Abstract
Background:
In recent years, the combination of CDK4/6 inhibitors (CDK4/6i) and endocrine therapy (ET) has emerged as the standard first-line treatment for hormone receptor positive (HR+) and human epidermal growth factor receptor 2 negative (HER2−) metastatic breast cancer (MBC) patients. However, the comparison between the efficacy of CDK4/6i has been poorly explored before. Moreover, it remains unclear about the optimal choice of CDK4/6i in the first-line treatment for HR+/HER2− MBC patients in Asian, especially Chinese populations.
Objectives:
Our study aims to compare the efficacy of three CDK4/6i widely used in the Chinese population (palbociclib, abemaciclib, and dalpiciclib) in the real world.
Design:
From 2020 to 2023, the medical records of patients diagnosed with HR+/HER2− MBC were retrospectively assessed in seven institutions in China. Patients who received first-line palbociclib, abemaciclib, or dalpiciclib plus ET were included.
Methods:
Demographic and clinical data were retrospectively collected and analyzed. Real-world progression-free survival (rwPFS), overall survival (OS), and objective response rate were used to analyze the clinical outcome.
Results:
In total, 209 HR+/HER2− MBC patients were eligible for this study. Eighty-eight (42.1%), 79 (37.8%), and 42 (20.1%) patients were administered first-line palbociclib, abemaciclib, or dalpiciclib plus ET. The overall median rwPFS was 19 months, with no significant difference between these three CDK4/6i (p = 0.84). The results were similar even after propensity score matching. The median OS was not reached. Cox univariate and multivariate regression analysis identified that higher KI67 index, liver metastasis, and primary endocrine resistance were independent risk factors for rwPFS in patients with initial CDK4/6i plus ET.
Conclusion:
This study presents a comparison of the real-world efficacy between three CDK4/6i widely used in the Chinese population. Palbociclib, abemaciclib, and dalpiciclib demonstrated comparable efficacy in Chinese patients with advanced HR+/HER2− MBC.
Trial registration:
ClinicalTrials.gov identifier: NCT06344780.
Keywords
Introduction
Breast cancer is a significant global health concern that poses a serious threat to women’s health worldwide. 1 Since metastatic breast cancer (MBC) is virtually incurable, it is crucial to delay disease progression and improve quality of life. Nearly 75% of all MBCs have a molecular subtype of hormone receptor positive (HR+) and human epidermal growth factor receptor 2 negative (HER2−), and the standard first-line treatment for which is CDK4/6 inhibitors (CDK4/6i) combined with endocrine therapy (ET).
A series of prior phase III randomized studies has proven CDK4/6i plus ET can significantly extend HR+/HER2− MBC patients’ progression-free survival (PFS). PALOMA-2 study showed the first-line application of palbociclib plus letrozole can contribute to a significantly longer median PFS (mPFS) than letrozole alone, 2 and the subsequent PALOMA-3 trial also confirmed palbociclib plus fulvestrant had similar benefits. 3 In the next few years, phase III studies of abemaciclib and ribociclib also demonstrated promising outcomes in the treatment of HR+/HER2− MBC patients.4 –8 It is noteworthy that the phase III study of dalpiciclib also showed its viability as a CDK4/6i option for HR+/HER2− MBC patients in China.9,10 To date, palbociclib, abemaciclib, and ribociclib for HR+/HER2− MBC patients’ first-line treatment have been approved by the U.S. FDA and the National Medical Products Administration (NMPA) of China, respectively. Also, dalpiciclib was approved by the NMPA in December 2021. However, which CDK4/6i is the best first-line treatment option for HR+/HER2− MBC patients has been long debated. The toxicity profiles of each CDK4/6i are primary considerations for physicians when selecting the best fit CDK4/6i for first-line treatment. Physicians must also comprehensively evaluate the specific clinical conditions of each patient, such as comorbidities. Additionally, whether a CDK4/6i provides an overall survival (OS) benefit is also a factor for physicians to consider. For example, in the PALOMA-2, PALOMA-3, and MONARCH-3 trials, palbociclib and abemaciclib did not demonstrate an OS benefit.11 –13 Currently, there is only one ongoing phase III trial directly comparing ribociclib and palbociclib (NCT05207709), which primarily targets the HER2-enriched (HER2-E) population. 14 Moreover, Asian populations in most landmark CDK4/6i trials were limited 15 and the frequencies of potential grade 3 and 4 adverse effects in Asian populations is higher, 16 posing challenges when attempting to select the most appropriate CDK4/6i for a patient.
