Abstract
Even though significant improvements in the treatment of colorectal cancer (CRC) have been made in recent years, survival rates for metastatic colorectal cancer (mCRC) are poor. Effective treatment options for metastatic colorectal cancer remain limited, and new therapeutic strategies are desperately needed. Several tyrosine kinase inhibitors (TKIs) and monoclonal antibodies (mAbs) that target angiogenesis, a critical process for facilitating tumor cell growth, invasion, and metastasis, are either approved or in clinical development for the treatment of mCRC. Many of these agents have shown efficacy in mCRC, both as single agents and in combination with standard chemotherapy regimens. However, there is a need for predictive markers of response to identify those patients most likely to benefit from antiangiogenic therapy, and, to date, no markers of this type have been validated in patients. Additionally, because antiangiogenic agents typically cause cytostatic as opposed to cytotoxic antitumor effects, it is important to determine the best strategies for evaluating therapeutic response in mCRC to ensure maximum clinical benefit. In this review, we summarize the efficacy and tolerability of approved and investigational antiangiogenic agents for the treatment of mCRC. We also discuss potential markers of response to antiangiogenic agents and how these markers, along with appropriate endpoint selection, can improve clinical trial design.
Introduction
Colorectal cancer (CRC) is currently the third most common cancer and the second leading cause of cancer-related death in men and women combined in the United States [American Cancer Society, 2015]. When localized, CRC is highly treatable and often curable with surgery. When detected at a localized stage, 5-year survival for CRC is 90%; however, only 40% of CRC cases are diagnosed at an early stage [American Cancer Society, 2015]. A substantial proportion of patients present with metastatic CRC (mCRC) at diagnosis [American Cancer Society, 2015], and about half of patients who have undergone surgery for early-stage CRC with curative intent will develop mCRC [Young et al. 2014]. If cancer has spread to nearby organs or lymph nodes, 5-year survival drops to 71%, and if disease has spread to distant organs, 5-year survival is only 13% [American Cancer Society, 2015]. Despite significant improvements in treatment options for mCRC, outcomes remain poor, emphasizing the need for new agents with efficacy and tolerability in mCRC.
Angiogenesis is an important process for the growth, progression, and metastasis of various solid tumor types, such as CRC, and involves various growth factors and their receptors, including vascular endothelial growth factor and its receptors (VEGF/VEGFRs), platelet-derived growth factor and its receptors (PDGF/PDGFRs), and fibroblast growth factor and its receptors (FGF/FGFRs) [Capdevila et al. 2014]. Mechanisms of targeting angiogenesis include monoclonal antibodies (mAbs) and small molecule tyrosine kinase inhibitors (TKIs). Several antiangiogenic agents are currently approved or in clinical development for the treatment of mCRC, many of which are multitargeted TKIs with varying inhibition signatures (Table 1). This review article summarizes the efficacy and tolerability of approved antiangiogenic agents for mCRC and new antiangiogenic agents under clinical development for mCRC; additionally, we discuss potential strategies to improve clinical trial design (e.g. selection of appropriate surrogate endpoints) and tools for assessing response to antiangiogenic therapy.
