Abstract
The current standard of care for the management of estrogen receptor (ER)-positive and human epidermal growth factor receptor 2 (HER2)-negative breast cancer has been redefined by the introduction of cyclin-dependent kinase 4/6 (CDK4/6) inhibitors. Although adults aged 65 years and older account for the majority of patients with breast cancer, limited data are available about the age-specific dosing, tolerability, and benefit of CDK4/6 inhibitors in this growing population. Older adults are under-represented in clinical trials and as a result, clinicians are forced to extrapolate from findings in younger and healthier patients when making treatment decisions for older patients. In this article, we review the limited age-specific evidence on the efficacy, toxicity, and quality of life (QoL) outcomes associated with the use of CDK4/6 inhibitors in older adults. We also describe ongoing trials evaluating CDK4/6 inhibitors in the older population and highlight that only a minority of adjuvant and metastatic trials of CDK4/6 inhibitors in the general breast cancer population includes geriatric assessments. Finally, we propose potential strategies to help guide decision making for fit and unfit older patients based on disease endocrine sensitivity, the need for rapid response and geriatric assessment.
Introduction
Aging is a risk factor for cancer whose incidence will increase in the next decades due to the increasing longevity of the population.1,2 Breast cancer is the most common cancer diagnosed in women. 3 According to the Surveillance, Epidemiology and End Results Program (SEER), there were 266,120 new breast cancer diagnoses in 2018 in the United States, which constitute 15.3% of new cancer diagnoses. 3 Over 40% of breast cancers are diagnosed in older patients (65–74 years: 24.1%, 75–84 years: 13.6%, 84+ years: 5.5%) and over 50% of breast cancer deaths occur in the older adult population (65–74 years: 24.1%; 75–84 years: 19.7%; 84+ years: 16.9%).
Approximately 80% of breast cancers are estrogen receptor (ER) positive and 20% are human epidermal growth factor receptor 2 (HER2) positive. 4 ER-positive breast cancer patients tend to be older, have well-differentiated disease, smaller tumor size and negative lymph node status, which, along with a more indolent biology, contributes to the better survival compared with other disease subtypes. 4 Yet, older women account for the majority of breast cancer deaths despite significant advances having been achieved in its diagnosis and treatment. In fact, older adults are historically under-represented in clinical trials, and reports indicate that the accrual rates of older adults have not improved in the last decade.5,6 The age of older patients enrolled in registration trials supporting United States Food and Drug Administration (FDA) approval from 2005 to 2015 continues to be low (65–74 years: 17%; 75–79 years: 3%; 80+: 1%). 7 Furthermore, older patients on clinical trials may be fitter and not necessarily representative of the older population as a whole. It is also well recognized that older patients with breast cancer may have impairments that correlate with chemotherapy toxicity 8 and are frequently underdetected in routine practice. 9 These discrepancies between patients seen in the real world and trial participants affect the applicability of therapeutic recommendations for older patients. 10
Cyclin-dependent kinase 4/6 (CDK4/6) inhibitors have been a significant breakthrough in the management of ER-positive, HER2-negative breast cancer. In this review, we discuss the rationale for targeting CDK4/6 and clinical trials evaluating CDK4/6 inhibitors, highlighting efficacy, toxicity, and quality of life (QoL) outcomes relevant to older adults based on age-specific data published in literature or provided by the manufacturing companies where available.
Targeting CDK4/6
One of the hallmarks of cancer is the ability to circumvent programs regulating cell proliferation, senescence, and apoptosis. 11 Each step of the cell cycle has checkpoints strictly governed by CDK, which include a cyclin (protein) and a kinase (enzyme). 12 A critical step is the transition from the G1 phase, in which cells grow, to the S phase, in which deoxyribonucleic acid (DNA) replication occurs. This step requires binding of cyclin D1 to CDK4 and/or CDK6, phosphorylation of the retinoblastoma protein (Rb), and activation of the E2F transcription factor, all of which promote cellular transition through the G1 phase (Figure 1). 13 In normal tissues, cell-cycle checkpoints provide mechanisms to halt cellular growth and to remove damaged cells by apoptosis. In malignant cells, however, checkpoint defects lead to uncontrolled growth and tumorigenesis. 14
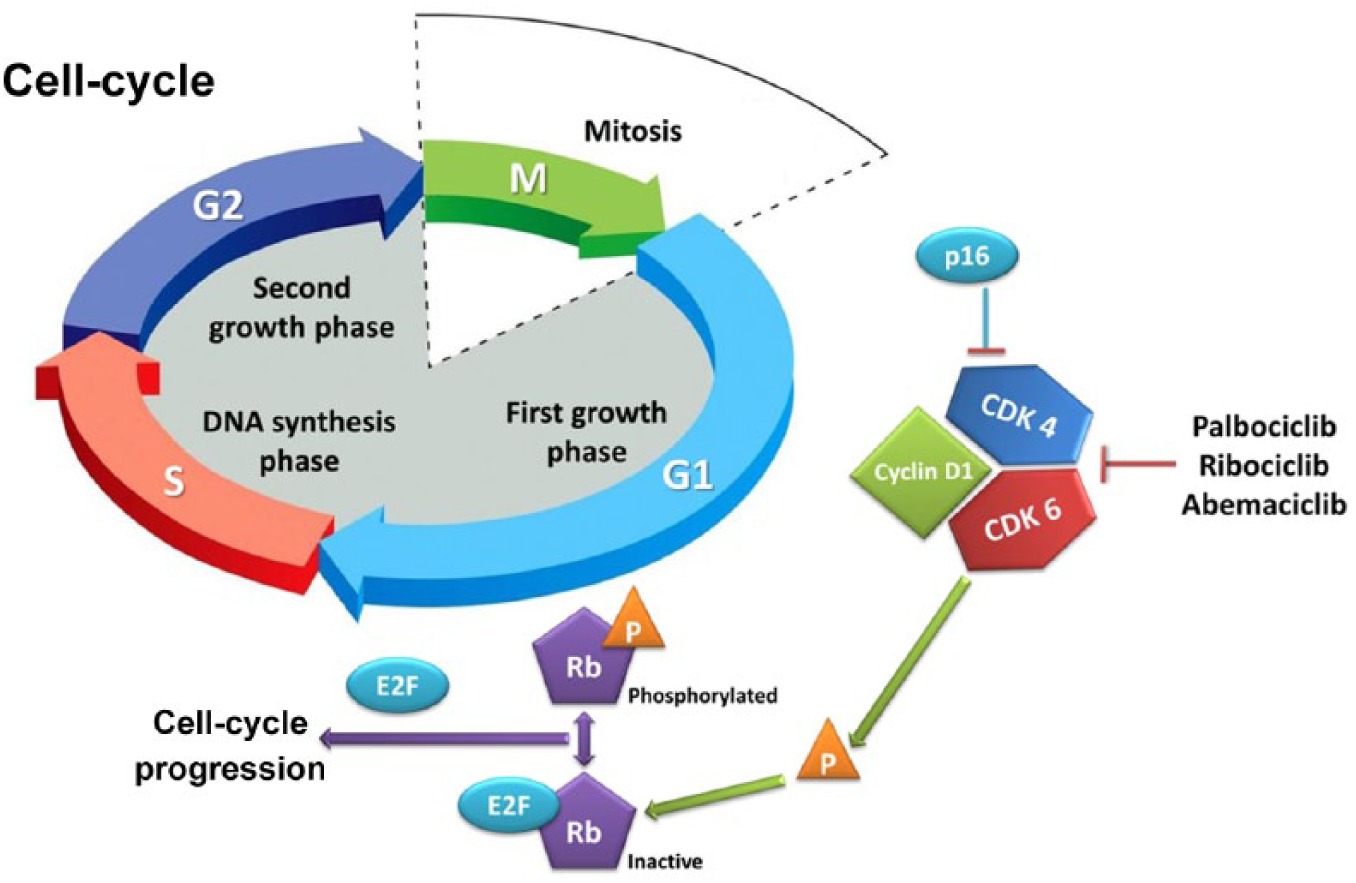
The retinoblastoma-E2F pathway.