Real-world evidence, derived from analysis of real-world data (RWD) containing treatment patterns and outcomes, has gained increasing attention in recent years. Real-world studies often encompass a broader patient population and can generate new insights following phase III studies and the approval of new drugs and combination therapies. A recent real-world study based on the Flatiron Health Analytic Database published by Rugo et al. 17 reported that first-line palbociclib plus ET rather than ET monotherapy could significantly prolong OS in HR+/HER2− MBC patients in real-world settings. In addition, real-world studies can often include populations overlooked by phase III studies, providing important clinical evidence and reflecting treatment patterns in real-world settings. For example, some recent real-world studies have focused on populations typically underrepresented in phase III studies, such as elderly patients.18,19 Here, we designed this study to evaluate RWD on three commonly used CDK4/6i (palbociclib, abemaciclib, and dalpiciclib) in China with the goal of providing insight into real-world practices and inform clinical decision-making in the management of patients with MBC.
Methods
Study design and participants
This retrospective, observational multicenter study was approved by the Ethics Committee and Institutional Review Board (IRB) of Fudan University Shanghai Cancer Center (FUSCC) (IRB approval number: 1812195-6) and was conducted according to the tenets of the Declaration of Helsinki. The need for informed consent was waived, owing to the retrospective nature of the study in accordance with the national legislation and institutional requirements. This study was retrospectively registered with ClinicalTrials.gov (ClinicalTrials.gov identifier: NCT06344780) and the other study ID number was YOUNGBC-28.
The medical records of patients diagnosed with HR+/HER2− MBC from June 2020 to October 2023 in seven institutions were retrospectively evaluated. The seven institutions were: FUSCC, Tumor Hospital of the Chinese Academy of Medical Sciences, Hunan Cancer Hospital, Sanhuan Cancer Hospital, The First Affiliated Hospital of the University of Science and Technology of China, Union Hospital of Tongji Medical College of Huazhong University of Science and Technology, and Renmin Hospital of Wuhan University. Patients who fulfilled the following criteria were enrolled in this study: (1) age ⩾18 years old; (2) diagnosed as HR+/HER2− recurrent unresectable (local or regional) or stage IV (M1) breast cancer; (3) received CDK4/6i (abemaciclib, palbociclib, or dalpiciclib) as first-line therapy for at least one cycle between June 2020 and October 2023; (4) complete medical records. Patients with the following conditions were defined as visceral crisis: (1) severe organ dysfunction with metastatic disease assessed by clinical symptoms and routine laboratory data; (2) rapid progression of metastatic disease; (3) other conditions met the criteria for visceral crisis judged by investigators largely based on ABC guidelines 7. 20 Endocrine-naïve or resistance was determined based on ABC guidelines 7. 20 Disease-free interval (DFI) was defined as the time from surgery to documentation of unresectable or metastatic recurrence. Baseline demographic features, treatment history, and survival outcomes were obtained by reviewing medical records. This study adheres to the STROBE guidelines for reporting observational cohort studies 21 (Supplemental File 1).
Outcomes
We used real-world progression-free survival (rwPFS), OS, and objective response rate (ORR) as outcome measures of treatment efficacy. The primary outcome measure of our study was rwPFS, and secondary outcome measures included OS, ORR, and safety. rwPFS was defined as the time from treatment initiation to disease progression or death. Disease progression was determined by the treating physician according to the medical history, radiologic examination, and pathology reports. Patients who were alive at the time of the last evaluation and without evidence of progression were censored. OS was defined as the time from treatment initiation to death or final follow-up. ORR was defined as the percentage of patients with complete response (CR) or partial response (PR). Computed tomography, magnetic resonance imaging, and physical examination were used to assess treatment response in accordance with the criteria of Response Evaluation Criteria in Solid Tumors 1.1. 22
Statistical analyses
The descriptive data comprised the information on demographics, medical history, and first-line treatment options. Clinical characteristics were described using medians (ranges) or percentages. T-tests, Chi-square tests, or Fisher’s exact tests were used to compare baseline demographics and clinical characteristics between treatment groups. Missing data were addressed by treating them as null values in the analysis. The Kaplan–Meier analysis and 95% confidence intervals (CIs) were used to estimate median rwPFS and median OS. Patients with loss to follow-up were censored. Cox proportional hazards model was used to estimate hazard ratios (HRs) and corresponding 95% CIs. Independent prognostic factors for rwPFS were obtained by univariate and multivariate Cox regression analysis. Propensity score matching (PSM) using the nearest neighbor matching with a caliper of 0.2 was performed to control the confounding factors and determine the robustness of the primary results. For further analysis of rwPFS, the total cohort was split into subgroups based on their endocrine resistance status or the presence of visceral crisis.
Two-tailed CIs and p-values were obtained. All statistical analyses were performed using R version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria). A p value less than 0.05 was considered statistically significant.