Inhibition signatures (IC50, in nmol/l) of antiangiogenic tyrosine kinase inhibitors currently approved or in clinical development for the treatment of metastatic colorectal cancer.
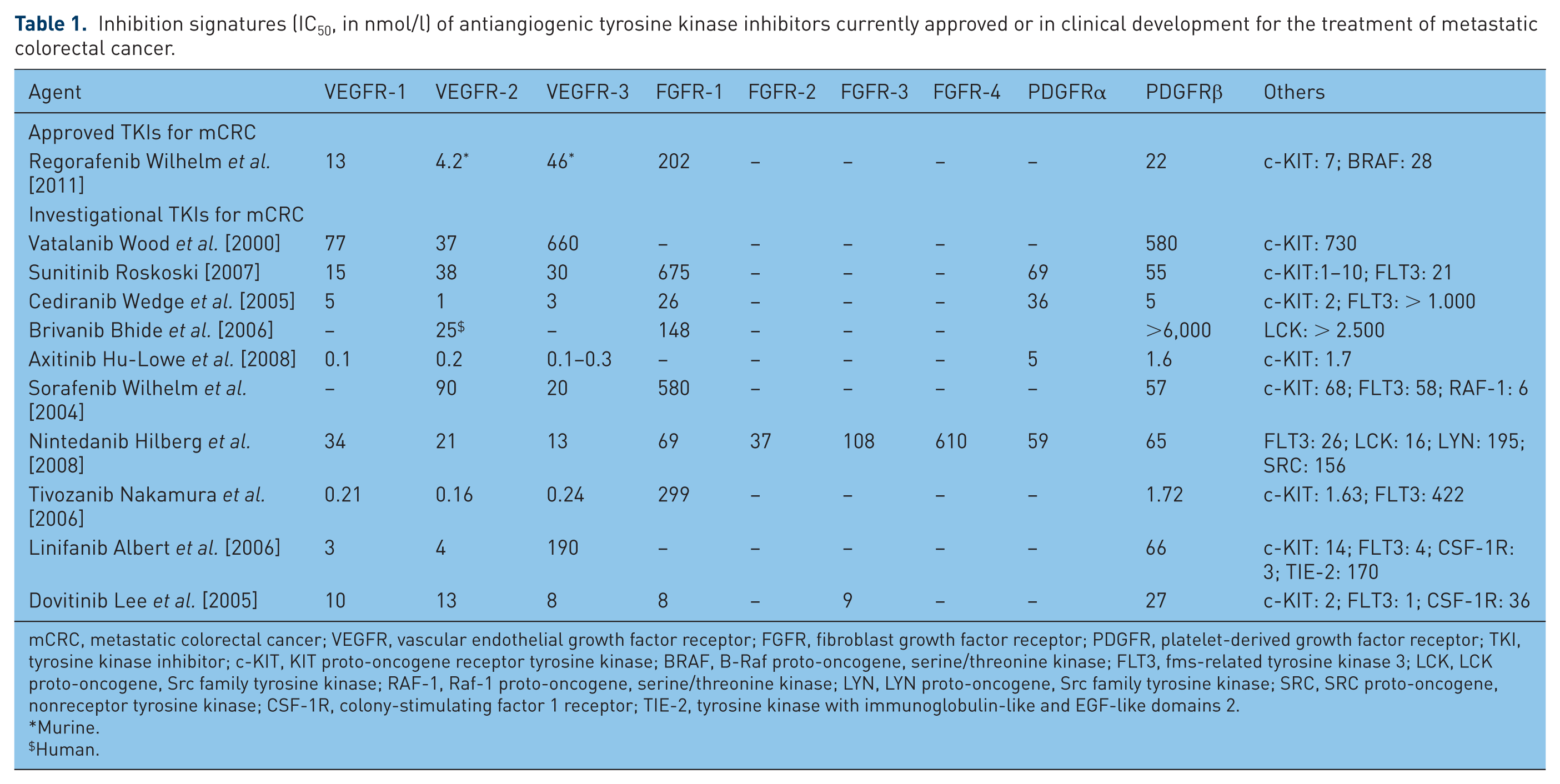
mCRC, metastatic colorectal cancer; VEGFR, vascular endothelial growth factor receptor; FGFR, fibroblast growth factor receptor; PDGFR, platelet-derived growth factor receptor; TKI, tyrosine kinase inhibitor; c-KIT, KIT proto-oncogene receptor tyrosine kinase; BRAF, B-Raf proto-oncogene, serine/threonine kinase; FLT3, fms-related tyrosine kinase 3; LCK, LCK proto-oncogene, Src family tyrosine kinase; RAF-1, Raf-1 proto-oncogene, serine/threonine kinase; LYN, LYN proto-oncogene, Src family tyrosine kinase; SRC, SRC proto-oncogene, nonreceptor tyrosine kinase; CSF-1R, colony-stimulating factor 1 receptor; TIE-2, tyrosine kinase with immunoglobulin-like and EGF-like domains 2.
Murine.
Human.
Efficacy and safety of approved antiangiogenic agents for metastatic colorectal cancer
Systemic chemotherapy has been the standard treatment modality for mCRC for several decades, particularly 5-fluorouracil (5-FU) [Cunningham et al. 2010]. More recently, two regimens, FOLFOX [5-FU + leucovorin (LV) + oxaliplatin] and FOLFIRI (5-FU + LV + irinotecan), were shown to have similar efficacy when given sequentially regardless of administration order, with a median overall survival (OS) of 20 months [Tournigand et al. 2004]. Combinations of a fluoropyrimidine (5-FU or oral capecitabine) with either oxaliplatin [FOLFOX or XELOX (capecitabine + oxaliplatin)] or irinotecan [FOLFIRI or XELIRI (capecitabine + irinotecan)] are widely accepted as standard chemotherapy regimens for first- and second-line treatment of mCRC [Colucci et al. 2005; Douillard et al. 2000; Goldberg et al. 2004; Saltz et al. 2000; Tournigand et al. 2004]. The National Comprehensive Cancer Network guidelines recommend a choice of six chemotherapy regimens for mCRC [FOLFOX, FOLFIRI, XELOX, infusional 5-FU/LV, capecitabine, or FOLFOXIRI (5-FU + oxaliplatin + irinotecan + LV)], with preferred initial therapy depending on the intensity of the treatment [National Comprehensive Cancer Network, 2015].
With the introduction of molecularly targeted agents, such as bevacizumab (an anti-VEGF-A mAb), or cetuximab or panitumumab [both anti–epidermal growth factor receptor (EGFR) mAbs], in combination with chemotherapy, median OS of metastatic CRC has improved to approximately 30 months, particularly in RAS wild-type cancers [Douillard et al. 2010; Hurwitz et al. 2004; Innocenti et al. 2015; Van Cutsem et al. 2011b]. Several antiangiogenic agents have been approved for the treatment of mCRC, and a brief overview of the efficacy and safety of these agents in mCRC is described below, with efficacy results summarized in Table 2.
Summary of efficacy results from phase III trials for approved antiangiogenic agents for metastatic colorectal cancer.

