Abstract
Background:
Radical nephrectomy is a recommended treatment for localized renal cell carcinoma when partial nephrectomy is not feasible. This approach is typically applied to patients with a normal contralateral kidney and no preexisting chronic kidney disease (CKD).
The primary concern with surgery is its potential long-term impact on renal function.
Objectives:
To assess the risk of development of chronic kidney disease after radical nephrectomy.
Design and methods:
This is a retrospective cohort study of patients with normal preoperative renal function and contralateral kidney who underwent radical nephrectomy for cancer. At the last follow-up, patients were categorized according to CKD-EPI guidelines as having normal renal function grades G1-2 or impaired renal function grade ⩾G3a. Univariable and multivariable Cox regression analyses were used to identify the significant risk factors for CKD-free survival.
Results:
We included 135 patients, 49.1 SD 12.9 years old, 62 women (45.9%), and 73 men (54.1%). Preoperative weight was 81 SD 18 kg, and eGFR was 99.2 SD 19.2 mL/min/1.73 m2. The last follow-up 32.8 SD 16.8 months later showed that 38 (28.1%) patients developed renal impairment. Univariable analysis showed that preoperatively age in years, age ⩾65, male gender, hypertension, Charlson Comorbidity score, cerebrovascular disease, low preoperative eGFR, serum creatinine, and baseline CKD EPI grade G2 were significant predictors of last follow-up renal impairment. Intraoperatively, anesthesia duration, surgery duration, and postoperatively, within the first 48 hours lower eGFR, creatinine, and CKD-EPI category G3-5 were significant risk factors predicting CKD. After exclusion of colinear risk factors, multivariable analysis, however, indicated that preoperative eGFR (p < 0.027), lower intraoperative nadir MAP (p = 0.006), and postoperative within 48-hour eGFR (p = 0.003) were independent predictors of CKD-free survival at the last follow-up.
Conclusion:
Significant risk factors for renal impairment after radical nephrectomy were a decreased preoperative eGFR, intraoperative hypotension and, within 48h, postoperative eGFR. No other preoperative or intraoperative risk factor independently predicted CKD.
Plain language summary
Why was this study done? Many people with kidney cancer need surgery to remove the affected kidney—a procedure called radical nephrectomy. Although this surgery often cures the cancer, some patients later develop long-term kidney problems, even if their remaining kidney looked healthy beforehand. We wanted to understand why this happens and how to better predict and prevent it. What did the researchers do? We looked at 135 adults who had one kidney removed for cancer. All patients had normal kidney function and a healthy remaining kidney before surgery. We reviewed their medical records to identify changes in kidney function over time and investigated whether any factors before, during, or soon after surgery were linked to a higher risk of kidney problems later. What did the researchers find? About 3 in 10 people developed long-term kidney problems after surgery. People with lower kidney function before surgery, even if still within the “normal” range, were more likely to develop problems. Another strong risk factor was how the kidney function changed within 48 hours after surgery. Patients whose kidney function dropped early were more likely to have permanent problems. Other factors—like age, blood pressure, or how the surgery was done—were not independent predictors when we analyzed the data closely. What do the findings mean? This study shows that even small differences in kidney function before or just after surgery can have a long-term impact. By identifying people at higher risk early, doctors can monitor and manage kidney health more closely. The results may help guide surgical and anesthesia decisions, improve patient counseling, and support better long-term care planning.
Keywords
Introduction
Radical nephrectomy (RN) is a recommended treatment for localized renal cell carcinoma (RCC), particularly when partial nephrectomy (PN) is not feasible. 1 This approach is typically applied to patients with a normal contralateral kidney and no preexisting chronic kidney disease (CKD), offering excellent long-term oncological outcomes. However, the balance between preserving renal function and ensuring optimal oncological results remains a subject of debate when comparing RN and PN. 2
The primary concern with RN is its potential long-term impact on renal function, particularly the risk of acute kidney injury (AKI) and subsequent development of CKD.3–5 CKD, in turn, is a well-established risk factor for cardiovascular events and increased mortality. 6 Studies indicate that up to 52% of patients who undergo RN or PN develop new-onset CKD, with RN being identified as an independent risk factor. 3 Furthermore, RN has been linked to a higher risk of progression to end-stage renal disease (ESRD), which contributes to poorer overall survival outcomes. 5
Some studies suggest that preexisting glomerular filtration rate (GFR) and CKD, rather than nephron loss from surgery, are more critical factors driving postoperative CKD development. 7 Most previous research has focused on preoperative clinical parameters and surgical type, frequently including patients with preexisting renal impairment. Identified risk factors for CKD include preoperative GFR, older age, male gender, obesity, high BMI, hypertension, smoking, and undergoing RN.3,5,8 The heterogeneity of input data, methods, and outcome measures made reaching a generally accepted prediction model of renal impairment after RN complex. 9
Although some recent studies have explored intraoperative factors affecting renal function after nephrectomy,10,11 others have focused on immediate postoperative outcomes. 12 Few studies have specifically targeted patients with normal preoperative renal function.12,13 Nonetheless, including both RN and PN in some analyses complicates the interpretation of results. 13
Our planned study aimed to address these limitations by examining the impact of RN on renal function in patients with a normal contralateral kidney. We aimed to account for factors that had confounded previous studies, such as preexisting CKD, varying surgical approaches, inclusion of advanced RCC stages, and lack of detailed intraoperative data.5,8,14–17 We also planned to use the CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) formula, which is more accurate than serum creatinine alone in estimating GFR, to avoid including patients with CKD, which had affected earlier studies.13,18
Our primary objective is to identify risk factors contributing to CKD development after RN. By examining clinical and perioperative factors in a homogeneous patient cohort and incorporating comprehensive intraoperative data, we aim to offer more precise insights into the role of RN in renal function decline and identify strategies to mitigate the risk of CKD in this patient population.
