Abstract
Renal cell cancer (RCC) continues to be among the most lethal malignancies in the USA. Introduction of anti-vascular epidermal growth factor receptor tyrosine kinase inhibitors over a decade ago resulted in improvement in disease outcomes, but further development of new therapies largely stagnated for many years. More recently, a better understanding of disease biology and treatment-resistance patterns has led to a second renaissance in drug development, with the anti-programmed cell death protein 1 immune checkpoint inhibitor, nivolumab, paving the way for additional therapies entering clinical trial testing in the treatment of RCC.
Keywords
Introduction
Metastatic renal cell cancer (RCC) continues to be among the most lethal malignancies in the USA, with an estimated 14,080 deaths alone in 2015. 1 Nephrectomy remains the mainstay of treatment for localized disease, but a subset of these patients eventually develops metastatic disease. 2 Prognosis to date remains poor, as RCC is predominantly resistant to cytotoxic chemotherapy. While cytokine immunostimulatory therapy can lead to disease control for some, significant toxicities have largely limited their use. The introduction of anti-vascular epidermal growth factor receptor (VEGF-R) tyrosine kinase inhibitors (TKIs) has resulted in improvement in disease outcomes, including overall survival (OS). However, for many years, development of treatment beyond VEGF-targeted therapies has largely stagnated. More recently, however, a better understanding of disease biology and treatment-resistance patterns is leading to a second renaissance in drug development, with the anti- programmed cell death protein 1 (PD-1) immune checkpoint inhibitor nivolumab and multitargeted TKI cabozantinib paving the way for additional therapies entering clinical trial testing for the treatment of RCC. Here, we will focus on emerging therapies for clear cell (cc) RCC beyond VEGF-R inhibition.
Treatment of metastatic ccRCC: historical challenges
Failure of cytotoxic chemotherapy
While chemotherapy is widely considered a standard treatment for patients with predominantly sarcomatoid and collecting duct RCC variants, its role in treating ccRCC is poorly defined. Several regimens have demonstrated only modest activity in early phase clinical trials, and there are almost no phase III clinical trials evaluating the utility of cytotoxic chemotherapy in metastatic RCC. 3
The mechanisms of resistance to chemotherapy in RCC remain undefined. Reduction of intracellular drug accumulation via overexpression of transporter efflux pump permeability glycoprotein was not seen to be an important mediator in in vitro studies. 4 Another hypothesis is that molecular drug target alterations may confer chemotherapy resistance in RCC. Acquired mutation in the Von Hippel–Lindau (VHL) gene is commonly seen in ccRCC, and its inactivation leads to the intracellular accumulation of hypoxia-inducible factor (HIF), a microtubule-dependent transcription factor of genes involved in angiogenesis and tumor cell proliferation and survival. 5 Antimicrotubule chemotherapies are ineffective in RCC,6,7 but no mutations have been identified in tubulin drug-binding sites for taxanes to account for this resistance. 8 One hypothesis is that RCC has altered expression of tubulin isotypes, resulting in changes in microtubule dynamics as well as drug binding. VHL has been associated with regulation of microtubule dynamics, suggesting that the loss of VHL may affect taxane resistance. 9 Microtubule inhibitors have been shown to be potent inhibitors of HIF-1 expression and activity, thus HIF inhibition downstream of microtubule perturbation is a key determinant in the clinical response to antitubulin therapy. 10 RCC is the lone tumor type where microtubule-targeting drugs fail to suppress HIF-1, independently of VHL functional status. 11 These data suggest that the HIF-signaling axis has been uncoupled from microtubule control in RCC, and thus deregulation of the microtubule/HIF axis in RCC may contribute to taxane resistance.