Results
Baseline characteristics
A total of 209 patients were included in our study. The baseline patient characteristics are shown in Table 1. The median age of palbociclib, abemaciclib, and dalpiciclib groups were 54.5, 55, and 57.5 years, respectively. Among these patients, 103 (49.3%) patients have more than 2 metastatic sites and 117 (56.0%) patients have visceral metastasis. Thirty (14.4%) patients were considered primary endocrine resistant and 112 (53.6%) patients were secondary endocrine resistant before initial treatment. Twenty-seven (12.9%) patients in the total cohort had visceral crisis at baseline. In total, 124 (59.3%), 83 (39.7%), and 2 (1.0%) patients were prescribed aromatase inhibitor (AI), fulvestrant, and tamoxifen as concomitant endocrine drugs. Patients with de novo metastatic disease were more likely to be administered dalpiciclib than palbociclib or abemaciclib (26.2% vs 17.0% for palbociclib and 6.3% for abemaciclib). Most baseline clinical and pathological characteristic features were approximately balanced except for DFI. In addition, although there were no statistically significant differences in baseline ET resistance status among the three groups (p = 0.207), there was still a trend indicating an imbalance in baseline ET primary resistance status among the three groups (14.8% vs 17.7% vs 7.1%).
Baseline patient characteristics.

Pearson’s Chi-squared test.
Fisher’s exact test for count data.
DFI, disease-free interval; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma.
Real-world progression-free survival
After a median follow-up of 21.8 months, 107 PFS events had occurred, and the overall median rwPFS was 19 (95% CI, 15–22.5) months. The median rwPFS was 19.0 (95% CI, 13.0–22.6) months in the palbociclib group, 20.0 (95% CI, 15.0–NA) months in the abemaciclib group, and 18.0 (95% CI, 15.1–NA) months in the dalpiciclib group. While it showed no significant difference between three cohorts (p = 0.84) using Kaplan–Meier analysis (Figure 1).

Kaplan–Meier curve of median rwPFS for patients receiving palbociclib, abemaciclib, or dalpiciclib.
Because of an imbalance in baseline characteristics (DFI), PSM was performed to compare the efficacy of palbociclib versus abemaciclib and palbociclib versus dalpiciclib. The following variables were considered in the matching: age, menstrual status, DFI, T stage, node status, endocrine resistance, pathological type, HER2 status, KI67 expression, adjuvant therapy, ECOG performance status, number of metastatic sites, metastatic sites, and visceral crisis. Table 2 shows the baseline features after PSM, the baseline characteristics were comparable between palbociclib group versus abemaciclib group or dalpiciclib group. After PSM, no significant difference was found in median rwPFS of palbociclib group and abemaciclib group ((HR, 0.981; 95% CI, 0.578–1.665; p = 0.94) (Figure 2(a))). And there was no substantial difference in median rwPFS between palbociclib group and dalpiciclib group either ((HR, 1.03; 95% CI, 0.502–2.112; p = 0.94) (Figure 2(b))).
Patient characteristics after PSM.

DFI, disease-free interval; ET, endocrine therapy; HER2, human epidermal growth factor receptor 2; PSM, propensity score matching.

Kaplan–Meier curves of median rwPFS for patients receiving palbociclib, abemaciclib, or dalpiciclib after PSM. (a) Median rwPFS of palbociclib group and abemaciclib group. (b) Median rwPFS of palbociclib group and dalpiciclib group.
We then analyzed the efficacy of these three CDK4/6i in endocrine-naïve and endocrine-resistant populations. Patients with endocrine-naïve, primary endocrine resistance, and secondary endocrine-resistance accounting for 32.1% (n = 67), 14.4% (n = 30), and 53.6% (n = 112) in all patients. Kaplan–Meier curves indicate patients with primary endocrine resistance had significantly shorter median rwPFS than endocrine-naïve group and secondary endocrine-resistance group (9.0 vs 22.0 vs 20.0 months) (Figure 3(a)). Then we assessed rwPFS in primary endocrine-resistance population, the results demonstrated these three groups with primary endocrine resistance had similar median rwPFS (p = 0.82) (Figure 3(b)). Similar results were also shown in endocrine-naïve and secondary endocrine-resistance populations (Figure 3(c) and (d)).
