Abstract
Postmastectomy radiotherapy (PMRT) has been shown to decrease locoregional recurrence and improve overall survival in patients with tumors greater than 5 cm or positive nodes. Because neoadjuvant chemotherapy (NAC) can cause significant downstaging, the indications for PMRT in the setting of NAC remain controversial and thus careful consideration of clinical stage at presentation, pathologic response to NAC, and other clinical characteristics, such as grade and biomarker status is required. The current review synthesizes both prospective and retrospective data to provide evidence for recommending PMRT after NAC for patients presenting with cT3–4 disease, cN2–3 disease, and residual nodal disease, as well as rationale for omitting PMRT in patients with cT1-2N0-1 disease who achieve a pathologic complete response. Other scenarios, including nodal complete response in the presence of other risk factors, are also explored. The topics of pre-NAC clinical staging and pathologic axillary nodal staging are reviewed, and radiation portal design is briefly discussed.
Introduction and general principles of postmastectomy radiation
The American College of Radiology (ACR) currently recommends consideration of postmastectomy radiation (PMRT) for patients with tumors greater than 5 cm or with at least four positive nodes [Taylor et al. 2009]. This recommendation is largely based on the results of three randomized controlled trials that demonstrated an overall survival (OS) benefit for PMRT in patients with lymph node positive disease (pN+), tumors greater than 5 cm in size, and pectoralis fascia/skin involvement [Overgaard et al. 1997, 1999; Ragaz et al. 2005]. This OS benefit is thought to stem from the fact that PMRT reduces the risk of locoregional recurrence (LRR) by approximately two thirds [Clarke et al. 2005]. The most recent iteration of the Early Breast Cancer Trialist’s Collaborative Group (EBCTCG) meta-analysis showed that among 3086 women, PMRT reduced the 10-year first LRR rate from 26.0% to 8.1%, with a corresponding reduction in 20-year breast-cancer mortality from 66.4% to 58.3% [McGale et al. 2014]. Notably, equivalent gains in LRR and breast cancer mortality were noted among subsets with one to three nodes positive or at least four nodes positive. An earlier EBCTG analysis suggested that an absolute reduction in 5-year LRR of over 10% would be necessary for a survival benefit to be shown at 15 years [Clarke et al. 2005]. However, some suggest employing PMRT even when the absolute LRR risk (not the absolute risk reduction) is greater than 10%, citing the expected long-term survival of these patients.
Despite the availability of three randomized trials and meta-analyses to guide PMRT decision making in the absence of neoadjuvant chemotherapy (NAC), PMRT in this setting is not without controversy. Several institutional series have noted very low rates of locoregional failure among select women with T3N0 or T2N1 disease being treated in the modern era [Taghian et al. 2006; Yu et al. 2008; Floyd and Taghian, 2009; Moo et al. 2013; McBride et al. 2014]. Other series have suggested expanding the role of PMRT to include select patients with T1–2N0 breast cancer who have particularly high-risk features (e.g. young age, high grade, triple negative) based on high documented risk of LRR in the absence of PMRT [Rowell, 2009; Abdulkarim et al. 2011]. The role of PMRT remains widely debated among women with T3N0 or T1-2N0-1 disease, with most practitioners individualizing radiation decision making based on perceived risk.
The decision to deliver PMRT depends on both the absolute risk of LRR and the magnitude of reduction in this absolute risk. Prospective data are lacking to inform PMRT decision making following NAC and, given that NAC commonly alters postmastectomy pathologic findings with at least 20–40% of patients receiving NAC having axillary downstaging [Fisher et al. 1997], it is unclear if we can extrapolate findings from the available data informing PMRT decision making in the absence of NAC. In 2008, the National Cancer Institute published a consensus statement suggesting that PMRT after NAC be delivered for patients with clinical stage III disease (i.e. T4, N2–N3, or T3N1) or ypN+ disease [Buchholz et al. 2008]. However, a survey of practicing radiation oncologists reported heterogeneity in adherence to the latter portion of the recommendation, and in decision making with regards to patients with stage II disease in general [Beriwal et al. 2013].
The purpose of this focused review is to analyze the relevant literature examining the benefits or lack thereof for PMRT in the setting of NAC. Additionally, the subjects of pre-NAC staging workup, pathologic axillary staging, and radiation portal design will be briefly considered. Conclusions regarding situations for which PMRT following NAC should be considered will be provided.
Indications for PMRT after NAC: literature review
Lessons from retrospective series
Natural history of LRR: implications for PMRT
PMRT following NAC is inherently controversial due to the lack of prospective data available to guide decision making. We have thus relied on retrospective data to inform treatment decisions. Retrospective analyses have typically examined LRR risk in subsets of women who have not received PMRT following NAC to assess overall LRR risk and risk according to specific patient and tumor features. When possible, retrospective series have attempted to compare women receiving and not receiving PMRT. We have in general learned from these studies, which will be detailed below, that careful consideration of pre-NAC clinical stage, post-NAC pathologic stage, and individual patient and tumor factors is critically important to rendering appropriate radiation therapy recommendations.
