Abstract
Background:
The endocannabinoid system (ECS) regulates homeostasis, inflammation, and organ-specific function. In the kidney, ECS activity modulates renal hemodynamics, and its overactivation is linked to chronic injury. However, the importance of the ECS involvement in acute kidney injury (AKI) remains unclear. This study aimed to characterize changes in circulating endocannabinoid (eCB) levels before and after relief of upper urinary tract obstruction (UUTO), to better understand ECS dynamics during acute renal dysfunction. These findings may inform the development of novel biomarkers and therapeutic targets for renal injury.
Objectives:
To characterize changes in circulating eCB levels before and after relief of UUTO, and to compare responses between patients with and without AKI.
Design:
Prospective observational cohort with paired, within-person sampling.
Methods:
Patients presenting to the emergency department with acute renal colic due to obstructive urolithiasis who underwent kidney decompression within 24 h were prospectively enrolled. Clinical, laboratory, and imaging data plus paired blood samples for eCB analysis were collected pre and post-drainage. Patients were divided into two groups: those who had AKI at presentation, and non-AKI controls. Serum eCBs were quantified, and fold changes compared using nonparametric analysis.
Results:
Twenty-two patients enrolled (10 had AKI and 12 served as non-AKI controls). Serum N-acylethanolamines (NAEs) showed divergent responses between the two groups. In AKI, N-arachidonoylethanolamine (AEA), N-palmitoylethanolamine, and N-oleoylethanolamine increased following drainage (p = 0.06, 0.008, 0.08). In contrast, patients without AKI demonstrated a reduction in NAE levels, with a significant AEA drop (p = 0.03) after obstruction relief. Notably, the fold-change in NAE levels post-drainage was significantly higher in patients with AKI compared to those without AKI.
Conclusion:
Circulating NAEs increase following relief of obstruction in patients with acute renal dysfunction, suggesting a potential role for ECS activation in the pathophysiology of UUTO-induced kidney injury. These findings highlight the ECS as a promising target for further investigation as a possible therapeutic avenue in AKI.
Trial registration:
Not applicable.
Plain language summary
A kidney stone can obstruct the urinary tract, causing severe pain and sometimes a sudden drop in kidney function. Doctors treat this by placing a tube or stent to relieve the obstruction so urine can flow again. What’s less known is how the body’s own chemistry responds to this stressful event-and whether that response differs when the kidneys are injured. Our bodies make small, cannabis-like chemicals (called endocannabinoids) that help regulate pain, inflammation, and organ function. We asked: do these natural signals change after an obstructing stone is relieved, and are the changes different in people with acute kidney injury? We studied 22 adults who needed urgent drainage for a kidney stone that was obstructing urine flow. Ten had acute kidney injury; twelve did not. We took a blood sample before drainage and another afterward and measured the levels of these cannabis-like signals. People with acute kidney injury showed an increase in several of these natural signals after the obstruction was relieved. In contrast, people without kidney injury tended to have stable or lower levels after treatment. What does this mean? The body may turn up these natural, cannabis-like signals when the kidneys are stressed, possibly as a protective response. If confirmed in larger studies, these changes could help doctors detect kidney injury earlier and might even point to new treatments that harness the body’s own chemistry to protect the kidneys.
Introduction
The endocannabinoid system (ECS) is a complex cell-signaling network that plays a critical role in maintaining physiological homeostasis and modulating inflammatory responses. 1 It consists of endogenous ligands known as endocannabinoids (eCBs), notably N-arachidonoylethanolamine (AEA) and 2-arachidonoylglycerol (2-AG), along with the enzymes responsible for their synthesis and degradation, and their receptors, primarily cannabinoid-1 receptor (CB1R) and cannabinoid-2 receptor (CB2R). In addition to classical eCBs, the ECS includes related N-acylethanolamines (NAEs), such as N-palmitoylethanolamine (PEA) and N-oleoylethanolamine (OEA), which do not activate CB1R and CB2R but share biosynthetic and degradation pathways 2 (Figure 1). The ECS is implicated in the regulation of numerous physiological processes across multiple organ systems, underscoring its therapeutic potential.