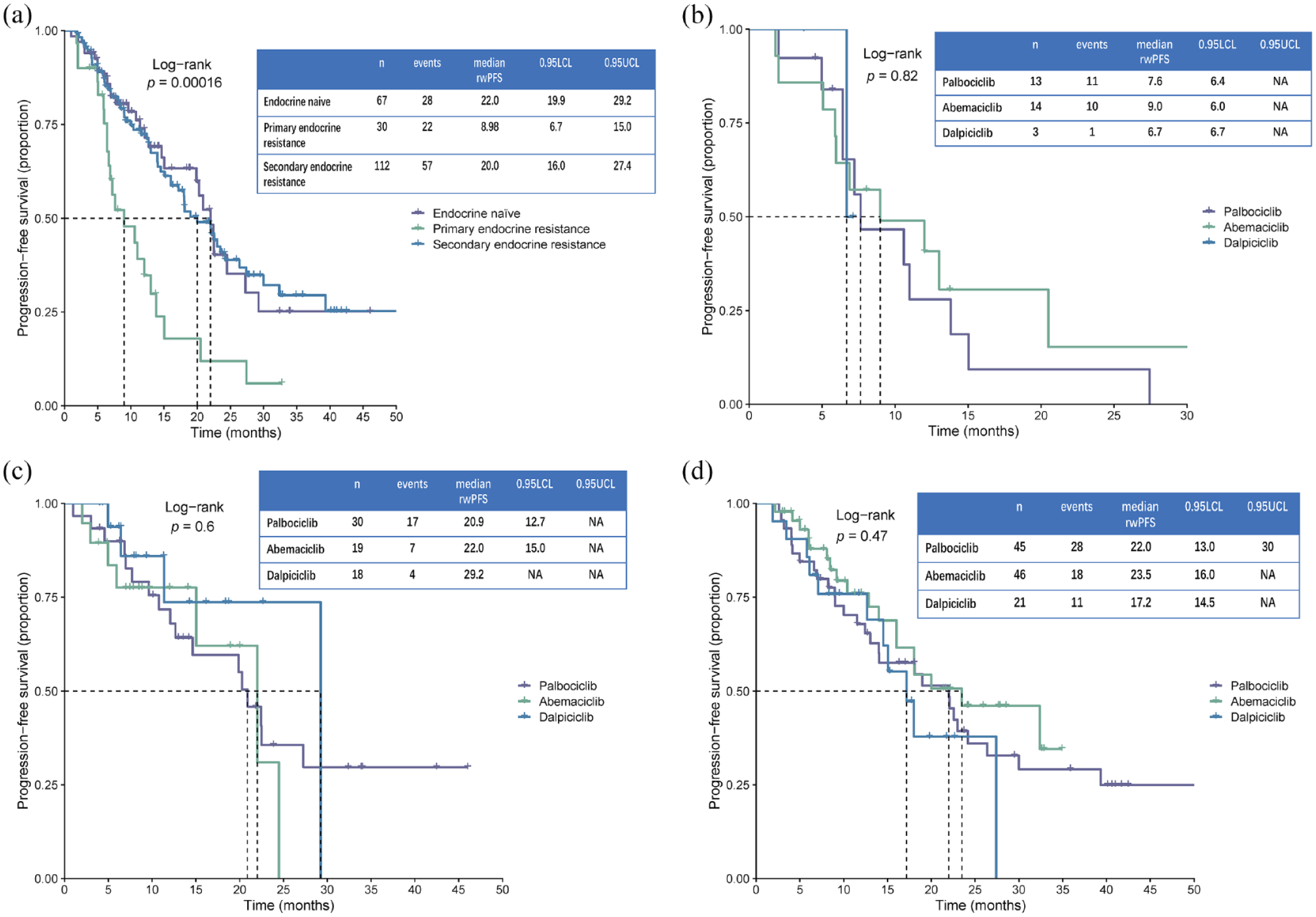
Kaplan–Meier curve of median rwPFS, stratified by endocrine-naïve, primary endocrine resistance, or secondary endocrine resistance. (a) Median rwPFS of patients with endocrine-naïve, primary endocrine resistance, or secondary endocrine resistance. (b) Median rwPFS of patients with primary endocrine resistance. (c) Median rwPFS of patients with endocrine-naïve. (d) Median rwPFS of patients with secondary endocrine resistance.
In this study, we also included 27 (12.9%) patients with visceral crisis. The Kaplan–Meier curve showed that patients with visceral crisis tended to have a shorter median rwPFS, but this was not statistically significant (p = 0.19) (Figure 4(a)). And in patients with visceral crisis or not, there was no significant difference among these three CDK4/6i (Figure 4(a) and (b)).

Kaplan–Meier curve of median rwPFS, stratified by visceral crisis. (a) Median rwPFS of patients with visceral crisis or not. (b) Median rwPFS of patients with visceral crisis. (c) Median rwPFS of patients without visceral crisis.
To screen out the independent prognostic factors for rwPFS in individuals with first-line CDK4/6i plus ET, univariable and multivariable Cox regression analysis were performed. The prognostic factors for rwPFS obtained by univariate Cox regression analysis were examined by multivariate Cox regression analysis. As shown in Table 3 and Figure 5, patients with primary endocrine resistance, higher KI67 expression, and liver metastasis were predicted to have shorter rwPFS.
Results of Cox univariate and multivariate regression analysis.
