Abstract
Background:
Prostate-specific membrane antigen (PSMA) PET is becoming the preferred imaging technique for prostate cancer (PCa) because of its superior sensitivity and specificity compared to traditional imaging methods. This article evaluates the methodological and reporting quality of literature regarding the cost-effectiveness of PSMA-positron emission tomography (PET)/computed tomography (CT) or MRI for detecting and staging PCa.
Objective:
To describe the methodological and reporting quality of the published cost-effectiveness studies on PSMA-PET.
Methods:
MEDLINE and EMBASE were searched from inception to December 05, 2025. Two researchers independently screened all retrieved articles according to inclusion and exclusion criteria. Studies were appraised for methodological quality using the Quality of Health Economic Studies checklist and for reporting quality using the Consolidated Health Economic Evaluation Reporting Standards checklist.
Results:
A total of seven studies were examined, representing both private and public healthcare systems. Overall, these studies demonstrate high methodological and reporting quality. When compared to existing imaging technologies or standard care practices, PSMA-PET/CT or MRI is cost-effective within country-specific willingness-to-pay thresholds.
Conclusion:
Cost-effectiveness evaluations may not be widely generalizable due to significant variability among geographical regions concerning resource availability, costs, morbidity and mortality, and standards of practice. As PSMA-PET/CT imaging technology becomes more widely available in additional countries in the coming years, we expect to generate more country-specific data.
Plain language summary
Prostate-specific membrane antigen (PSMA) PET has emerged as the preferred imaging technique for prostate cancer (PCa) due to its superior sensitivity and specificity compared to traditional imaging methods. We reviewed full economic evaluation studies of PSMA PET/CT or MRI, searching MEDLINE and EMBASE from their inception until May 2025. Two researchers independently screened all retrieved articles based on inclusion and exclusion criteria. We assessed the methodological quality of the studies using the Quality of Health Economic Studies (QHES) checklist and the reporting quality using the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist. In total, we examined seven studies that represented both private and public healthcare systems. Overall, these studies showed high methodological and reporting quality. When compared to existing imaging technologies or standard care practices, PSMA PET/CT or MRI is considered cost-effective within country-specific willingness-to-pay thresholds. However, the results of cost-effectiveness evaluations may not be universally applicable due to significant variations among geographical regions in terms of resource availability, costs, disease morbidity and mortality, and standards of practice. As PSMA PET/CT imaging technology becomes more widely available in additional countries in the coming years, we anticipate the generation of more country-specific data.
Introduction
Prostate cancer (PCa) is one of the most common non-cutaneous cancers diagnosed in men worldwide.1,2 In addition, PCa is one of the leading causes of cancer-related death in men worldwide.1,2 The 5-year relative survival rate of PCa is 90%; however, relative survival decreases significantly once the cancer has spread to other parts of the body (34%). 3 Black men are more likely to be diagnosed with PCa and twice as likely to die from PCa compared to non-Hispanic White men. 4 PCa is diagnosed mainly in men aged 65 years or older. 5 As the aging population continues to grow, the burden of PCa on the healthcare system will continue to grow. 5
The accurate detection of PCa and the extent to which the cancer has spread in the body (i.e., staging) is critical to inform treatment decision-making. Traditionally, multi-parametric magnetic resonance imaging (mpMRI), computed tomography (CT), and bone scans are used to stage the disease to decide upon a treatment regimen. However, these traditional imaging technologies lack sensitivity and specificity to detect and stage PCa accurately. 6
Prostate-specific membrane antigen (PSMA) is overexpressed in PCa tissue and neovasculature associated with the tumour. 6 PSMA-positron emission tomography (PET)/computed tomography (CT) or MRI, a novel advanced imaging technique, has demonstrated superiority to traditional imaging (mpMRI, CT, or bone scan) to detect and stage PCa. 6 PSMA-PET/CT has a better detection rate and diagnostic accuracy than conventional imaging technologies for biochemical recurrence and metastasis. 6 There are various PSMA-PET radiotracers (68Gallium or 18Fluorine) currently available, enabling this advanced imaging technology to be available in various countries.