Overview of the ECS. The ECS comprises two main eCBs, 2-AG and N-arachidonoylethanolamine (AEA; anandamide), which act as endogenous ligands for cannabinoid receptors 1 and 2 (CB1R and CB2R). 2-AG is synthesized by DAGL and primarily degraded by MAGL. AEA is primarily synthesized by NAPEPLD and degraded by FAAH. Additional NAEs, including OEA and PEA, share similar biosynthetic and degradative pathways with AEA but do not directly activate CB1R or CB2R. These NAE compounds are often referred to as endocannabinoid-like lipids because of their structural similarity and functional overlap with classical eCBs.
Components of the ECS, including eCB, their receptors, and associated enzymes, are widely expressed throughout the urinary tract (Figure 2). In the kidney, CB1R is present in endothelial cells, glomeruli, proximal and distal tubules, and collecting ducts. 3 The bladder also expresses ECS components, including CB1R in urothelial cells, detrusor muscle, and nerve fibers.4–7 Key eCB ligands such as AEA and 2-AG, along with their respective degrading enzymes fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase, respectively, are detected in both the kidney3,8,9 and bladder.10–13 Functionally, the renal ECS modulates hemodynamics, tubular reabsorption, proteinuria, and immunomodulation of kidney cells. 3 Emerging evidence supports a pathogenic role for ECS overactivation in chronic kidney disease (CKD), particularly via CB1R, which promotes oxidative stress, inflammation, fibrosis, and tissue damage.3,14 We and others have demonstrated that pharmacological blockade or genetic deletion of CB1R ameliorates kidney damage in models of obesity- and diabetes-induced CKD.15–20

Expression of ECS components in the urinary tract. CB1Rs are widely expressed throughout the urinary tract, including the endothelial cells, glomeruli, proximal and distal tubules, and collecting ducts of the kidney. In the bladder, CB1R is found in urothelial cells, the detrusor muscle, and nerve fibers innervating the bladder. The eCB anandamide (AEA) and its primary degrading enzyme, FAAH, are also present in both the kidney and bladder. Similarly, 2-AG and its degrading enzyme, MAGL, are expressed in these tissues, highlighting the presence of a functional ECS across the urinary tract.
In contrast, the role of the ECS in acute kidney injury (AKI) remains poorly defined.3,14 Alterations in eCB levels have been reported in various rodent models of AKI; however, the specific ligands involved and the direction of these changes differ across studies.9,21,22 Similarly, the contribution of CB1R and CB2R to AKI pathogenesis appears context-dependent, with studies reporting both protective and deleterious effects depending on the underlying etiology. 23 Recently, we reported an acute elevation in circulating classical eCBs and NAEs during renal ischemia in patients undergoing partial nephrectomy, 24 further supporting ECS involvement in acute renal insults.
In this study, we present, for the first time, a distinct systemic eCB response profile in patients with acute renal dysfunction secondary to upper urinary tract obstruction (UUTO), compared to non-AKI patients. Specifically, we examined dynamic changes in circulating eCBs before and after obstruction relief in patients presenting with renal colic and acute obstructive uropathy. These findings highlight the potential of the ECS both as a biomarker of acute kidney dysfunction and as a therapeutic target for preventing AKI associated with nephrolithiasis.
Materials and methods
Study design and ethical approval
The study was approved by the Hadassah Medical Organization Institutional Review Board (Helsinki Committee), approval number HMO-0177-20. Initial approval was granted on April 20, 2020, with annual continuing approval in place through May 2023. The study period fell entirely within this approval window. Written informed consent was obtained from all participants before any study procedures. Adult patients hospitalized with acute UUTO due to urolithiasis who underwent renal drainage during their hospital stay were eligible for inclusion. Exclusion criteria included pregnancy and cannabis use within 30 days prior to enrollment. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 25 The completed STROBE checklist is provided as a Supplemental File.