One of the earliest series suggesting a locoregional control benefit for PMRT after NAC focused on 55 patients with locally advanced breast cancer (i.e. cT3–4 or cN2–3) [Abdel-Wahab et al. 1998]. Forty-two patients received PMRT and 13 did not. With a median follow up of 47 months, the LRR rates were 31% for patients treated without PMRT and 7% for patients treated with PMRT. This translated to a significant 3-year OS benefit of 88% versus 46%.
The MD Anderson Cancer Center (MDACC) then published a series of reports detailing their findings from retrospective analysis of robust institutional data. In one of the earliest series examining risk factors for LRR among patients undergoing mastectomy after NAC, Buccholz and colleagues analyzed outcomes of 150 such patients who did not receive PMRT [Buchholz et al. 2002b]. The 10-year actuarial LRR rate was 27% in this cohort, which was largely composed of patients with locally advanced disease (55% of patients had clinical stage IIIA disease or higher). Predictors of LRR included clinical stage IIIB disease or greater at presentation, at least four positive nodes pathologically, and no use of tamoxifen. The MDACC group subsequently compared 5-year LRR patterns between the same 150 patients treated with NAC and no PMRT and 1031 patients treated with adjuvant chemotherapy and no PMRT [Buchholz et al. 2002a]. Despite a large imbalance in clinical stage at presentation (55% stage IIIA or higher in NAC versus 9% in adjuvant), the overall pathologic tumor size and number of involved axillary nodes were the same. However, the overall 5-year LRR rate was greater for patients receiving NAC (27% versus 15%), with statistically significant differences seen in subsets of patients with tumors matched for size or magnitude of nodal disease. Taken together, these data strongly suggested that the risk of LRR depended on both initial clinical factors and pathologic factors at the time of surgery (i.e. pathologic findings consistent with response to NAC do not negate more locally advanced clinical presentation).
The MDACC group next demonstrated the potential benefits of PMRT by comparing outcomes of 542 patients treated with NAC, mastectomy, and PMRT with those of 134 patients treated with NAC and mastectomy alone (essentially the same 150 patients originally studied, less the 16 who experienced an early recurrence within 2 months of treatment) [Huang et al. 2004]. The 10-year LRR rate was 22% in the absence of PMRT and 11% with PMRT with a hazard ratio (HR) of LRR without PMRT of 4.7. The 10-year cause-specific survival (CSS), however, was identical between the two groups (58% with PMRT and 50% without), but on multivariate analysis PMRT provided a significant benefit, with a HR of 2.0. Significant improvement in CSS was detected for patients with at least stage IIIB disease, cT4 tumors, and at least four involved nodes. The authors subsequently reported that five factors predicted for 10-year LRR in this cohort: skin/nipple involvement, supraclavicular nodal disease, no tamoxifen use, extracapsular extension, and estrogen-receptor negative disease [Huang et al. 2005]. For patients with two or fewer of these factors (74% of all patients), the 10-year LRR rate was less than 8%, but for those with at least three risk factors, the 10-year LRR rate was 28%.
More recently, Wright and colleagues reported the outcomes of 464 patients who received PMRT after NAC and mastectomy at the University of Miami [Wright et al. 2013]. Of these, 17.5% received tangent-only radiation [the remainder had radiation to both the chest wall and supraclavicular fossa; only a minority were estimated to have radiation covering the internal mammary nodes (IMNs)]. The investigators noted an overall 5-year LRR rate of 5.8%, with a rate of 1.9% for patients with ypN0 disease. On multivariate analysis, tangent-only PMRT, ypN+ status and triple negative histology were significantly associated with LRR (HRs of 3.39, 10.23 and 8.5, respectively). Clinical stage III versus stage II disease was a significant predictor of LRR on univariate analysis only. The authors concluded that they could not identify a single subtype of patients for whom supraclavicular irradiation offered no locoregional control benefit, though they noted that the low rate of LRR in patients with a nodal pathologic complete response (pCR) suggests a limited absolute benefit in that cohort.
Finally, Nagar and colleagues reported the outcomes of 161 patients treated with NAC and mastectomy, with or without PMRT [including full regional nodal irradiation (RNI)] from 2003 to 2010 [Nagar et al. 2015]. The overall 5-year LRR rate was 16.1%, with PMRT providing a significant reduction in LRR (HR 0.25), translating into an improved 5-year disease-free survival (91.3% versus 64.8%). Residual tumor and nodal status were significantly associated with an increased risk of LRR, but no pre-NAC clinical factors (e.g. clinical T stage or N stage) were predictive. The authors posit that more accurate staging leads to upstaging or downstaging with respect to what would have been considered the clinical stage in the past, thereby diluting the predictive effect of upfront clinical stage. Though provocative, the hypothesis requires validation, and it should also be recognized that many patients may not have had extensive staging workups prior to NAC.