Materials and methods
Patients and design
We conducted a retrospective cohort study including all patients who underwent RN in our tertiary care hospital for a unilateral, organ-confined renal cortical tumor presumed to be primary malignancy based on clinical evaluation from February 2017 to December 2021.The Institution’s Ethics Committee has approved the study. We included only patients ⩾18 years old with preoperative eGFR ⩾60 mL/min/1.73 m2 and having a normal contralateral kidney in imaging studies. Patients with persistent albuminuria, urine sediment abnormality, or electrolyte disturbance were excluded. We retrieved data from the electronic patient charts, including preoperative, intraoperative, and postoperative clinical and laboratory data. Electronic automated intraoperative data were available on the anesthesia machines starting Feb 2017, and patients before this date were excluded.
Procedure
All patients underwent volatile general anesthesia according to standard practice. Induction was carried out using propofol 1–2 mg/kg with opioids, while sevoflurane (2–4 vol %) was used for maintenance. Patients were mechanically ventilated with a volume-controlled mode with a tidal volume of 6–8 mL/kg and a FiO2 of 0.4 to 0.5. Patients underwent an open or minimally invasive RN based on the surgeon’s assessment of the anatomical complexity of the tumor.
Definition of variables
eGFR was calculated using the CKD-EPI 2021 formula. 19 Hypertension was defined as BP ⩾120/80 mmHg. AKI within 48 after surgery was defined as an absolute increase in serum creatinine level by 0.3 mg/dL or ⩾5 0% increase from the preoperative value. 20 Postoperative eGFR was calculated at 3, 6, 12, and 24 months and the last follow-up. Postoperative last follow up CKD category was assigned according to the CKD-EPI formula into grades G1-G2 with eGFR ⩾60 mL/min/1.73 m2 or G3a and above with eGFR < 60 mL/min/1.73 m2. 18
Statistical analysis
Sample size calculation
This study employed a retrospective cohort design, and no a priori sample size or power calculation was performed. The sample size was determined by the number of consecutive patients who underwent RN for RCC during the study period and met the predefined inclusion criteria.
Analysis
All data frequencies were reported using descriptive statistics and crosstabulation. The target variable was the CKD category at the last follow-up G1-2 versus G3a and above. CKD-free survival was studied using the Cox univariable and multivariable analyses for risk factors.
Preoperative data were analyzed separately from the perioperative and postoperative data in the multivariable analysis. Codependent variables were excluded, leaving only one source value. To account for potential collinearity among clinically relevant predictors we performed variance inflation factor (VIF) analysis to test for multicollinearity for all predictors in univariate analysis (Tables 1 and 2). Variables with a VIF > 5 were considered collinear. To identify which variables are correlated we studied the “Collinearity Diagnostics” table. We used Eigenvalue near zero, Condition Index > 30, and variance proportion in each dimension > 0.5 to identify variables with strong collinearity. Next, we performed pairwise correlation using bivariate analysis for suspected pairs with high correlation to confirm the results. Pairs with Pearson’s correlation coefficients r > 0.7 were considered for removal of one variable from the final multivariable analysis. To test which of the two variables age or preoperative eGFR is an independent predictor of CKD, using Cox regression, these variables were modeled separately then together.
Continuous risk factors descriptive and univariable statistics.

Univariable Cox regression CKD free survival at last follow up. The event is category CKD ⩾ G3a at last follow-up. Text in bold indicates significant risk factors.
AKI, acute kidney injury; BMI, body mass index; BW, body weight; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration formula; CVA-D, cerebrovascular accident or peripheral arterial disease; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; HR, hazard ratio; LOS, length of stay; MAP, mean arterial pressure; VIF, variance inflation factor.
Categorical risk factors descriptive and univariable statistics.