Cyclin D1 is overexpressed or amplified in a significant proportion of breast cancer, particularly in the ER-positive subtype.15,16 Additionally, antiestrogen medications rapidly downregulate cyclin D1, and deregulated cyclin D1 expression may cause resistance to these agents. Since CDK4 and 6 are important for cyclin-mediated G1–S transition, their inhibition represents an attractive therapeutic option, particularly in ER-positive breast cancer. 17 The first CDK4/6 inhibitor, palbociclib, inhibited the growth of ER-positive breast cancer cell lines, and subsequently demonstrated its efficacy in clinical trials. Since the advent of palbociclib, two additional CDK4/6 inhibitors (ribociclib and abemaciclib) have been approved for use in ER-positive breast cancer. 18
The cell cycle represents a convergent point for both cancer and aging, 19 suggesting that normal aging may interact with cell-cycle targeting. CDK4/6 inhibition leads to a senescent-like cellular phenotype, which, in contrast with that induced by aging, may be reversible upon drug discontinuation. 18 Aging has various effects on cell-cycle components which may also influence the efficacy of CDK4/6 inhibitors. The expression of the cell-cycle regulator (and CDK4/6 inhibitor) p16Ink4a in lymphocytes, for example, exponentially increases with chronological age. 20
Although CDK4/6 inhibitors act on one of the interfaces between cancer and aging, the effect of cellular senescence on their efficacy has not been adequately studied. Additionally, little is known about the influence of chronological and biological age on CDK4/6 inhibitors’ toxicity.
Potential challenges of CDK4/6 inhibition in older patients
The most common toxicity of CDK4/6 inhibitors palbociclib and ribociclib is myelosuppression, since CDK6 is responsible for the promotion of hematologic precursor proliferation. 21 Neutropenia is mediated by cell-cycle arrest without apoptosis, a mechanism distinct to that seen in chemotherapy-related agranulocytosis. 22 It is dose related, not cumulative, and its severity often decreases in consecutive cycles. Generally, for grade 1/2 toxicities, no treatment interruptions or dose adjustments are needed. In the case of grade 3 neutropenia on palbociclib, dose adjustments are not required except on day 1 or with concomitant fever. In the case of ribociclib, dose should be held until recovery to grade ⩽ 2 and ribociclib should be resumed on a reduced dose. For abemaciclib, dose should be held until recovery to grade ⩽ 2 and resumed on a reduced dose if grade 3 neutropenia is recurrent. In the case of grade 4 neutropenia, all three drugs require dose interruption until recovery to grade ⩽ 2 followed by dose reduction.23–25 Despite the high rates of neutropenia, febrile neutropenia remains rare, reflecting the mechanistic differences compared with chemotherapy. Other toxicities associated with CDK4/6 inhibitors include: fatigue, mucositis, gastrointestinal toxicities, liver function test abnormalities, and QT prolongation. The rates and significance of these in older patients with the individual agents is discussed in the following sections.
Palbociclib undergoes hepatic metabolism involving oxidation and sulfonation through the cytochrome P450 3A4 (CYP3A) and sulfotransferase enzyme SULT2A1. 23 Ribociclib and abemaciclib are also metabolized in the liver, mainly through oxidation via CYP3A4.24,25 Feces is the major route of excretion of CDK4/6 inhibitors. No dose adjustments are required in cases of mild hepatic impairment [total bilirubin ⩽ upper limit of normal (ULN) and AST > ULN, or total bilirubin > 1.0–1.5 × ULN and any AST], which does not impact on their exposure; however, the pharmacokinetics of palbociclib has not been studied in the presence of moderate to severe hepatic impairment. In patients with Child–Pugh B or C, it is recommended to start ribociclib at a reduced dose (400 mg). In patients with Child–Pugh C, it is recommended that abemaciclib be administered once daily.23–25 Similarly, no dose adjustments are required in cases of mild to moderate renal impairment [30 ml/min ⩽ creatinine clearance (CrCl) < 90 ml/min]. The impact of severe renal impairment (CrCl < 30 ml/min) on the pharmacokinetics and exposure of the three drugs is unknown.23–25 Of interest, abemaciclib was shown to inhibit renal tubular secretion transporters and can increase serum creatinine without affecting the glomerular filtration. 26 This typically occurs within the first 28 days of treatment and is reversible upon discontinuation. In older adults, this may be erroneously interpreted as renal impairment. Therefore, the measurement of alternative markers including urea, cystatin C and calculated glomerular filtration rate may be considered.