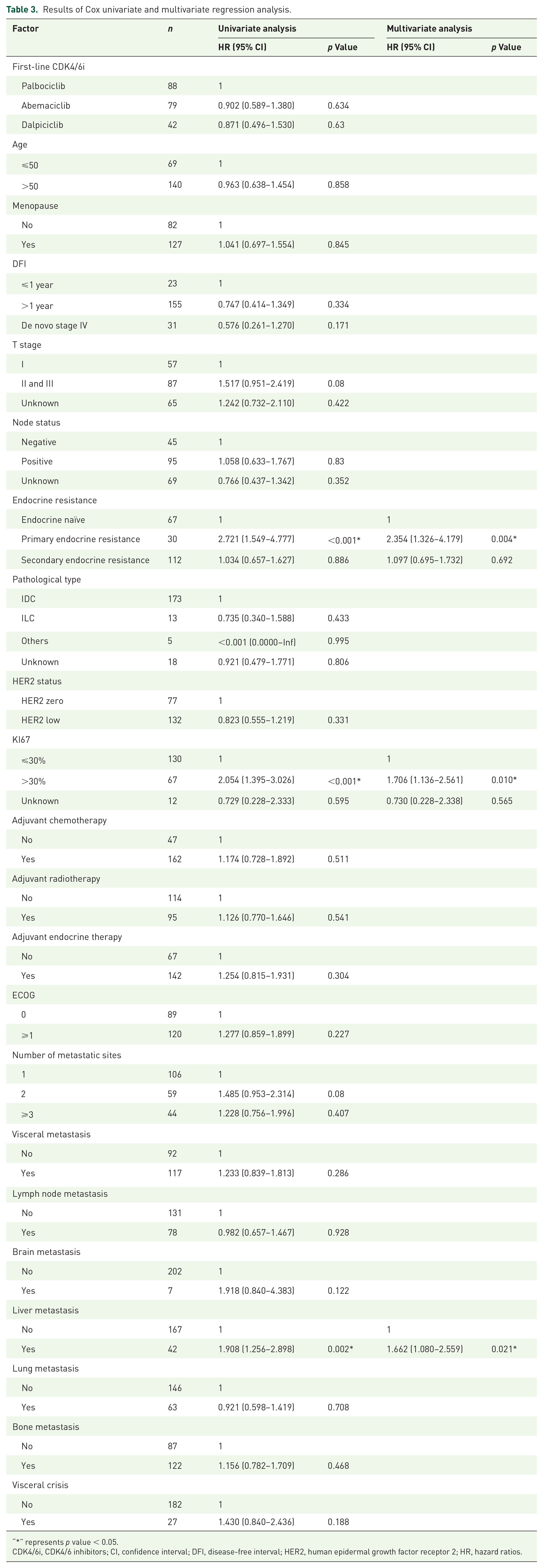
“*” represents p value < 0.05.
CDK4/6i, CDK4/6 inhibitors; CI, confidence interval; DFI, disease-free interval; HER2, human epidermal growth factor receptor 2; HR, hazard ratios.

Forestplot of multivariate Cox regression analysis.
Secondary outcomes
Median OS was not reached in these three groups. Seventy-two (34.95%) patients in overall population achieved objective response and three patients’ response were not evaluable. The ORR in palbociclib group, abemaciclib group, and dalpiciclib group were 39.08% (n = 34), 32.47% (n = 25), and 30.95% (n = 13), as illustrated in Figure 6. There were 1 (1.15%) and 2 (2.60%) patients in palbociclib and abemaciclib group achieved CR. No statistical significance in ORR was observed between the three groups (p = 0.56) using Chi-squared test.