Importance of a pCR
Achieving a pCR after NAC, typically defined as having no residual invasive disease in the breast or nodes on surgical pathology, has been associated with improved survival in both of the prospective trials discussed below, as well as numerous other studies [Rastogi et al. 2008; Cortazar et al. 2014]. A recent meta-analysis of 1955 patients confirmed that, on a patient-level analysis, achieving ypT0-TisN0 status was significantly associated with improved event-free-survival and OS [Cortazar et al. 2014]. The implications of pathologic response on LRR rates warrant detailed review. One could hypothesize that a pCR could negate the need for PMRT, however there are data to suggest high rates of LRR despite the achievement of pCR in select subsets.
The aforementioned series from Huang and colleagues found that the 46 patients with clinical stage III disease or higher who experienced a pCR still had significantly lower 10-year LRR rates with PMRT (33% versus 3%) [Huang et al. 2004]. This contrasted with patients with clinical stage I and II disease and a pCR, for whom PMRT did not seem to affect LRR rates. In a follow-up series, McGuire and colleagues specifically investigated LRR rates following pCR in 106 patients with noninflammatory breast cancers treated with NAC and mastectomy [McGuire et al. 2007]. The 10-year LRR rate was 0% for the 32 patients with clinical stage I–II disease, regardless of PMRT use. For the 74 patients with stage III disease, however, PMRT significantly improved the 10-year LRR rate (33.3% versus 7.3%), and this improvement in LRR translated into significant improvements in CSS (40% versus 87%) and OS (33.3% versus 77.3%). Notably, few patients in this group had cT3N0 disease, precluding evaluation of the prognostic significance of achieving a pCR in this subset of patients. While these data strongly suggest that PMRT is indicated for patients with clinical stage III disease regardless of their pathologic response, other studies have provided evidence to the contrary. Two recent analyses have examined LRR among women achieving a pCR in the nodes (as opposed to pCR in the breast and nodes). Investigators at the Institut-Curie evaluated 134 patients with a nodal pCR after NAC, of whom 78 underwent mastectomy followed by PMRT and the remainder had mastectomy alone [Le Scodan et al. 2012]. The investigators found numeric differences in 10-year LRR (3.8% with PMRT and 13.2% without) and OS (77.2% and 87.7%) that did not reach statistical significance. No difference was also noted in a subset analysis restricted to the 50 patients who had clinical stage III disease at presentation. Presence of residual tumor in the breast did portend poorer outcomes. Finally, a recent report from Korea found no difference in 5-year LRR rates among 151 patients with clinical stage II–III disease who had a nodal pCR after NAC, regardless of PMRT receipt [Shim et al. 2014]. In this study, both age (⩽40 years old versus >40 years old) and pathologic T stage were associated with higher risks of LRR and CSS, while PMRT was not associated with either. An analysis based on stage was not performed, though with 5-year LRR rates of 1.9% and 7.7% with and without PMRT, respectively, it is unlikely that a difference would have been identified.
Overall, these data suggest that for patients with stage II disease who achieve a pCR, PMRT is unlikely to provide a significant LRR benefit. For patients with stage II or stage III disease who achieve a nodal pCR, the presence of residual breast disease, or young patient age, may portend higher LRR rates, with conflicting evidence regarding the need for PMRT particularly in patients with stage III disease. Findings from two prospective trials also suggest a significant prognostic value of nodal pCR for LRR risk (see below) [Mamounas et al. 2012]. Two ongoing trials address this issue directly. The NSABP51/RTOG 1308 trial randomizes patients with cT1–3N1 (pathologically proven) who achieve a nodal pCR after NAC and undergo mastectomy to either PMRT or no PMRT, with stratification factors including breast pCR, hormone receptor status, Her2 status, and receipt of adjuvant chemotherapy (http://www.nsabp.pitt.edu/B-51.asp). The RAPCHEM study in the Netherlands is a prospective nonrandomized study in which patients with cT1–2N0-1 (histologically proven nodal disease, but excluding patients with more than three nodes on imaging) who receive NAC undergo risk-adapted radiotherapy after surgery (https://clinicaltrials.gov/ct2/show/NCT01279304). Patients with nodal pCR are considered low risk and, after mastectomy, will not receive PMRT.