The aging process is associated with decreased physiologic reserve of multiple systems, which may also be affected by comorbidities, drug interactions and cancer itself. Data evaluating the pharmacokinetics and safety of CDK4/6 inhibitors in older adults are lacking. Of particular importance is the fact that the decreased bone marrow reserve seen in older patients may enhance the risk of myelosuppression, which is a common side effect of this class of drugs. Liver metabolism also decreases with aging and this can lead to increased drug exposure and adverse events. The progressive decreases in glomerular filtration rate and renal blood flow may potentially enhance the severity of dehydration in older patients experiencing diarrhea or nausea as a result of treatment. The prolongation of the corrected QT (QTc) interval intrinsic with aging needs to be considered, as it may possibly increase the risk of cardiac adverse events in older patients receiving ribociclib.
A key consideration prior to initiating CDK4/6 inhibitors and when managing toxicities is the potential for drug interactions. This is of particular importance in older patients who are more likely to be taking concurrent medications. Particular care must be taken when drugs affecting the function of CYP3A4 are used concomitantly with CDK4/6 inhibitors. Their hepatic metabolism may be affected by various pharmacological agents which are frequently used in older adults (Table 1). 27 Concomitant use of strong CYP3A4 and CDK4/6 inhibitors should be avoided, and alternative therapeutic approaches should be considered. However, if CYP3A4 inhibitors must be administered then dose reductions of CDK4/6 inhibitors are recommended. A list of potentially relevant drugs is shown in Table 1. One particularly common scenario would be the use of direct oral anticoagulants concurrently with CDK4/6 inhibitors. CYP3A4 is involved in the metabolism of apixaban and rivaroxaban, with a minimal role in the metabolism of edoxaban, which is eliminated instead mainly via the efflux transporter P-glycoprotein (P-gp)/ABCB1 system. Palbociclib, ribociclib and abemaciclib inhibit P-gp but are only weak, time-dependent inhibitors of CYP3A4. No data about interactions with edoxaban are available, although the inhibition of P-gp mediated by CDK4/6 inhibitors can theoretically lead to greater exposure to edoxaban. Therefore, either apixaban or rivaroxaban would be a better choice if patients require concomitant oral anticoagulation. While the consideration of concurrent medications is relevant to all patients, the higher rate of comorbid health conditions and therefore polypharmacy in older patients makes this issue particularly pertinent in this population.
Potential drug interactions with CDK4/6 inhibitors and their management. 27

AVP, arginine vasopressine; CDK4/6, cyclin-dependent kinase 4/6; 5-HT, 5-hydroxytryptamine; mTOR, mammalian target of rapamycin; MTP, microsomal triglyceride transfer protein; SERM, selective estrogen receptor modulator; SSRI, selective serotonin reuptake inhibitor; PDE, phosphodiesterase.
Palbociclib
Palbociclib was the first CDK4/6 inhibitor to be investigated. However, specific evidence to guide its use in older adults is limited. The phase II PALOMA-1 study tested the safety and efficacy of palbociclib in combination with letrozole versus letrozole alone in the first-line setting for patients with ER-positive HER2-negative advanced breast cancer. 28 A total of 76 out of 165 enrolled patients were aged 65+ and derived a median progression-free survival (PFS) of 26.2 months [95% confidence interval (CI) 12.6–not reached (NR)] with palbociclib plus letrozole versus 12.9 months (95% CI 5.7–22.2) with letrozole alone. Grade 3–4 adverse events, dose reduction and discontinuation rates were not influenced by age.
Based on the positive results from PALOMA-1, a large phase III randomized double-blind study (PALOMA-2) was initiated. This study recruited 666 postmenopausal treatment-naïve women with ER-positive, HER2-negative advanced breast cancer. Compared with letrozole and placebo, the combination of palbociclib and letrozole was associated with improved PFS [24.8 versus 14.5 months; hazard ratio (HR) 0.58; 95% CI 0.46–0.72] and objective response rate (ORR; 42% versus 35%). 29 Neutropenia was common with the combination compared with letrozole alone (89.5% versus 6.3%), along with increased rates of fatigue, nausea, and alopecia. Overall survival (OS) data are still immature. The PALOMA-2 study enrolled 262 patients (39.3%) aged 65+, who represented 40.8% (n = 181) of those enrolled in the combination arm and 36.5% (n = 81) in the placebo arm. The median age in the experimental arm was 62 years (range: 30–89) and 61 years (range: 28–88) in the placebo arm. In the subgroup analysis, the benefit in PFS was maintained in patients 65+ with an HR of 0.57 (95% CI 0.39–0.84) versus 0.57 (95% CI 0.43–0.74) in patients younger than 65. No age-specific data about toxicities in first line are available.
The double-blind phase III PALOMA-3 randomized 521 women with ER-positive, HER2-negative advanced breast cancer to either palbociclib and fulvestrant or placebo and fulvestrant.30,31 These patients either relapsed during or within 1 year after completion of adjuvant endocrine therapy or progressed on prior palliative hormonal treatment. The combination improved median PFS (9.5 versus 4.6 months; HR 0.46; 95% CI 0.36–0.59), but with higher rates of neutropenia (65% versus 1%, respectively) and fatigue (39% versus 28%). OS data are still immature. One hundred and twenty-nine patients (24.8%) were aged 65+. In the experimental arm, the median age was 57 (range: 30–88) and in the placebo arm it was 56 (range: 29–80). Individuals aged 65+ represented 24.8% (86 patients) and 24.7% (43 patients) of the group, respectively. In the subgroup analysis, older patients derived PFS benefit with an HR of 0.35 (95% CI 0.19–0.62) compared with 0.44 (95% CI 0.32–0.61) in patients younger than 65.
In the PALOMA-3 study, age did not significantly increase the risk of grade 3–4 neutropenia. In patients aged younger than 50, 50–69, and 70+, grade 3–4 neutropenia occurred in 28.7, 57.4, and 31.9%, respectively. 32 In multivariate analysis, a trend for association of grade 3–4 neutropenia and infections was detected in patients aged 70+ versus 50–69 and in patients aged 70+ versus younger than 50, although it was not statistically significant.