To assess differences in eCB profiles between patients with and without renal function impairment, participants were stratified into two groups: Group 1—AKI Group: Patients who met criteria for AKI, defined according to KDIGO guidelines as an increase in baseline serum creatinine of ⩾50%. Baseline serum creatinine was defined as the most recent stable value documented in the patient’s institutional electronic medical record within the 3 months preceding the acute episode. For the purposes of study inclusion and group stratification, the serum creatinine value at the time of hospital admission was used to define the presence of AKI. Group 2—Non-AKI Group: Patients who underwent renal drainage despite having stable renal function. Indications for drainage in this group included refractory pain, urinary tract infection, or as a preparatory step prior to ureteroscopy. In this study, “renal drainage” refers specifically to acute decompression via either the retrograde insertion of a ureteral stent or the antegrade placement of a percutaneous nephrostomy (PCN) tube.
Study period: Participants were prospectively enrolled between June 2020 and December 2022.
Sample size and power: This was an exploratory prospective cohort intended to estimate within-person changes in circulating eCBs after relief of obstruction and to generate effect-size estimates for future confirmatory studies. A formal a priori power calculation was not performed because reliable variance estimates were unavailable for these analytes in this acute clinical setting, and enrollment was constrained by the emergency care pathway. A target of approximately 20–25 consecutive eligible patients during the planned accrual window was pre-specified as feasible. Effect sizes and 95% confidence intervals are reported to inform future sample size calculations.
Clinical evaluation and indications for drainage
Upon admission, all patients underwent comprehensive clinical evaluation, including physical examination, laboratory testing, and imaging assessments. The decision to proceed with renal drainage was made based on standard clinical criteria, including the presence of urinary tract infection, impaired renal function, bilateral obstruction, or intractable pain unresponsive to conservative treatment.
Patient enrollment and data collection
Following written informed consent, demographic, clinical, laboratory, and imaging data were prospectively collected for all participants. Pain intensity during hospitalization was assessed using the Visual Analog Scale. All analgesic medications administered during the hospital stay were recorded, with a specific focus on non-steroidal anti-inflammatory drugs (NSAIDs). Information regarding medication use prior to hospital admission was not available.
Blood sampling and study design
To account for inter-individual variability in circulating eCB levels, a paired-sample design was employed in which each participant served as their own control. Two fasting blood samples were obtained from each participant: Sample 1: Collected during hospitalization, prior to obstruction relief, following an overnight fast. Sample 2: Collected after relief of obstruction via renal drainage. This design enabled the assessment of intra-individual changes in eCB levels in response to decompression, while also allowing for inter-group comparisons between patients with and without AKI.
Endocannabinoid extraction and measurement by LC-MS/MS
Serum eCBs were extracted, purified, and quantified using stable isotope dilution liquid chromatography-tandem mass spectrometry (LC-MS/MS), as previously described. 24 Briefly, serum proteins were precipitated with ice-cold acetone and 50 mM Tris buffer (pH 8.0). Next, an ice-cold extraction buffer [1:1 MeOH/Tris Buffer + an internal standard (IS; d4-AEA)] was added to the samples. Lipids were then extracted using an ice-cold CHCl3:MeOH mixture (2:1, v/v), followed by triple chloroform washes. Extracts were dried under a stream of nitrogen and reconstituted in methanol for analysis.
LC-MS/MS was performed on an AB Sciex QTRAP® 6500+ mass spectrometer (Framingham, MA, USA) coupled to a Shimadzu UHPLC System (Kyoto, Japan). Chromatographic separation was achieved by injecting 5 µL of each sample onto a Kinetex 2.6 µm C18 column (100 × 2.1 mm; Phenomenex, Torrance, CA, USA). The autosampler temperature was maintained at 4°C, and the column at 40°C throughout the analysis.