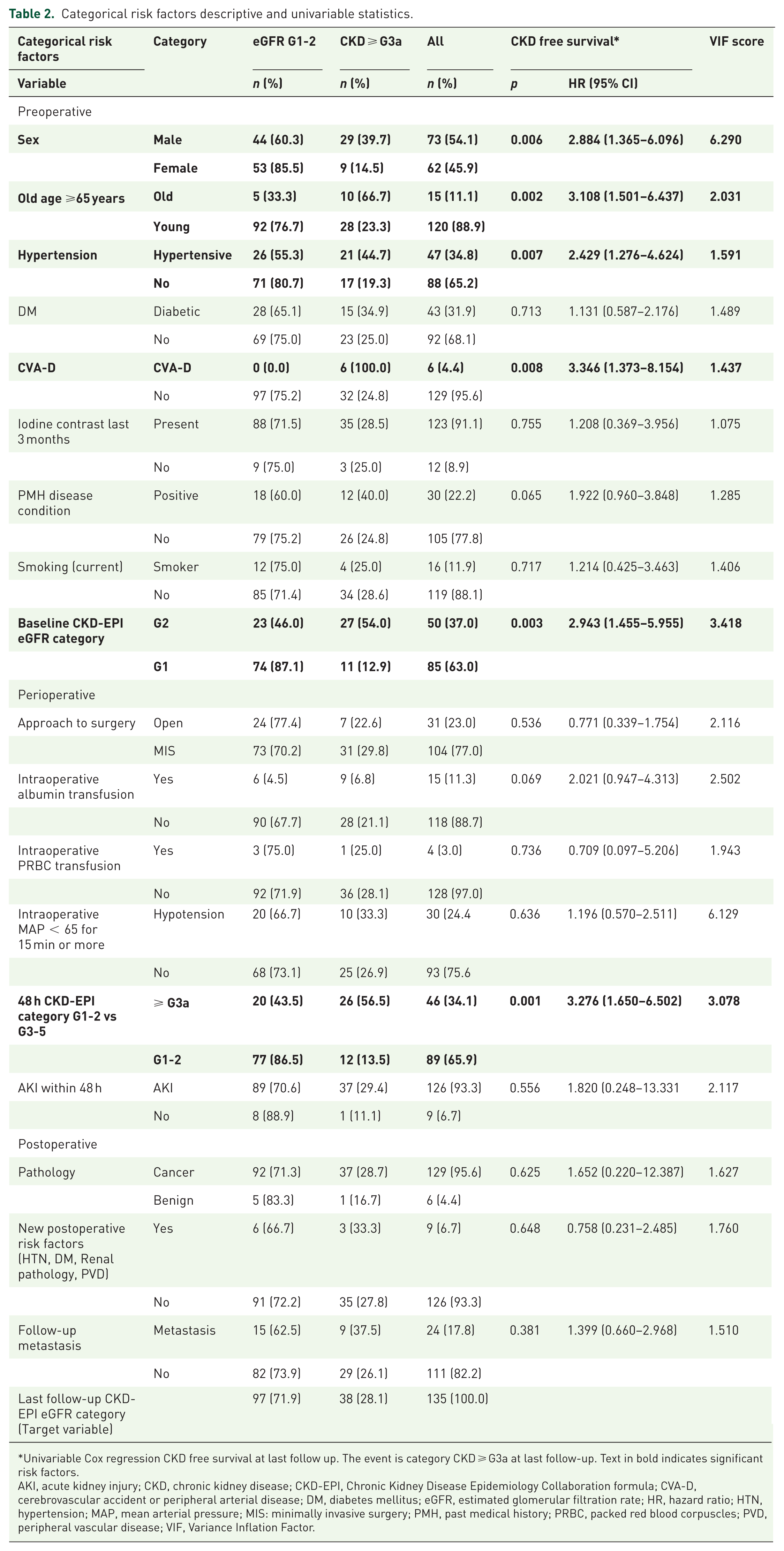
Univariable Cox regression CKD free survival at last follow up. The event is category CKD ⩾ G3a at last follow-up. Text in bold indicates significant risk factors.
AKI, acute kidney injury; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration formula; CVA-D, cerebrovascular accident or peripheral arterial disease; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; HR, hazard ratio; HTN, hypertension; MAP, mean arterial pressure; MIS: minimally invasive surgery; PMH, past medical history; PRBC, packed red blood corpuscles; PVD, peripheral vascular disease; VIF, Variance Inflation Factor.
Significant values were considered when p < 0.05.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement available at http://www.strobe-statement.org. The checklist from the statement is submitted as a Supplemental File 1.
Results
Descriptive statistics
Preoperative data
One hundred thirty-five patients, 49.1 SD 12.9 years old, 62 women (45.9%), and 73 men (54.1%), were included (Tables 1 and 2). Preoperative weight was 81 SD 18 kg, and eGFR was 99.2 SD 19.2 GFR mL/min/1.73 m2.
Operative data
Anesthesia: All patients had induction anesthesia with intravenous propofol and narcotics. Maintenance anesthesia was conducted with sevoflurane inhalation in all patients except three, who had intravenous propofol maintenance. The choice of open surgery versus minimally invasive surgery was not random and was determined by the surgeon based on the technique’s feasibility. Minimally invasive surgery included robotic-assisted or laparoscopic nephrectomy.
Intraoperative variables were not uniformly available across the cohort; intraoperative nadir MAP was documented in 123 patients, whereas urine output data were available in 104 patients; analyses of these variables were therefore performed using available-case data
Postoperative data
The last follow-up 32.8 SD 16.8 months later showed that 97 (71.9%) patients maintained normal renal function while 38 (28.1%) developed renal impairment.
Risk factors for CKD
Univariable analysis
Univariable analysis showed that preoperatively age in years (p = 0.001), age ⩾65 (p = 0.002), male gender (p = 0.006), hypertension (p = 0.007), Charlson Comorbidity score (p = 0.007), cerebrovascular accident or peripheral arterial disease (CVA-D, p = 0.008), preoperative eGFR (p < 0.001), serum creatinine (p < 0.001) and baseline CKD EPI grade G2 (P = 0.003) were significantly associated with last follow up renal impairment (Tables 1 and 2). When modeled separately with preoperative risk variables, higher preoperative eGFR was independently associated with a lower risk of CKD at last follow-up (HR 0.963 per 1 ml/min/1.73 m2 increase, 95% CI 0.937–0.990; p = 0.007), whereas age was not (HR 1.031 per year increase, 95% CI 0.983–1.081; p = 0.214). This association persisted when both variables were entered simultaneously into the model, with preoperative eGFR remaining independently associated with CKD risk (HR 0.965 per 1 ml/min/1.73 m2 increase, 95% CI 0.938–0.992; p = 0.011), while age remained nonsignificant (HR 1.022 per year increase, 95% CI 0.975–1.071; p = 0.366).
The Cox survival curve shows the significant relation between baseline CKD EPI grade and CKD-free survival at the last follow-up (Figure 1).

Cox regression showing baseline CKD-EPI category as a predictor of CKD-free survival at last follow-up.