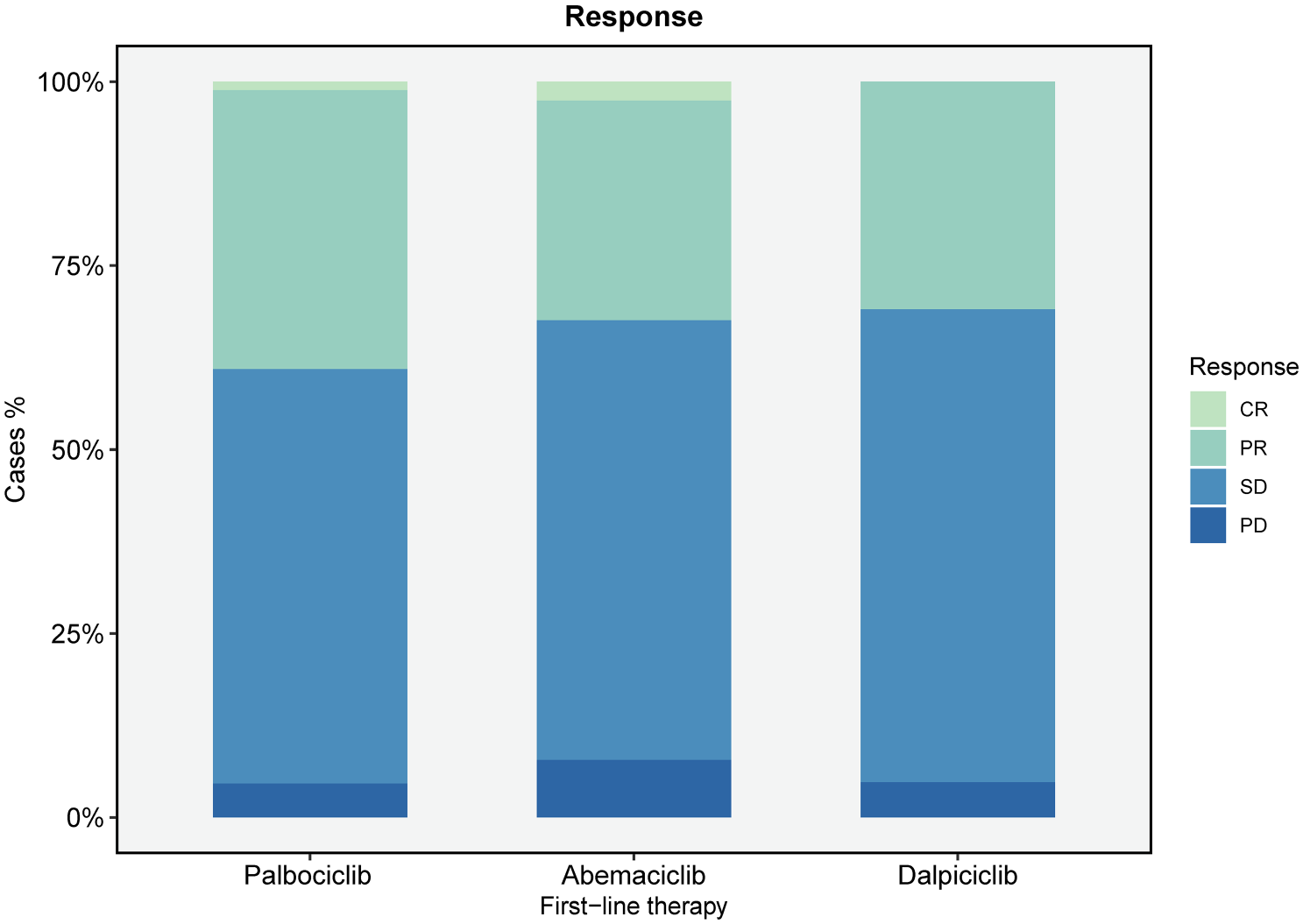
Response rate of patients receiving palbociclib, abemaciclib, or dalpiciclib.
As for adverse events (AEs), 158 (75.60%) patients experienced at least one all-grade AEs. Grade 3 and higher AEs were reported in 27.27% (n = 24), 24.05% (n = 19), and 28.57% (n = 12) of patients in palbociclib group, abemaciclib group, and dalpiciclib group, respectively. The most common grade 3 and higher AE was neutropenia in all groups (20.45% for palbociclib group, 11.39% for abemaciclib group, and 21.43% for dalpiciclib group). Notably, patients using abemaciclib in this study showed more all-grade diarrhea than the other two groups (12.66% for abemaciclib group, 0.00% for palbociclib group, and 2.38% for dalpiciclib group), which is consistent with the known safety profile. Overall, the incidence of grade 3 or higher AEs was similar among the three groups. Other AEs of the patients were reported in Table 4.
Adverse events of palbociclib, abemaciclib, and dalpiciclib group.