Value of innovative health technologies
Increasingly, governments and/or payers require an economic evaluation to inform decisions regarding the adoption of innovative health technologies to demonstrate their value for money. 7 One of the most used methodologies is cost-effectiveness or cost-utility analyses. Cost-effectiveness (or utility) analysis evaluates the incremental costs and effectiveness of a new health technology compared to existing health technologies or standards of care. For example, the cost-effectiveness analysis express result as cost per life-years gained. Whereas the cost-utility analysis expresses results as cost per quality-adjusted life-years (QALYs), which decision-makers prefer since QALYs as a measure of effectiveness can be used for almost all health technologies. QALY is a product of quality and quantity (survival) of life. Quality of life is measured in terms of “utility,” ranging from “1” representing “perfect health” to “0” representing “death.” Cost-effectiveness (or utility) is expressed as an incremental cost-effectiveness ratio (ICER), the ratio of the difference in costs (numerator) divided by the difference in effects (denominator) between new and existing (standard of care) health technologies. Most developed nations have country-specific willingness-to-pay (WTP) threshold, indicating the maximum amount a decision-maker will pay per additional gain in QALY. For example, the WTP threshold values used in the USA, Canada, and UK are US$100,000 and US$150,000/QALY, 8 CAD$50,000/QALY, 9 and £20,000 and £30,000/QALY, 10 respectively. Although WTP threshold is not the only criteria used to adopt health technologies. The aim of this review was to describe the methodological and reporting quality of the published cost-effectiveness studies on PSMA-PET.
Methods
Literature search and article selection
The systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 11 A comprehensive literature search of MEDLINE (PubMed) and EMBASE databases was conducted from inception to December 05, 2025. Search term included “prostate specific membrane antigen,” “prostate specific membrane antigen-positron emission tomography,” “prostate specific membrane antigen-computed tomography,” “prostate specific membrane antigen-magnetic resonance imaging,” “cost,” “treatment cost,” “healthcare cost,” “direct cost,” and “economic burden.” Boolean operators (AND and OR) were used to augment the search. Duplicate citations were identified and excluded using the Zotero bibliographic management software.
Two reviewers (CS, RR) independently searched the databases and screened the search results to identify potentially relevant studies. They reviewed the title, abstract, and full text of each article, reaching consensus during the screening process. The articles were screened for relevance using these inclusion and exclusion criteria. Inclusion criteria were (1) a peer-reviewed article published in English and (2) provides a cost-effectiveness (e.g., cost per life-years gained) or cost-utility analysis (e.g., cost per QALY gained) of PSMA. Exclusion criteria were (1) is a conference abstract, commentary, letter to the editor, review article without original data, or gray literature, and (2) provides only a cost estimate.
For all potentially eligible articles identified during first-level screening, the full text was reviewed to ensure that the article met all eligibility criteria. The reference lists of eligible articles were reviewed for articles not identified by computerized searches.
Data abstraction
The data abstracted independently by the two reviewers from each eligible article included the first author/year of publication, country, study population, new health technology, comparator(s), perspective, decision analytic model, time horizon, discount rate, currency, year of costing, ICER, and cost-effective at WTP threshold. The findings from the included studies were synthesized using descriptive and qualitative approaches. Studies were appraised for methodological quality using the Quality of Health Economic Studies (QHES) checklist and for reporting quality using the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist.
Results
The initial literature search identified 526 articles as outlined in the diagram in Figure 1. Duplicate articles (n = 105) retrieved by the computerized search were excluded. On review of the article title and abstracts, 385 articles were excluded because they were unrelated and did not meet the eligibility criteria. A full-text review of the remaining 36 articles identified 7 studies for inclusion in the final review.

PRISMA flowchart for study selection.
Characteristics of literature on PSMA-PET imaging technology
The peer-reviewed publications summarized in Table 1 have investigated the cost-effectiveness of PSMA-PET compared to existing imaging technology or standard of care to diagnose and stage PCa. These studies were conducted in Australia (n = 3),12,13,17 The Netherlands (n = 3)16,18,24 and USA (n = 3).14,15,25 These studies represent a range of PCa patients, such as newly diagnosed high-risk PCa,12,17 intermediate- to high-risk PCa with or without lymph node metastasis,13,17,18 men diagnosed with PCa, 14 or newly diagnosed with local or regional PCa. 15 The new health technology (advanced diagnostic imaging) investigated by the emerging literature was mostly PSMA PET/CT,13–18 and one study examined PSMA PET/MRI 12 compared to existing imaging technologies, which include CT, MRI, and bone scan. Radiopharmaceutical tracers represented by studies were 68Ga12,13,16,17,18 and 18F.13–15 Most of the studies were conducted from the perspective of the healthcare system,12–14,16 and one from a third-party payer 15 and societal. 17 One of the studies 17 that adopted a societal perspective included the cost of time out of work to receive the diagnostic imaging (i.e., out-of-pocket cost).