mCRC, metastatic colorectal cancer; ORR, overall response rate; PFS, progression-free survival; TTP, time to progression; OS, overall survival; IFL, irinotecan + 5-fluorouracil (5-FU) + leucovorin (LV); HR, hazard ratio; XELOX, capecitabine + oxaliplatin; FOLFOX4, 5-FU + LV + oxaliplatin; mFOLFOX6, bolus and infusion 5-FU + LV + oxaliplatin; bFOL, bolus 5-FU + low-dose LV + oxaliplatin; FOLFIRI, 5-FU + LV + irinotecan; mIFL, irinotecan + bolus 5-FU/LV; CapeIRI, irinotecan + capecitabine; NR, not reported; CT, chemotherapy; BSC, best supportive care.
FOLFOX4 + bevacizumab versus FOLFOX4 alone.
Log-rank test.
Bevacizumab
Bevacizumab, a humanized IgG1 mAb directed against VEGF-A, was the first approved antiangiogenic agent for the treatment of mCRC, indicated in combination with chemotherapy [Genentech Inc., 2014]. Bevacizumab has demonstrated clinical benefit in combination with chemotherapy in the first-line [Cao et al. 2009; Cassidy et al. 2011; Fuchs et al. 2008, 2007; Hochster et al. 2008; Hurwitz et al. 2004, 2013, 2014; Lv et al. 2013; Petrelli et al. 2013; Saltz et al. 2008; Tebbutt et al. 2010; Van Cutsem et al. 2009] and second-line treatment settings [Bennouna et al. 2013; Cunningham et al. 2013a; Giantonio et al. 2007]. The overwhelming body of positive, albeit modest, evidence supports the use of bevacizumab in the first- and second-line setting (although trial outliers exist) [Passardi et al. 2015]. Moreover, results from the ML18147 study provided the first evidence supporting continuation of bevacizumab after disease progression on standard bevacizumab-based first-line treatment [Bennouna et al. 2013]. Further evidence supporting continuation of bevacizumab beyond progression was observed in the phase III BEBYP study [Masi et al. 2015]. Data also support the use of maintenance treatment with bevacizumab associated with a fluoropyrimidine after induction chemotherapy [Diaz-Rubio et al. 2012; Hegewisch-Becker et al. 2015; Simkens et al. 2015; Stein et al. 2016]. Class-related adverse events (AEs) associated with bevacizumab include hypertension, proteinuria, arterial thrombosis, mucosal bleeding, gastrointestinal perforation, and wound healing problems; importantly, adjusted for time on therapy, bevacizumab does not increase the incidence of chemotherapy-related AEs [Van Cutsem et al. 2014].
Ziv-aflibercept
Ziv-aflibercept is a soluble fusion protein with high affinity for VEGF-A/B and placental growth factor (PlGF) [Van Cutsem et al. 2012]. It is approved in combination with FOLFIRI for patients with mCRC who are resistant to or have progressed following an oxaliplatin-containing regimen, based on the results from the VELOUR trial [Van Cutsem et al. 2012; Sanofi-Aventis, 2012]. In this phase III randomized trial, ziv-aflibercept in combination with FOLFIRI significantly improved median OS, progression-free survival (PFS), and response rate compared with FOLFIRI plus placebo in patients with mCRC previously treated with an oxaliplatin-containing regimen. Benefit of ziv-aflibercept in combination with FOLFIRI was observed in the subgroup of 30% of patients overall who had received previous bevacizumab. Ziv-aflibercept has also been studied in the first-line setting in combination with modified FOLFOX6 (mFOLFOX6) in the phase II randomized AFFIRM study, and appeared to show similar efficacy to mFOLFOX6 alone [Pericay et al. 2012]. The most common grade 3/4 AEs associated with ziv-aflibercept include side effects associated with VEGF inhibition (e.g. hypertension, proteinuria) and other AEs associated with irinotecan-based chemotherapy (e.g. neutropenia, diarrhea, asthenia, stomatitis) [Van Cutsem et al. 2012].
Ramucirumab
Ramucirumab, a human VEGFR-2 mAb, is approved for the treatment of mCRC in combination with FOLFIRI for patients with disease progression on or after prior therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine [Eli Lilly and Company, 2015]. Results from the phase III, randomized, double-blind RAISE study showed that ramucirumab plus FOLFIRI significantly improved OS versus placebo plus FOLFIRI as second-line treatment [Tabernero et al. 2015b]. The most common grade 3/4 AEs in the ramucirumab group were neutropenia, hypertension, diarrhea, and fatigue.
Regorafenib
Regorafenib is a multitargeted TKI that inhibits VEGFR-1/2/3, PDGFRβ, FGFR-1, TIE-2, c-KIT, RET, RAF-1, and BRAF [Wilhelm et al. 2011] and is approved for the treatment of patients with mCRC who have been previously treated with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy, an anti-VEGF therapy, and, if Kirsten rat sarcoma viral oncogene homolog (KRAS) wild type, an anti-EGFR therapy [Bayer Inc., 2015]. Results from the phase III, randomized, placebo-controlled CORRECT and CONCUR trials show improvements in PFS and OS with regorafenib monotherapy versus placebo in patients who have progressed after all standard therapies [Grothey et al. 2013; Li et al. 2015]. Grade 3/4 AEs associated with regorafenib include VEGF-related hypertension, hand–foot skin reaction, fatigue, diarrhea, and rash/desquamation. Regorafenib-related AEs generally occurred during the first one-to-two cycles of treatment and were manageable with dose reduction or interruption.