Intraoperatively, duration of anesthesia (p = 0.043) and duration of surgery (p = 0.025) were significant risk factors predicting CKD (Table 1). Postoperatively, within the first 48 hours, eGFR (p < 0.001), creatinine (p < 0.001), and CKD-EPI category G3-5 (p = 0.001) were significant risk factors predicting CKD (Tables 1 and 2). The Cox survival curve shows a significant relation between the 48-hour CKD-EPI category and CKD-free survival at the last follow-up (Figure 2).
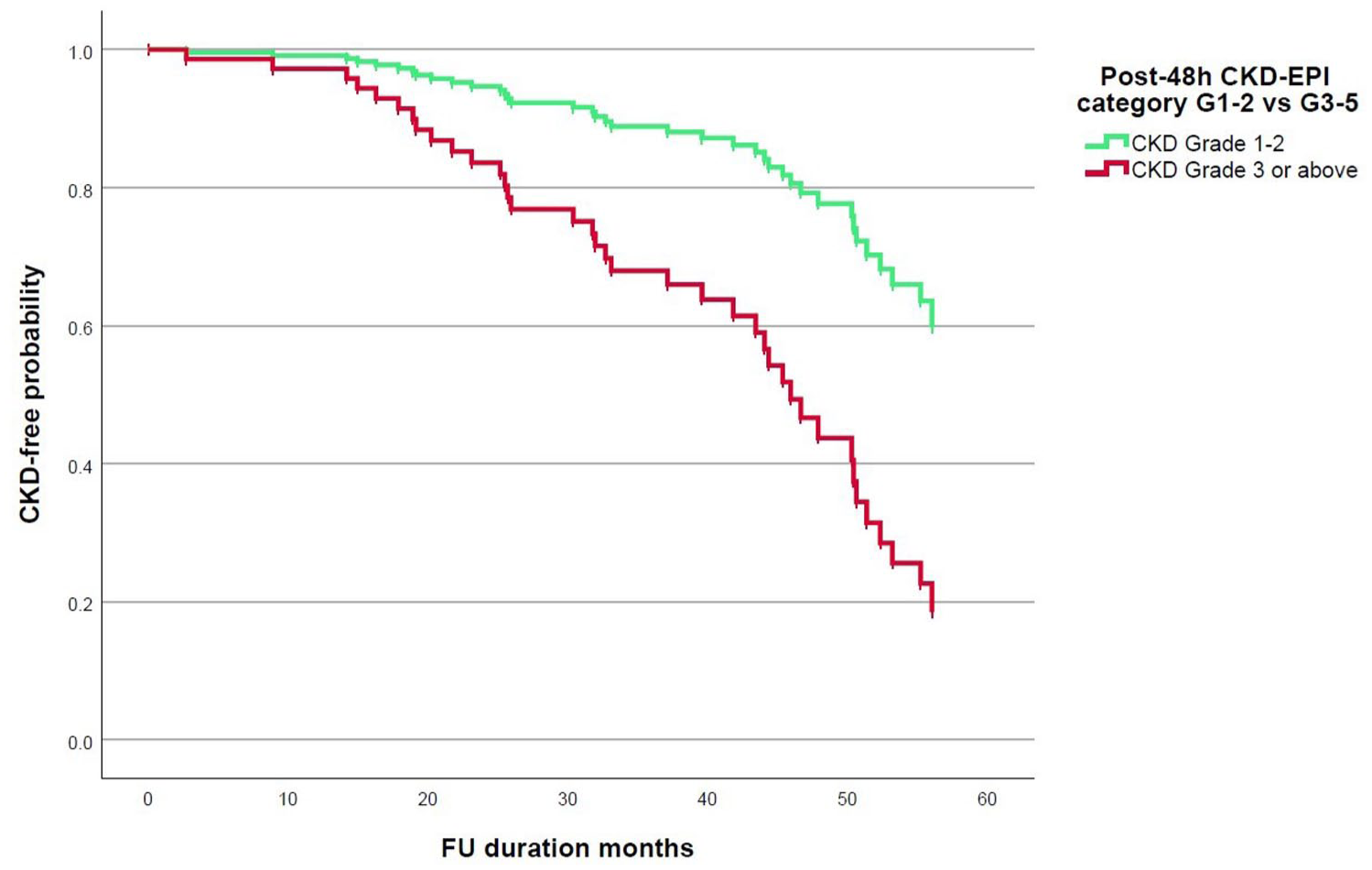
Cox regression showing 48-h CKD-EPI grade as a predictor of CKD-free survival at last follow-up.
Multivariable analysis
Based on the results of the VIF analysis, we identified variables that showed strong collinearity. These were age at surgery years correlating with preoperative eGFR mL/min/1.73 m2, BMI correlating with BW Kg, duration surgery min correlating with duration anesthesia min, intraoperative duration hypotension min correlating with intraoperative mean arterial pressure (MAP) < 65 for 15 min or more, 48h creatinine µmol correlating with 48h eGFR mL/min/1.73 m2, and finally sex correlated with both preoperative eGFR and BW independently. Therefore, we removed the following variables from the final multivariable analysis: Age at surgery, BMI, duration surgery min, intraoperative duration hypotension min, 48h creatinine µmol, and sex.
Preoperative risk factors: The multivariate analysis singled out the preoperative eGFR as the most significant risk factor for the development of CKD (Table 3).
Preoperative multivariable Cox regression.

The event is category CKD ⩾ G3a at last follow-up. Text in bold indicates significant risk factors.
AKI, acute kidney injury; BW, body weight; CKD, chronic kidney disease; CVA-D, cerebrovascular accident or peripheral arterial disease; DM, diabetes mellitus; Hb, hemoglobin; eGFR, estimated glomerular filtration rate; HR, hazard ratio; PMH, past medical history.