In a pooled analysis of data from 872 patients enrolled in the randomized phase II and III studies (PALOMA-1, -2 and -3) investigating safety and efficacy of palbociclib plus letrozole or fulvestrant in ER-positive, HER2-negative breast cancer, 221 (25%) of the participants were aged 65–74 and 83 (10%) were aged 75+. 33 In the first-line setting for all patients, median PFS was 24.4 months (95% CI 22.0–26.2) in the combined palbociclib and letrozole group versus 13.6 months (95% CI 11.1–16.4) in the placebo and letrozole group with an HR of 0.53 (95% CI 0.44–0.64). In the pretreated group, median PFS was 9.5 months (95% CI 9.2–11.0) in the combined palbociclib and fulvestrant group versus 4.6 months (95% CI 3.5–5.6) in the placebo and fulvestrant group with an HR of 0.46 (95% CI 0.36–0.59). The PFS benefit was observed in patients aged 65–74 and 75+. No new safety concerns were identified in the older population, and no more than 5% had grade 3–4 adverse events except for neutropenia and leukopenia. Neutropenia occurred in 81% of patients treated with palbociclib compared with 5% in the control group; these were consistent in both age groups (77% versus 1% in patients aged 65–74; 90% versus 3% in those aged 75+). Eleven patients (1%) experienced febrile neutropenia on palbociclib compared with none in the placebo group. A total of 166 patients (19%) treated with palbociclib developed serious adverse events of any grade (25% of those aged 65–74 and 30% of those 75+), and 77 patients (9%) had to discontinue it owing to adverse events compared with 22 (5%) in the control group. Neutropenia remained the most frequent reason for discontinuation in 14 patients (1.6%) [4 patients (1.8%) aged 65–74; 4 patients (4.8%) aged 75+]. The average clearance of palbociclib was lower for older patients, but the observed differences were unlikely to be clinically relevant in view of the observed safety and efficacy profile.
An FDA pooled analysis of two studies of CDK4/6 inhibitors also suggested similar efficacy and safety in older women (aged 65+ and 70+) compared with their younger counterparts, although greater serious adverse events and discontinuations occurred in patients aged 65+. 34 A recent retrospective analysis of 160 patients aged 65+ (including 92 aged 70+) revealed similar findings. 35
The QoL data for palbociclib trials have not been reported by age. In PALOMA-3, using the EORTC QLQ-C30 and BR23 measures, patients in the palbociclib group had less reduction in global QoL from baseline, and experienced a longer time to deterioration of QoL, compared with the placebo group. 36 Notably, both groups experienced significant worsening of cognitive and role functioning from baseline. In the palbociclib group, there was a significant decrease in pain from baseline (which was also shown in the PALOMA-2 trial), and improvement in emotional functioning. In PALOMA-2, the two treatment groups did not differ in the QoL change from baseline using Functional Assessment of Cancer Therapy—Breast cancer (FACT-B), Functional Assessment of Cancer Therapy (FACT)—General, and EuroQOL—5 dimensions (EQ-5D) scales. 37 However, the palbociclib group experienced significant improvement in pain scores. In both treatment groups, patients without progression of disease had delayed worsening of QoL measured by FACT-B. To what extent these QoL benefits are experienced by older patients has not been described. However, it seems reasonable to conclude that similar benefits are likely, in particular, as use of a CDK4/6 inhibitor may defer the time to cytotoxic chemotherapy in older patients.
Ribociclib
Ribociclib is a CDK4/6 inhibitor with similar preclinical properties to palbociclib. To date, ribociclib has been evaluated in three pivotal trials: MONALEESA-2 and MONALEESA-3, and MONALEESA-7.38–40 Based on the findings of the MONALEESA-2 trial, ribociclib was approved by the FDA in March 2017 41 and the European Medicines Agency in August 2017 42 for use in combination with an aromatase inhibitor (AI) as a first-line treatment for HR-positive/HER2-negative advanced or metastatic breast cancer in postmenopausal women. Recently, the FDA has also approved its use in combination with fulvestrant in first or second line, based on the MONALEESA-3 study findings. 24 Despite pronounced changes in drug metabolism, absorption, and distribution with increasing age, limited evidence exists to guide therapy with ribociclib in the increasing number of older adults with breast cancer. Clinical trials evaluating ribociclib primarily included women less than age 70. Thus, there exists a significant knowledge gap in the safety and tolerability of delivering ribociclib to older adults with cancer.
In the phase III, randomized, double-blinded MONALEESA-2 study, 668 postmenopausal women ranging from ages 23–91 (median: 62 years) with advanced ER-positive/HER-2 negative breast were assigned to receive either letrozole and ribociclib or letrozole and placebo. Of 334 patients who received ribociclib and letrozole, 150 patients (45%) were 65+, and per the package insert, only 35 patients (11%) were 75+. Moreover, there has been no published cohort data for enrolled patients aged 70+. Similarly, in MONALEESA-3, among the 484 patients who received ribociclib and fulvestrant, the median age was 63 (range: 31–89), but unlike in MONALEESA-2, there are currently no published data on women 65+ who participated in this trial.
A subgroup analysis of the efficacy and safety of MONALEESA-2 patients was conducted based on prespecified age cut off of 65, which included 373 patients less than age 65 years and 295 patients aged 65+.43,44 Among patients receiving ribociclib and letrozole, the median PFS for patients aged 65+ was NR (95% CI 19.3–NR) compared with 18.4 months (95% CI 15.0–NR) in older patients who received letrozole alone. There was no significant difference in the effect of ribociclib treatment on PFS between older (65+; HR 0.608; 95% CI 0.394–0.937) and younger (younger than 65; HR 0.523; 95% CI 0.378–0.723) patients. ORR was greater in the ribociclib arm across both age groups [younger than 65: ribociclib arm, 44% (95% CI: 36–51) and letrozole-only arm 25% (95% CI: 19–31); 65+ years: ribociclib arm, 37% (95% CI: 30–45) and letrozole-only arm, 31% (95% CI: 24–39)]. 44 The maintained PFS benefit in patients 65+ was confirmed on a more updated analysis with an HR of 0.658 (95% CI 0.466–0.928). 38
Among the three pivotal trials studying ribociclib, the most common adverse events reported are neutropenia, leukopenia, and fatigue.38–40 In particular, MONALEESA-2 patients receiving combination ribociclib and letrozole versus experienced grade 3–4 neutropenia (59.3% versus 0.9% in letrozole alone), leukopenia (21% versus 0.6% in letrozole alone), and fatigue (2.4% versus 1% with letrozole). 38 Patients receiving ribociclib with letrozole experienced a pulmonary embolism at a rate of 0.6%. Among patients in the ribociclib and letrozole group, 7.5% discontinued due to adverse events, whereas only 2.1% of the letrozole group discontinued the study. In MONALEESA-2, The safety profile of the subgroup analysis by the prespecified age cut off of 65 did not show any apparent differences in systemic exposure related to age. Nausea, alopecia, diarrhea, and vomiting were adverse events reported in >10% of patients in the ribociclib arm over the placebo arm, regardless of the age subgroup. However, there was a >10% increase in the incidence of fatigue and grade 1–2 anemia reported in the ribociclib plus letrozole over the placebo plus letrozole arm in patients 65+. 44 The incidence of anemia, hypertension, and asthenia were also higher in older patients, although this was irrespective of treatment arm.