Potential biomarkers for antiangiogenic therapies in metastatic colorectal cancer
In recent years, research efforts have focused on identifying potential predictive biomarkers to provide information on tumor response to antiangiogenic therapy for mCRC. However, the complexity of tumor–host interactions coupled with the complexity of the VEGF pathway have made the identification of predictive biomarkers for antiangiogenic therapies challenging [Khan et al. 2015]. While several markers have appeared promising in preclinical models, they have failed as predictors of response in human trials [Crawford et al. 2009; Li et al. 2007; Pan et al. 2007]. Thus, to date, no biomarkers have emerged that are able to predict the efficacy of antiangiogenic agents. Several studies show that VEGF plasma levels are considered prognostic in patients with mCRC, but they are not predictive biomarkers for anti-VEGF therapy [Cascinu et al. 2002; Davies et al. 2000; Lee et al. 2000; Maeda et al. 2000; Nakayama et al. 2002].
There has been little advancement in identifying predictive markers for bevacizumab [Kasi et al. 2015]. In a comprehensive evaluation of four phase III trials of bevacizumab in CRC, lung cancer, and renal cell carcinoma (RCC), pretreatment total circulating VEGF-A was prognostic but not predictive for bevacizumab-based treatment benefit [Hegde et al. 2013]. In the phase III MAX study, KRAS and BRAF mutations were not predictive of bevacizumab benefit [Price et al. 2011]. Similarly, results from retrospective subset analyses suggest that the survival benefit observed with bevacizumab plus irinotecan/5-FU/LV as first-line therapy is independent of KRAS, BRAF, or p53 mutation status [Ince et al. 2005]. Likewise, in the ML18147 trial, KRAS mutation status did not predict efficacy of second-line bevacizumab plus chemotherapy in patients progressing after first-line bevacizumab and standard chemotherapy [Kubicka et al. 2013]. In another analysis of tissue samples from patients who participated in the MAX trial, Bruhn and colleagues used a multiplex protein assay to determine tumor expression levels of proangiogenic proteins; however, no proangiogenic proteins were predictive of PFS benefit with bevacizumab [Bruhn et al. 2014].
Recent studies suggest other types of potential biomarkers that may be of interest for bevacizumab in mCRC. Results from the Nordic ACT trial and subsequent analyses confirm the prognostic value of miRNA-126 in mCRC and suggest that the VEGFR-1 319 C/A single nucleotide polymorphism is a potential predictive biomarker for bevacizumab plus chemotherapy for mCRC [Hansen et al. 2012, 2013]. Additionally, changes in circulating miRNA-126 have been associated with response to therapy and may represent a resistance biomarker in the setting of first-line chemotherapy plus bevacizumab [Hansen et al. 2015]. There is some evidence for the predictive and possibly prognostic value of endothelial cell and angiogenic markers for mCRC treatment [Hansen et al. 2012, 2014; Liu et al. 2013], and these observations merit future follow-up studies. Results from a study comparing chemotherapy combined with bevacizumab as first-line treatment for mCRC suggest potential predictive ability of VEGF genotypes in patients with mCRC; however, these results need to be validated prospectively in larger populations [Koutras et al. 2012]. In a retrospective review of patients with mCRC who received bevacizumab-containing first-line regimens, the nonspecific tumor marker carcinoembryonic antigen (CEA) was the only circulating biomarker with accuracy for identifying patients with worse outcome during bevacizumab treatment [Martinetti et al. 2014]. Additional studies have suggested that variants in angiogenesis-related genes and serum VEGF-A levels may be associated with response to bevacizumab plus chemotherapy in mCRC, and gene expression profiling has identified gene signatures that may have predictive value for response [de Haas et al. 2014; Hayashi et al. 2014; Pentheroudakis et al. 2014].
Hypertension, a common AE associated with anti-VEGF therapy, has been evaluated as a potential surrogate biomarker for efficacy from antiangiogenic agents, but results have been conflicting. In a meta-analysis of nine studies, bevacizumab-related hypertension was associated with increased response rate and improved PFS and OS, suggesting that hypertension may be a surrogate of bevacizumab efficacy in mCRC; however, additional prospective trials are needed to confirm these findings [Chen et al. 2014]. In a retrospective analysis of patients with mCRC treated with bevacizumab or a TKI in combination with chemotherapy, development of proteinuria during treatment was associated with poorer survival, while hypertension was predictive of significantly higher response rates [Khoja et al. 2014].
Biomarker analyses are also being conducted with regorafenib in mCRC. In an exploratory biomarker subgroup analysis of the phase III CORRECT trial using BEAMing technology, regorafenib was associated with clinical benefit versus placebo across mutational subgroups defined by KRAS and PIK3CA mutational status or circulating DNA concentration [Tabernero et al. 2015a]. The only plasma protein associated with longer OS was high versus low concentrations of TIE-1; however, this association was not significant in multivariable analyses. Del Prete and colleagues analyzed the role of lactate dehydrogenase (LDH) serum levels in predicting clinical outcome in pretreated mCRC patients receiving regorafenib, with results suggesting a role for LDH as a marker of clinical outcome [Del Prete et al. 2014]. In another analysis of pretreated patients with mCRC, high platelet count and high neutrophil/lymphocyte ratio were predictive factors for clinical outcome in regorafenib-treated patients [Del Prete et al. 2015]. However, both high LDH and platelet levels most likely reflect larger tumor burden.