Discussion
In the last decade, the emergence of CDK4/6i has revolutionized the treatment of HR+/HER2− MBC patients. In preclinical studies, the activation of cyclin-dependent kinases has been found to participate in the occurrence and development of breast tumors. Cyclin D1, a CDK4/6 binding protein, tends to be extremely overexpressed in ER+/HER2− breast cancer, resulting in continuous activation of the D1-CDK4/6 complex, leading to poor prognosis.23,24 The mechanism of action of CDK4/6i is that they inhibit CDK4/6 and thus dephosphorylate retinoblastoma (Rb) protein, which in turn leads to its dissociation from the transcription factor E2F, preventing cell-cycle progression. 25 Substantial evidence from prior phase III randomized studies and real-world studies have demonstrated that the combination of CDK4/6i with ET significantly prolongs PFS compared to endocrine monotherapy.2,3,7,8,10,26,27 Moreover, the potential OS benefits of different CDK4/6i for HR+/HER2− MBC patients have recently received increased attention. Although the OS benefit of palbociclib and abemaciclib was not significant in the PALOMA-2 (median OS: 53.9 vs 51.2 months, p = 0.3378), 11 PALOMA-3 (median OS: 34.9 vs 28.0 months, p = 0.09), 12 and MONARCH-3 (median OS: 66.8 vs 53.7 months, p = 0.0664) 13 studies, ribociclib and abemaciclib have been reported to provide significant OS benefits in the MONALEESA-2 (median OS: 63.9 vs 51.4 months, p = 0.008), 28 MONALEESA-3 (median OS: 53.7 vs 41.5 months), 29 MONALEESA-7 (median OS: 58.7 vs 48.0 months), 30 and MONARCH-2 (median OS: 46.7 vs 37.3 months, p = 0.01) 31 studies. This inconsistency in OS benefit among the CDK4/6i poses a challenge for physicians in selecting the appropriate CDK4/6i. Currently, there remains only one ongoing clinical trial that directly compares palbociclib and ribociclib (NCT05207709), with the main cohort being the HER2-E population. 14 Therefore, the optimal selection of the CDK4/6i in different clinical situations for HR+/HER2− MBC patients remains to be determined.
At present, there are three CDK4/6i extensively used in China: palbociclib, abemaciclib, and dalpiciclib. Although ribociclib was approved by the NMPA in January 2023, it was not covered by Chinese health insurance until January 2024, thus ribociclib is not widely used in the Chinese populations currently and therefore was not included in this study. Unlike developed countries such as the United States, CDK4/6i were approved by the NMPA in China 3–5 years later than the U.S. FDA. Dalpiciclib approved by the NMPA has not been approved by the FDA. For the reasons above, the HR+/HER2− MBC populations in China only began to widely use CDK4/6i plus ET as the standard treatment in the past 3–4 years. Also, the types of CDK4/6i used by the Chinese populations are slightly different from those in the United States and other countries. To date, no real-world studies have compared the efficacy of these three CDK4/6i in Chinese populations.
In this real-world study, we retrospectively analyzed the RWD from June 2020 to October 2023 in seven institutions. In our cohort, 42.1% (n = 88), 37.8% (n = 79), and 20.1% (n = 42) of patients were prescribed palbociclib, abemaciclib, and dalpiciclib as initial treatment. The difference in the patient numbers may be due to the later launch of dalpiciclib in China compared with the other two CDK4/6i. This difference also reflects physicians’ preferences to some extent. In the present study cohort, 117 (56.0%) patients had visceral metastasis, which is in line with the proportions reported in published real-world studies.26,32,33 Notably, 27 (12.9%) patients in this study cohort had visceral crisis, which is often excluded from phase III studies and poorly investigated.
The rwPFS of these three drugs observed in this real-world study are partially in line with a series of published phase III randomized studies.34 –36 Median rwPFS of our palbociclib group (n = 88) was 19.0 months, which is comparable with the mPFS (21.5 months) reported in PALOMA-4 study, which mainly included Asian populations. 34 The proportion of patients with visceral metastases included in the PALOMA-4 study (55.9%) was similar to this study (56.0%), but included more patients with de novo metastasis (19.4%) and DFI ⩽12 months (32.1%) than this study. 34 Moreover, the median rwPFS of palbociclib reported in this study showed a similar trend to the mPFS of subgroup analyses of Asian patients in the PALOMA-2 (22.2 months) and PALOMA-3 (13.6 months) studies.35,36 The median rwPFS of abemaciclib group in this study was reported as 20 months, which was shorter than the mPFS reported in MONARCH-3 study (29.4 months).7,13 This discrepancy may be due to the fact that the MONARCH-3 study enrolled more de novo metastatic patients (39.8%) than this study (14.8%), who had not been exposed to adjuvant ET before and might be more responsive to CDK4/6i plus ET treatment. 20 The rwPFS of dalpiciclib reported in this study (18 months) was also inconsistent with mPFS previously reported in DAWNA-2 study (30.6 months), possibly because of the small sample size of dalpiciclib group (n = 42) in this study and the inclusion of more de novo metastatic patients (41.4%) in DAWNA-2. 10 Furthermore, unlike most previous phase III studies that used aromatase inhibitors (AI) as a concomitant drug of CDK4/6i, some patients (39.7%) in this study used fulvestrant as a concomitant drug of CDK4/6i. Since this study included some patients with visceral crisis, it is reasonable to expect deviations between its findings and those of previous phase III studies.