Intratumoral heterogeneity
While some systemic therapies may lead to tumor shrinkage and alleviation of symptoms in metastatic (m) RCC, resistance ensues when the disease resorts to alternative pathways to drive angiogenesis and cell proliferation, either through adaptation of individual tumor cells or selection of surviving cells that are already using alternative pathways. Ideally, one can characterize tumors individually based on DNA sequencing and other means, leading to selection of the specific pathways that drive cell proliferation, an approach also known as ‘personalized medicine’. However, this methodology has largely been limited by extensive intratumoral heterogeneity; varying gene expression signatures can be detected even within the same tumor specimen in samples of those with RCC.12,13 This largely limits the utility of prognostic impact of biomarkers and, in addition, spatial variation in genetic and phenotypic properties poses a significant obstacle for optimal personalized treatment. 14
Immunostimulatory therapy: cytokine immunotherapy
The lack of sensitivity to cytotoxic chemotherapy for most patients with mRCC prompted research efforts for novel approaches. Spontaneous tumor regressions in mRCC after cytoreductive nephrectomy, with the observation that immune cells often infiltrate the tumor tissue, suggested that adaptive immunity might play an important role in this malignancy.15,16 This hypothesis led to immunostimulatory trials with interleukin-2 (IL-2) and interferon alpha (IFN-α).
IL-2
In phase II clinical trials, high-dose recombinant IL-2 achieved 7% complete responses (CR) and 8% partial responses (PR) in 25 patients with mRCC.17,18 Long-term durability of the treatment response in this small subset of responding patients was likely due to the ability of IL-2 to modulate T-cell proliferation and differentiation. Patients that achieved CR had a median duration of response exceeding 80 months, and those that achieved PR, 20 months. 19 Though these figures remain unrivaled by current-generation VEGF-targeted therapy, the general adoption of high-dose IL-2 has been hindered by its low response rate and significant toxicity profile, including a 4% risk of treatment-related mortality.20,21
IFN-α
The ability of IFN-α to regulate anti-angiogenesis and immune functions translates to a response rate of approximately 12% when used as monotherapy in mRCC, and confers a modest survival benefit of 3.8 months in the pooled data of four clinical trials over the control arm, although cases of long-term durable responses have been reported.22,23 Combining IFN-α with other agents has typically yielded additional modest clinical benefits. 24 Most notably, the AVOREN and CALGB 90206 trials randomized treatment-naïve mRCC patients to receive IFN-α with or without bevacizumab.25,26 While both phase III studies found a consistent improvement in progression-free survival (PFS) (AVOREN: 10.2 months versus 5.4 months, hazard ratio [HR] = 0.63; CALGB 90206: 8.5 months versus 5.2 months, HR = 0.71) and overall response rates (ORR) (AVOREN: 31% versus 13%; CALGB 90206: 25.5% versus 13.1%),8,9 neither met significance for OS benefit (AVOREN: 23.3 months versus 21.3 months, HR = 0.86; CALGB 90206: 18.3 months versus 17.4 months, HR = 0.86).
VEGF and mechanistic target of rapamycin inhibitors: a paradigm shift for mRCC therapy
Deeper insight into the somatic genetics of hereditary ccRCC led to the identification of the VHL tumor suppressor gene. 27 Its interaction with HIF plays a key role not only in inherited disease, but also in sporadic forms of RCC. 28 Inactivation or loss of the VHL gene product prevents ubiquitin-mediated degradation of HIF-1a/2a leading to upregulation and constitutive expression of pro-angiogenesis agents, including VEGF, erythropoietin, platelet-derived growth factor (PDGF), transforming growth factor-beta, and carbonic anhydrase IX. These angiogenic signals confer the ability to proliferate under hypoxic conditions, driving oncogenesis, and accounts for the hypervascular nature of VHL-associated neoplasms. 29 This was the basis for exploring anti-angiogenic therapy targeting VEGF. Multiple VEGF pathway inhibitors have been subsequently developed and approved for mRCC. As a therapeutic class, VEGF pathway inhibitors have consistently outperformed IFN or placebo in randomized phase III trials, and have become accepted as the preferred first-line therapy for mRCC.