Perioperative risk factors: Multivariable analysis of perioperative variables included only 123 patients with complete anesthesia data (Table 4). The multivariable analysis indicated that lower intraoperative nadir MAP was independently associated with increased risk of CKD at last follow-up (HR 0.894 per mmHg increase, 95% CI 0.825–0.969; p = 0.006), indicating that deeper intraoperative hypotension confers higher long-term renal risk. In addition, in the multivariable analysis, higher eGFR at 48 h postoperatively was independently associated with a lower risk of CKD at last follow-up (HR 0.896 per 1 ml/min/1.73 m2 increase, 95% CI 0.833–0.964; p = 0.003; Table 4).
Perioperative multivariable Cox regression.

The event is category CKD ⩾ G3a at last follow-up. Text in bold indicates significant risk factors.
AKI, acute kidney injury; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration formula; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; HR, hazard ratio; LOS, length of stay; MAP, mean arterial pressure; PRBC, packed red blood corpuscles.
There is a positive linear relationship between preoperative eGFR and last follow-up eGFR (Figure 3).
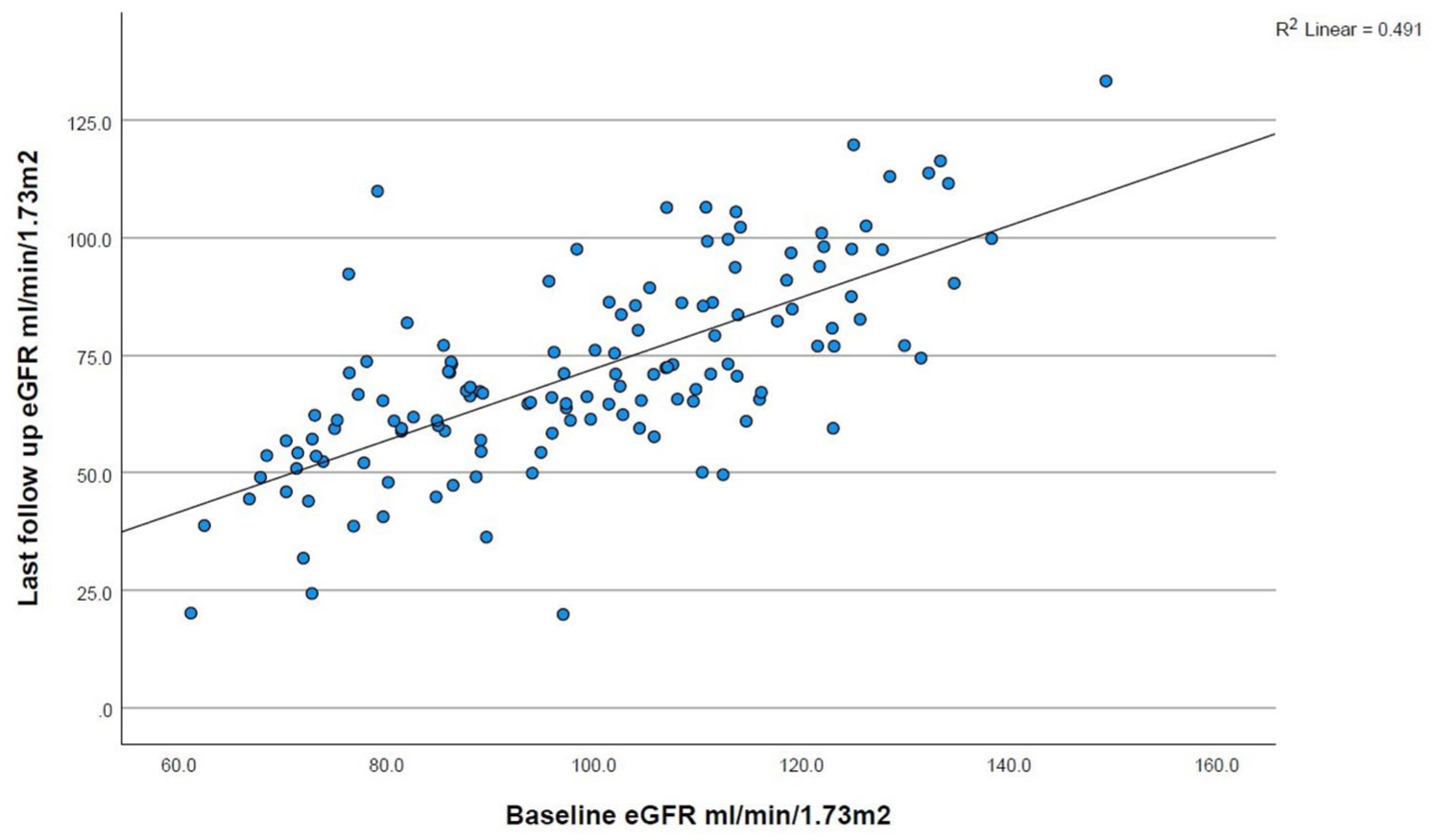
Scatter plot shows the relation between baseline and last follow-up eGFR.
A linear relationship exists between 48-hour eGFR and the last follow-up eGFR (Figure 4). Pathology, new risk factors, or metastasis were not predictors of CKD (Tables 2 and 5).
