Abstract
Background:
Fournier’s gangrene (FG) is a rapidly progressive necrotizing infection with high mortality. Early recognition of high-risk patients is critical for improving outcomes.
Objectives:
To evaluate the prognostic value of the Inflammatory Burden Index (IBI)—a composite marker derived from C-reactive protein, neutrophil, and lymphocyte counts—for predicting mortality, intensive care requirements, and clinical course in FG.
Design and methods:
This retrospective cohort included 130 patients diagnosed with FG and treated at a tertiary referral center between 2015 and 2025. Demographic, clinical, and laboratory data were collected at admission. IBI was calculated using first-draw laboratory values. Primary outcomes were disease-specific mortality and intensive care unit (ICU) admission; secondary outcome was length of hospital stay. Univariable and multivariable logistic regression analyses were performed, and discriminatory capacity was evaluated using receiver operating characteristic analysis.
Results:
Nonsurvivors exhibited significantly higher IBI values than survivors (p = 0.001). On multivariable analysis, IBI independently predicted in-hospital mortality (p = 0.017), disease-specific mortality (p = 0.004), and ICU admission (p = 0.008). Higher IBI was also associated with prolonged hospitalization (p = 0.012). Discriminative performance was acceptable, with an area under the curve of 0.757 for overall mortality and 0.856 for disease-specific mortality.
Conclusion:
Admission IBI is an independent predictor of mortality, ICU requirement, and hospital stay in FG. As a simple, cost-effective, and rapidly available biomarker, IBI may support early risk stratification and decision-making in clinical practice.
Plain language summary
Fournier’s gangrene is a rapidly progressing and life-threatening infection that affects the genital and perineal region. Early detection of patients who are likely to become severely ill is crucial for saving lives. In this study, researchers evaluated a blood-based marker called the Inflammatory Burden Index (IBI), which combines three commonly measured laboratory values: C-reactive protein (CRP), neutrophil count, and lymphocyte count. This index reflects how strongly the body’s immune system is reacting to infection. The research included 130 patients who were treated for Fournier’s gangrene between 2015 and 2025. The study found that patients with higher IBI values when they arrived at the hospital were more likely to die from the infection, require intensive care treatment, and stay in the hospital longer. Even after adjusting for other health conditions and age, IBI remained a strong and independent predictor of poor outcomes. Because IBI is calculated from standard blood tests that are available in almost every hospital, it can be used easily and immediately when patients are first admitted. Doctors may use this information to identify high-risk patients earlier, decide on intensive monitoring or surgical management, and improve overall treatment planning. In short, the study suggests that the Inflammatory Burden Index is a simple, inexpensive, and powerful tool to predict which patients with Fournier’s gangrene are at greatest risk of severe illness or death.
Introduction
Fournier’s gangrene (FG) is a necrotizing fasciitis of the perineum and genital region—a rare but fulminant infection with high mortality if not promptly treated. 1 This polymicrobial soft-tissue infection progresses rapidly through fascial planes, leading to systemic toxicity and sepsis. 2 Due to its aggressive course and lethal potential, early identification of disease severity is critical. Various prognostic scoring systems and laboratory markers have been explored to stratify risk in FG patients.3–5 Infection-based hematologic indices have prognostic value in severe infections. In FG, elevated neutrophil-to-lymphocyte ratio (NLR) is linked to poor outcomes; patients with NLR ⩾8 showed higher risk of ventilation or death, highlighting NLR as a rapid, cost-effective tool. 6 Such findings underscore the importance of systemic inflammatory indices as accessible markers of disease severity in FG and other critical infections.