Prospective trials are needed to further evaluate these potential predictive biomarkers and to identify new biomarkers that may help incorporate antiangiogenic agents into the treatment paradigm for mCRC. Potential biomarkers are a common theme of study for agents with approved indications in mCRC and for new antiangiogenic agents under clinical development. It is very unlikely that a single molecular alteration in CRC will be predictive of benefit to antiangiogenic agents because these agents target the tumor microenvironment, which is a genetically stable system. Initial preclinical studies suggested that angiogenic inhibitors help normalize blood vessels that supply the tumor, decreasing vessel permeability and intratumoral pressure, and thus facilitating the delivery of antineoplastic agents [Jain, 2001, 2005]. However, this concept has recently been challenged in other models and, in fact, a decrease in the delivery of chemotherapy after anti-VEGF therapy has been observed [van der Veldt et al. 2012]. It also appears that the benefits provided by antiangiogenic agents have reached a plateau; effects are generally modest and, in clinical practice, we do not see specific subgroups of patients who achieve outstanding responses. The Colorectal Cancer Subtyping Consortium recently analyzed more than 30 patient cohorts and came to a clear consensus on four CRC molecular subtypes enriched for key clinical, pathway, and molecular traits [Guinney et al. 2015], which may provide a potential predictive way to measure efficacy for antiangiogenic agents.
Other antiangiogenic agents for refractory metastatic colorectal cancer
Numerous antiangiogenic agents are under clinical development for the treatment of mCRC. Results from these clinical studies are summarized below and in Table 3.
Summary of efficacy results from select phase II/III trials for investigational antiangiogenic agents for metastatic colorectal cancer.

mCRC, metastatic colorectal cancer; ORR, overall response rate; PFS, progression-free survival; TTP, time to progression; OS, overall survival; FOLFOX, ; FOLFOX4, 5-fluorouracil (5-FU) + leucovorin (LV) + oxaliplatin; NR, not reported; NS, not significant; HR, hazard ratio; FOLFIRI, 5-FU + LV + irinotecan; XELOX, capecitabine + oxaliplatin; mFOLFOX6, bolus and infusion 5-FU + LV + oxaliplatin.
Several multitargeted TKIs have been evaluated in combination with chemotherapy for mCRC but failed to meet the primary endpoint in phase III studies. Vatalanib, an angiogenesis inhibitor that blocks VEGFR-1 and -2, did not significantly improve PFS or OS when combined with FOLFOX4 versus placebo plus FOLFOX4 as first-line therapy (CONFIRM 1) [Hecht et al. 2011], and did not improve OS in combination with FOLFOX4 as second-line therapy (CONFIRM-2) [Van Cutsem et al. 2011a] for mCRC. In both studies, there was a higher incidence of AEs leading to treatment discontinuation with vatalanib and lower FOLFOX4 dose intensity.
Sunitinib, a multitargeted inhibitor of VEGFR-2, PDGFR-β, c-KIT, and FLT3 that is approved for advanced RCC and gastrointestinal stromal tumor after disease progression on, or intolerance to, imatinib mesylate, and progressive, well-differentiated pancreatic neuroendocrine tumors in patients with unresectable locally advanced or metastatic disease [Pfizer Inc., 2011], did not significantly improve PFS or OS in combination with FOLFIRI versus placebo plus FOLFIRI as first-line therapy for mCRC, with more grade ⩾ 3 AEs (particularly diarrhea, stomatitis/oral syndromes, fatigue, hand–foot syndrome, neutropenia, thrombocytopenia, anemia, and febrile neutropenia), deaths as a result of toxicity, dose delays/reductions, and treatment discontinuations in the sunitinib arm [Carrato et al. 2013].
Cediranib, a TKI with activity against all 3 VEGFRs, failed to improve OS when combined with chemotherapy (FOLFOX or capecitabine/oxaliplatin) versus chemotherapy alone in the phase III HORIZON II first-line mCRC study, but did improve PFS; median chemotherapy dose intensity was decreased by about 10% in patients treated with cediranib, which may have compromised the results [Hoff et al. 2012]. In the phase III HORIZON III first-line mCRC study, OS and PFS were comparable with cediranib plus mFOLFOX6 versus bevacizumab plus mFOLFOX6, but the predefined boundary for PFS noninferiority was not met [Schmoll et al. 2012]. Patients treated with cediranib completed fewer chemotherapy cycles versus patients treated with bevacizumab and had significantly less favorable patient-reported outcomes [Schmoll et al. 2012]. Similar results were observed in the phase III HORIZON I second-line mCRC study, in which PFS and OS were similar with cediranib 20 or 30 mg plus mFOLFOX6 versus bevacizumab plus mFOLFOX6 [Cunningham et al. 2013b]. Grade ⩾ 3 AEs were more common with cediranib 30 mg versus cediranib 20 mg or bevacizumab. Exploratory biomarker analyses from the HORIZON studies did not reveal any predictive biomarkers for cediranib response [Jurgensmeier et al. 2013]. One factor that may have contributed to the phase III studies of vatalanib, cediranib, and sunitinib plus chemotherapy not meeting their efficacy endpoints was the reduction in chemotherapy administration because of increased toxicity with these agents [Capdevila et al. 2014].