VEGF-R TKIs
Multiple VEGF-R TKIs have demonstrated efficacy in RCC (Table 1), with sunitinib and pazopanib both approved for first-line treatment in those with metastatic disease. Sunitinib showed improved PFS (11 months versus 5 months, HR = 0.42, 95% confidence interval [CI]: 0.32–0.54), ORR (47% versus 12%), and trend for superior OS (26.4 months versus 21.8 months, HR = 0.82, 95% CI: 0.67–1.01) compared with IFN-α in treatment-naïve mRCC. 30 Pazopanib similarly demonstrated a first-line PFS benefit (9.2 months versus 4.2 months, HR = 0.46, 95% CI: 0.34–0.62) and ORR improvement (30% versus 3%) compared with placebo, though no OS improvement was seen (22.9 months versus 20.5 months, HR = 0.91, 95% CI: 0.71–1.16), likely due to the high crossover rate.31,32 Head-to-head comparison of these TKIs in the phase III COMPARZ trial showed that pazopanib was noninferior to sunitinib for the primary endpoint of PFS (8.4 months versus 9.5 months, HR = 1.05, 95% CI: 0.71–1.16) and secondary endpoint of OS (28.3 months versus 29.1 months, HR = 0.92, 95% CI: 0.79–1.01).33,34 However, health-related quality of life measures and patient preference favored pazopanib in two different randomized studies. 35
Phase III trials in metastatic renal cell carcinoma.

HR, hazard ratio; IFN-α, interferon alfa; mOS, median overall survival; mPFS, median progression-free survival; N/A, not applicable; NR, not reached; ORR, overall response rate.
Axitinib and sorafenib both demonstrate PFS benefit as second-line agents, though axitinib outperformed sorafenib (AXIS: 8.3 months versus 5.7 months, HR = 0.66, 95% CI: 0.55–0.78), whereas sorafenib was only compared with placebo (5.5 months versus 2.8 months, HR = 0.44, 95% CI: 0.35–0.55) in randomized trials.36–38 It is noteworthy that the PFS benefit of axitinib in AXIS is less prominent in patients previously treated with first-line VEGF-TKI compared with those treated with first-line cytokine therapy.
VEGF-R monoclonal antibody
Bevacizumab is a monoclonal antibody that binds the ligand, VEGF, preventing its interaction with VEGF-R and its downstream effects. Two large phase III trials (AVOREN and CALGB 90206) randomized treatment-naïve mRCC patients to receive bevacizumab plus IFN-α or placebo plus IFN-α. A consistent PFS benefit was associated with bevacizumab in both AVOREN (10.2 months versus 5.4 months, HR = 0.63, 95% CI: 0.52–0.75) and CALGB 90206 (8.5 months versus 5.2 months, HR = 0.71, 95% CI: 0.61–0.83), as well as ORR improvement (AVOREN: 31% versus 13%; CALGB 90206: 25.5% versus 13.1%).39,40 Neither trial met significance for OS benefit, though a trend towards better survival was observed in both studies.41,42 Combining bevacizumab with sorafenib or temosirolimus has not been shown to improve PFS compared with bevacizumab alone, and no trials to date have compared bevacizumab plus IFN-α with bevacizumab monotherapy. 43
Mechanistic target of rapamycin inhibitors
In addition to VEGF pathway inhibition, upstream target mechanistic target of rapamycin (mTOR) was identified as another therapeutic target for mRCC. mTOR has the potential to inhibit tumor growth via regulating cell growth, cell-cycle progression, angiogenesis, and response to hypoxic stress through the HIF-1a pathway. 44 However, recent data suggest a more modest role for mTOR inhibitors in the management of mRCC. While temsirolimus originally demonstrated a significant OS benefit over IFN-α (10.9 months versus 7.3 months, HR = 0.73, 95% CI: 0.58–0.92) in the first-line setting for poor-risk mRCC, as a second-line therapy it was associated with a worse OS when compared with sorafenib (12.3 months versus 16.6 months, HR = 1.31, 95% CI: 1.05–1.63).45,46 Similarly, everolimus failed to show noninferiority for PFS against sunitinib (7.4 months versus 10.7 months, HR = 1.4, 95% CI: 1.2–1.8) as first-line therapy. 47 For years, treatment after progression on first-line VEGF-R TKI remained controversial; while everolimus is generally better tolerated, axitinib is associated with a higher response rate albeit similar PFS benefit. With more promising therapies becoming available in the armamentarium of RCC treatment, both therapies have been relegated to later lines of therapy.