Recently, a novel composite index of inflammation—the Inflammatory Burden Index (IBI)—has been introduced and increasingly applied in clinical research. The IBI is calculated by multiplying serum C-reactive protein (CRP) concentration by the absolute neutrophil count and dividing the result by the lymphocyte count (IBI = CRP × neutrophils/lymphocytes). This formula reflects both the magnitude of the acute-phase response and the degree of lymphocyte suppression. 7 By combining an acute-phase reactant (CRP) with two immune cell parameters, IBI reflects an individual’s overall inflammatory and immune status. 8 In essence, a high IBI indicates an exaggerated inflammatory response (elevated CRP and neutrophilia) coupled with relative lymphopenia, a pattern often seen in severe infections. The clinical significance of IBI has been demonstrated across diverse conditions. In acute ischemic stroke, for instance, elevated IBI was significantly associated with higher 90-day morbidity, indicating worse neurological outcomes. 9 In oncology, a multicenter study of 6359 cancer patients found that those with high IBI had markedly reduced survival rates (45.7% vs 69.1% in 5-year survival, p < 0.001) compared to patients with low IBI. 10 IBI has outperformed traditional markers in cancer prognosis; in nonsmall cell lung cancer, it showed the highest accuracy (C-statistic 0.64) and independently predicted overall survival. 11 Chronic inflammatory diseases show similar trends: for example, in patients with chronic inflammatory airway disease, higher IBI levels correlated with increased all-cause and respiratory mortality. 8 These findings show that IBI reflects disease severity and prognosis, often outperforming single biomarkers.
Given its composite nature, IBI has also been investigated in infectious and critical illness settings. Recent evidence in sepsis is particularly compelling. In a large intensive care unit (ICU) cohort, septic patients with higher IBI had significantly greater mortality risk. 7 There was a near-linear relationship between IBI level and 28-day mortality, and importantly, IBI demonstrated better discriminatory power for death than CRP alone. 7 These data suggest that IBI captures the severity of systemic inflammation in sepsis more effectively than conventional markers, likely because it integrates both the magnitude of the acute-phase response and the degree of immune cell perturbation. Because FG represents a state of overwhelming inflammatory activation with concurrent immune dysregulation, a composite index such as IBI may theoretically capture disease severity more reliably than single-parameter markers. Consistent with these findings, a recently published study further confirmed that elevated IBI values are strongly associated with disease severity and mortality in patients with severe infectious conditions, supporting the use of IBI as a robust indicator of systemic inflammatory stress. 12
We hypothesized that IBI could indicate disease severity in FG, as it reflects key features of severe infection. Yet no study has assessed IBI in FG. Thus, we aimed to evaluate whether elevated IBI at presentation predicts worse outcomes or advanced disease, and whether it can serve as a prognostic biomarker to improve early risk stratification and management in this life-threatening condition.
Materials and methods
This retrospective single-center cohort included all consecutive adults (⩾18 years) admitted with a clinical diagnosis of FG between August 2015 and August 2025. A total of 292 patients were screened. We excluded individuals with missing baseline inflammatory markers required for IBI calculation, incomplete outcome data, insufficient documentation, or recurrent/nonindex admissions. We also excluded patients with hematologic malignancy, active autoimmune disease, or systemic immunosuppressive therapy at presentation to avoid confounding of inflammatory parameters. After applying these predefined criteria, 130 patients were included in the final analytic cohort.
Ethics
The study protocol was approved by the Institutional Ethics Committee of Izmir Katip Celebi University (Approval No.: 2025-SAEK-0852, Date: 09.09.2025) and conducted in accordance with the Declaration of Helsinki. All patients signed institutional treatment consent forms, including authorization for anonymized use of clinical information for scientific research and publication. As this work was based on retrospectively collected, de-identified records, additional study-specific consent was not required.
Collected data and outcome measures
Demographic and clinical data were abstracted from electronic records: age, sex, body mass index, smoking and alcohol status, major comorbidities (hypertension (HT), diabetes, coronary artery disease, peripheral arterial disease, history of malignancy), number of debridements, use and duration of vacuum-assisted closure, fecal or urinary diversion, and total length of hospital stay. Baseline laboratory values at first presentation—ideally before initial debridement—included complete blood count (neutrophils, lymphocytes, hemoglobin, platelets), serum creatinine, estimated glomerular filtration rate (eGFR), serum bicarbonate, pH, lactate, and CRP. The IBI was calculated from first-draw values as:
Clinical outcomes were predefined. Overall mortality was death from any cause during the index hospitalization. Disease-specific mortality was death primarily attributable to FG or its septic complications, adjudicated from physician documentation and discharge summaries by two investigators with consensus. ICU admission was recorded whenever organ support or invasive monitoring was required. Length of stay was measured in days from admission to discharge; patients transferred to other acute-care facilities were censored on the transfer date.