Gradient elution was performed using mobile phases consisting of 0.1% formic acid in water (phase I) and 0.1% formic acid in acetonitrile (phase II). Detection was conducted in a positive ion mode using electrospray ionization (ESI) and multiple reaction monitoring (MRM). Quantification of 2-AG, AEA, OEA, PEA, and arachidonic acid (AA) was performed using calibration curves and expressed as pmol/mL of serum.
Statistical analysis
All statistical analyses were performed using GraphPad Prism version 10 (GraphPad Software, CA, USA) and Winpepi version 11.65. Continuous variables are presented as median with range, while categorical variables are expressed as proportions. Intergroup comparisons were performed using Fisher’s exact test for categorical variables and the Mann–Whitney U test for continuous variables. Changes in individual serum eCB concentrations before and after kidney drainage were analyzed, and fold changes were calculated to facilitate intergroup comparisons between patients with and without AKI. Intra-individual changes in eCB levels following renal drainage were evaluated using the Wilcoxon signed-rank test. A two-tailed p-value < 0.05 was considered statistically significant for all analyses.
Results
A total of 22 patients were enrolled in the study. Based on renal function at presentation, patients were categorized into two groups: ten patients met the criteria for AKI (Group 1), while twelve patients maintained stable renal function and were assigned to the non-AKI group (Group 2). Baseline demographic and clinical characteristics were similar between the two groups, with the exception of parameters related to kidney function, such as pre-drainage serum creatinine and blood urea nitrogen levels, as detailed in Table 1. The AKI group presented with significantly higher median serum creatinine (1.86 mg/dL; range 1.39–2.67) compared to the non-AKI group (0.94 mg/dL; range 0.61–1.39; p < 0.001). Hemodynamic parameters, including median systolic and diastolic blood pressures, were comparable between cohorts. Imaging assessments confirmed moderate-to-severe hydronephrosis in the majority of the cohort (70% in AKI vs 83.3% in non-AKI; p = 0.35), with most obstructions located in the proximal ureter. Clinically, all patients in the AKI group (100%) demonstrated significant improvement in renal function following decompression and were successfully discharged without requiring renal replacement therapy.
Patient groups characteristics: AKI versus non-AKI.

AKI, acute kidney injury; NSAID, non-steroidal anti-inflammatory drug; VAS, Visual Analog Scale.
Bold values and asterisks (*) indicate statistical significance at the p < 0.05 level.
Regarding medication and analgesic history, 30% of AKI patients and 41.7% of non-AKI patients were on antihypertensive therapy. Thiazide diuretic and NSAID use were minimal; no patients in the AKI group were receiving thiazides or NSAIDs prior to or during the study period. In the non-AKI group, one patient (Patient #7) was on a chronic thiazide regimen, and one patient (Patient #17) was administered a single dose of an NSAID (Ketorolac) prior to the first blood draw. Precise urine output quantification was not consistently available for all participants in the emergency setting.
The median time elapsed between drainage until second blood draw was 21 h (12–22 h) with no significant difference between the two groups (p = 0.29). Analysis of circulating eCB levels before and after relief of obstruction revealed distinct patterns between the two patient groups. In the AKI group, post-drainage measurements showed a trend toward increased levels of NAEs, including AEA and OEA, with a statistically significant elevation in PEA levels (p = 0.008), compared to pre-drainage values (Figure 3(a)–(e)). No significant changes were observed in levels of 2-AG or AA, a downstream eCB metabolite.