Other RWD support the results of this study. The median rwPFS (19.0 months) of palbociclib group (n = 88) in this study was consistent with the unadjusted median rwPFS in the published P-reality (19.7 months) and P-Reality X (19.8 months) study.17,27 In 2024, a real-world study reported a median rwPFS of 24.5 months for HR+/HER2− MBC people using palbociclib plus ET as first-line therapy in the Japanese population, which had a similar trend to the present study. 33 This Japanese population-based study also documented an ORR of 37.9% in first-line treatment group, similar to the ORR of palbociclib (39.1%) reported in this study. 33 Other published RWD showed similar trends to this study.19,26,37
Dalpiciclib, as a widely used CDK4/6i only in the Chinese population, appeared relatively late compared with other CDK4/6i. In 2021, the DAWNA-1 study showed that dalpiciclib plus fulvestrant rather than fulvestrant monotherapy in HR+/HER2− patients after ET treatment progression could significantly prolong HR+/HER2− MBC patients’ mPFS (15.7 vs 7.2 months). 9 And subsequent DAWNA-2 study also reported that first-line dalpiciclib plus AI could also significantly prolong patients’ mPFS compared with AI monotherapy (30.6 vs 18.2 months). 10 So far, no studies have reported the efficacy of dalpiciclib in real-world settings. This study is the first to report the real-world efficacy of first-line dalpiciclib plus ET in HR+/HER2− MBC patients.
In the analyses of the present study, we did not observe a significant difference among the median rwPFS of palbociclib, abemaciclib, and dalpiciclib (p = 0.84). This insignificance was also observed in the visceral crisis subgroups and the endocrine resistance subgroups. To minimize confounding factors among the cohorts, we performed PSM using the nearest neighbor matching with a caliper of 0.2. However, the efficacy of palbociclib versus abemaciclib and palbociclib versus dalpiciclib remained nonsignificant after PSM. A real-world study published in 2023 reported the first-line efficacy of palbociclib, abemaciclib, and ribociclib in the real world, which also showed no difference in median rwPFS among the three drugs, but abemaciclib significantly prolonged rwPFS compared with the other two drugs in patients without visceral metastasis or with endocrine resistance. 32 However, this phenomenon was not found in our study, which possibly be due to the difference in the baseline features of two studies and the difference in follow-up time (median follow-up: 21.8 vs 27.6 months). To explore the independent prognostic factors for rwPFS, we performed Cox univariate and multivariate regression analysis for all populations in this study and found that higher expression of KI67, liver metastasis, and primary endocrine resistance was associated with unfavorable rwPFS. These three independent risk factors were in line with those reported in previous studies.38,39
To our knowledge, this is the first real-world, multicenter study comparing the three CDK4/6i extensively used in China. Although the median rwPFS of the three drugs in this study did not show a significant difference, this study can still inform the decision making of physicians. Given the efficacy of the three drugs is similar, the side effects and cost-effectiveness of these drugs should be taken into account. As the follow-up period continues, it is possible that the advantages of one of the three drugs will become clearer. According to the findings of this study, higher KI67 index, liver metastasis, and primary endocrine resistance may be associated with shorter rwPFS and should be considered in clinical practice.
There are some limitations in this study, including a small sample size, retrospective study design, and relatively short median follow-up time (21.8 months). In addition, physician bias in the selection of CDK4/6i and patients’ compliance may affect the results of the study to a certain extent. Moreover, some sample sizes of the subgroups analyzed in this study may be inadequate. Although the application of PSM balanced the baseline between the groups, a small sample size could have prevented us from reaching valuable conclusions. However, this study is the first to provide a comparison of CDK4/6i in the Chinese population. Moreover, independent prognostic factors of rwPFS were obtained, which can provide certain references for subsequent studies and clinical decision-making. Considering the limitations of this study, prospective studies of head-to-head comparison between these CDK4/6i need to be conducted as soon as possible.
Conclusion
In this study, we found that the first-line efficacy of three CDK4/6i (palbociclib, abemaciclib, and dalpiciclib) in Chinese HR+/HER2− MBC patients was comparable in real-world settings. The efficacy remained similar across subgroups with different endocrine resistance status and visceral crisis. Notably, higher KI67 index, liver metastasis, and primary endocrine resistance were identified as independent prognostic factors for shorter PFS.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241302018 – Supplemental material for Real-world comparison of palbociclib, abemaciclib, and dalpiciclib as first-line treatments for Chinese HR+/HER2−metastatic breast cancer patients: a multicenter study (YOUNGBC-28)
Supplemental material, sj-docx-1-tam-10.1177_17588359241302018 for Real-world comparison of palbociclib, abemaciclib, and dalpiciclib as first-line treatments for Chinese HR+/HER2−metastatic breast cancer patients: a multicenter study (YOUNGBC-28) by Yifan Chen, Yizhao Xie, Die Sang, Ning Xie, Xinhua Han, Yanxia Zhao, Juanjuan Li, Jian Yue, Peng Yuan and Biyun Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