Statistical analysis
All analyses were performed in Jamovi (v2.6; The Jamovi Project, Sydney, NSW, Australia). Normality of continuous variables was assessed with the Shapiro–Wilk test and visual inspection. Continuous data are presented as mean ± SD or median (IQR), and categorical data as counts and percentages. Between-group comparisons used the Student’s t test or Mann–Whitney U test for continuous variables, and Chi-square or Fisher’s exact test for categorical variables.
Univariable analyses identified candidate predictors, which were entered into multivariable logistic regression to determine independent predictors of overall mortality, disease-specific mortality, and ICU admission. Covariates included age, comorbidity burden (Charlson Comorbidity Index (CCI)—not the age-adjusted version 13 or diabetes), renal function (creatinine or eGFR), serum bicarbonate, and IBI. Model fit and discrimination were assessed by the Hosmer–Lemeshow test and receiver operating characteristic (ROC) analysis.
To evaluate prognostic performance, ROC curves were generated for IBI, with optimal thresholds defined by the Youden index. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were reported. For length of stay, linear regression was applied; skewed data were log-transformed in secondary analysis. Results are presented as regression coefficients with 95% confidence intervals (CIs), and IBI effects expressed per 1000-unit increase. Statistical significance was set at p < 0.05 (two-sided). This research was reported in accordance with the STROBE guidelines for cohort studies.
Results
A total of 130 patients were analyzed. Of the 130 patients included, 80 survived (61.5%) and 50 died during hospitalization (38.5%). Nonsurvivors were older (65.2 ± 11.8 vs 55.1 ± 13.7 years, p < 0.001), had lower body mass index (BMI; 26.3 ± 5.2 vs 28.2 ± 5.4 kg/m2, p = 0.041), and higher CCI (4.4 ± 2.4 vs 2.7 ± 2.5, p < 0.001). Renal function was worse in nonsurvivors (creatinine 1.74 ± 1.27 vs 1.30 ± 1.10 mg/dL, p = 0.049; eGFR 62.4 ± 35.1 vs 80.1 ± 28.7 mL/min/1.73 m2, p = 0.002). Inflammatory markers were significantly elevated: NLR (18.3 ± 15.7 vs 11.6 ± 10.7, p = 0.028), platelet-to-lymphocyte ratio (PLR; 324.1 ± 157.5 vs 228.9 ± 157.8, p = 0.007), CRP (234.2 ± 100.0 vs 167.3 ±107.0 mg/L, p < 0.001), and IBI (4601.0 ± 4626.3 vs 2414.3 ± 3022.8, p = 0.001). Fournier’s Gangrene Severity Index (FGSI) scores were significantly higher in nonsurvivors compared with survivors (20.38 ± 5.49 vs 17.12 ± 3.59, p < 0.001). Acid–base status (pH, bicarbonate, lactate) did not differ (all p > 0.05). Among categorical variables, diabetes (p = 0.049) and smoking (p = 0.018) were more frequent in nonsurvivors; other comorbidities showed no differences. Baseline characteristics by survival status are summarized in Table 1.
Baseline clinical and laboratory characteristics by overall survival status.

Student’s t test (independent).
Mann–Whitney U test.
Fisher’s exact test.
χ2 test.
BMI, body mass index; CAD, coronary artery disease; CCI score, Charlson Comorbidity index score; CRP, C-reactive protein; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; FGSI, Fournier’s Gangrene severity index; Hb, hemoglobin; HT, hypertension; IBI score, inflammatory burden index score; NLR, neutrophil-to-lymphocyte ratio; PAD, peripheral artery disease; PLR, platelet-to-lymphocyte ratio; SD, standard deviation.