Characteristics of included studies.
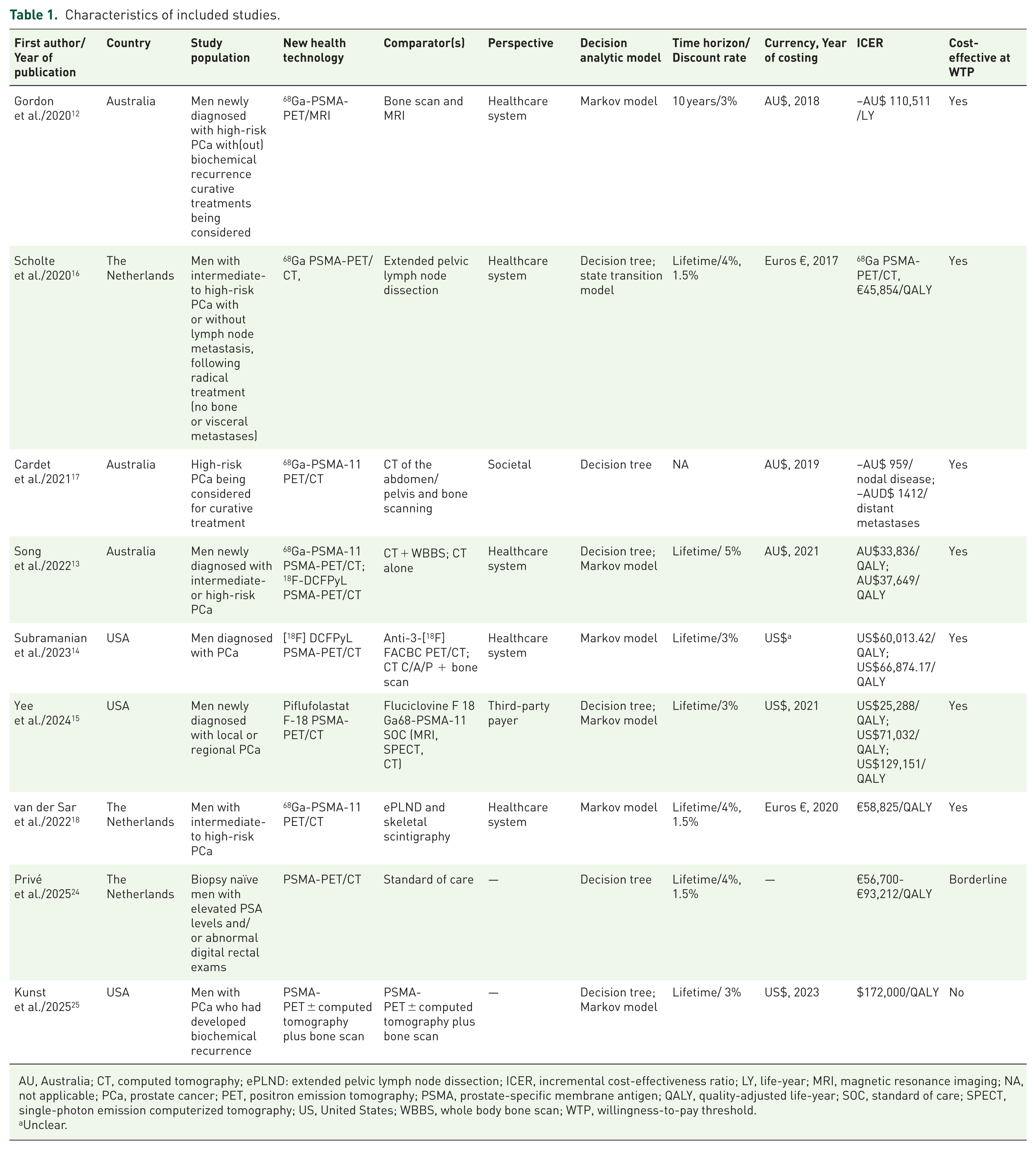
AU, Australia; CT, computed tomography; ePLND: extended pelvic lymph node dissection; ICER, incremental cost-effectiveness ratio; LY, life-year; MRI, magnetic resonance imaging; NA, not applicable; PCa, prostate cancer; PET, positron emission tomography; PSMA, prostate-specific membrane antigen; QALY, quality-adjusted life-year; SOC, standard of care; SPECT, single-photon emission computerized tomography; US, United States; WBBS, whole body bone scan; WTP, willingness-to-pay threshold.