Cardiac toxicity in the form of QT-interval prolongation has been reported with ribociclib, which may cause cardiac repolarization abnormalities by affecting subunits of voltage-gated channels. 45 Concomitant use of QT-prolonging medications, such as those shown in Table 1, should be avoided. In the phase III MONALEESA-2 trial, one case of sudden cardiac death was reported 46 and found to be related to concomitant use of methadone.46,47 An electrocardiogram (ECG) is required at baseline and treatment can be initiated in cases where the QTc interval is <450 msec on Fridericia’s formula (QTcF); repeat ECGs are recommended on cycle 1, day 14, and cycle 2, day 1, and subsequently as clinically indicated. In cases where the QTcF > 480 msec, a dose interruption is indicated until resolution. In cases where the QTcF > 500 msec, a dose interruption followed by a dose reduction is recommended. 24 For the same reason, serum electrolytes need to be monitored and abnormalities should be corrected, especially during the first six cycles of treatment. 24 Older adults have a higher baseline risk of QT prolongation due to increasing age itself, as well as pre-existing heart conditions.48–50 Therefore, in older patients with cardiac comorbidities and polypharmacy, ribociclib must be used with caution after a cardiology evaluation, or alternative options including palbociclib can be considered. In cases of long-QT syndrome, recent myocardial infarction, congestive heart failure, unstable angina, bradyarrhythmias or electrolyte abnormalities, the use of ribociclib should be avoided.
Health-related QoL (HRQoL) data in patients 65+ enrolled in MONALEESA-2 were presented at the 2018 American Society of Clinical Oncology Annual Meeting. 38 Based on the EORTC QLQ-C30 and BR-23, HRQoL was similar in the two treatment groups. A clinically meaningful improvement was noted for pain in the ribociclib group through the first year of treatment. Both groups had an approximate 28-month median time to deterioration of HRQoL.
Although age-specific data show that patients 65+ benefit from ribociclib and have similar toxicity profile compared with younger patients, very few patients 70+ were included. Because of this, further efforts are needed to prospectively evaluate the safety and tolerability of ribociclib and endocrine therapy in older adults 70 years and above. In this respect, the phase IIIb COMPLEEMENT-1 trial has recruited over 3000 patients, of whom over 20% were aged 70+, and all of whom received letrozole and ribociclib. The results of this study are awaited.
Abemaciclib
One single-arm phase II single-agent study (MONARCH-1) and two randomized studies of abemaciclib plus endocrine therapy have been published (MONARCH 2 and 3). Despite this, the evidence to guide the use of abemaciclib in older patients with breast cancer is, again, limited.
The phase III, randomized, double-blinded MONARCH-2 study included 669 patients with ER-positive, HER2-negative advanced breast cancer who progressed on adjuvant or neoadjuvant endocrine therapy, or progressed following first-line palliative endocrine therapy. 51 The study included 245 patients (36.6%) aged 65+. Results demonstrated a statistically significant improvement in PFS of 16.4 versus 9.3 months (HR 0.55; 95% CI 0.45–0.68). Subgroup analyses showed no statistically significant differences in benefit between younger and older patients (HR 0.52 and 95% CI 0.42–0.68 in patients younger than 65 versus HR 0.62 and 95% CI 0.41–0.94 in patients 65+).
The phase III, randomized, double-blinded MONARCH-3 study included 493 patients with ER-positive, HER2-negative advanced breast cancer. The median age of patients was 63 years (range: 32–88) and they had an Eastern Cooperative Oncology Group performance status of 0 or 1. There was a statistically significant improvement in median PFS with abemaciclib in combination with endocrine treatment versus placebo plus endocrine treatment (HR 0.54; 95% CI 0.41–0.72). 52 Subgroup analyses showed no differences in efficacy between patients younger than 65 and 65+ (HR 0.53 and 95% CI 0.33–0.77 versus HR 0.57 and 95% CI 0.36–0.90, respectively).
There are no age-specific toxicity data which have been published. In the MONARCH-1 study, all patients had at least one treatment-related adverse event; most importantly, diarrhea (grade 3 in 19.7% of cases), fatigue, nausea, and grade 1–3 renal dysfunction. Similarly, 98.8% of patients treated with abemaciclib in the MONARCH-2 trial and 98.5% in the MONARCH-3 study had an adverse event (grade 3 in 48.5% and grade 4 in 6.4% of patients), especially diarrhea, neutropenia, fatigue, and infections.
It is worth noting that gastrointestinal toxicities, including nausea, vomiting and diarrhea, are more commonly associated with abemaciclib than with the other CDK4/6 inhibitors (although there are no direct comparative data). This different toxicity profile may be due to the fact that abemaciclib has higher selectivity for CDK4, which is the main regulator of the intestinal cell cycle. 53 Therefore, while abemaciclib-associated myelosuppression is less prevalent, gastrointestinal toxicity and fatigue are more common.51,52 Gastrointestinal toxicities should be managed with standard nonpharmacological interventions and antidiarrhoeal agents, and clinically relevant drug interactions should be considered (Table 1). 22 Prophylactic antidiarrhoeal treatment is usually not necessary, but clinicians should consider that among older patients, even low-grade toxicities such as grade 2 diarrhea may lead to functional decline, and more aggressive interventions may be necessary in this population. 54 Data recently presented from MONARCH-2 using the EORTC QLQ-C30, BR-23, and Brief Pain Inventory short form showed no significant difference in HRQoL between the two treatment groups; however diarrhea, appetite loss, and nausea/vomiting were worse in the abemaciclib arm. 55 These data were not specific to older patients. Further analysis indicated appetite loss and nausea/vomiting were worse with early cycles and returned to near baseline after cycle 7, whereas diarrhea returned to near baseline post-treatment.
