Abstract
Background:
Many metastatic prostate cancer prognostics have been suggested, but few are validated. Nodal metastasis burden and baseline biochemical characteristics are overlooked in the currently accepted stratifications for metastatic hormone-sensitive prostate cancer (mHSPC). Prostate-specific membrane antigen positron emission tomography/computed tomography (PSMA PET/CT) is likely to increase the incidence of pelvic nodal and mHSPC undetected by conventional scans. However, there is no consensus on managing regional nodal metastasis (N1M0) and no separate guidelines for non-regional nodal (M1a) and low-volume bone (M1b) spread but collectively as a part of low-volume CHAARTED disease.
Objectives:
To assess the different prognostic factors for stage IV disease classified as CHAARTED low-volume on a real-world series of patients and to examine treatment preference for each of the disease subcategories.
Methods and design:
This retrospective cross-sectional study included patients diagnosed with HSPC at stage IV, with low-volume disease according to the CHAARTED criteria. Data were collected from the database of Portsmouth and St. Mary NHS Hospitals between February 2017 and August 2023. Patient characteristics were analysed, and prognostic factors were evaluated using Cox regression analysis. 5-year progression-free survival (PFS) was the primary outcome measure.
Results:
Data on 126 patients were analysed. Seven patients (6%) had N1M0, 28 (22%) M1a, and 91 (72%) M1b. 5-year PFS was 80.9% for M1a and 54.9% for M1b metastases, p = 0.3. High prostate-specific antigen (PSA) value (⩾25) was identified as an independent prognostic factor for PFS with HR = 2.80 (95% CI: 1.19–6.56), p = 0.0179. Variable treatment preference for each subclass reflects the uncertainty regarding the best regimen and the importance of consolidation prostate radiotherapy (cRT) in clinical practice.
Conclusion:
Early results of our data analysis underscore the significance of baseline PSA as an independent prognostic factor alongside anatomical tumour extent of spread in stage IV low-volume metastasis prostate cancer. There is no agreement on treatment for each subcategory, necessitating further real-world studies and clinical trials. Further follow-up would assess the prognostic benefit of cRT.
Keywords
Introduction
Prognostic factors aim to group patients who have similar clinical outcomes. This facilitates the conduction of clinical trials involving relatively similar patient groups, the comparison of clinical and pathological data from various hospitals worldwide, and the formulation of treatment recommendations for these patient populations.1,2 The eighth edition of the AJCC-TNM classifications of cancer spread outside the prostate depends mainly on the site of metastasis. It considers prostate cancer at stage IV in the presence of metastases in pelvic lymph nodes (N1M0) or abdominal lymph nodes (M1a), alongside the presence of bone (M1b) or visceral metastases (M1c). 3 For a long, systemic treatment with androgen deprivation therapy (ADT) has been considered the core of treatment for these subgroups. Afterwards, efforts to improve treatment effectiveness, including concurrent use of local or systemic therapies with ADT, have emerged. A heterogeneity among studies was highly significant for all outcomes for mHSPC after ADT, 4 which reflects the prognostic differences among the disease subgroups.
Different factors predicting survival have been suggested for metastatic prostate cancer, such as the number and the site of metastases, the International Society of Urological Pathology (ISUP) grade group, alkaline phosphatase levels, performance status, and initial and nadir prostate-specific antigen (PSA). However, only a small number of these factors have been confirmed.5–9
The most accepted CHAARTED criteria sub-grouped metastatic disease according to the presence or absence of visceral metastases and number of bones spread, examining the stratified benefit of chemotherapy and other different ADT additions on the different metastatic burden. 8 Unlike risk stratification for high-risk M0 patients,2,10 factors overlooked in these classifications include nodal metastasis burden and information on baseline biochemical and pathological characteristics of metastatic hormone-sensitive prostate cancer (mHSPC), which could influence the prognosis.