Specific subsets: stage I/II, molecular subtype, and young age
Investigators at MDACC have issued a series of reports examining outcomes in specific subsets of interest. Among 132 patients with clinical stage I or II disease who did not receive PMRT after NAC, the 10-year LRR rate was 10% [Garg et al. 2004]. For patients with cT1–2 disease and one to three positive nodes, the 5-year LRR was 5%; however, cT3N0 disease, at least four nodes at surgery, and age up to 40 were significant predictors of LRR. Nagar and colleagues compared outcomes between 119 patients with cT3N0 disease who underwent PMRT after NAC and mastectomy with 43 who did not [Nagar et al. 2011]. More patients who received PMRT were up to 40 years of age and had ypN+ disease, but despite this, the 5-year LRR rate was 24% for patients without PMRT and 4% for patients who received it. In a subset analysis, PMRT had a significant locoregional control benefit for patients with ypN+ or high-grade disease. The authors did note that the 5-year LRR for patients with cT3N0, ypN0 disease who did not receive PMRT was 14%, nearly double the rate of isolated LRR among patients with pT3N0 disease receiving adjuvant chemotherapy followed by PMRT in an earlier series by Taghian and colleagues [Taghian et al. 2006]. This discrepancy has been hypothesized to be due to occult nodal disease in patients presenting with cT3N0 disease [Meric et al. 2000].
Data for other specific subsets are fairly limited. Clinical multifocality and multicentricity do not appear to be associated with increased risk of LRR [Oh et al. 2006], nor does Her2 positivity [Buchholz et al. 2004; Nagar et al. 2015]. Triple negative status, however, does appear to increase the risk of LRR [Huang et al. 2005; Wright et al. 2013]. In the aforementioned study by Wright and colleagues, the authors reported that seven of nine regional recurrences occurred in patients with triple negative histology [Wright et al. 2013].
A focused study of LRR and OS outcomes among 107 patients younger than 35 years treated with NAC and mastectomy found improved 5-year LRR (12% versus 37%) and OS (67% versus 48%) for patients who received PMRT [Garg et al. 2007]. These improvements were present despite a greater preponderance of adverse features among patients receiving PMRT. On specific subset analysis, the survival benefit was only statistically significant for patients with clinical stage IIB disease, though similar trends were seen in all stages. This finding is consistent with the aforementioned Korean study, which had also found age up to 40 to be a significant predictor of LRR [Shim et al. 2014].
In a recent comprehensive review, wherein appropriateness of PMRT after NAC was determined using the ACR Appropriateness Criteria modified Delphi methodology, it was concluded that patients with T1–2N0–1 who were aged over 40 and had estrogen receptor positive disease, with less than four positive axillary nodes without lymphovascular space invasion or extracapsular extension have a 5-year LRR of less than 10% and thus may not benefit from PMRT [Fowble et al. 2012].
Lessons from NSABP-18 and NSABP-27
Two large prospective randomized controlled trials of NAC, NSABP-18 and NSABP-27 also allow an examination of the natural history of LRR following mastectomy, as both prohibited patients from receiving PMRT [Fisher et al. 1997; Bear et al. 2003; Rastogi et al. 2008]. In a recent pooled analysis, the overall 10-year LRR rate was 11.1% (8.4% local, 2.7% regional; 71% of all LRR were local) among the 1947 patients undergoing mastectomy [Mamounas et al. 2012]. In a multivariate analysis, significant predictors of LRR included clinical tumor size (>5 cm versus ⩽5 cm; HR 1.58), clinical nodal status (cN+ versus cN0; HR 1.53), and pathologic nodal status/breast tumor response (HR 2.21 for ypT+N0 versus total pCR, and 4.48 for ypN+ versus total pCR). Interestingly, for patients treated with breast-conserving therapy, clinical tumor size was not a significant predictor, but age (<50 versus ⩾50) was. Table 1 depicts the 10-year LRR rates stratified by the predictive risk factors; notably, among patients with cN+ disease, only 32 of 424 patients (7.5%) had a pCR (in the breast and nodes), so the observed 10-year LRR of 0.0% in that subset of patients must be kept in context. Among patients with ypN+ disease, 10-year LRR rates were greater than 10% regardless of clinical nodal status, clinical tumor size or pathologic N stage. The analysis was limited by lack of information regarding molecular subtypes of tumors; no patient received Her2/neu-directed therapy, and all patients in NSABP-27 received tamoxifen concurrently with chemotherapy, which was subsequently shown to be inferior to sequential administration [Albain et al. 2009].
Ten-year locoregional recurrence rates for patients undergoing neoadjuvant chemotherapy followed by mastectomy.