Ongoing research
Major efforts are ongoing to further refine the role of CDK4/6 inhibitors in the management of breast cancer, as shown in Table 2.
Ongoing trials of CDK4/6 inhibitors.

Please note that this study has not been registered with ClinicalTrials.gov yet and available information has been derived from a press release. 56
AEs, adverse events; aTPP, activated partial thromboplastin time; CBR, clinical benefit rate; cfDNA, plasma-cell-free deoxyribonucleic acid; CrCl, creatinine clearance; DCR, disease control rate; DFS, disease-free survival; DoR, duration of response; ECOG, Eastern Cooperative Oncology Group; FGFR1, fibroblast growth factor receptor; GFR, glomerular filtration rate; Hb, hemoglobin; HER2−, Human epidermal growth factor receptor 2 negative; HR+, hormone receptor-positive; HRQoL, health-related quality of life; INR, international normalized ratio; LVEF, left ventricular ejection fraction; ORR, objective response rate; OS, overall survival; PDE, ; PFS, progression-free survival; PK, pharmacokinetic; PS, performance status; QoL, quality of life; QTcF, corrected QT interval using Fridericia’s formula; RFS, recurrence-free survival; SAEs, serious adverse events; T-DM1, trastuzumab emtansine; TTF, time to treatment failure; TTP, time to progression; ULN, upper limit of the institutional normal range; VTE, venous thromboembolism.
The sequence of treatments is a key question being addressed by two ongoing studies, and this question is likely to be of great relevance to older patients. It is unclear at this point whether patients should be treated with a CDK4/6 inhibitor in the first-line setting, or after first progression on an AI. The implication for efficacy and QoL needs to be further investigated. To address this, the SONIA trial [ClinicalTrials.gov identifier: NCT03425838] is comparing the use of a CDK4/6 inhibitor plus a non-steroidal AI in first line followed by fulvestrant in second line with a nonsteroidal AI in first line followed by fulvestrant plus CDK4/6 inhibition in second line. The primary endpoint is PFS after two lines of treatment. The observational Treat ER+ight trial [ClinicalTrials.gov identifier: NCT02753686] is also focusing on the treatment patterns and sequence as well as the effectiveness and safety of various treatment options (including CDK4/6 inhibitors) for postmenopausal ER-positive, HER2-negative advanced breast cancer patients in a real-world setting.
Palbociclib trials
A phase II, single-arm trial is investigating the role of palbociclib specifically in an older population of patients [ClinicalTrials.gov identifier: NCT02760030], aimed at determining the treatment failure-free survival of its combination with fulvestrant in women aged 70+ with surgically or medically inoperable ER-positive, HER2-negative breast cancer.
Although not specific for the older population, an observational study [ClinicalTrials.gov identifier: NCT03280303] is evaluating the real-world use of palbociclib and including geriatric assessments for participants aged 70+. In the adjuvant setting, the multicenter, randomized, phase III PALLAS study [ClinicalTrials.gov identifier: NCT02513394] is evaluating the addition of 2 years of palbociclib to letrozole for patients, over letrozole alone. This study is highly relevant to patients with ER-positive, HER2-negative early breast cancer at higher risk of recurrence, which includes also older adults. In the palliative setting, palbociclib is being evaluated in combination with a number of options: tucatinib, an anti-HER2 tyrosine kinase inhibitor (TKI), in first and second line in combination with letrozole for ER-positive, HER2-positive disease in a phase Ib/II study [ClinicalTrials.gov identifier: NCT03054363]; fulvestrant and erdafitinib, a TKI-inhibiting FGFR, in a phase Ib study [ClinicalTrials.gov identifier: NCT03238196]; tamoxifen in a multicenter, nonrandomized, phase II trial [ClinicalTrials.gov identifier: NCT0266866]. Possible differences in pharmacokinetics and pharmacodynamics of palbociclib in postmenopausal Asian patients are being evaluated in the phase III PALOMA-4 trial [ClinicalTrials.gov identifier: NCT02297438] of palbociclib plus letrozole compared with placebo plus letrozole. However, the upper age limit here is 70 years and this may well limit generalizability of its findings. Finally, the phase II PACE (Palbociclib After CDK and Endocrine Therapy) study [ClinicalTrials.gov identifier: NCT03147287] is investigating the role of palbociclib in combination with fulvestrant with or without avelumab upon progression on CDK4/6 inhibitors.
Ribociclib trials
Ribociclib will be specifically evaluated in a population aged 70+ in a single-arm, phase IIa study [ClinicalTrials.gov identifier: NCT03477396] to assess its safety and tolerability along with AIs.
Other ongoing trials are not age specific. In the adjuvant setting, ribociclib is being assessed in the phase II, pilot LEADER study [ClinicalTrials.gov identifier: NCT03285412] along with standard endocrine therapy. In the metastatic setting, the phase I/II TRINITI-1 trial [ClinicalTrials.gov identifier: NCT02732119] is determining if the continued use of ribociclib beyond progression along with second-line everolimus and exemestane is still effective. The multicenter, randomized MAINTAIN trial [ClinicalTrials.gov identifier: NCT02632045] is also assessing ribociclib in combination with fulvestrant upon progression, on AI plus a CDK4/6 inhibitor. Another randomized phase Ib trial [ClinicalTrials.gov identifier: NCT01857193] is exploring the same triplet, as well as the combination of ribociclib with exemestane.
Abemaciclib trials
No trials specifically addressing the role of abemaciclib in older patients are currently ongoing. In the adjuvant setting, abemaciclib is being assessed within the monarchE study [ClinicalTrials.gov identifier: NCT03155997] in combination with standard adjuvant endocrine therapy in high-risk, node-positive patients. As in the PALLAS study, this study is highly relevant to older patients. In the metastatic setting, the MONARCH plus study [ClinicalTrials.gov identifier: NCT02763566] is randomizing postmenopausal women to abemaciclib plus nonsteroidal AI (NSAI) or fulvestrant versus placebo plus NSAI or fulvestrant to compare efficacy of these approaches in first line. Abemaciclib is also being evaluated along with tamoxifen in the phase II nextMONARCH-1 trial [ClinicalTrials.gov identifier: NCT02747004] in pretreated women with ER-positive, HER2-negative breast cancer. In the ER-positive, HER2-positive disease population, the results of the phase II monarcHER trial [ClinicalTrials.gov identifier: NCT02675231] of abemaciclib plus trastuzumab with or without fulvestrant or chemotherapy are awaited.