A new era in immunotherapy in mRCC: anti-PD1/programmed death ligand-1 checkpoint inhibitors
The interaction between PD-1 (present on T cells), and one of its ligands (PD-L1) (present on antigen-presenting cells and tumor cells) constitutes an immune checkpoint through which tumors can induce T-cell tolerance and avoid immune destruction. A retrospective analysis of 306 patients who underwent nephrectomy for ccRCC at the Mayo Clinic from 1990 to 1994 revealed that patients whose tissue demonstrated expression of PD-L1 (B7-H1) had significantly worse 5-year cancer-specific survival (41.9%) when compared with those whose tumors did not express PD-L1 (82.9%), suggesting that blocking the interaction of PD-L1 with PD-1 might reverse tumor-induced immune tolerance and serve as an attractive approach for RCC therapy. 48 Similar to results seen in murine models, initial phase I studies of MDX-1106/BMS-936558/ONO-4538 (nivolumab) (Bristol-Myers Squibb, New York, USA) demonstrated that antibody-mediated blockade of PD-1 induced durable tumor regression and prolonged stabilization of disease in patients with various advanced cancers, including mRCC. 49 A subsequent phase II study evaluated 168 patients with mRCC who had received 1–3 prior anti-angiogenic therapies, randomized to receive nivolumab at doses of 0.3 mg/kg (n = 60), 2.0 mg/kg (n = 54), or 10.0 mg/kg (n = 54) every 3 weeks until disease progression or unacceptable toxicity. 50 A manageable safety profile was seen across all three doses, with notable antitumor activity seen, supporting phase III testing.
CheckMate 025 is a randomized, open-label, phase III study that compared nivolumab with everolimus in patients with mRCC who had received one or two prior anti-angiogenic therapies. 51 A total of 821 patients with metastatic ccRCC were randomly assigned in a 1:1 ratio to receive 3 mg/kg of nivolumab intravenously every 2 weeks or a 10 mg everolimus tablet once daily. At the first interim analysis, median OS was 25.0 months (95% CI, 21.8–not reached) with nivolumab and 19.6 months (95% CI: 17.6–23.1) with everolimus, with a HR for death of 0.73 (95% CI: 0.57–0.93; p = 0.002), meeting the prespecified criterion for superiority (p ⩽ 0.0148). The ORR (25% versus 5%) favored nivolumab, despite similar median PFS (4.6 months versus 4.4 months). Grade 3/4 treatment-related adverse events occurred in 19% (most common: fatigue 2%) of patients receiving nivolumab and in 37% (most common: anemia 8%) of the patients receiving everolimus. These remarkable results demonstrated for the first time that a treatment for mRCC could provide definitive OS benefit after progression on first-line anti-angiogenic therapy, establishing nivolumab as the new standard of care in this setting. The US Food and Drug Administration approved nivolumab for the treatment of metastatic ccRCC after progression on VEGF-R TKI on 25 November 2015. Other anti-PD-1 antibodies (MK-3745) and anti-PD-L1 (MPDL3280A) inhibitors are currently in clinical trial testing, as are combinations with other modalities (cytokine therapy, VEGF-R inhibitors, and other immune checkpoint inhibitors) (Table 2).