While LATITUDE is another validated classification of risk for M1 disease which includes ISUP grade of cancer, to our knowledge, it has not been regularly used as a criterion for stratification of results of the vast high yield mHSPC trials except for Abiraterone Acetate plus Prednisolone (AAP) addition.9,11 Furthermore, there is no need to fulfil all but only two of the criteria to classify as high risk; therefore, the site of metastasis and pathological diagnosis can be overlooked in many situations for low-risk patients.
The advent of PSMA PET/CT is anticipated to increase the incidence of pelvic nodal and low-volume mHSPC, which the conventional scans could not detect. 12 However, there is no consensus about the best treatment regimen for the subgroups of mHSPC yet. In addition, no guidelines provide separate management for M1a or low-volume M1b, but the recommendations are for both as a part of CHAARTED low-volume disease (LVD).2,10 The reason is mainly the focus of the interventional phase III trials to use the CHAARTED and LATITUDE criteria as a stratification tool for their results.
Our study’s early analysis aimed to evaluate, in a real-world cohort of patients, the prognostic value of the clinical variables at baseline and treatment choice by clinicians for different subgroups of stage IV, LVD mHSPC.
Methods
This retrospective cross-sectional collaborative study recruited 126 patients diagnosed with nodal and low-volume HSPC at the Queen Alexandra Hospital in Portsmouth and St Mary’s Hospital on the Isle of Wight, both NHS-affiliated institutions in the United Kingdom. The study assessed all the patients who underwent treatment for metastatic prostate cancer from February 2017 to August 2023 at the hospitals.
Data collection was conducted by the two Hospitals’ physicians. Eligibility included patients with confirmed metastatic prostate cancer with N1M0, M1a or M1b with three or fewer bone metastases confined to axial bone. Confirmation of the primary prostate cancer was performed after biopsy diagnosis for most of our patients. Five patients had no biopsy taken with a digital rectal exam and PSA test used to confirm the diagnosis. Exclusion criteria include any 8th edition AJCC stage except stage IV N1M0, M1a and M1b. M1b patients with more than three axial bone metastases with other concurrent extra-axial and/or visceral metastasis were excluded. Disease volume assessment was conducted meticulously by the medical practitioners through a comprehensive analysis of staging CT, bone, MRI and PET images. The radiological diagnosis of cancer substage and progression as per RECIST criteria was concluded from the formal reports written by the radiology consultant and the outcome of the urology multidisciplinary team meetings held in the aforementioned hospitals, both of whom are not investigators of the study. Bony metastases were documented with attention to their location, number, and morphology, along with evaluation of any lymph node metastases.
Clinical data collection encompassed various clinical parameters including dates of diagnosis, biopsy, disease progression, mortality, initial Gleason score, PSA value at the time of metastatic disease, disease stage according to the 8th edition of the AJCC – TNM classification and details of administered treatments.
The primary outcome was measuring progression-free survival (PFS). PFS was defined as the duration from diagnosis of metastatic disease, rather than treatment, to radiologically documented disease progression (local or distant), death, or the last known contact date when the patient was alive. Patients who discontinued treatment for reasons unrelated to disease progression were censored. PFS was specifically calculated for patients undergoing first-line treatment. Confirmation of metastasis was used as the starting point rather than treatment initiation because of the difficulty of determining the accurate date of therapy start and our main aim, which was primarily evaluating the prognosis in metastatic disease rather than the impact of the treatments. In addition, with doublets or triplets, the treatment with Androgen Receptor Pathway Inhibitors (ARPI) and/or docetaxel could start even 3 months after the initiation of ADT and cRT could begin up to 6 months later. Also, ADT was sometimes initiated based on PSA levels before the diagnostic process concluded the presence and definition of metastatic disease.
A relatively high number of patients were censored because of the limited time from inclusion to our early analysis. Statistically significant results with unequal censoring or without sufficient sample size remaining before the clinical outcome was reached were excluded. Further analysis will be published after further follow-up of our data.