Unclear.
All studies have used decision analytic modeling involving a decision tree with or without a Markov (state transition) model to compare costs and effects between novel imaging technologies (e.g., PSMA-PET/CT or MRI, 68Ga or 18F) to existing imaging technologies. None of the studies has justified using a specific model type. For example, whether authors used the Markov model to simulate recurring events. 19 The time horizon represented by these studies ranges from 10 years to a lifetime (Table 1). Of note, one of the studies 17 investigated only the diagnosis of PCa and did not include the impact of treatment strategies on outcomes. In contrast, the other studies investigated the diagnostic aspect with curative treatment and its impact on costs and effects. As such, there is a lack of standard guidance on limiting the analysis to the diagnosis of the cancer or combining it with curative treatments to compare costs and effects between new and existing imaging technologies.
Studies that have included radical prostatectomy or radiotherapy as one of the curative treatments did not clearly indicate the distribution of different types of surgery (open, laparoscopic, or robotic) or radiotherapy (external or internal) modeled and the corresponding ICER generated by the model. At the country-specific WTP threshold, the ICER of the base case of most of the studies was cost-effective (Table 1). Authors have identified various parameters ICER was sensitive to, lesion detection rate 68Ga-PSMA, 12 cost of follow-up in usual care group, 12 lower sensitivity of extended pelvic lymph node dissection (ePLND), 16 loss of QALYs to ePLND, 16 efficiency of the gallium-68 generator to produce the PSMA radioisotope and associated costs, 17 diagnostic cost, diagnostic sensitivity, regional/distant disease distribution, starting PSA distribution, and the utilities of having no disease or local disease. 15 Of note, none of the studies reported costs, effects, and ICER by risk groups and by different type of prostatectomy or radiotherapy.
Critical assessment of methodological quality
The Quality of Health Economic Studies (QHES) checklist 20 was used to assess the methodological quality of the studies. The QHES checklist is a reliable and valid instrument that is easy to use.21,22 The QHES checklist consists of 16 criteria, each with a weighted score ranging from 1 to 9. 20 The quality of a study can range from 0 to 100, where 0 represents the lowest quality and 100 is the highest quality. 20 One adaptation was made to the QHES system if a criterion was not relevant to a study that was assigned “not applicable.”
Table 2 summarizes the findings of the QHES methodological assessment. Notable methodological issues were that for most of the studies, the data on clinical evidence was gathered either from RCT, observational study, or systematic review-/meta-analysis on primary studies prone to various biases or a high degree of heterogeneity (question#3).12–14,16 The authors did not justify the selection of clinical evidence data for model development from sources that vary in the data quality (RCT vs observational study). None of the studies have adequately described measuring or estimating resource quantities used, corresponding unit costs, and calculating total costs (question#9). Only two studies adequately described health outcome measurement used valid and reliable scales, EQ-5D-3L 13 and EQ-5D-5L 15 (question#11). None of the studies have justified the choice of the decision analytic model they have used in their model (question#13), and the direction and magnitude of potential biases (question#14).
Results of QHES assessment.

QHES checklist questions to examine methodological quality 19 :
Q1—Was the study objective clearly defined?
Q2—Were the study perspective and rationale for selecting the specific perspective stated?
Q3—Were parameters used in the analysis based on the best available source of data?
Q4—Was subgroup analysis prespecified at the beginning of the study?
Q5—Was uncertainty assessed (statistical analysis for random events and sensitivity analysis for assumptions)?
Q6—Was incremental analysis conducted between comparators?
Q7—Was the methodology of data abstraction stated?
Q8—Was an appropriate time horizon used? Were costs and outcomes discounted for a time horizon beyond one year?