Ongoing clinical trials for renal cell carcinoma.
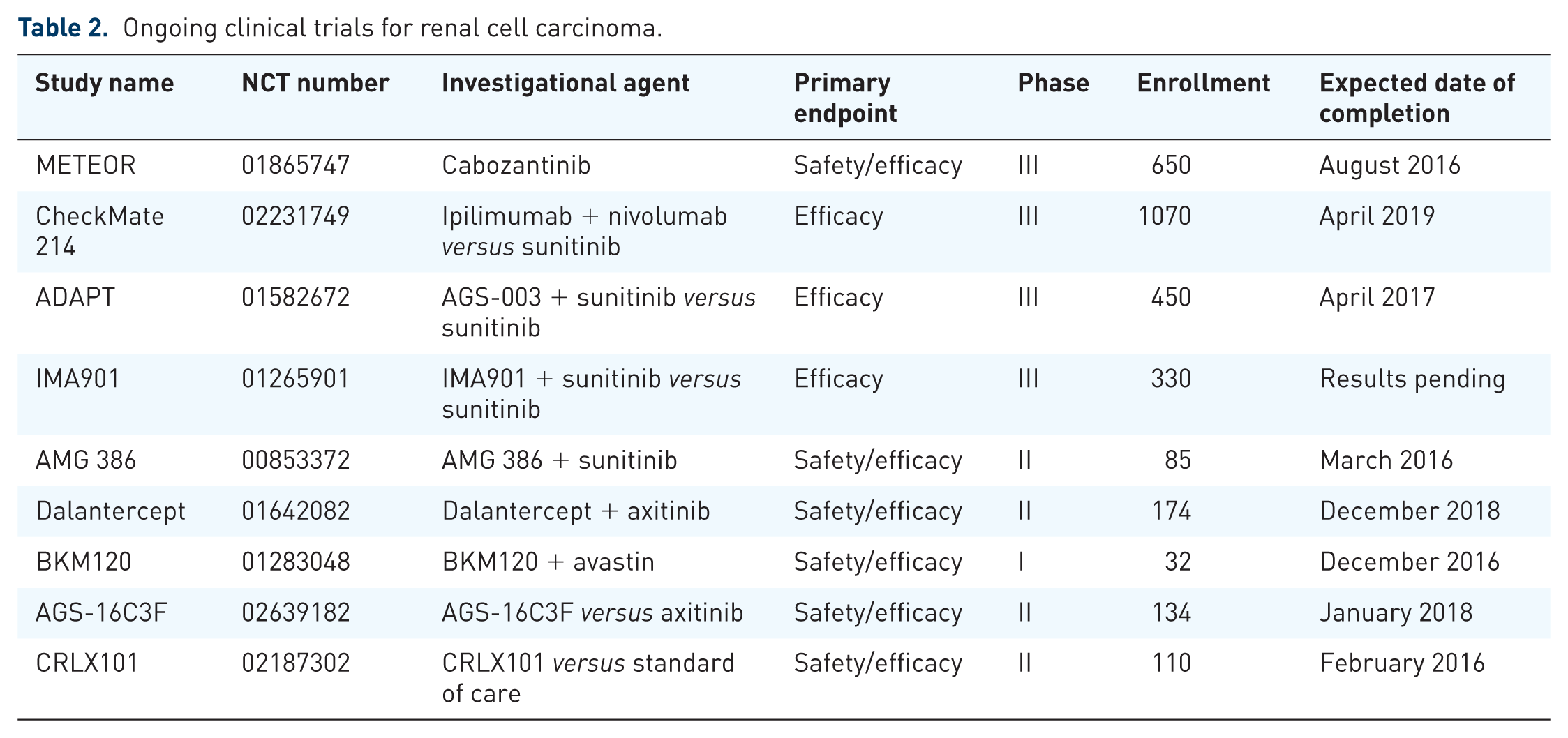
Promising novel therapies: the next steps
For years, development of treatment beyond VEGF-targeted therapies had been largely unsuccessful. More recently, better understanding of disease biology and treatment-resistance patterns has led to the introduction of effective therapies adding to inhibitors of VEGF-R signaling (Figure 1). Here, we will focus on those emerging therapies for ccRCC beyond VEGF-R inhibition.