The Strengthening the Reporting of Observational Studies in Epidemiology statement (STROBE) was used to report this study (Supplemental File 1). 13 Ethical guidelines were strictly followed throughout the study, adhering to the principles outlined in the Declaration of Helsinki. The study protocol was registered as an audit at Portsmouth Hospitals University NHS Trust (Audit ID No. 5621, dated July 20, 2023) under the title ‘Choice of management in patients with low-volume metastatic prostate cancer’. Informed consent for this analysis was not obtained as no additional information beyond what was already available was considered in the analysis. Patients signed a written consent for treatment that includes the possibility of sharing information about their clinical history for audit purposes.
Statistical analysis
Categorical variables were presented as proportions, while continuous data were summarized using measures of central tendency, including means, medians and measures of dispersion such as standard deviations.
To explore potential associations between categorical variables, appropriate statistical tests were employed, including Fisher’s exact test or the Chi-square test. Additionally, the Kruskal–Wallis test was utilized to investigate relationships among various continuous variables in our study. Kaplan–Meier curves were generated to illustrate survival outcomes for categorical variables. Median PFS times were estimated using the product-limit method, while follow-up time was determined using the Kaplan–Meier reverse method.
Log-rank test p-values were computed to compare PFS distributions across different categories. The median follow-up times were compared using the non-parametric Wilcoxon test. The prognostic significance of clinical variables was assessed through univariate Cox regression analysis for PFS. Subsequently, a multivariable stepwise Cox regression analysis was performed, incorporating clinical baseline prognostic factors.