Brivanib, a small molecule TKI of VEGFR and FGFR, was combined with cetuximab in a phase III study of patients with wild-type KRAS mCRC previously treated with combination chemotherapy [Siu et al. 2013]. While PFS and response rates were significantly better with the combination versus cetuximab alone, addition of brivanib did not significantly improve OS (primary endpoint) and increased toxicity [Siu et al. 2013]. Fewer patients taking brivanib received ⩾90% of the dose intensity of cetuximab versus those receiving placebo.
Axitinib is a selective inhibitor of VEGFR-1/2/3, PDGFRβ, and c-KIT [Choueiri, 2008] that is approved for the treatment of advanced RCC after failure of one prior systemic therapy [Pfizer Inc., 2012]. In a phase II study of axitinib, bevacizumab, or both in combination with mFOLFOX6 as first-line therapy for mCRC, neither axitinib nor axitinib plus bevacizumab combined with mFOLFOX6 improved PFS, OS, or response rate versus bevacizumab plus chemotherapy [Infante et al. 2013]. Patients who received axitinib had fewer treatment cycles and more frequent hypertension and headache episodes [Infante et al. 2013]. Similarly, in a phase II study of second-line axitinib or bevacizumab with mFOLFOX6 or FOLFIRI, axitinib did not improve outcomes versus bevacizumab plus chemotherapy and was less well tolerated [Bendell et al. 2013]. Initial data suggest maintenance axitinib may be effective after first-line mFOLFOX plus bevacizumab for mCRC [Joseph et al. 2015].
Sorafenib, a TKI of VEGFR-1/2/3, PDGFR, c-KIT, FLT3, and RAF that is approved for unresectable hepatocellular carcinoma, advanced RCC, and locally recurrent or metastatic, progressive, differentiated thyroid carcinoma refractory to radioactive iodine treatment [Bayer HealthCare Pharmaceuticals, 2015], is being studied in both the first- and second-line treatment settings for mCRC. In a phase I/II study, sorafenib plus irinotecan showed promising activity as second- or later-line treatment in patients with heavily pretreated KRAS-mutated mCRC [Samalin et al. 2014]. In the phase II RESPECT trial of sorafenib plus mFOLFOX6 as first-line treatment, the addition of sorafenib did not improve PFS or OS versus chemotherapy alone; KRAS and BRAF status did not seem to impact treatment outcomes [Tabernero et al. 2013]. The most common grade 3/4 AEs with sorafenib were neutropenia, peripheral neuropathy, and grade 3 hand–foot skin reaction, with a lower dose intensity in the sorafenib group. New data were recently presented for the ongoing phase II trial of sorafenib plus irinotecan versus sorafenib or irinotecan monotherapy in KRAS-mutated mCRC after failure of all approved active drugs at the time of study (NEXIRI 2) [Samalin et al. 2016]; the study demonstrated a higher rate of nonprogression at 2 months of treatment with the combination treatment versus irinotecan or sorafenib monotherapy (59% versus 23% versus 22%), along with a modest increase in median PFS (3.7 versus 1.9 versus 2.1 months) and median OS (7.0 versus 6.3 versus 5.1 months). Elevations in grade 3/4 neutropenia, febrile neutropenia, and diarrhea were observed with the combination compared with the irinotecan or sorafenib monotherapy. Ongoing trials of sorafenib in mCRC are summarized in Table 4.
Summary of ongoing phase II/III clinical trials using investigational antiangiogenic agents for metastatic colorectal cancer.

mCRC, metastatic colorectal cancer; BSC, best supportive care; CRC, colorectal cancer.
Information was last accessed in August 2016.
New antiangiogenic agents
Nintedanib, a triple angiokinase inhibitor of VEGFR-1/2/3, PDGFRα/β, FGFR-1/2/3, FLT3, and members of the src family [Hilberg et al. 2008], is also in phase III development for mCRC. Nintedanib is approved in the European Union in combination with docetaxel for the treatment of patients with locally advanced, metastatic, or locally recurrent non-small cell lung cancer of adenocarcinoma histology after first-line chemotherapy [European Medicines Agency, 2015a]; it is also approved in the European Union and in the United States for the treatment of patients with idiopathic pulmonary fibrosis [European Medicines Agency, 2015b; Boehringer Ingelheim Pharmaceuticals, 2014]. Phase I results of nintedanib monotherapy in 21 patients with refractory mCRC showed one confirmed partial response and clinically relevant antivascular effect in 67% of patients [Mross et al. 2014]. In a phase II study of alternating 7-day treatment periods, first with nintedanib and then with afatinib (an ErbB family blocker), 43.5% of patients maintained stable disease and 15.2% remained progression free at 16 weeks of treatment [Bouche et al. 2011]. In a phase I/II study, nintedanib demonstrated preliminary efficacy versus bevacizumab when combined with mFOLFOX6 as first-line therapy for mCRC, although no statistical comparisons between groups were performed [Van Cutsem et al. 2015]. There was a lower proportion of patients discontinuing treatment with nintedanib versus bevacizumab for AEs or serious AEs, and nintedanib did not decrease treatment exposure of mFOLFOX6 treatment compared with bevacizumab. Ongoing trials include the phase III LUME-Colon 1 study [ClinicalTrials.gov identifier: NCT02149108] in refractory CRC and a phase I/II study in combination with capecitabine [ClinicalTrials.gov identifier: NCT02393755].