Mechanisms of action of current treatments in metastatic renal cell cancer. c-MET, tyrosine-protein kinase Met; mTOR, mechanistic target of rapamycin; PD-1, programmed cell death protein 1; PD-L1 programmed cell death ligand 1; PDGF, platelet-derived growth factor; PDGF-R, platelet-derived growth factor receptor; VEGF, vascular epidermal growth factor; VEGF-R, vascular epidermal growth factor receptor.
Targeted TKIs
MET/AXL inhibition
The MET pathway plays a crucial role in the activation of key oncogenic signaling pathways (RAS, PI2K, STAT3) and angiogenesis, while the AXL pathway is involved in epithelial-mesenchymal transition and cancer metastasis. 52 Cabozantinib (formerly known as XL-184) inhibits MET and AXL in addition to VEGF-R, as their upregulated gene expression by VHL inactivation has been implicated as a resistance pathway to anti-angiogenic therapy. 53 An initial phase I study demonstrated promising antitumor activity and safety with cabozantinib in patients with mRCC resistant to VEGF-R and mTOR inhibitors. 54 This paved the way for a landmark phase III study METEOR, in which 658 mRCC patients who had received at least one VEGF-R-targeting TKI were randomized to receive either second-line cabozantinib or everolimus. 55 A significant PFS benefit was seen with cabozantinib compared with everolimus (7.4 months versus 3.8 months, HR = 0.58, 95% CI: 0.45–0.75), with benefit consistently observed regardless of number of prior lines of VEGF-R inhibitors, and across all Memorial Sloan-Kettering Cancer Center risk subgroups. A subsequent analysis showed median OS favored caboazantib (21.4 months versus 16.5 months, HR = 0.67, 95% CI: 0.53–0.83; p = 0.0003). 56 These promising results provided proof of principle that co-inhibition of the MET/AXL pathways is effective and clinically meaningful, and cabozantinib has now received regulatory approval for the treatment of mRCC after progression on VEGF-R TKI therapy in the USA.
Multitargeted kinase inhibition
Lenvatinib is an oral TKI that inhibits multiple targets, including VEGF-R1–3, fibroblast growth factor receptors 1–4, PDGF-Rα, RET, and KIT. Using mouse xenograft models of human RCC showed that the combination of lenvatinib plus everolimus, rather than either alone, resulted in a synergistic treatment response. 57 In a phase Ib study, lenvatinib in combination with everolimus showed manageable toxicity and promising activity: 15 of 18 patients in both lower dose and maximal tolerated dose cohorts achieved either PR (n = 6) or stable disease (SD) (n = 9). 58 This led to a phase II study, as patients with progressive cc mRCC following one prior VEGF-targeted therapy were randomized 1:1:1 to lenvatinib (24 mg/day), everolimus (10 mg/day), or lenvatinib plus everolimus (18 mg/day and 5 mg/day) in 28-day cycles. 53 A total of 153 patients were enrolled: lenvatinib plus everolimus prolonged PFS versus everolimus alone (HR = 0.40; 95% CI: 0.24–0.68; p < 0.001). Lenvatinib monotherapy also prolonged PFS versus everolimus (HR = 0.61; 95% CI: 0.38–0.98; p = 0.048). OS analysis showed a statistically significant survival benefit favoring the combination compared with everolimus alone (HR = 0.51; 95% CI: 0.30–0.88; p = 0.024). For the combination treatment, the most common treatment-emergent adverse events were diarrhea (84%), decreased appetite (51%), and fatigue (47%), with ⩾ grade 3 adverse events of diarrhea (20%), hypertension (14%), and fatigue (10%). Based on these impressive results, this combination is now approved for the treatment of mRCC for those who have progressed on first-line VEGF-R TKI therapy. A phase III randomized trial of the combination in mRCC is currently being planned.