Conclusion
CDK4/6 inhibitors have revolutionized the management of advanced ER-positive, HER2-negative breast cancer. Nonetheless, specific data about their efficacy and safety profile in older patients are limited and mostly derived by subgroup analyses of the pivotal trials. Based on these analyses, CDK4/6 inhibitors appear to be equally effective in older as in younger patients, with very similar outcomes among the three drugs as shown in Table 3. There are limited data regarding adverse events and toxicity in older patients but available data suggest that older patients seem to derive similar efficacy with either similar to or slightly increased toxicity from these agents compared with their younger counterparts. QoL measures are also lacking; incorporating QoL in trials is important because some older patients may value QoL as important or more important than survival. 57 No trials have specifically evaluated the role of CDK4/6 inhibitors in an older population of patients, who are typically subject to a shorter life expectancy, competing comorbidities, polypharmacy and an increased risk of treatment-related toxicities. Moreover, with only a few exceptions listed in Table 2, many ongoing trials are at risk of excluding older women due to narrow eligibility criteria or the lack of inclusion of geriatric parameters. These will hinder the applicability of their findings to a large population of patients with ER-positive, HER2-negative breast cancer. The lack of biomarkers able to define patients who are more likely to benefit from the addition of a CDK4/6 inhibitor 58 is likely to add further uncertainty to the decision-making process in the older subgroup.
Key efficacy and toxicity outcomes of CDK4/6 inhibitors in older patients based on available data from the pivotal trials.
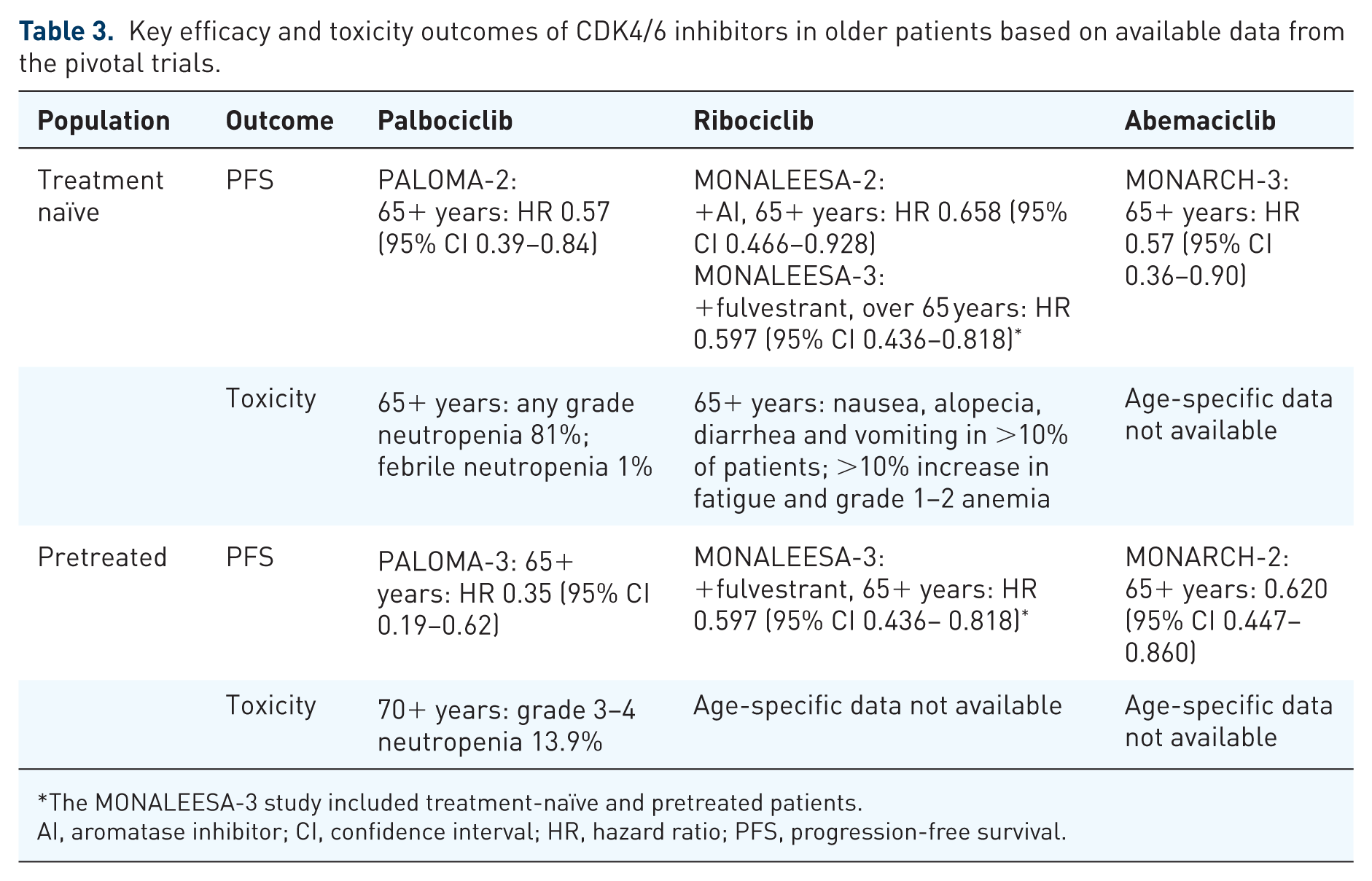
The MONALEESA-3 study included treatment-naïve and pretreated patients.
AI, aromatase inhibitor; CI, confidence interval; HR, hazard ratio; PFS, progression-free survival.