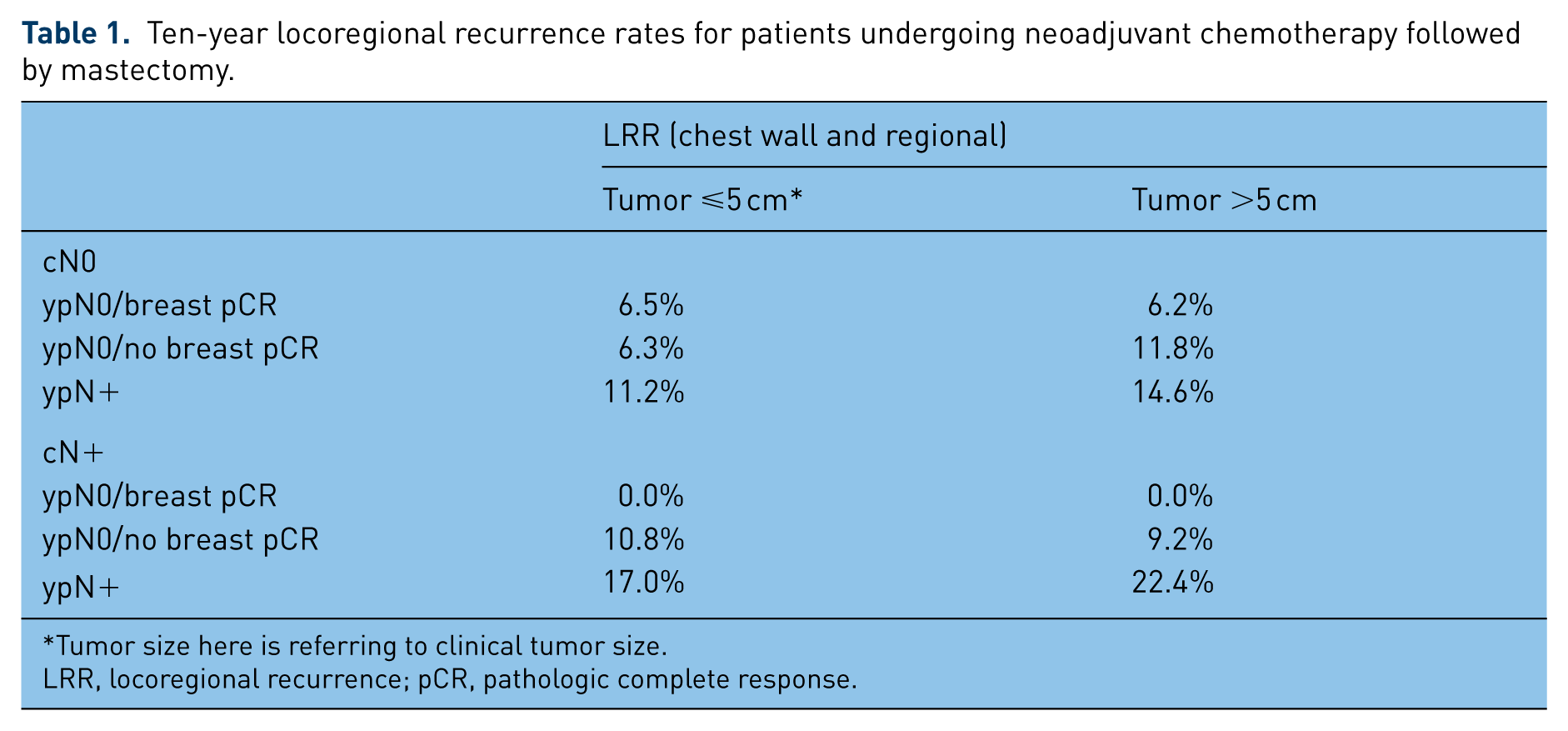
Tumor size here is referring to clinical tumor size.
LRR, locoregional recurrence; pCR, pathologic complete response.
These data suggest that the overall 10-year LRR rate following NAC and mastectomy is relatively low in the absence of PMRT, and have led some to suggest omission of PMRT in patients with clinical stage II disease who achieve a pCR [Bellon et al. 2012; Marks and Prosnitz, 2014; White and Mamounas, 2014]. It is imperative to note that the patients enrolled in NSABP-18 and 27 were generally at far lower risk of LRR overall than those in the retrospective studies referenced above: 55% had cT1–2N0 disease, 20% had cT1–2N1 disease, 16% had cT3N0 disease and only 9% had cT3N1 disease.
Summary
We recommend careful consideration of pre-NAC clinical stage, post-NAC pathologic stage and individual patient and tumor factors in order to individualize decision making regarding PMRT after NAC. In general, we recommend PMRT after NAC for patients who have presented with cT3–4 disease or cN2–3 disease regardless of pathologic extent of disease at the time of surgery. We also recommend PMRT for all patients who have residual nodal disease after NAC. For patients presenting with clinical stage II disease (excluding cT3N0) who achieve a pCR, we typically recommend omitting PMRT. For patients with clinical stage II disease (excluding cT3N0) who achieve a nodal pCR, we recommend enrollment on NSABP51; absent that, we would err towards offering PMRT for patients of young age (⩽40 years old), those with greater than 2 cm residual tumor in the breast, those with high-grade histology and those with triple negative histology. These recommendations are largely consistent with what others have proposed [Fowble et al. 2012; Hoffman et al. 2012]. We do recommend that all patients who undergo NAC meet with a radiation oncologist, preferably upfront, to facilitate their multidisciplinary management.