Q9—Was appropriate methodology used to estimate resource use and cost?
Q10—Was the primary outcome measure of the study clearly stated?
Q11—Were valid and reliable health outcome measurement scales used?
Q12—Were the economic model, methods, and analysis described clearly and transparently?
Q13—Were the choice of the modeling approach, assumptions, and limitations stated and justified?
Q14—Were the direction and magnitude of potential biases discussed?
Q15—Was there synergy between the study results and conclusions and recommendations?
Q16—Was the source of funding disclosed?
NA, not applicable; QHES, Quality of Health Economic Studies.
Critical assessment of reporting quality
The reporting quality of the studies were evaluated using the CHEERS checklist 2022, 23 which has 28 items categorized into the following groups: (1) title, (2) abstract, (3) introduction, (4) methods, (5) results, (6) discussion, and (7) other relevant information (source of funding and conflict of interest). 23 Each item on the CHEERS checklist was assigned “yes,” indicating reported; “no,” indicating not reported; “partially met,” indicating not adequately reported; “not applicable,” indicating if that item was not relevant; and “unknown,” indicating if the authors considered that item.
Table 3 summarizes the quality of reporting of the emerging literature. Notable deficiencies observed in reporting were that most studies did not explicitly describe the sociodemographic characteristics of the target population represented by their research (item 5). For example, sociodemographic characteristics such as age, gender, race, ethnicity, income level, and educational attainment, among others, were not mentioned in most studies.12,13,15,16 Such details are essential to understand the target population represented by the model and help with the generalizability of findings. Half of the studies12,14,16 did not mention the valuation of health outcomes included in the study (item 13). For example, studies that reported QALYs did not describe whether valid and reliable multiattribute utility instruments (e.g., EQ-5D-3L, EQ-5D-5L) were used to generate scores converted to utilities to value health outcomes. In addition, the population characteristics on which utilities were measured and how that aligns with the author's study were not mentioned. Most studies12,14,16,17 did not describe the measurement and valuation of resources, and the corresponding unit costs used to generate total costs (item 14). Hence, it can be challenging to know if all key cost components were included in cost calculations, which makes it harder to compare costs reported by different studies.
Results of CHEERS 2022 assessment.
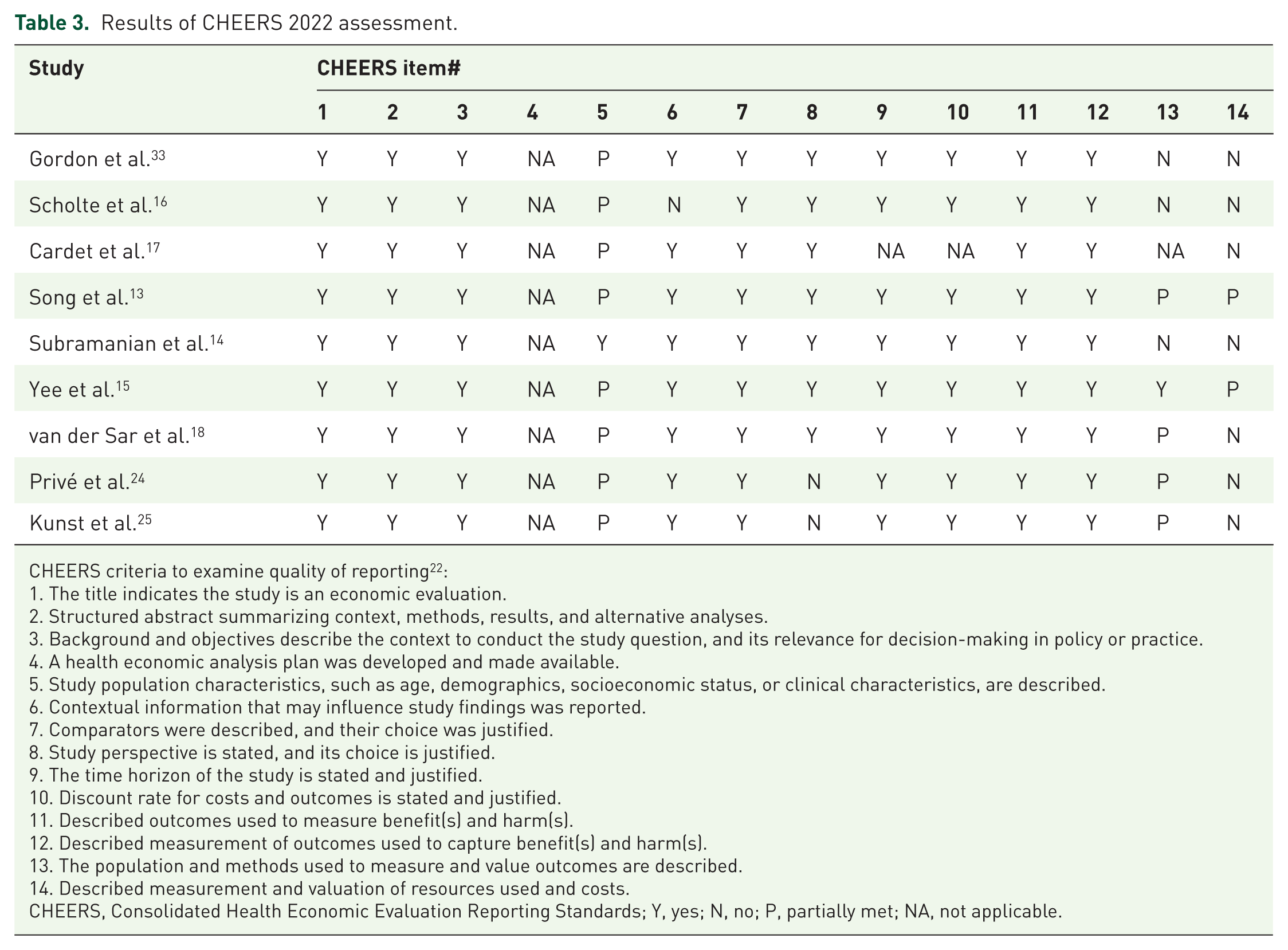
CHEERS criteria to examine quality of reporting 22 :
The title indicates the study is an economic evaluation.
Structured abstract summarizing context, methods, results, and alternative analyses.