The multivariable logistic regression for in-hospital mortality is summarized in Table 2. Age (odds ratio (OR) 1.05; p = 0.004) and IBI (p = 0.017) were independently associated with mortality, whereas eGFR (p = 0.467) and diabetes (p = 0.109) were not. ROC analysis identified an optimal IBI cut-off value of approximately 2118 for predicting mortality, yielding 52.0% sensitivity, 86.3% specificity, and an area under the curve (AUC) of 0.757 (Figure 1).
Multivariable binomial logistic regression for overall survival status.
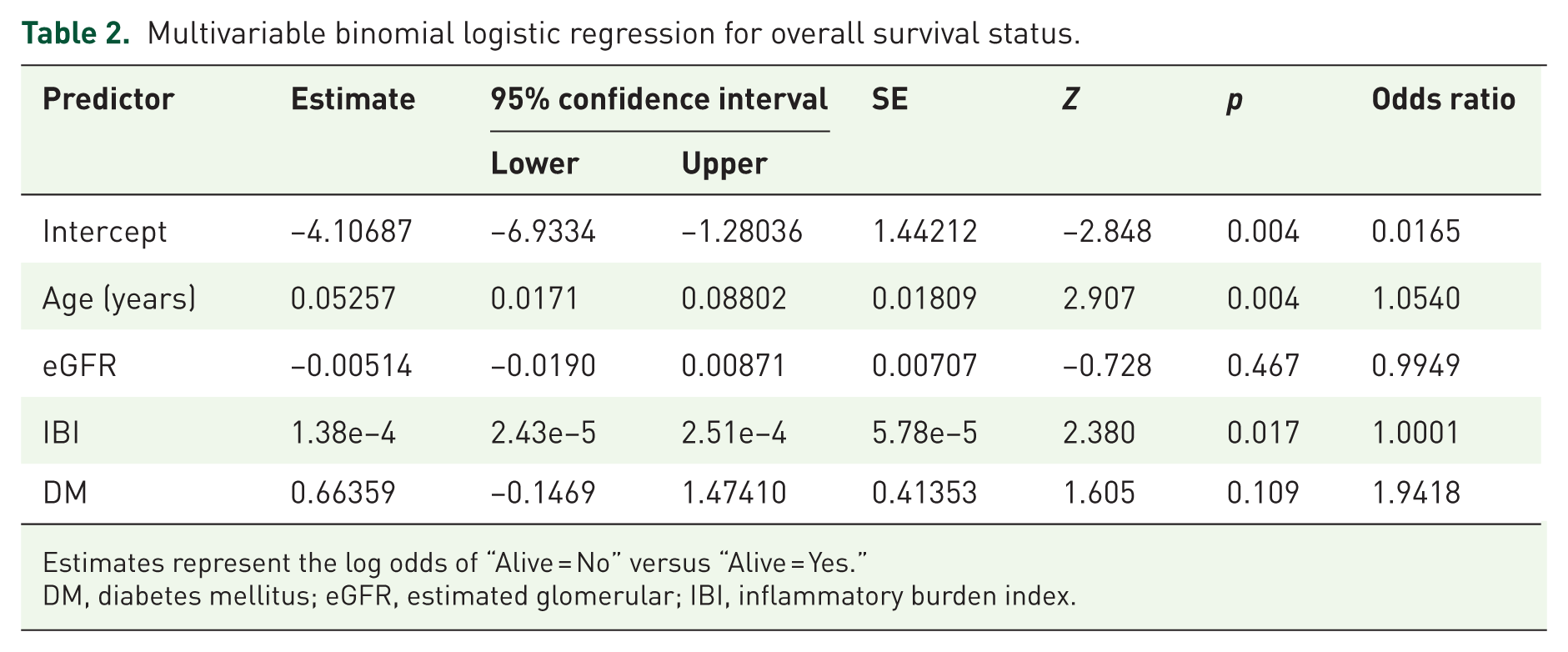
Estimates represent the log odds of “Alive = No” versus “Alive = Yes.”
DM, diabetes mellitus; eGFR, estimated glomerular; IBI, inflammatory burden index.