Preneoadjuvant chemotherapy imaging and nodal assessment
Because both pathological and pre-NAC clinical factors impact risk of LRR following NAC, accurate pre-NAC staging is imperative. Prior to initiating NAC, it is crucial to establish clinical T and N stage as definitively as possible in order to optimally inform post-NAC locoregional therapy decision making. Physical examination alone is unreliable and upwards of 30% of patients who are considered to have cN0 on examination will have occult nodal disease found by sentinel lymph node biopsy (SLNB) [Chung and Giuliano, 2010; Kuehn et al. 2013]. Axillary ultrasound is a helpful imaging modality for detecting nodal disease, with concerning features including a longitudinal/transverse greatest dimension ratio less than 2, absent hilum, eccentrically widened cortex, and cortical thickening [Mainiero, 2010]. Altered morphology, rather than size, may provide a greater specificity for detecting malignancy [Alvarez et al. 2006]. Fine-needle aspiration (FNA) of suspicious-appearing nodes provides a specificity of nearly 100% for malignant disease [Baruah et al. 2010; Park et al. 2011; Bazan and White, 2015].
Although imperfect, more advanced imaging modalities, such as magnetic resonance imaging (MRI) and 18-fluorodeoxyglucose positron emission tomography (18FDG-PET) can be particularly helpful in evaluating nodal disease burden [Kvistad et al. 2000; Greco et al. 2001; Luciani et al. 2004; Wahl et al. 2004; Mortellaro et al. 2009; Koolen et al. 2012]. An early study reported that MRI had a sensitivity of 83% and a specificity of 90% for nodal disease [Kvistad et al. 2000], while a prospective study of 18FDG-PET reported a sensitivity of 61% and a specificity of 80% [Wahl et al. 2004]. Our recommendation is to obtain an axillary ultrasound for all patients with an invasive breast cancer diagnosis; suspicious nodes should be biopsied and clipped. If positive nodes are found, then advanced imaging is recommended, particularly PET-CT which can delineate regional nodal disease that may benefit from targeted radiation therapy [Bazan and White, 2015]. However, even for patients without histologically confirmed nodes we routinely request MRI imaging to better determine the extent of disease in the breast.
The use of advanced imaging to estimate response is an area of active investigation. In a retrospective review, axillary ultrasound was the most sensitive test for detecting ypN+ disease after NAC, with a sensitivity of 69.8% compared with 63.2% for 18FDG-PET and 61.0% for MRI [Hieken et al. 2013]. However, accuracy was the highest for 18FDG-PET at 71.9%, followed by ultrasound at 65.1% and MRI at 60.2%. Two recent studies also suggest that 18FDG-PET may be particularly helpful in restaging patients with triple negative histology [Straver et al. 2010; Koolen et al. 2014], and carry prognostic significance in certain situations [Groheux et al. 2012; Groheux et al. 2013]. A recently published analysis from the American College of Surgeons Oncology Group (ACOSOG) Z1071 (ALLIANCE) trial examined axillary ultrasound after NAC and its impact on sentinel node biopsy [Boughey et al. 2015]. The authors concluded that employing a strategy of limiting SNB to those with negative axillary ultrasound post NAC could serve to reduce false negative rates with SNB to below 10%. As Dialani and colleagues concluded in a recent review of imaging after NAC, there is no currently accepted standard with regards to imaging method for monitoring response to NAC [Dialani et al. 2015]. In general, we recommend repeat MRI (or ultrasound if initial MRI is not performed) after the fourth cycle of NAC, as this might also indicate which patients have not responded well and should move forward with local therapy.
A further detailed review of this workup is beyond the scope of this focused review and interested readers are directed to other detailed reviews [Pilewskie and King, 2014; Bazan and White, 2015; Dialani et al. 2015].
Pathologic nodal staging
In recent years, SLNB has replaced full axillary lymph node dissection (ALND) in both patients with cN0 disease and in patients with cT1–2 disease and involvement of up to two nodes on initial SLNB [Krag et al. 2010; Giuliano et al. 2011; Galimberti et al. 2013]. Additionally, the recently published European Organisation for Research and Treatment of Cancer (EORTC) 10981–22023 AMAROS randomized trial has shown that axillary radiotherapy can replace ALND in patients with a positive SLNB and a 0.5–3 cm primary [Donker et al. 2014]. Because NAC can lead to axillary downstaging in up to 20–40% of cases [Fisher et al. 1997], the use of SLNB in the context of NAC has generated controversy akin to that seen with the use of PMRT. Initially, SLNB was discouraged after NAC, but the recent American Society of Clinical Oncology guidelines suggest that it can be offered before or after NAC, with the standard of care for confirmed nodal disease after NAC being a full ALND [Lyman et al. 2005, 2014]. A detailed discussion of the various methods of pathological axillary staging in the context of NAC is beyond the scope of this focused review (interested readers are directed to recent excellent reviews [Mamounas, 2014; Lyman, 2015], but a brief overview will be presented).