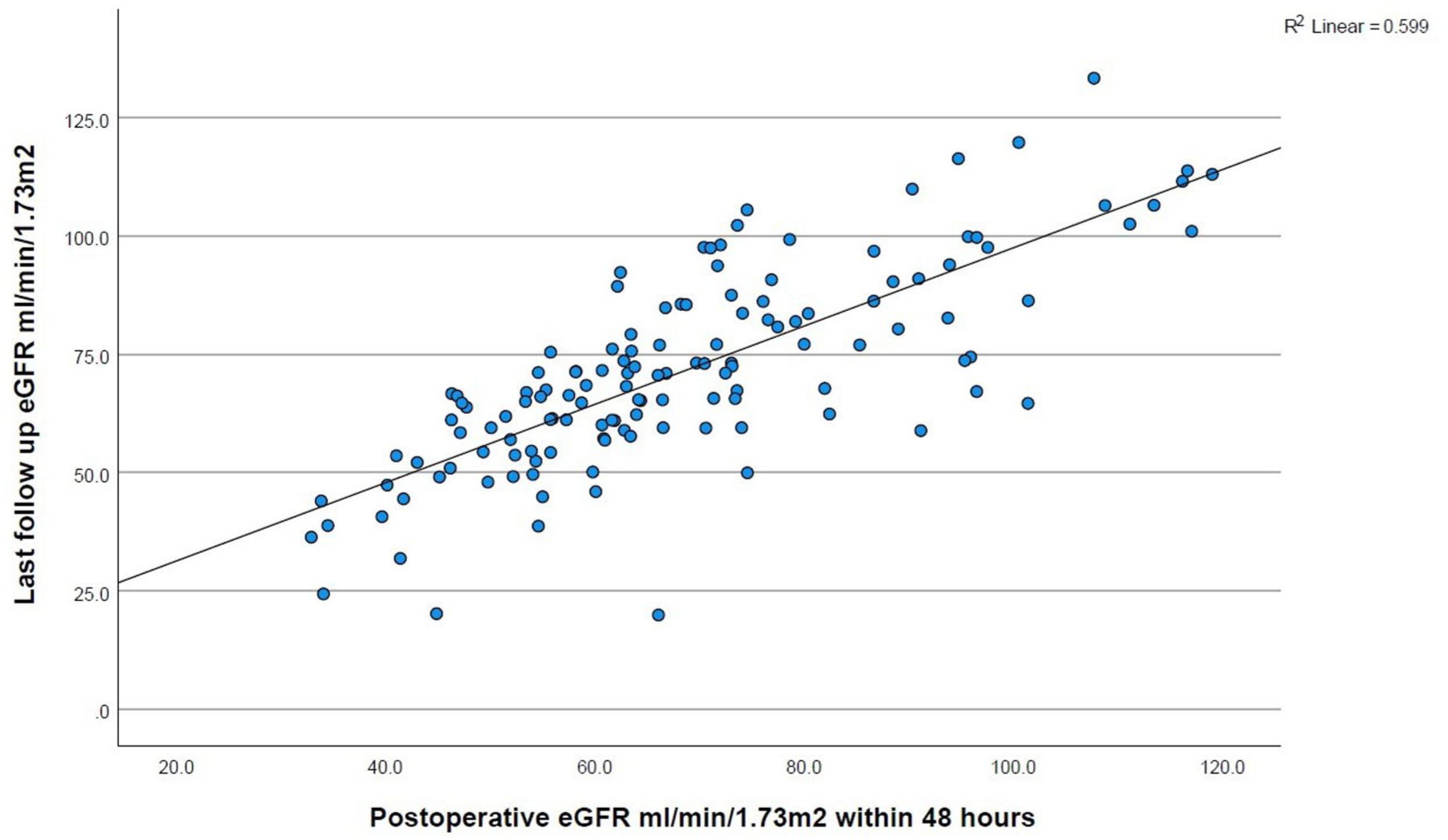
Scatter plot showing the relation between immediate postoperative and last follow-up eGFR.
Postoperative multivariable Cox regression.

The event is category CKD ⩾ G3a at last follow-up.
CKD, chronic kidney disease; DM, diabetes mellitus; HR, hazard ratio; HTN, hypertension; PVD, peripheral vascular disease.
Discussion
In this study, specific inclusion and exclusion criteria were established to eliminate factors that could complicate the interpretation of the impact of RN on renal function and had confounded previous studies.5,8,9,13–17 Patients who were excluded from the study included those with prior CKD, contralateral renal abnormalities, PN, and advanced RCC stages.
The inclusion criteria focused on patients without preoperative CKD, as determined by eGFR and the absence of diagnosed kidney disease.
According to the 2024 Kidney Disease: Improving Global Outcomes (KDIGO) guideline, the criteria for diagnosing and classifying CKD include the cause, decreased GFR, albuminuria, and any other marker of kidney damage. 18 Additionally, patients with structurally normal contralateral kidneys, as confirmed by CT imaging, were included in the study. Furthermore, none of the included patients showed any laboratory evidence of CKD, including albuminuria, preoperative urinary sediment abnormalities, electrolyte imbalances indicative of tubular disorders, or a history of renal transplantation.
We used the CKD-EPI formula to estimate the eGFR, known for its accuracy in evaluating kidney function across all ranges. This aligns with nephrology literature, which defines CKD with a cutoff value of 60 mL/min/1.73 m2, in contrast to the 45 mL/min/1.73 m2 cutoff suggested by some other surgical studies.13,21,22 Indeed, in other studies, a postoperative GFR cutoff of < 45 mL/min/1.73 m2 was associated with increased probabilities of adverse outcomes, including a 50% decline in GFR, the need for dialysis, and higher 5-year all-cause mortality.7,21,22 CKD stage 3a was considered clinically significant, given the higher hospitalization rate associated with eGFR values between 45 and 59 mL/min/1.73 m2. 6
It was found that 28.1% of patients with previously healthy contralateral kidneys developed stage 3 CKD after an average follow-up period of 33 months. The incidence of postoperative renal impairment observed in this study was lower than that reported in several other studies.