ROC of the multivariable model for overall survival status. The ROC curve displays sensitivity versus 1 – specificity across all predicted-probability thresholds of the logistic regression model. The positive class was death during the index hospitalization. Model predictors were age, estimated glomerular filtration rate, IBI, and diabetes. The diagonal line denotes the line of no discrimination. ROC analysis identified an optimal IBI cut-off value of approximately 2118 for predicting mortality, corresponding to the highest Youden Index (J = 0.612) and yielding 52.0% sensitivity and 86.3% specificity. The overall discriminatory performance of the model was moderate, with an AUC of 0.757.
ICU patients were older (64.1 ± 15.4 vs 58.1 ± 13.5 years, p = 0.035), had higher comorbidity (CCI 4.3 ± 1.9 vs 3.2 ± 2.6, p = 0.027), worse renal function (creatinine 2.20 ± 1.27 vs 1.36 ± 1.14 mg/dL, p = 0.001; eGFR 46.4 ± 30.9 vs 77.6 ± 30.6 mL/min/1.73 m2, p < 0.001), and higher inflammatory indices: NLR (24.8 vs 12.5, p = 0.001), PLR (398.4 vs 244.2, p = 0.014), CRP (264.7 vs 181.5 mg/L, p = 0.003), and IBI (6414.7 vs 2747.5, p < 0.001). Bicarbonate was lower in ICU patients (20.0 vs 22.6 mEq/L, p = 0.006), while pH and lactate showed no difference (both p > 0.20; Table 3).
Baseline clinical and laboratory characteristics by intensive care unit admission.

Mann–Whitney U test.
Student’s t test (independent).
Fisher’s exact test.
χ2 test.
BMI, body mass index; CAD, coronary artery disease; CCI score, Charlson comorbidity index score; CRP, C-reactive protein; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; HT, hypertension; IBI score, inflammatory burden index score; NLR, neutrophil-to-lymphocyte ratio; PAD, peripheral artery disease; PLR, platelet-to-lymphocyte ratio.
The multivariable logistic regression for ICU admission is summarized in Table 4. IBI (p = 0.008) and serum bicarbonate (p = 0.047) were independently associated with ICU admission, whereas age (p = 0.426) and CCI (p = 0.477) were not. Model discrimination is depicted in Figure 2 (AUC = 0.783).
Multivariable binomial logistic regression for intensive care unit admission.

Estimates represent the log odds of “need for intensive care = Yes” versus “Need for Intensive Care = No.”
CCI, Charlson comorbidity index; IBI, inflammatory burden index; SE, standard error.

ROC curve of the multivariable model for ICU admission. The ROC curve plots sensitivity versus 1 − specificity across all predicted-probability thresholds of the logistic regression model. The positive class was ICU admission (yes). Model predictors were age, CCI, IBI, and serum bicarbonate. The AUC was 0.783. At the probability cut-off of 0.50, specificity was 0.982 and sensitivity 0.167.
The multivariable logistic regression for disease-specific mortality is summarized in Table 5. IBI was independently associated with disease-specific mortality (p = 0.004), as was age (OR 1.09 per year; p = 0.048), whereas CCI was not (p = 0.880). Model discrimination is shown in Figure 3 (AUC = 0.856).
Multivariable binomial logistic regression for disease-specific mortality.

Estimates represent the log odds of “Death due to disease = Yes” versus “Death due to disease = No.”
CCI, Charlson Comorbidity Index; IBI, Inflammatory Burden Index.

ROC curve of the multivariable model for disease-specific mortality. The ROC curve plots sensitivity versus 1 – specificity across all predicted-probability thresholds of the logistic regression model. The positive class was death due to disease during the index hospitalization. Model predictors were age, IBI, and CCI. The AUC was 0.856. At a probability cut-off 0.50, specificity was 1.00 and sensitivity 0.111.
As shown in Table 6, IBI correlated with Length-of-stay (LOS; Pearson r = 0.179, p = 0.042; Spearman ρ = 0.290, p < 0.001). In multivariable analysis with log-transformed LOS, both IBI (β = 4.92 × 10−5, p = 0.012) and urinary diversion (β = 0.735, p = 0.012) predicted longer stay, while age and CCI were not significant. Model fit was modest (R = 0.310, R2 = 0.096).