Multiple single-institution reports and multicenter studies have suggested that the sentinel node identification rate following NAC is lower than in the upfront setting [Mamounas et al. 2005; Classe et al. 2009; Hunt et al. 2009; Kelly et al. 2009]. A recent-meta-analysis reported an overall sentinel node identification rate of 89% after NAC, with an overall false negative rate (FNR) of 14% [Fu et al. 2014]. The relatively higher FNR after NAC, compared with the upfront FNR of less than 10%, was initially cause for concern, but results from three recently published trials provide some insight into minimizing this FNR [Boughey et al. 2013; Kuehn et al. 2013; Boileau et al. 2015].
In the phase II ALLIANCE trial, 649 women with cT0–4N1–2 disease who received NAC underwent a SLNB prior to a completion ALND, with the primary outcome being the FNR of SLNB [Boughey et al. 2013]. No sentinel node was found in 7% of patients, and only one node was found in 12% of patients. Overall, the FNR was 12.6%; for patients with only two sentinel nodes removed, the FNR was 21.1%, compared with 9.1% for patients with at least three sentinel nodes removed. If a dual identification method was used (blue dye and radiolabeled colloid), the FNR dropped to 10.8%. In the four-arm prospective SENTINA study, patients were stratified to receive SLNB before NAC (for cN0 that remained ycN0), before and after NAC (for cN0 that were found to be pN0 by upfront SLNB) or after NAC (for cN+ which converted to ycN0) [Kuehn et al. 2013]. The primary outcome was the FNR in patients who had a SLNB done after NAC in patients with cN+ disease that converted to ycN0, which was 14% (with a detection rate of 80%). The investigators found that if patients only had one or two sentinel nodes removed, the FNRs were 24% and 18%, respectively, but if they had at least three nodes removed, it was less than 5%. If dual tracer methodology was used, the FNR was 8.6%. Notably, both the ALLIANCE and the SENTINA studies focused on patients treated with breast-conserving therapy.
Most recently, the prospective SN-FNAC study evaluated the accuracy of SLNB after NAC in 153 patients with biopsy-proven T0–3N1–2 disease [Boileau et al. 2015]. The investigators found a sentinel node identification rate of 87.6%, and an FNR of 8.4%. Notably, however, immunohistochemistry was mandated and patients with ypN0(i+) disease were considered to have positive sentinel nodes. Had these patients been considered to have negative sentinel nodes, then the FNR would increase to 13.3%. For patients with only one node identified, the FNR was 18.2%; it was also 14.2% for patients with T3 tumors.
Taken together, the data suggest that a FN rate of less than 10% can be achieved with SLNB after NAC in women with cN+ disease if certain criteria are met: at least three sentinel nodes are identified and removed, dual identification method is used, and biopsy proven nodes pre NAC are clipped and excised. For patients with positive sentinel nodes after NAC, the current standard of care is a completion ALND; however, the current A011202 phase III clinical trial [ClinicalTrials.gov identifier: NCT01901094] will randomize patients with stage II and IIIA breast cancer who have a positive sentinel node after NAC to axillary radiotherapy versus ALND. For patients with at least three sentinel nodes found to be negative, and for whom PMRT is indicated, the radiation portal design can be adjusted accordingly (see below). If up to three sentinel nodes are removed and found to be negative, we recommend completion ALND for full workup, and if this cannot be done, we suggest a low threshold for providing PMRT with axillary radiotherapy. In the case of patients with histologically confirmed nodal disease prior to NAC, if treatment effect is not commented on or a clip is not seen and had been placed at the time of the original biopsy, then the case should be reviewed by the surgeon and pathologist to ensure adequate axillary sampling has been performed.
Radiation portal design
The three large randomized trials that demonstrated an improved OS with PMRT all included RNI covering the supraclavicular fossa as well as the IMNs [Overgaard et al. 1997, 1999; Ragaz et al. 2005]. However, in patients without certain adverse pathologic features (e.g. lymphovascular space invasion, >50% nodes positive, at least four nodes positive overall, and gross extranodal extension >2 mm), the risk of supraclavicular failure is low [Strom et al. 2005]. The risk of isolated IMN failure is similarly low in most cases, leading to debate over the necessity of IMN irradiation [Taghian et al. 2004; Jagsi and Pierce, 2013]. The EORTC 22922/10925 trial, which included women undergoing either mastectomy or lumpectomy with ALND or SLNB who either had medial tumors (regardless of nodal status) or pN+ disease, randomized patients to receive RNI or no RNI [Poortmans et al. 2014]. The National Cancer Institute of Canada Clinical Trials Group (NCIC CTG) MA.20 trial randomized women with node-positive or high-risk node-negative disease (defined as T3 or T2 with <10 nodes removed in addition to at least one of the following: grade 3 histologic categorization, estrogen receptor negativity or lymphovascular invasion) after breast conserving surgery and surgical nodal evaluation (either SLNB or ALND) to RNI or no RNI [Whelan et al. 2015]. In the MA.20 trial, the RNI included supraclavicular fossa/level III axilla, the IMNs and level I–II of the axilla in selected circumstances; in the EORTC trial, the RNI included the medial supraclavicular nodes and the IMNs, and radiation of the axilla was performed in a small subset (7.4–8.3%) of both arms. Neither trial found a 10-year OS benefit with the addition of RNI, although in both trials disease-free survival, locoregional control and distant metastasis-free survival were modestly but significantly improved with the addition of RNI. The decision to provide RNI versus tangent-only radiation is a topic of significant controversy and a detailed discussion is beyond the scope of this focused review. However, experience related specifically to the situation of radiation portals for PMRT after NAC is fairly limited and will be reviewed here.