Both univariate and multivariate Cox regressions were conducted for both continuous and categorical variables, with results reported as hazard ratios (HRs) and corresponding 95% confidence intervals (CIs). Multivariate Cox analysis is used to account for bias and confounding factors. Statistical significance was determined at the 0.05 level for all analyses. Data were extracted from electronic medical records and anonymized data were analysed using R Statistical Software (version 4.2.3; R Core Team, 2023).
Results
Patient characteristics and treatment
Out of 126 patients with stage IV CHAARTED low-volume mHSPC, 7 had N1 (6%), 28 (22%) M1a and 91 (72%) M1b. The patient characteristics according to the site of metastases are shown in Table 1. The only significant difference among these three categories of patients was found in treatment with ADT only (p < 0.001). While 2 out of 7 patients with N1 (29%) received a combination of ADT and androgen receptor-targeted agents (ARTA) or docetaxel, 9 out of 28 patients (32%) with M1a and 8 out of 91 patients (9%) with M1b received ADT alone.
Baseline clinical characteristics of the patients stratified by metastatic site.
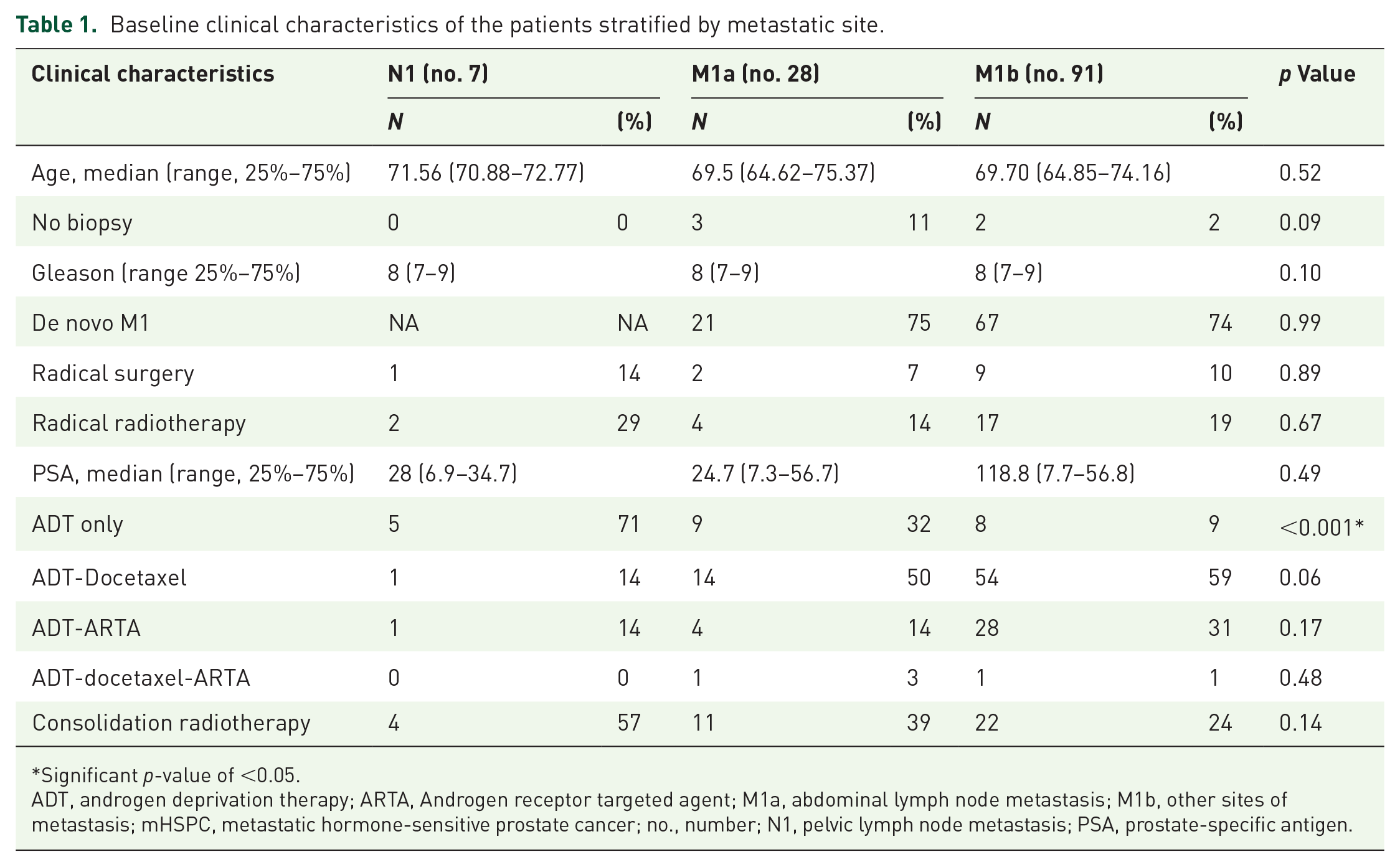
Significant p-value of <0.05.
ADT, androgen deprivation therapy; ARTA, Androgen receptor targeted agent; M1a, abdominal lymph node metastasis; M1b, other sites of metastasis; mHSPC, metastatic hormone-sensitive prostate cancer; no., number; N1, pelvic lymph node metastasis; PSA, prostate-specific antigen.
Although not statistically significant, radiotherapy, either as radical or consolidation treatment, was offered more frequently to patients with N1 (29% and 57%, respectively) compared to M1a (14% and 39%) or M1b (19% and 24%) (p = 0.67 and p = 0.14, respectively). Radical radiotherapy used here refers to radiotherapy to prostate and metastatic lesions with curative intention; on the other hand, consolidation radiotherapy refers to radiotherapy to the prostate only to improve prognosis.
Clinical outcome (progression-free survival)
With a median follow-up of 27.0 months (95% CI, 12.9–49.2), the 5-year PFS rate was 63.1% for all patients and, respectively, 100% (95% CI, 100%–100%) for patients with N1, 80.9% (95% CI, 62.8–100) for those with M1a and 54.9% (95% CI, 38.5–78.4) for those with M1b (p = 0.3) (Figure 1, Table 2).