Multivariable linear regression for log-transformed length of stay and its association with the IBI and clinical covariates.
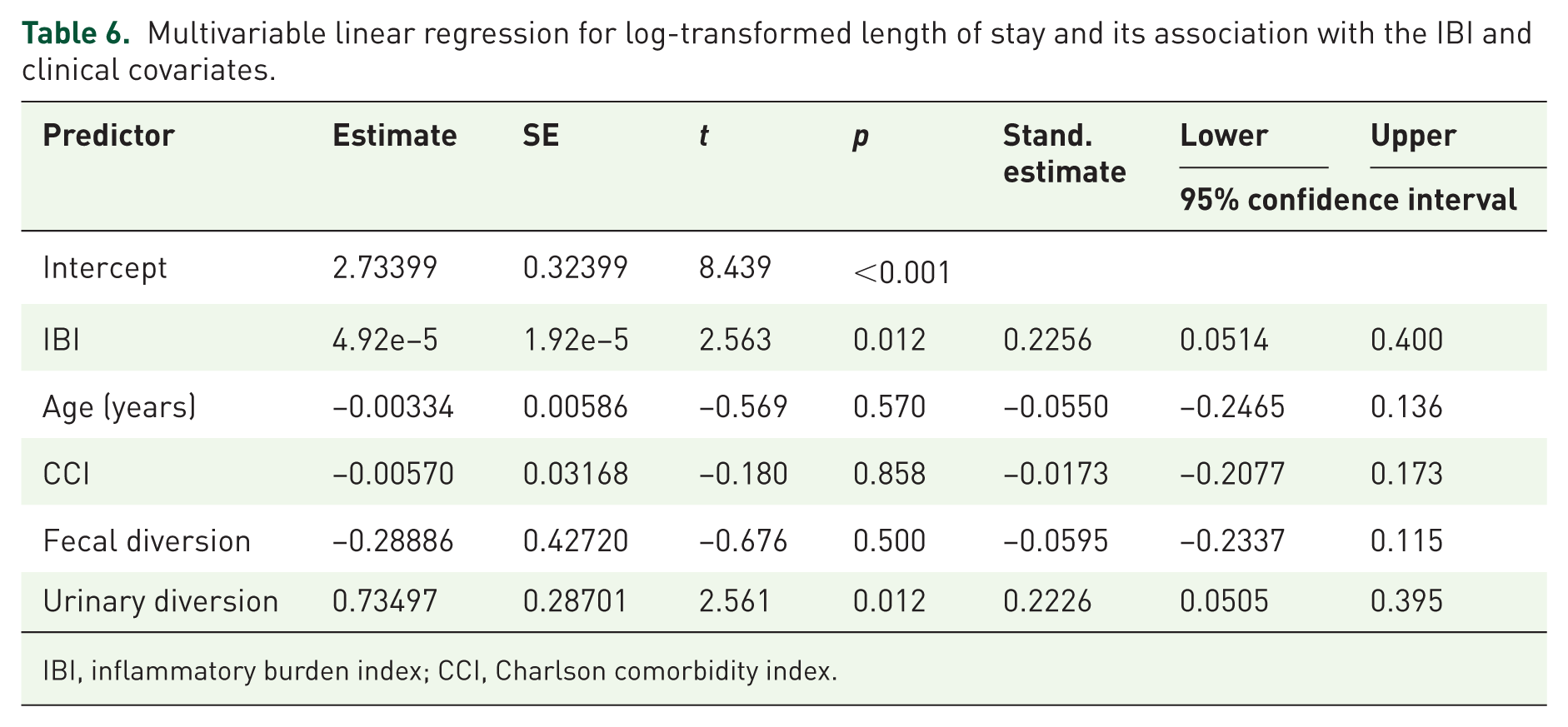
IBI, inflammatory burden index; CCI, Charlson comorbidity index.