Immunotherapy
CTLA-4 checkpoint inhibition
CTLA-4 is an inhibitory receptor expressed by T cells, which upon ligand binding, initiates T-cell proliferation and function, and maintenance of immune tolerance. 59 A phase II study of the anti-CTLA-4 monoclonal antibody ipilimumab was conducted in patients with mRCC. 60 A total of 5 out of 40 patients who received ipilimumab 3 mg/kg every 3 weeks achieved PR with durations ranging from 7 months to 21 months; the longest responses were among patients who had no prior treatment with IL-2. Immune-related adverse effects grade 3/4 or lower were reported in 17 of 40 patients, with the most common effect being enteritis. There was a highly significant association between autoimmune events and tumor regression (response rate = 30% with autoimmune event, 0% without autoimmune event; p = 0.009). These promising data have led to CheckMate 214, a randomized phase III study evaluating nivolumab combined with ipilimumab versus sunitinib in previously untreated advanced or mRCC. 61
Dendritic cell vaccines
AGS-003 is an autologous dendritic cell vaccine, prepared from matured and optimized monocyte-derived dendritic cells, which are co-electroporated with amplified tumor RNA plus synthetic CD40L RNA. AGS-003, was administered in combination with sunitinib in a phase II study to 21 patients with intermediate and poor risk mRCC, and demonstrated a 62% clinical benefit rate (9 PRs, 4 SD) and median PFS of 11.2 months. 62 Notably, five patients (24%) survived more than 5 years, and AGS-003 was well tolerated with only mild injection-site reactions. The magnitude of the increase in CD8(+) CD28(+) CD45RA(–) effector/memory T cells (CTLs) correlated with OS. Given these encouraging results, a phase III randomized study ADAPT has been launched, with 450 patients randomized in a 2:1 fashion to either sunitinib with AGS-003 or sunitinib alone for those with de novo mRCC (NCT 02639182).
Multipeptide vaccines
IMA901 is a multipeptide vaccine, developed based on the selection of nine human leukocyte antigen (HLA)-class I- and one HLA-class II-binding tumor-associated peptides. 63 A comparison was performed of RNA and HLA ligands derived from normal tissue versus tumor tissue from 32 patients with RCC; immunogenicity was assessed using peripheral blood mononuclear cells derived from healthy donors. A phase II clinical study evaluated disease control rate, OS, and safety of IMA901 intradermal vaccinations administered with or without low-dose cyclophosphamide 300 mg/m2. 64 Disease control rates at 6 months were 31% in the postcytokine group (n = 40), and OS rates were 87%, 79%, and 68% at 6 months, 12 months, and 18 months, respectively. Notably, myeloid-derived suppressor cells, apolipoprotein A-I, and chemokine ligand 17 were predictive for both immune response to IMA901 and OS. A multicenter, open-label, randomized, phase III study with OS as the primary endpoint is ongoing in patients with metastatic or locally advanced RCC when IMA901 is added to first-line therapy with sunitinib (NCT01265901).
Other novel targeted therapies in early clinical development
Ang/Tie-2 inhibition: trebananib
The Ang/Tie-2 pathway is involved in both basal angiogenesis in response to hypoxia in RCC and in vascular stability in the setting of VEGF blockade.65,66 Trebananib (AMG-386) is an anti-angiopoietin peptibody that can disrupt the Ang/Tie-2 axis. A phase II study of sunitinib plus sequential cohorts of trebananib at either 10 mg/kg or 15 mg/kg reported PFS of 13.9 months and 16.0 months, with ORRs at 58% and 59%, suggesting enhanced antitumor activity for the combination. 67 Importantly the toxicity profiles seen were predominantly attributable to sunitinib. A phase II study evaluating trebananib with various anti-VEGF TKIs is currently ongoing.