Background and objectives describe the context to conduct the study question, and its relevance for decision-making in policy or practice.
A health economic analysis plan was developed and made available.
Study population characteristics, such as age, demographics, socioeconomic status, or clinical characteristics, are described.
Contextual information that may influence study findings was reported.
Comparators were described, and their choice was justified.
Study perspective is stated, and its choice is justified.
The time horizon of the study is stated and justified.
Discount rate for costs and outcomes is stated and justified.
Described outcomes used to measure benefit(s) and harm(s).
Described measurement of outcomes used to capture benefit(s) and harm(s).
The population and methods used to measure and value outcomes are described.
Described measurement and valuation of resources used and costs.
CHEERS, Consolidated Health Economic Evaluation Reporting Standards; Y, yes; N, no; P, partially met; NA, not applicable.
Results of CHEERS 2022 assessment (continued).

CHEERS criteria to examine quality of reporting 22 :
Reported currency used to measure costs, unit costs, and year of conversion.
Described the model in detail and why it is used.
Described methods used to transform data, extrapolating data, and model validation.
Described methods used to estimate if results varied across subgroups.
Described how impacts are distributed across different individuals or adjusted to reflect priority populations.
Described methods used to characterize any sources of uncertainty in the analysis.
Described approaches to engage patients or service recipients, the public, communities, or stakeholders (e.g., clinicians or payers) to design the study.
Reported all analytic inputs (e.g., model input values, ranges, and references), including uncertainty or distributional assumptions.
Reported the mean values of costs and outcomes of interest and summarized them in an appropriate overall measure.
Described whether uncertainty influenced model findings.
Reported any difference in patient/service recipient, the public, community, or stakeholder involvement made to the approach or findings of the study.
Reported key findings, limitations, ethical or equity considerations not captured, and how these could affect patients, policy, or practice.
The source of funding and the role of the funder in the identification, design, conduct, and reporting of the analysis are reported.
Conflicts of interest of authors are reported.
CHEERS, Consolidated Health Economic Evaluation Reporting Standards; Y, yes; N, no; P, partially met; NA, not applicable; UNK, unknown.
The clinical literature indicates that specific equity-deserving groups are disproportionately affected by PCa. One of the studies 14 has considered patient demographics and socioeconomic status while interpreting their findings (item 19). Most of the studies12–14,16 were not explicit about whether they engaged with clinicians to design their study (item 21).