Progression-free survival stage IV low-volume mHSPC patients stratified by the metastatic site.
Clinical outcome of stage IV low-volume mHSPC patients stratified by the metastatic site.

CI, confidence interval; FU, follow-up; M1a, abdominal lymph node metastasis; M1b, other sites of metastasis; mHSPC, metastatic hormone-sensitive prostate cancer; N1, pelvic lymph node metastasis; no., number; PFS, progression-free survival.
Prognostic factors
Significant prognostic variables in univariate analysis for PFS were a PSA value equal to or above the median value of 25 (HR 2.65, 95% CI, 1.13–6.22, p = 0.025) and administration of cRT (HR 0.12, 95% CI, 0.02–0.87, p = 0.036) (Table 3).
Univariate and multivariate analyses for PFS by baseline clinical characteristics of stage IV low-volume mHSPC patients.

Significant p-value of <0.05.
ADT, androgen deprivation therapy; ARTA, Androgen receptor targeted agent; CI, confidence interval; cRT, consolidation prostate radiotherapy; H, high; HR, hazard ratio; L, low; M1a, abdominal lymph node metastasis; M1b, other sites of metastasis; M, metastases; mHSPC, metastatic hormone-sensitive prostate cancer; N1, pelvic lymph node metastasis; PFS, progression-free survival; PSA, prostate-specific antigen.
Multivariate analysis confirmed high PSA value (HR 2.80, 95% CI, 1.19–6.56, p = 0.0179) and cRT (HR 0.11, 95% CI, 0.02–0.80, p = 0.029) as independent prognostic factors for PFS (Table 3). These results are generated data and external validation remains necessary.
Baseline PSA
There was no significant difference in follow-up between the patients with PSA below and above 25 at the initial diagnosis (median of 33.2 vs 26.8 months, p = 0.14). Patients with lower baseline PSA showed a significantly higher 5-year PFS (74.9%, 95% CI, 56.9–98 vs 51.9%, 95% CI, 43.9–77.1, p = 0.02) (Figure 2, Table 4).

Progression-free survival stage IV low-volume mHSPC patients stratified by the baseline PSA.
Clinical outcome of stage IV low-volume mHSPC patients stratified by level of PSA at initial presentation.
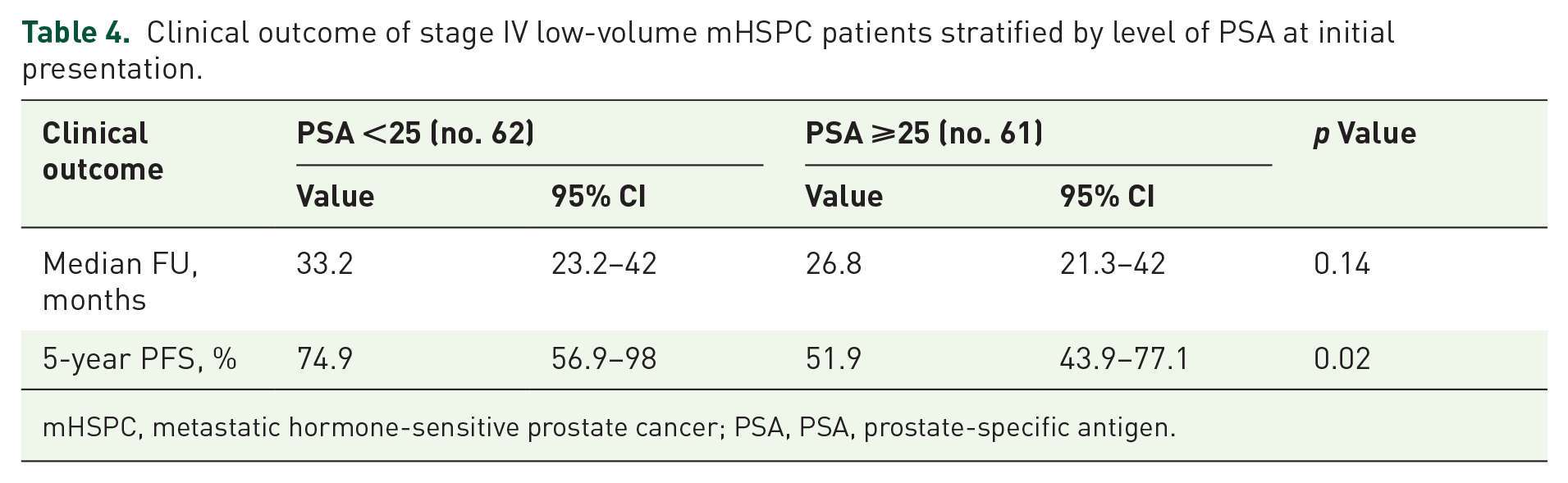
mHSPC, metastatic hormone-sensitive prostate cancer; PSA, PSA, prostate-specific antigen.