Tivozanib (AV-951), a TKI of VEGFR-1/2/3, was evaluated in combination with everolimus, a mammalian target of rapamycin inhibitor, in a phase II study in patients with refractory mCRC [Wolpin et al. 2013]. Stable disease was achieved in 50% of patients, and the combination was well tolerated. Patients who developed grade 1+ hypertension had increased stable disease rates and longer OS times, suggesting that hypertension may be a predictive marker for tivozanib efficacy. The most common grade 3/4 AEs were thrombocytopenia and hypophosphatemia.
Linifanib (ABT-869), a selective TKI of VEGFR and PDGFR families, in combination with mFOLFOX6, was compared with bevacizumab plus mFOLFOX6 as second-line treatment for mCRC [O’Neil et al. 2014]. While efficacy results were similar with linifanib plus mFOLFOX6 compared with bevacizumab plus mFOLFOX6, grade 3/4 AEs were more common with linifanib, and median cycles of 5-FU were reduced with linifanib versus bevacizumab. Therefore, the authors concluded that use of linifanib, as second-line treatment for mCRC is not supported.
Icrucumab (IMC-18F1), a human mAb against VEGFR-1, showed potential antitumor activity against advanced solid tumors, including CRC, in a phase I study [LoRusso et al. 2014]. Results from a phase II randomized study of mFOLFOX6 with or without icrucumab or ramucirumab as second-line therapy after first-line irinotecan-based therapy are eagerly awaited [ClinicalTrials.gov identifier: NCT01111604].
Dovitinib, a TKI that inhibits VEGFR-1/2, FGFR-1/3, PDGFRβ, FLT3, and c-KIT, has also shown preclinical activity in CRC cells regardless of KRAS or BRAF mutation status [Gaur et al. 2014; Lee et al. 2015], but initial results from a phase II study in patients with advanced CRC previously treated with anti-VEGF therapy [ClinicalTrials.gov identifier: NCT01676714] showed no evidence of clinical activity [Semrad et al. 2016].
New strategies for clinical trial design and imaging techniques for metastatic colorectal cancer
Unlike cytotoxic chemotherapy, angiogenic TKIs cause cytostatic effects and, thus, using Response Evaluation Criteria In Solid Tumors (RECIST) response criteria may not provide accurate assessment of the clinical benefits gained from these agents [Khan et al. 2015]. For example, patients treated with regorafenib may present significant cavitation of tumor lesions, although this does not correlate with objective response [Lim et al. 2015]. Additionally, response rates may not always translate to clinical benefit in terms of PFS and possibly OS benefits in mCRC [Gill et al. 2011]. Because cytostatic drugs result in stable disease, response rate might be low with these agents, but benefits in terms of PFS and possibly OS may still be observed [Wilson et al. 2015]. For example, in an analysis of patients with colorectal liver metastases treated with bevacizumab-containing chemotherapy regimens, response determined by RECIST criteria did not correlate with survival [Chun et al. 2009]. Moreover, RECIST response criteria likely detect late rather than early progression, so new forms of measuring early progression are crucial for drug development in solid tumors. It is important to remember that the strength of antiangiogenic agents is delaying tumor progression rather than inducing tumor shrinkage and, thus, time-related endpoints need to be chosen.
Imaging techniques may be useful in determining response or resistance to antiangiogenic therapies, as they can be used to verify tumor morphology and blood flow [Khan et al. 2015]. Dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) appears to be a new imaging biomarker that could serve as a useful tool for determining response to antiangiogenic agents in mCRC [Jackson et al. 2007; Morgan et al. 2003; Tuncbilek et al. 2004]. Diffusion-weighted MRI (DW-MRI) may also provide useful information in determining response to antiangiogenic therapies [Li and Padhani 2012; Padhani et al. 2009]. Another potential imaging biomarker is positron emission tomography (PET) [Khan et al. 2015; Nagengast et al. 2011]. There is growing interest in fluorodeoxyglucose (FDG) PET/computed tomography (CT) to assess response to therapy, but this technique needs to be validated in prospective clinical trials [Wilson et al. 2015]. Authors of a systematic review of published literature concluded that 18F-FDG PET has a high predictive value and should be implemented into clinical practice for the management of CRC [de Geus-Oei et al. 2009]. In a study comparing eight metabolic indices derived from an FDG PET/CT scan for early assessment of tumor response in 40 patients with mCRC, all standardized uptake values uncorrected for partial volume effect and calculated in early PET scans (2 weeks after starting chemotherapy) accurately predicted later response (after 6–8 weeks on therapy) using RECIST response criteria in CT scans [Maisonobe et al. 2013]. Another study that looked at intra-individual metabolic responses via standardized FDG-PET/CT among patients with mCRC treated with sorafenib plus capecitabine (SoMore) showed that the presence of ⩾1 metabolically refractory lesion was associated with a poorer outcome among patients than the absence of metabolically refractory lesions, suggesting that metabolic response heterogeneity could be considered as a predictive efficacy biomarker for this treatment regimen [Hendlisz et al. 2015]. While these imaging biomarkers still require validation, these tools are becoming increasingly available in many research centers, which could allow imaging biomarkers to be available and validated within the near future [Khan et al. 2015].