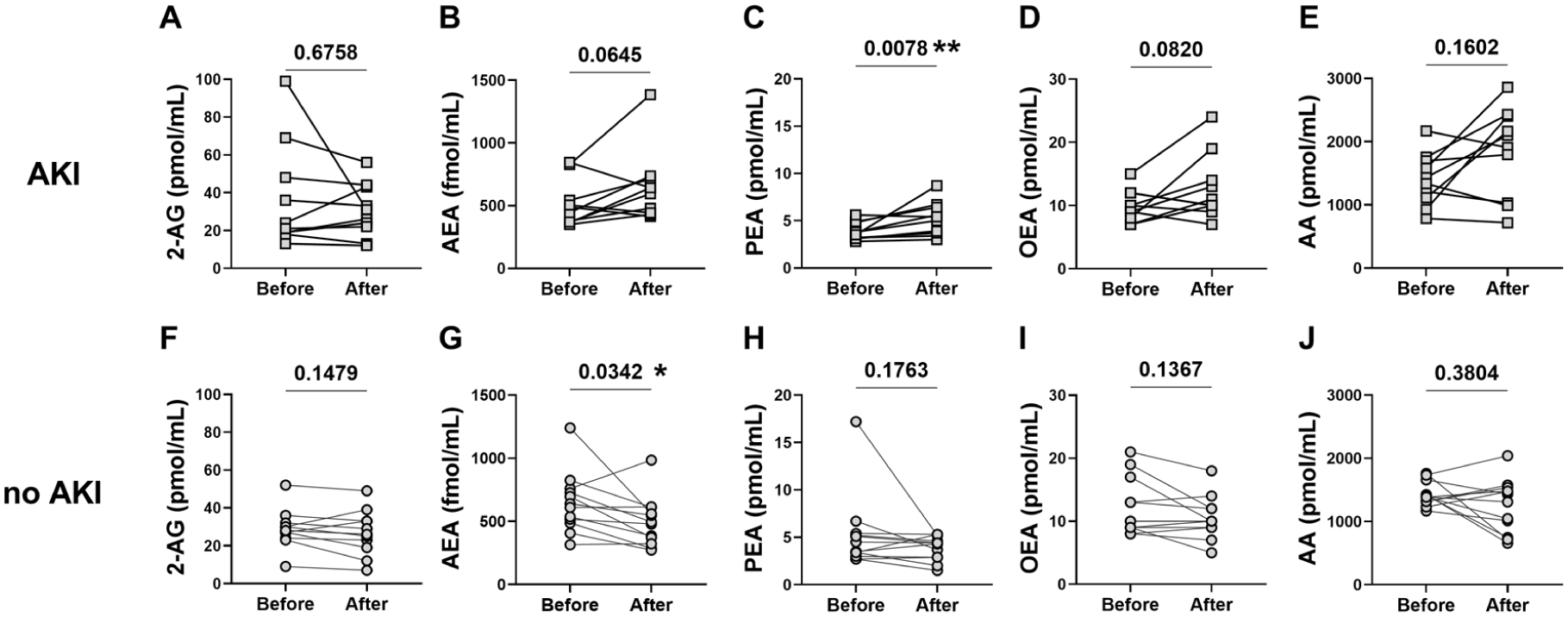
Dynamic changes in serum endocannabinoid levels following kidney stone drainage. Paired measurements of serum eCBs and related lipids before and after obstruction relief in patients with kidney stones. Panels show levels of: (a, b, f, g) classical eCBs, 2-AG and anandamide (AEA); (c, d, h, i) eCB-like compounds, PEA and OEA; (e, j) the downstream metabolite AA. Patients were stratified by clinical indication for drainage: those with AKI are shown in squares, while those undergoing drainage for other indications (e.g., infection, pain, or pre-surgical preparation, no AKI) are shown in circles. Statistical comparisons were performed using the Wilcoxon matched-pairs signed-rank test.
In contrast, the non-AKI group exhibited a different response. Specifically, serum AEA levels significantly decreased following relief of obstruction (p = 0.03), while levels of PEA, OEA, 2-AG, and AA remained unchanged (Figure 3(f)–(j)).
When directly comparing the fold changes in eCB levels between groups, the AKI group demonstrated a significantly greater increase in NAEs, particularly AEA (p = 0.0026) and PEA (p = 0.0036), with a borderline increase in OEA (p = 0.0523; Figure 4(b)–(d)). No significant differences were observed in the changes in 2-AG or AA levels (Figure 4(a) and (e)).