RN for small renal tumors was linked to the new onset of CKD in 20.1% of patients, 20.1% acute renal failure, and 27.7% chronic renal insufficiency. 23 The five-year rates were 18% for new onset CKD, 18% for acute renal failure, and 26% for chronic renal insufficiency. In one study that included only patients with normal preoperative renal function, the cumulative 5-year incidence of CKD following RN was 43.3%. 4
Similarly, in a study that included less than 1% preexisting CKD, 43.1% of patients undergoing RN developed new CKD Stage 3 one year after surgery. 24 In another study that included PN, the new onset CKD was 40%. 25 Finally, in patients with normal opposite kidneys and no preexisting CKD who underwent RN for renal tumors 4 cm or smaller, 66.7% developed new-onset CKD. 26 Notably, no cases of end-stage renal disease (ESRD) were observed among the patients. In contrast, other reports indicated a 5-year incidence of ESRD post-RCC diagnosis at 2.4%. 5 However, these results may be affected by the inclusion of patients with preexisting CKD, high tumor stage, and different surgical treatment modalities. 5
Renal functional decline after unilateral nephrectomy in our RCC cohort was comparable in magnitude to that reported after living donor nephrectomy. 27 In the Florence series of robotic living donor nephrectomy, median eGFR declined from 88 to 57.4 ml/min/1.73 m2 at 24 months (median ΔeGFR −25.3 ml/min/1.73 m2), while in our RCC cohort mean eGFR decreased from 99.2 to 71.5 mL/min/1.73 m2 at a mean follow-up of 33 months (mean ΔeGFR −27.7 mL/min/1.73 m2). The close similarity in absolute eGFR reduction across these distinct populations supports the biological plausibility of our findings and confirms that the observed impact of RN on long-term renal function is robust and not attributable to cohort-specific bias.
The findings from the univariable analysis align with those reported in previous studies. Several reports indicated that age, male gender, hypertension, preoperative eGFR, and early postoperative GFR decline were risk factors for developing CKD.5,8,14 These conclusions, however, were based on a subset analysis that included patients with preexisting CKD and PN. Some studies focused on patients with normal preoperative renal function, including our present study.4,12,14,23,28,29 Across all analyses, including the present study, preoperative eGFR emerged as an independent predictor of postoperative deterioration in renal function.4,12,14,28,29 The robustness of preoperative eGFR as a predictor of postoperative CKD suggests its significance across various study designs, endpoints, and statistical analysis methods. However, there was no consensus on the role of age, obesity, hypertension, diabetes, Charlson Comorbidity Index, and gender as independent preoperative risk factors for renal impairment after RN.4,12,14,28,29 None of these factors were identified as independent predictors of CKD in the present study. The lack of agreement in these studies on the role of these risk factors likely arises from the diverse study methodologies. In fact, one study showed that excluding preoperative comorbidity did not affect the value of a nomogram predicting postoperative renal impairment. 28
There is a lack of studies examining the impact of intraoperative factors related to anesthesia and hemodynamic changes on the development of CKD.10,11 In the univariable analysis, the only significant intraoperative risk factors for CKD were the duration of anesthesia and surgery (Tables 1 and 2). While factors such as hemorrhage, hypotension, and prolonged surgery may intuitively be associated with the development of CKD, none were found to be significant predictors of CKD in the univariable regression analysis. Other studies have reported similar findings.4,20 However, in multivariable analysis, after exclusion of colinear risk factors, lower intraoperative nadir MAP was independently associated with increased risk of CKD at last follow-up (Table 4). This finding suggests that the severity of intraoperative hypotension, as reflected by the nadir MAP, is a critical determinant of long-term renal outcomes after RN, likely by exceeding renal autoregulatory thresholds in the remaining kidney and predisposing to ischemic injury.
Some other studies have indicated that low MAP during surgery was not significantly linked to AKI. 11 However, it’s worth noting that the latter study had 21% of patients with preexisting CKD, which could have influenced the conclusions. 11
Postoperative AKI is a well-documented independent risk factor predicting CKD grade ⩾3, with 43.9% of patients developing AKI postnephrectomy and 8.9% progressing to CKD grade ⩾3.20 A significant 70.2% of these patients developed CKD.20 Additionally, another study reported that 65.1% of patients with preoperative normal eGFR developed AKI after RN or PN for RCC, but more commonly with RN. 14
In this study, unlike 48-h postoperative eGFR, AKI which affected 93.3% of patients did not significantly influence the development of CKD at the final follow-up. The lack of statistical significance for AKI as a risk factor may be attributed to the disproportionate number of patients who experienced AKI compared to those who did not.
Postoperative AKI was found to be linked with preoperative dehydration rather than intraoperative hypotension, which was not identified as a significant risk factor, in line with our research. 30 One possible reason is that tertiary care centers may adhere to high-quality anesthesia protocols that effectively manage patients during surgery. It is, therefore, not surprising that surgical procedures conducted in rural areas, the private sector, and low-volume centers were associated with a higher incidence of postoperative CKD. 25
The type of anesthesia may represent a potential risk factor contributing to postoperative CKD in this study. Studies have shown that propofol anesthesia was associated with a lower risk of AKI and CKD than volatile anesthesia, with no significant differences between sevoflurane and desflurane on renal function.10,31 In this study, all but three patients received maintenance sevoflurane volatile anesthesia, limiting the ability to make a meaningful comparison between anesthesia groups.