With respect to trial endpoints, while improvement in OS is easily measured, unambiguous, and objective, and is generally considered the gold standard measurement of treatment efficacy in oncology, it is also associated with several limitations [Gill et al. 2011; Wilson et al. 2015]. When considered as an endpoint for trials that were conducted in an early-line setting, the measurement of OS requires large patient numbers, prolonged follow-up time periods, and can be confounded by other factors such as subsequent therapies and crossover within randomized trials from control to investigational drug. PFS is considered a sensitive indicator of treatment efficacy for mCRC [Gill et al. 2011; Wilson et al. 2015]. In comparison with OS, PFS can be evaluated earlier, requires fewer patients, and is not confounded by crossover and subsequent lines of therapy. While some data suggest that PFS is an acceptable surrogate endpoint and is strongly associated with improvements in OS in chemotherapy trials for mCRC [Buyse et al. 2007; Tang et al. 2007], the availability of active post-first-line progression therapies negates that finding in the modern era. In a recent analysis of 16,762 patients from 22 first-line mCRC studies, including 12 studies testing antiangiogenic or anti-EGFR agents, there was a positive but only modest correlation between PFS and OS [Shi et al. 2015]. Moreover, PFS is associated with measurement error and bias, as determining time of disease progression is an approximation, and evaluation of progression using radiographic scans is subjective [Gill et al. 2011]. Ultimately, the choice of appropriate endpoint (OS versus PFS) for mCRC depends on the setting in which the trial is being performed. For first-line trials, it is not reasonable to choose OS as the primary endpoint when the predicted magnitude of effect is modest (for example, a hazard ratio of 0.80) due to the influence of subsequent lines of therapy. However, in a salvage-therapy setting, OS is an appropriate endpoint for clinical studies in mCRC.
Another endpoint worthy of consideration is patient-reported outcomes, which are difficult to measure in the refractory mCRC setting since patients are typically asymptomatic. Time to clinical deterioration is an interesting and clinically relevant endpoint in trials of refractory cancers. However, its definition is subjective and influenced by treatment-related toxicities. Quality of life endpoints are also complex to measure as they are subject to culture variability, methodological problems (e.g. handling missing data), and difficulty in conducting intention-to-treat analyses.
Conclusion
In recent years, significant progress has been realized in the management of mCRC. The consistent, albeit modest, evidence of clinical efficacy observed with antiangiogenic agents in the treatment of mCRC provides the rationale for targeting angiogenesis as a treatment strategy for this disease, with several antiangiogenic agents being approved for the treatment of mCRC within the last decade. As results become available from ongoing clinical trials of newer antiangiogenic agents currently in development, clinicians will need to determine the most appropriate and beneficial role for these agents within the mCRC treatment continuum and perhaps decide whether future research should continue to target the VEGF pathway or focus on other possible treatment pathways or combinations. Preliminary results from a phase 1b study suggest the potential benefit of combining immunotherapy with atezolizumab, an antiprogrammed death-ligand 1 mAb, with bevacizumab with or without FOLFOX in patients with mCRC [Bendell et al. 2015]. Validated predictive biomarkers are critical to identify patients who will most likely benefit from treatment with antiangiogenic agents, as well as from any cancer-directed therapy for mCRC. In this regard, stratifying patients according to their molecular subtype might potentially identify subgroups of patients whose disease is more sensitive to antiangiogenics. Retrospective analyses of tumor tissue collected in phase III trials, prospective studies, and meta-analyses of biomarker data are needed. The drug development process for new molecularly targeted agents for mCRC, including antiangiogenic agents, will need to take into account the new forms for measuring response/progression and then reliably assess tumor control through PFS as an endpoint, particularly in the first-line treatment setting.
Footnotes
Acknowledgements
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors received no direct compensation related to the development of the manuscript. Writing and editorial support were provided by Melissa Brunckhorst, PhD, of MedErgy, which was contracted and funded by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). BIPI was given the opportunity to review the manuscript for medical and scientific accuracy, as well as intellectual property considerations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Writing and editorial support were provided by Melissa Brunckhorst, PhD, of MedErgy, which was contracted and funded by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI).
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rachel Riechelmann: research grants from Bayer; honoraria for lectures from Bayer, Roche, and Amgen. Axel Grothey: research grants were provided to the Mayo Clinic Foundation by Bayer, Boehringer Ingelheim, Boston Biomedicals, Eisai, Genentech, and Sanofi.