ALK-1 inhibition: dalantercept
The role of ALK-1 in angiogenesis is complex and context dependent, but emerging data suggest that although VEGF is critical for early endothelial proliferation and sprouting, it has a major role in the development of mature, functional vascular beds.68,69 ALK-1 inhibitors under development include dalantercept (ACE-041) and PF-03446962. Dalantercept inhibits angiogenesis in several preclinical models, and was shown to enhance the efficacy of VEGF-R TKI in RCC xenograft models.70,71 Dalantercept is currently being studied in a phase I/II clinical trial in combination with axitinib in patients with VEGF-R TKI-resistant advanced RCC (NCT01642082).
mTOR complex 1 and 2 and phosphatidylinositol 3 kinase
Currently approved mTOR inhibitors primarily inhibit TORC1 and have less activity against TORC2. The translation of HIF-2a, the dominant oncogene in RCC, is more dependent on the activity of TORC2, and a potential mechanism for acquired resistance to the mTOR complex (mTORC) 1 inhibitors is loss of negative feedback loops that are normally operative when mTORC1 is active potentially leading to phosphatidylinositol 3 kinase (PI3K), AKT, TORC2, and ultimately HIF-2 upregulation. 72 Dual TORC1 and TORC2 inhibition have been hypothesized to enhance the efficacy associated with TORC1 inhibition in patients with RCC.
Several novel dual mTORC1/2 inhibitors have been studied in preclinical models of human RCC. One example is GDC-0980, a dual PI3K/mTORC1/2 inhibitor, which was associated with high rates of adverse effects. BEZ235, showed enhanced efficacy relative to a rapamycin in RCC xenograft models, but development has been halted due to a profound side-effect profile. BKM120, another PI3K inhibitor, is now being tested in combination with bevacizumab in patients with advanced RCC (NCT01283048).
Novel drug conjugates
ENPP3 is a protein expressed in greater than 90% of ccRCCs. AGS-16C3F is a fully human immunoglobulin G 2k monoclonal antibody conjugated to the microtubule disrupting agent monomethyl auristatin F via a plasma-stable linker, which binds to ENPP3. In a phase I study of patients with ccRCC (median prior treatment = 3), AGS-16C3F was well tolerated at 1.8 mg/kg and showed antitumor activity, with a median disease control of nore than 23 weeks and durable PR in 2 of 10 of those with ccRCC. 73 A phase II study with AGS-16C3F at 1.8 mg/kg is currently ongoing (NCT01672775).
Another drug conjugate therapy, CRLX101, is a novel camptothecin containing nanoparticle drug conjugates that deliver sustained levels of active camptothecin into cancer cells while reducing systemic exposure and toxicity. CRLX101 also durably inhibits HIF-1a, and in preclinical models is seen to be synergistic with bevacizumab. Presented at ASCO 2015, an ongoing phase I/II study has thus far enrolled 22 patients with a median of two prior therapies. The median PFS was 9.9 months, with no dose-limiting toxicities observed. 74 ORR was 23%, and 85% experienced clinical benefit (PR or SD). A randomized phase II clinical trial in mRCC is currently ongoing (NCT01625936).
Future directions
With better understanding in pathogenesis and mechanisms of treatment resistance in RCC, there is hope that treatment beyond VEGF inhibition will continue to expand. Understanding the relationship between the tumor microenvironment, mechanisms of resistance to specific treatments, and identifying driver/activating mutations of RCC will require real-time, large-scale genomic analyses of tumor samples at different disease states. Importantly, using the Cancer Genome Atlas analyses can be useful to identify novel driver mutations/activated pathways, and future clinical trials incorporating tissue sampling of a metastatic site before and after disease progression will be key to continuing clinical development of RCC therapy going forward.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