The findings of this study may support preoperative counseling regarding surgical options and the likelihood of postoperative CKD. The results also highlight a multifactorial approach to reducing the risk of CKD following RN. While univariable analysis identified several risk factors that were not independent predictors of CKD, these risk factors may be clinically significant and, most importantly, the only modifiable factors in many situations. Therefore, thorough preoperative assessment and optimization of renal function are crucial. Managing comorbidities like hypertension may help improve outcomes. Intraoperative strategies should focus on shortening the duration of anesthesia and surgery and maintaining stable hemodynamics during surgery.
Additionally, considering the choice of anesthesia, with a preference for intravenous methods when feasible, could potentially reduce renal impairment. Postoperative monitoring should be rigorous, especially within the first 48 h, as early declines in eGFR are critical indicators of future renal function. Implementing these recommendations can help reduce the incidence of CKD in patients undergoing RN.
The study has both strengths and weaknesses that need to be discussed. One strength lies in the thorough selection of patients, including only those with normal preoperative kidney function and normal contralateral kidneys. This reduces variables and makes the study more reliable. Additionally, the study used a precise method for estimating kidney function, providing more accurate results than other studies. The mean follow-up of 32.8 months and the applied multivariable analyses provide useful insight into renal functional outcomes after RN and allow identification of relevant risk factors for CKD. However, this follow-up duration may not fully capture very late renal functional decline, and longer-term studies extending beyond 5 years are warranted. However, the study has areas for improvement. A limitation of this study is the absence of a formal a priori sample size or power calculation, which is inherent to its retrospective design. Nevertheless, the cohort size and number of outcome events were sufficient to allow multivariable analysis and to identify statistically and clinically meaningful predictors of long-term CKD. The nonrandomized selection of surgical techniques may introduce bias, and the limited variety in anesthesia types makes it difficult to compare their effects on renal outcomes. The incomplete intraoperative data and the absence of a quantitative evaluation of proteinuria are also limitations that reduce the power of the analysis. CKD staging was based solely on eGFR, as albuminuria or proteinuria data were not routinely available in this retrospective cohort, and future prospective studies should incorporate systematic proteinuria assessment to fully align with KDIGO recommendations. We acknowledge the absence of detailed fluid balance analysis, as review of the available intraoperative records revealed that input and output volume data were inconsistently documented across cases. This limitation is an important consideration for future prospective research. Intraoperative nadir MAP was not uniformly documented across all cases, and analyses were therefore conducted using available-case data; this may have reduced statistical power, but in the absence of evidence for differential missingness and given the consistency of the observed effect, it is not expected to have materially altered the primary association. Although detailed hemodynamic variability was not analyzed, we focused on clinically relevant measures of intraoperative hypotension, nadir MAP and sustained hypotension, which capture the severity of perfusion compromise most relevant to renal injury.
Lastly, certain risk factors may have been underestimated, indicating the need for further investigation.
There is a concern of over-reliance on eGFR as a Proxy Variable rather than age where in the univariable analysis both show significant association with CKD. With collinearity analysis we identified that age and preoperative eGFR are strongly correlated. We chose to exclude age from the final multivariable analysis based on clinical and statistical evidence. Clinically, age is a distal, indirect determinant of renal function, whereas preoperative eGFR is a proximal and integrative physiological marker reflecting nephron mass, vascular health, and cumulative comorbidity burden. When collinearity exists, retaining the proximal biological variable is statistically and clinically preferable. When modeled separately, preoperative eGFR but not age was associated with CKD risk. Furthermore, when entered simultaneously, eGFR remained significant whereas age was still insignificant, indicating that the effect of age on postoperative CKD is largely mediated through baseline renal function.
Another concern is that systemic therapies for metastatic RCC, including immune checkpoint inhibitors and targeted agents, may be associated with renal adverse events. However, these effects are typically infrequent and are often reversible with treatment interruption or corticosteroids. Importantly, conventional cytotoxic chemotherapy, which is associated with cumulative nephrotoxicity, is not recommended for RCC due to lack of efficacy. In our cohort, metastatic disease was not significantly associated with CKD at last follow-up in univariable analysis, suggesting that metastatic status, and by extension, systemic therapy exposure, did not materially confound the primary renal outcome.
An additional limitation is the absence of oncological outcomes and the lack of a comparative analysis with PN, which could be addressed in future studies using a multiarm design.
In conclusion, while the study provides valuable insights, addressing these limitations in future research could enhance the robustness and applicability of the findings.
Conclusion
The study examines the impact of RN on kidney function in patients with normal kidney function and a healthy opposite kidney. It identifies risk factors and perioperative parameters contributing to the development of CKD, emphasizing the importance of careful patient selection and monitoring to reduce CKD risk after nephrectomy. Further research is needed to explore modifiable factors for improved patient outcomes.
Supplemental Material
sj-docx-1-tau-10.1177_17562872261429816 – Supplemental material for New-onset renal impairment following radical nephrectomy for renal cell carcinoma: a retrospective cohort study of clinical, intraoperative, and postoperative risk factors
Supplemental material, sj-docx-1-tau-10.1177_17562872261429816 for New-onset renal impairment following radical nephrectomy for renal cell carcinoma: a retrospective cohort study of clinical, intraoperative, and postoperative risk factors by Raouf M. Seyam, Alhasan M. Abduldaem, Omar N. Alfehaid, Abdulrhman M. Almaslamani, Fatimah S. Alghsham, Noora M. Alassaf, Nora M. Alghanam, Ahmed G Sayed, Aya I. Elnegali, Andrew M. Norris and Hassan M. Alzahrani in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