One study from the Centre Jean-Perrin compared outcomes between 39 patients who received PMRT, including RNI with 37 patients who had tangent-only PMRT [Gilliot et al. 2010]. The investigators found that there was no difference in 10-year LRR rates (95% without and 91% with RNI). However, in the group that did not receive RNI, the 10-year OS was 96%, while in the group receiving RNI, 10-year OS was 75% (p < 0.05). Distant-metastasis-free survival was similarly higher in the patients who did not receive RNI (97% versus 78%). The treatment groups were significantly unbalanced, however, with significantly more patients in the group treated with RNI having cT3–4 tumor and having significantly larger residual tumors at surgery. However, the considerably larger study by Wright and colleagues discussed above suggested a benefit to including a supraclavicular field [Wright et al. 2013]. Though Wright and colleagues were not able to determine whether the IMNs were irradiated for all patients, they estimated that less than 5% of patients received IMN radiation. While specific studies investigating omission of IMN irradiation in patients receiving NAC are lacking, a report from the MDACC suggested that up to 10% of patients with locally advanced disease have clinically detectable IMN adenopathy by imaging, and in those patients, directed IMN irradiation achieves excellent 5-year IMN and locoregional control [Zhang et al. 2010]. The current randomized NSABP-51 trial randomizes women with cT1–3N1 breast cancer who undergo lumpectomy or mastectomy and are found to have ypN0 to RNI versus no RNI. The Alliance A011202 trial randomizes women with cT1–3N1 breast cancer who remain ypN+ on SNB following NAC to axillary node dissection and RNI versus RNI alone. Among women with clinically node positive disease pre NAC, these trials are aiming to answer the questions of whether RNI is necessary in women who become pathologically node negative and whether RNI can replace axillary surgery in women who remain pathologically node positive.
Our recommendation is to proceed with RNI after NAC for any patient with ypN+ disease, and for patients presenting with cN2–N3 disease. We would not cover the fully dissected axilla in the absence of negative risk factors (<10 nodes removed, >50% nodes positive, vascular adherence). For patients with cN0–N1 disease who remain or convert to ypN0 disease after NAC and have had adequate axillary staging at the time of surgery (see above), if treating, we would cover the chest wall and determine the need for supraclavicular or IMN coverage based on careful consideration of adverse risk factors (e.g. lymphovascular space invasion, age, extent of node positivity pre NAC, size and biologic features of residual disease in the breast). For patients without adequate axillary staging, we would err on the side of caution and provide RNI (including the undissected axilla).
Conclusion
The decision to provide or omit PMRT after NAC is a complicated one and a consultation with a radiation oncologist is highly recommended, preferably before NAC is even started. We recommend careful consideration of pre-NAC clinical stage, post NAC pathologic stage, and individual patient and tumor factors in order to individualize decision making regarding PMRT after NAC. Synthesis of both retrospective and prospective data suggests that the risk of LRR following mastectomy is highest for patients with cT3–4 disease, cN2–3 disease and ypN+ disease, and thus we recommend PMRT for these patients. For patients with stage III disease, we would recommend PMRT regardless of pathological response. For patients presenting with clinical stage II disease (excluding cT3N0) who achieve a pCR, we typically recommend omitting PMRT. For patients with clinical stage II disease who achieve a nodal pCR, we recommend enrollment on NSABP51; absent that, we would err towards offering PMRT for patients with adverse features, including young age, greater than 2 cm residual tumor in the breast, high-grade or triple negative histology. Implicit in these recommendations is the requirement for accurate pre-NAC staging. We recommend axillary ultrasound with FNA of suspicious nodes and preferably MRI for all patients and PET-CT for all node-positive patients. Performing a SLNB after NAC is reasonable as long as at least three nodes are removed and a dual-tracer method is used. Radiation portal design is governed by similar principles as in the standard PMRT situation.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author(s) declare(s) that there is no conflict of interest.