Discussion
Our study demonstrates that the IBI, a composite marker of systemic inflammation (calculated as CRP × neutrophil count/lymphocyte count), is a powerful independent predictor of outcomes in patients with FG. Patients who succumbed to FG or required ICU support had significantly higher admission IBI values, underscoring a strong association between heightened inflammatory response and disease severity. This finding is biologically plausible given that the early systemic effects of FG result from an excessive inflammatory reaction of the host to the invasive infection.
This observation is in line with recent evidence showing that the IBI is closely associated with adverse clinical outcomes in severe infectious and inflammatory conditions, further supporting the concept that IBI reflects the magnitude of systemic inflammatory stress rather than disease-specific pathology alone. 14
Mortality in FG remains appreciable—historically reported between ~16% and 40%—even as modern specialized centers have reduced disease-specific fatality to around 10%.15,16 Our results suggest that IBI, as a quantification of the initial inflammatory burden, can help stratify which FG patients are at greatest risk for these poor outcomes.
These findings align with evidence supporting IBI as a prognostic marker. As a composite of CRP, neutrophils, and lymphocytes, IBI offers a broader measure of systemic inflammation. 17 Previous studies have noted that IBI offers more precise prognostic value for patient outcomes than individual inflammatory markers. 10 For instance, Pelc et al. 18 reported that elevated IBI correlated with higher postoperative complication rates and mortality in gastric cancer patients undergoing multimodal therapy. In a large prospective NSCLC study, Xie et al. 11 showed that IBI had the greatest discriminatory power among inflammatory indices and independently predicted overall survival. Notably, the same study showed that a high IBI also predicted other adverse outcomes such as 90-day postoperative complications, prolonged hospital stays, and increased hospitalization costs. 11 To our knowledge, this is the first study to assess IBI in necrotizing soft tissue infections such as FG. The strong prognostic value observed extends prior findings to acute surgical infections, underscoring systemic inflammation as a key determinant of outcomes across oncologic, neurologic, and infectious diseases.
In FG, various prognostic tools have been studied. The FGSI and its modifications (Uludağ FGSI, simplified FGSI) remain widely used and prognostically validated. 19 FGSI incorporates vital signs and labs to predict mortality, with higher scores indicating worse outcomes. Yet, its calculation can be cumbersome and performance inconsistent. Simpler lab-based markers are gaining attention; for instance, the CRP/albumin ratio recently differentiated survivors from nonsurvivors with accuracy comparable to FGSI. 19
Similarly, NLR—a component of IBI—has been validated as a simple marker of systemic inflammation. A 2025 meta-analysis 15 showed higher NLR in non-survivors (mean difference ~+4.5), with values >8–10 conferring a 1.7–2.3-fold increased mortality risk. These findings mirror those of smaller single-center studies that noted higher NLR (and related indices) among nonsurvivors and support the notion that an exaggerated innate immune response portends worse prognosis. 20 Our study extends prior work by assessing IBI, which integrates CRP and NLR to reflect both acute-phase response and immune cell shifts. Its superior prognostic performance in FG supports the value of combining multiple inflammatory signals into a single index, consistent with reports in other diseases. 17 Notably, some investigators have reported that neither FGSI nor NLR alone was an adequate predictor of mortality on multivariate analysis in FG, highlighting that single measures or scores might miss aspects of risk. 21 By contrast, the IBI’s ability to remain an independent predictor in our multivariable model (which included traditional risk factors) underscores its robustness and potential clinical utility.
Our multivariate analysis shows that IBI complements established risk factors, with age and comorbidity burden (CCI) also linked to worse FG outcomes.22,23 Advanced age remains a well-recognized mortality risk. 24 The presence of multiple chronic conditions synergistically impacts survival; indeed, an age-adjusted CCI score >5 has been linked to significantly higher mortality in FG patients. 20
Similarly, impaired renal function was linked to poor outcomes, as patients with lower eGFR fared worse. This is consistent with evidence that kidney injury predicts prognosis; in one large FG series, elevated BUN independently predicted mortality alongside disease extent and malignancy.16,25 Our findings confirm established prognostic factors but show that IBI remained significant after adjustment for age, eGFR, CCI, and other variables. This indicates that IBI reflects systemic inflammatory stress beyond demographics or comorbidities. Clinically, high IBI may flag patients at high risk despite younger age or fewer comorbidities, while modest IBI in older or comorbid patients may signal a more favorable prognosis. Thus, IBI provides incremental value for risk stratification.