Enhanced fold-change of serum NAEs in AKI patients following drainage. Comparison of fold changes in serum lipid mediators before and after kidney drainage between patients with AKI and those without AKI. (a, b) Classical eCBs, 2-AG and anandamide (AEA); (c, d) eCB-like compounds, PEA and OEA; (e) the downstream metabolite AA. Data are presented as fold-change post-drainage and were analyzed using the Mann–Whitney U test.
Discussion
Renal colic is an acute physiological event characterized by UUTO and, in many cases, a temporary reduction of nephron activity. This process begins with elevated pressure within the urinary collecting system, which translates to increased intratubular and intraglomerular pressure, leading to a transient decline in filtration and subsequent structural alterations within the nephron. If unresolved, the obstruction can initiate inflammatory and apoptotic pathways, eventually resulting in irreversible renal damage. 26
The ECS has emerged as a key regulator of renal function and pathology, exerting a “dual role” that is highly dependent on the specific receptor activated and the timing of the insult. Dysregulation of this system has been implicated in the progression of CKD, with both preclinical and clinical evidence supporting the role of CB1R activation in promoting renal inflammation, fibrosis, and injury.3,15,16,18–20 This pathway represents the detrimental arm of the ECS. Moreover, our group previously reported elevated systemic AEA levels in non-obese patients following partial nephrectomy, suggesting a dynamic eCB response to acute renal ischemia. 24 However, it is still unclear if the ECS dysregulation in AKI is a compensatory mechanism to overcome the damage or is it merely a consequence of a maladaptive process. Evidence regarding CB1R activation versus inhibition in AKI is conflicting; studies in cisplatin 21 and gentamycin 27 -induced nephrotoxicity indicate that CB1R blockade mitigates early renal damage, whereas CB1R agonism or elevation of eCB levels has been shown to confer protection against AKI.9,22,28,29 These discrepancies highlight the functional divergence between receptor types; while CB1R signaling often drives pathology, NAE-mediated signaling is frequently supportive. These discrepancies likely reflect distinct pathophysiological mechanisms underlying different forms of renal injury, suggesting that ECS effects are highly context- and etiology-dependent. Importantly, CB1R-independent pathways are central to the renoprotective arm of the ECS, contributing to the supportive effects of NAEs through their interaction with alternative receptors, including PPARα, PPARγ, GPR119, GPR55, and TRPV1. 30
Although the role of the ECS in AKI is less well-defined, several animal models have demonstrated ECS perturbations in response to ischemia, nephrotoxicity, and obstructive injury.9,21,22 In particular, studies using the unilateral ureteral obstruction (UUO) model have shown that ECS dysregulation contributes to the development of fibrosis and tubular damage.15,31 Despite this growing body of evidence, the involvement of the ECS in acute obstructive nephropathy because of kidney stones in humans remains largely unexplored.
This study is the first to investigate dynamic changes in circulating eCB levels in patients with acute UUTO, with and without AKI. Given the high inter-individual variability in serum eCB concentrations, 32 a paired-sample design was employed, wherein each patient served as their own control. Blood samples were collected during obstruction and again following renal decompression to assess intra-individual changes in eCB levels.
Our findings indicate that patients with AKI exhibited significant increases in serum NAEs, including AEA, PEA, and OEA, following relief of obstruction. In contrast, patients with obstruction but preserved renal function did not exhibit this elevation; in fact, serum AEA levels declined post-drainage. These NAEs share common biosynthetic and degradation pathways and are primarily catabolized by the enzyme FAAH. 2 Experimental studies have shown that increasing renal levels of NAEs through FAAH inhibition or genetic knockout provides protection against AKI and subsequent fibrosis in rodents.33,34 Specifically, exogenous administration of PEA has been shown to reduce inflammation, mitigate tubular injury, and preserve renal function in ischemia-reperfusion models. 35 Together, these findings support a potential renoprotective role for NAEs and suggest that their post-drainage elevation in AKI patients may reflect an intrinsic, compensatory response to renal injury, one that could be harnessed therapeutically. Interestingly, we have previously reported a rapid increase in circulating NAEs as early as 10 min after ischemic insult in patients undergoing partial nephrectomy, 24 further supporting the idea that ECS activation is an early systemic response to acute renal stress.
