFX, while elderly patients may still benefit from second-line mFFX. However, our study included only three patients with an ECOG score of 2, all of whom received the SOX regimen. Of these three patients with an ECOG score of 2, safety data were available for 2, and one of them developed grade 3 AE (thrombocytopenia). The proportion of elderly patients in the mFFX group was relatively small. Therefore, caution is still needed when attempting the mFFX regimen in elderly individuals with poor performance status. On the other hand, although the dosage of oxaliplatin used in this study is consistent with that used in previous studies on second-line treatment of PDAC, 12 it is lower than that used for advanced gastric cancer.26,27 Considering the acceptable safety of the SOX regimen, increasing the dose of oxaliplatin may achieve better efficacy for fit patients.
In addition to survival outcomes, the treatment responses of patients with advanced PDAC receiving second-line chemotherapy were also explored. Considering that advanced PDAC patients achieve less PR or CR after chemotherapy,8,12,28 we focused on the DCR and CA19-9 response. Our results suggested that the DCR in the mFFX group was significantly greater than that in the SOX group (68.8% vs 40.0%, p = 0.005), but there was no significant difference in the CA19-9 response (defined as a ⩾30% decrease from the baseline) rate between the two groups (33.3% vs 31.0%). The DCR achieved by second-line chemotherapy is numerically greater than the CA19-9 response rate, which indicates the dilemma of treating advanced PDAC. It is still difficult to reduce the tumor dimension and CA19-9 response to intensive chemotherapy (mFFX). Thus, novel therapeutic targets and drugs are urgently needed to improve the treatment response of patients with PDAC. Moreover, although the mFFX group had better survival outcomes, they did not achieve higher CA19-9 response rates, indicating the need to optimize the definition and threshold of the CA19-9 response. 29
A comprehensive understanding of the molecular features of PDAC is urgently needed to guide treatment strategies and discover promising prognostic markers. 30 We described the molecular features of 45 PDAC patients receiving second-line chemotherapy with available NGS genetic testing data. There was no artificial selection bias in this subgroup. We found that ARID1A, INPP4A, NTRK2, and PTPRS alterations may be prognostic factors for poor prognosis, while HR pathway alterations were associated with a trend toward longer survival. We also attempted to explore the role of gene mutations in guiding the selection of second-line chemotherapy regimens. However, regardless of the presence of gene mutations, patients receiving mFFX had better survival outcomes than those treated with SOX. ARID1A alterations occur in approximately 6% of cancers, and the alteration rate may exceed 10% in PDAC.31,32 Consistent with our results, it has been suggested that ARID1A alterations may mediate resistance to platinum-based chemotherapy. 31 Moreover, ARID1A alterations were not associated with PFS in patients treated with first-line GnP (data not shown). Therefore, for patients who carry ARID1A alterations, alternative chemotherapy options should be prioritized over platinum-based chemotherapy. Moreover, PTPRS alterations activate the ERK and AKT signaling pathways and contribute to tumor cell survival and proliferation, which are associated with poor prognosis in cancer patients.33,34 In contrast, the role of INPP4A and NTRK2 mutations in PDAC has not been confirmed. It should be noted that none of these biomarkers can indicate the use of FDA-approved drugs. Moreover, the sample size of patients who underwent NGS testing is limited; therefore, the clinical utility of the aforementioned molecular markers has certain limitations.
In the present study, we reported the real-world outcomes of patients with advanced PDAC refractory to GnP therapy treated with SOX or mFFX as second-line chemotherapy at a single center. This was the first study to compare the efficacy and safety of SOX with those of mFFX. However, there were several limitations to this study. First, this was a retrospective and nonrandomized study with inherent selection bias. Second, the incidence of AEs might be affected by monitoring bias. Additionally, we were unable to collect dose adjustment or dose intensity data, which will inevitably affect the interpretation of safety data. Third, this study only reviewed patients from a single center, and the sample size was too small to draw reliable conclusions even after adjusting for confounding factors using PSM. Furthermore, there may be differences in the pharmacodynamics and pharmacokinetics of S-1 between Caucasian and East Asian patients. 35 Consequently, the efficacy of S-1 in combination with gemcitabine as a first-line chemotherapy regimen warrants re-evaluation in non-Asian patients. Randomized controlled trials (RCTs) are mandatory to overcome the above limitations and confirm the conclusions of this study. In addition, the NAPOLI-1 study revealed that nal-IRI/FL achieved a significant survival benefit compared with 5-FU/LV monotherapy in advanced PDAC patients who were previously treated with gemcitabine-based therapy. 36 However, nal-IRI/FL was not included in this study due to insufficient sample size, and this therapy has not been widely used in China. RCTs directly comparing mFFX with nal-IRI/FL in the second-line setting are warranted to facilitate the selection of an optimal regimen for patients with gemcitabine-refractory PDAC. On the other hand, the NAPOLI-3 study found that NALIRIFOX as first-line treatment for metastatic PDAC showed significantly better efficacy than GnP, supporting the early application of effective combination regimens; furthermore, optimizing drug structure or dosages may bring better prognosis to pancreatic cancer patients. 37
Conclusion
In summary, the results of the present study suggested that the mFFX regimen, a second-line chemotherapy after GnP therapy failure, was associated with significantly longer survival than the SOX regimen but was associated with a greater incidence of severe AEs. Caution is still needed when attempting the mFFX regimen in elderly individuals with poor performance status. Further prospective studies are warranted to determine the optimal second-line chemotherapy regimen for patients with advanced PDAC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251378698 – Supplemental material for S-1 plus oxaliplatin versus modified FOLFIRINOX for advanced pancreatic adenocarcinoma after gemcitabine plus nab-paclitaxel failure
Supplemental material, sj-docx-1-tam-10.1177_17588359251378698 for S-1 plus oxaliplatin versus modified FOLFIRINOX for advanced pancreatic adenocarcinoma after gemcitabine plus nab-paclitaxel failure by Hui Tang, Zhengxia Li, Tingting You, Jinrong Ying, Mingming Yuan, Yuejuan Cheng, Yingyi Wang, Huanwen Wu and Chunmei Bai in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Thanks to the help offered by Jingya Zhou from the Department of Medical Record of Peking Union Medical College Hospital for providing the data retrieval services through the Electronic Medical Record Analytical Database (PUMCH-EMERALD).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