The management of ER-positive, HER2-negative advanced breast cancer should always take into account a number of factors, including patients’ performance status and preferences, life expectancy, comorbidities, previous lines of treatment and derived benefit, disease burden and extent, current symptoms or risk for developing symptoms, and organ function. Differentiating the apparently fit, older individual who is likely to benefit from and tolerate standard therapy, from the seemingly frail, older patient who is likely to experience unexpected side effects and requires a more personalized strategy, is important to reduce over- and undertreatment. 59
Frailty is an increasingly recognized clinical state of vulnerability to stressors such as treatments or serious illness and involves an increased risk for adverse health outcomes, including functional decline and mortality. 60 Despite frailty being increasingly prevalent in older age, chronological age alone does not define frailty. There is no gold standard for diagnosing frailty and multiple assessment tools have been developed, 61 although commonly used measurements are physical function, gait speed, and cognition. 62 The Balducci frailty criteria have traditionally defined vulnerable those individuals with some degree of dependence in routine activities and no more than two comorbidities; whereas frail adults are defined as dependent in activities of daily living (ADLs) and affected by either three or more comorbidities or a geriatric syndrome. 63 On the other hand, the Fried frailty criteria identify frail individuals based on the presence of three or more of the following: unintentional weight loss (⩾10 lbs in the past year), self-reported exhaustion, weakness (grip strength), slow observed walking speed, or low physical activity.60
Aging is a highly individualized, multidimensional process and a comprehensive geriatric assessment (CGA) can fill the gap of knowledge to guide management of older cancer patients and the incorporation of novel treatment approaches, including CDK4/6 inhibitors, in the treatment of older adults, and offer the opportunity to look at all the factors potentially impacting on therapy outcomes, to address their needs, and ultimately to select patients suitable for a more aggressive approach. A CGA provides a detailed evaluation of medical, psychosocial, and functional problems as detailed in Table 4. 64 Based on a CGA, frailty is defined as fulfilling one or more of the following criteria: dependency in ADLs; presence of severe comorbidities according to the Cumulative Illness Rating Scale; cognitive dysfunction (Mini Mental Status Examination score <24); depression (Geriatric Depression Scale score >13); malnutrition (Mini Nutritional Assessment score <17); polypharmacy (more than seven concomitant daily medications); or incontinence. 65 An increasing amount of evidence supports the use of CGA to predict treatment adverse events,66,67 estimate survival, 68 aid cancer treatment decisions, 69 detect problems usually neglected by routine assessments, 70 and improve mental health and well-being and pain control. 71 CGA is recommended by the International Society of Geriatric Oncology (SIOG) 72 and by the National Comprehensive Cancer Network guidelines. 73 Nonetheless, its use is still limited as it is perceived to be time consuming and difficult to implement in a busy oncology practice. Hence, various screening tools have been used to select patients that will benefit from a formal CGA. 74 For example, an abbreviated CGA, the Vulnerable Elders Survey-13, the Geriatric 8 tool, and the Flemish version of the Triage Risk Screening Tool have been developed. 75 A CGA should also be a key resource for clinical trial design in order to improve the evidence base for management of older patients with cancer and the applicability of study findings. 76
Comprehensive geriatric assessment domains and tools and abnormal scores useful for the diagnosis of frailty. 64

ADL, Activities of Daily Living; IADL, Instrumental ADL.
Therefore, we propose the algorithm shown in Figure 2 to guide the incorporation of CDK4/6 inhibitors in the management of ER-positive, HER2-negative advanced breast cancer in older patients based on a CGA. Fit patients with endocrine-sensitive disease should receive first-line AIs in combination with CDK/6 inhibitors; however, in the presence of endocrine-resistant breast cancer and if rapid response is not required, the association of fulvestrant and CDK4/6 inhibitors as per PALOMA-3 study findings may be used. For fit patients requiring rapid response due to severe symptoms, the use of first-line chemotherapy may be considered. It is debatable whether response rates are actually superior in a matched population of postmenopausal women with ER-positive, HER2-negative disease. Following induction chemotherapy, maintenance endocrine therapy plus CDK4/6 inhibition may be appropriate and this approach is currently under investigation in the CompLEEment-1 study, in which 22% of patients are aged 70+ and 19% received first-line chemotherapy. 101 Less fit patients may be better suited to receive endocrine treatment alone, particularly if their life expectancy is thought to be short owing to competing causes of mortality. Vulnerabilities in CGA domains, such as functional status, cognition, polypharmacy, mood, nutrition and geriatric syndromes, may impact on the decision to use of CDK4/6 inhibitors in frail adults due to the risk of potential toxicities and increased burden of medications, required hospital visits, and blood tests. In certain cases, endocrine treatment alone without CDK4/6 inhibitors may be used to minimize the impact of potential toxicities (such as myelosuppression and diarrhea) and drug interactions on QoL. Nonetheless, frail patients should not be denied CDK4/6 inhibitors a priori as CGA may also help identifying those suitable for multidisciplinary interventions that have the potential to improve their vulnerabilities for consideration of treatment. Moreover, in patients who are frail as a consequence of cancer itself, another potential option is considering the introduction of CDK4/6 inhibitors at a later stage, once their disease responds to endocrine agents and their fitness subsequently improves, in order to maximize their chances of long-term remission. Endocrine therapy alone may also be a reasonable option in less fit older patients if they are asymptomatic, treatment naïve, and have predominantly bony metastatic involvement, but who are likely to remain fit enough for a CDK4/6 inhibitor in the second-line setting. Nevertheless, the merits of treatment sequencing are still under investigation in the SONIA trial. More studies investigating the tolerance of this class of drugs in real-world adults and their role in frail patients in the context of their preferences and values guided by shared decision making are warranted. Supportive care should also be incorporated early in the management of any older cancer patients, along with locoregional approaches if required and upon multidisciplinary discussion.

Proposed initial approach to the management of ER-positive, HER2-negative advanced breast cancer in older patients.
Based on the available data showing similar efficacy and toxicity profile in the older and young age groups, CDK4/6 inhibitors are an attractive option for older patients with advanced ER-positive, HER2-negative breast cancer. Ongoing adjuvant trials will further define their role in the management of early-stage disease. Nonetheless, better evidence is needed to guide their incorporation in the treatment strategy for this population, and trials specifically enrolling older adults that include geriatric parameters are warranted. Prospective observational studies and real-world experiences may also fill the current gap of knowledge.
Footnotes
Acknowledgements
The authors wish to acknowledge the support of Colm Doody and Jane Crimmin, pharmacists at The Royal Marsden NHS Foundation Trust, London, UK, for their help in outlining drug interactions relevant to CDK4/6 inhibitors. The authors wish to also acknowledge the support of Pfizer and Novartis in highlighting age-specific data when available. NMLB and AR wish to acknowledge the support of the National Institute for Health Research Biomedical Research Center for Cancer (The Royal Marsden NHS Foundation Trust and Institute of Cancer Research). All authors wrote the review and contributed to the final version. All authors reviewed the final version.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
AR has received speaker fees from Novartis and Pfizer and advisory board fees from Novartis, Pfizer and Lilly. The remaining authors declare no competing interests.