Beyond mortality, we examined ICU need and hospital stay as indicators of severity and resource use. IBI was significantly higher in patients requiring ICU care, underscoring its clinical relevance in identifying the sickest FG patients, who are at risk of rapid progression to septic shock and multiorgan failure.15,26 Our findings indicate that IBI, as an early marker of systemic inflammation, identifies FG patients at risk for ICU need and deterioration, supporting timely intensive management. Higher IBI also predicted prolonged hospitalization, reflecting more severe disease and complications. These results parallel oncology data, where elevated IBI was linked to longer stays and poorer survival in NSCLC. 11 In FG, prolonged hospitalization often stems from repeat surgeries, intensive wound care, and complications. IBI’s prediction of longer stays may aid resource planning and counseling, and its link to ICU need underscores its value in capturing both mortality and morbidity burden.
IBI offers practical advantages in FG: it is inexpensive, derived from routine labs, and available at admission for rapid risk assessment. High IBI can prompt early escalation of care, unlike more complex scores with variable performance. By summarizing systemic inflammation in a single metric, IBI could enhance prognostic models and guide ICU admission or surgical decisions. Further studies should validate its impact, but our findings support IBI as a robust marker of FG severity.
Limitations
This study has limitations. Its retrospective, single-center design and modest sample size may reduce generalizability and power. No a priori sample size calculation or power analysis was conducted, as the study included all eligible patients within the defined time period, and the absence of predetermined power estimation may limit the generalizability of the effect size interpretation. Although clear inclusion criteria were applied, selection and information bias remain possible. Although the mortality rate was relatively high (38.5%), the absolute number of outcome events (n = 50) still limits multivariable model stability, and therefore these estimates should be interpreted as exploratory rather than definitive. Additionally, no internal or external validation or calibration analyses were performed; therefore, generalizability remains limited and the predictive performance of IBI should be verified in larger prospective cohorts. Clinical heterogeneity in presentation, comorbidity, and prior treatment could not be fully standardized in this retrospective cohort; however, the prognostic performance of IBI despite this variability suggests real-world applicability. We only evaluated admission IBI, without analyzing serial trends that might provide additional prognostic insight. Moreover, we did not compare IBI directly with all other proposed indices (e.g., CAR, SII, CONUT), so superiority cannot be assumed. CRP levels may also be influenced by unrelated conditions, though in our cohort acute FG was the dominant driver. Another limitation of our study is the absence of data on the interval between hospital admission and surgical intervention. Early debridement is known to influence outcomes in FG; however, as this parameter was unavailable for all patients, its potential effect on IBI-related prognosis could not be assessed. Finally, outcomes were limited to in-hospital and 30-day endpoints; long-term morbidity and quality of life were not assessed. Future multicenter prospective studies should validate IBI, define optimal cut-offs, and test its integration into multifactorial risk models.
Conclusion
In conclusion, the IBI appears to be a promising prognostic biomarker in FG, independently predicting disease-specific mortality and the need for intensive care. Incorporating IBI into the initial evaluation of FG patients could improve risk stratification and guide early aggressive management, ultimately contributing to better clinical decision-making for this life-threatening condition.
Supplemental Material
sj-docx-1-tau-10.1177_17562872261430104 – Supplemental material for Prognostic value of the inflammatory burden index for disease-specific mortality and intensive care need in Fournier’s gangrene
Supplemental material, sj-docx-1-tau-10.1177_17562872261430104 for Prognostic value of the inflammatory burden index for disease-specific mortality and intensive care need in Fournier’s gangrene by Serkan Ozcan, Enis Mert Yorulmaz, Mertcan Dama, Kursad Donmez, Osman Kose, Sacit Nuri Gorgel and Yigit Akın in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