There was an equal number of patients who represented the two PSA subgroups with a relatively equal number of censoring and an adequate sample size (Supplemental Table 1). Nevertheless, around 45 were censored for both arms, which counts for this study’s limitation.
Discussion
Our analysis of data from 126 patients over more than 6 years suggests that the baseline PSA value may be a useful independent prognostic factor. Additionally, metastasis limited to lymph nodes could be of different prognosis compared to LVD M1b. Data also reflect the disparate clinical practice regarding the choice of management for this subgroup.
The cumulative results of the study demonstrated the best 5-year PFS with PSA lower than 25 regardless of age, Gleason score, sites of metastasis and treatment received for our selected group of patients. In post hoc analysis of SWOG1216, TITAN and LATITUDE, PSA level change was monitored after months of intensification treatments in which levels drop after treatment proved a better long-term survival.14–16 Our results, on the other hand, illustrated the benefit of PSA value at initial diagnosis. Therefore, it could be used in the stratification of this subgroup as a prognostic factor and trials used classification.
Research utilizing the SEER registry database has determined that elevated PSA levels at 60 or more are linked to poorer cancer-specific survival, 17 in contrast to our results with only a PSA level of 25 or more. Another important difference from previous studies, the analysis of this study was on N1M0, M1a and LVD M1b, which reflects the significance of PSA level consideration in CHAARTED LVD, rather than M1 disease.
In patients with M1a, approximately one-third received ADT only with an intermediate estimate of PFS between low-volume N1 and M1b disease. Our series may indicate that even within low-volume disease, there could be differences in outcome between M1a and M1b, which requires a more in-depth analysis and prognostic factors that can guide treatment choices. Other evidence from examining STAMPEDE arms A versus C showed the importance of nodal burden on prognosis with worse prognosis with five or more nodal metastases. 18 Therefore, these results question the need for examining and treating isolated nodal metastasis as a separate entity.
There was an almost equal tendency to use the same treatment line among the different subgroups included. The only significant statistical variability among the different clinical variables monitored was ADT use alone for most N1M0 patients. This contrasted with the current recommendation of ADT plus AAP.2,10 STAMPEDE analysis of arms A versus G and J for the addition of AAP provided a significant stratified metastasis-free survival. 19 Despite evidence of beneficial addition from STAMPEDE for AAP and radiotherapy (RT) separately to N1M0 treatment,19,20 the final effect of this combination remains uncertain, especially with respect to toxicity versus benefit profile.
Despite not being statistically different, cRT was preferably offered to N1M0 compared to LVD M1. RT benefit for this subgroup is supported by evidence from the STAMPEDE trial. 20 Compared to RT to prostate and lymph nodes used in STAMPEDE analysis, patients in our study received RT to the prostate only. For LVD, current results from phase III trials showed contradictory overall survival (OS) outcomes after adding RT between PEACE-1 and STAMPEDE (arm A vs H), where STAMPEDE favours use unlike PEACE-1.21,22 In addition, STAMPEDE M1a OS had HR of 0.60 and 95% CI: 0.33–1.09, compared to clear statistical significance for M1b until three bone metastases. 23 Further monitoring of the patient’s data remains necessary to estimate the 5-year PFS and thus the prognostic benefit of cRT for each of our target disease subgroups.