Discussion
The cost-effectiveness of novel health technologies is a comparative analysis of alternative strategies regarding their costs and consequences. 14 They can inform decision-making about adopting a novel health technology by policymakers, payers, healthcare providers, patients, and the public about choices that affect health resource use, costs, and consequences. 23 This review is the first to identify and summarize the methodological and reporting quality of the emerging literature on the cost-effectiveness of PSMA-PET imaging technology to detect and stage PCa to inform treatment decision-making. The assessment of the emerging literature identified certain methodological and reporting issues present in the peer-reviewed studies. The current evidence highlights that PSMA-PET advanced imaging technology is cost-effective compared to a wide range of existing imaging technologies or standards of care at the country-specific WTP threshold. This provides confidence in the growing evidence supporting the detection and staging of PCa. Most studies have used a Markov cohort approach instead of patient-level simulation. Therefore, variability in response is not considered.
Incorrect staging at the time of primary diagnosis of PCa can lead to increased costs due to repeat testing and suboptimal treatment pathways, as well as adversely impacting survival and quality of life. 16 One of the cost-effectiveness studies highlighted barriers such as race, household income, and insurance status that might impede access to this advanced imaging technology, which can minimize its benefit to control cancer at the population level in specific healthcare systems. 14 This can raise essential health policy issues about this advanced imaging technology being affordable and accessible to all who may benefit from this technology. 26 Not recognizing and accounting for such barriers will impede successful implementation, especially in underserved populations (e.g., equity-deserving groups or those with low socioeconomic status). This will require the development of appropriate regulations and health policies to reduce or eliminate health disparities, inequities, and to improve health and well-being.
The studies12–18 examined represent only three countries, including public and private healthcare systems. The results of the cost-effectiveness studies cannot be directly applied to other countries due to significant variations in health systems, resource availability, clinical practice patterns, and economic factors, which affect both costs and treatment outcomes. While these results are not directly transferable, they can be adapted or transferred with appropriate adjustments to local costs and country-specific clinical data using established frameworks and modeling techniques to make them locally relevant for decision-making. Hence, as this advanced imaging technology becomes available in other countries, cost-effectiveness studies should be conducted in those countries, taking into consideration the local context. Second, develop models representing the distribution of the type of prostatectomy (open, laparoscopic, or robotic) or radiotherapy (external or internal) in the local context and report the corresponding ICER specific to each type of treatment. Third, future studies should investigate out-of-pocket costs experienced by underinsured or uninsured patients, and if they live in rural areas and must pay for travel, lodging, meals, and time off work to get imaging done. Finally, health policy research is required to identify solutions to factors that impede access to the imaging technology for PCa control at the population level.
Limitations
Our review has several limitations. First, we limited the review to studies published in English. Second, assessing the methodological and reporting quality of the studies using the QHES and CHEERS checklists involved subjective judgments from the reviewers, which may have influenced the outcomes. Third, publication bias could impact the findings, as studies with negative results are less likely to be published. Lastly, the included studies varied in terms of settings, interventions, and service utilization, making it challenging to compare them for identifying cost-effective services.
Conclusion
Cost-effectiveness evaluations may not be widely generalizable due to significant variations in health systems, resource availability, clinical practice patterns, and economic factors. Although preliminary results suggest that PSMA-PET can lead to cost savings in the management of prostate cancer patients, further assessments of its cost-effectiveness across different healthcare systems are necessary. Current literature indicates that decision analytic models evaluating the cost-effectiveness of PSMA-PET for diagnosis and staging have predominantly used patient cohorts instead of focusing on individual patients. Future research should adopt patient-level simulation approaches to more accurately reflect the complexities encountered in routine clinical practice, thereby facilitating better decision-making.
Supplemental Material
sj-docx-1-tau-10.1177_17562872261436874 – Supplemental material for Cost-effectiveness of PSMA-PET imaging technology in diagnosing and staging of prostate cancer: a systematic review
Supplemental material, sj-docx-1-tau-10.1177_17562872261436874 for Cost-effectiveness of PSMA-PET imaging technology in diagnosing and staging of prostate cancer: a systematic review by Chiranjeev Sanyal and Ricardo Rendon in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