Our findings regarding NAE elevation post-drainage in AKI patients highlight the complex “dual role” of the ECS, distinguishing the supportive, acute NAE response from the detrimental signaling associated with chronic CB1R activation in kidney pathology. While chronic activation of CB1Rs by ligands such as AEA has been linked to fibrosis and damage, other components may be protective. As highlighted in a recent review by Dubey et al., PEA exhibits significant therapeutic potential in renal damage by modulating inflammatory cytokines and reducing oxidative stress. 30 The specific surge in PEA we observed in the AKI cohort suggests that the system may prioritize these protective pathways in the acute phase of recovery. Furthermore, we considered potential enzymatic influences on these levels. Recent in silico and in vitro data suggest that thiazide diuretics can stabilize NAPE-PLD, the primary enzyme for NAE synthesis. In our study, thiazide use was limited to a single patient in the non-AKI group. Because no patients in our AKI group were receiving thiazides, the observed significant elevation in AEA and PEA within that cohort is likely a direct physiological consequence of the AKI and its resolution, rather than drug-induced stabilization of synthetic enzymes. Notably, the presence of a thiazide user in the non-AKI group did not prevent the significant post-drainage decline in AEA observed in that group, further suggesting that the clinical dynamics of obstruction relief outweighed potential pharmacological enzymatic stabilization.
Finally, the observed surge in circulating NAEs following drainage may have implications for post-obstructive diuresis. Activation of CB1R has been shown to modulate renal hemodynamics and tubular sodium handling, often promoting increased diuresis and natriuresis in experimental models. Therefore, the elevation of NAEs post-decompression in AKI patients could potentially contribute to the physiological diuretic response typically observed following the relief of high-grade urinary obstruction.
The present study has several limitations. The relatively small sample size may limit statistical power and generalizability. One important limitation is the lack of pre-obstruction baseline eCB measurements. Without these baseline values, it is not possible to determine whether the observed changes following drainage represent a true pathological shift induced by obstruction and subsequent relief, or merely a return to pre-obstruction homeostasis. In addition, the absence of serial follow-up samples restricts our ability to evaluate the long-term dynamics of circulating eCB levels beyond the immediate post-obstructive period. Moreover, the study focused solely on systemic (circulating) eCB levels and did not include tissue-level assessments from the affected kidneys, precluding localization of the source of eCB production, whether from the obstructed kidney, the contralateral kidney, or systemic compartments.
Despite these limitations, this study provides the first clinical evidence of dynamic changes in circulating NAEs in patients with AKI secondary to obstructive uropathy. While causality cannot be established from these observational data, our findings raise the possibility that the ECS plays an active role in modulating the renal response to acute obstruction. These results build on preclinical evidence linking NAEs to renal protection,33–35 and highlight the need for further translational research. In the future, ECS components, particularly NAEs, may serve as biomarkers to monitor renal function or predict recovery following obstructive injury. Moreover, pharmacological modulation of the ECS could represent a novel therapeutic strategy for AKI.
Supplemental Material
sj-docx-1-tau-10.1177_17562872261442624 – Supplemental material for Endocannabinoid responses to relief of obstruction in acute injured kidney: a prospective observational study
Supplemental material, sj-docx-1-tau-10.1177_17562872261442624 for Endocannabinoid responses to relief of obstruction in acute injured kidney: a prospective observational study by Ariel Rothner, Sharon E. Fishberg, Liad Hinden, Eyal Atias, Alina Nemirovski, Ofer N. Gofrit, Joseph Tam and Guy Hidas in Therapeutic Advances in Urology
Footnotes
Acknowledgements
We thank the emergency department and urology ward nursing teams for assistance with patient enrollment and sample collection.
Declarations
Availability of data and materials
De-identified data and analysis code are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