Most of the patients with LVD M1a and M1b in the study received doublet treatment with a slight tendency to use docetaxel plus ADT. While triplet therapy is supported by meta-analyses of phase III trials, including PEACE-1 and ARASENS, for CHAARTED high-volume disease, no stratified positive outcome was concluded for LVD.24–27 A meta-analysis conducted by Naqvi et al. 2022 showed the superiority of ARTI plus ADT over docetaxel plus ADT and triplet therapies for this subgroup. 25 However, the comparison between different ARTIs’ relative outcomes for cN1, M1a and LVD M1b remains uncertain. On the other hand, there was no difference in OS between AAP or docetaxel addition to ADT as per STAMPEDE sub-analysis for high-risk M0 and M1 with relatively similar side effects. 28 Therefore, evidence for phase III trials is still inconclusive for best systemic therapy for LVD.
Limitations of our study include its retrospective nature, the relatively small number of patients, namely N1M0, and the relatively short and different follow-up between patients who received cRT and who did not. Also, the number of censored patients was relatively high secondary to the nature of the study and the limited duration of follow-up especially for those included at the end of the study period. However, this early analysis aims to publish the reached significant outcome of our study with further follow-up and analysis expected. Despite the statistical significance of 5-year PFS after cRT, because of unequal censoring with many patients having not enough time for follow-up before data freeze, the results could not be validated. The same problem was noticed with N1M0 results. Thus, we decided to neglect these results interpretation. Follow-up of the patients would give more insight regarding the isolated prognosis of these subgroups.
In conclusion, early analysis of our data emphasizes the importance of baseline PSA as an independent prognostic factor, alongside anatomical tumour extent within LVD. Considering biochemical factors like PSA at presentation, a more precise stratification and a better framework for stage IV LVD management could be delivered, avoiding over- and undertreatment of its subgroups. Additionally, there was no absolute consensus regarding clinical practice treatment of choice for each subcategory. Following up our study’s patients could assess the prognostic benefit from cRT and confirm our findings.
@gbanna74, Dr Banna’s handle—the article’s supervisor—in the tweet.
Supplemental Material
sj-docx-1-tau-10.1177_17562872241297579 – Supplemental material for Prognostic factors and treatment choice for stage IV, low-volume metastasis hormone-sensitive prostate cancer: cross-sectional study of real-world data
Supplemental material, sj-docx-1-tau-10.1177_17562872241297579 for Prognostic factors and treatment choice for stage IV, low-volume metastasis hormone-sensitive prostate cancer: cross-sectional study of real-world data by Mohamed Ibrahim Elewaily, Marina Campione, Mona Ali Hassan, Shobana Anpalakhan, Naoko Atsumi, Benjamin Smalley, Anza Ashraf, Joanna Gale, Akash Maniam and Giuseppe Luigi Banna in Therapeutic Advances in Urology
Supplemental Material
sj-docx-2-tau-10.1177_17562872241297579 – Supplemental material for Prognostic factors and treatment choice for stage IV, low-volume metastasis hormone-sensitive prostate cancer: cross-sectional study of real-world data
Supplemental material, sj-docx-2-tau-10.1177_17562872241297579 for Prognostic factors and treatment choice for stage IV, low-volume metastasis hormone-sensitive prostate cancer: cross-sectional study of real-world data by Mohamed Ibrahim Elewaily, Marina Campione, Mona Ali Hassan, Shobana Anpalakhan, Naoko Atsumi, Benjamin Smalley, Anza Ashraf, Joanna Gale, Akash Maniam and Giuseppe Luigi Banna in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
