Abstract
Primary mucinous adenocarcinoma (PMAC) of the urethra is an exceptionally rare subtype of primary urethral carcinoma, with limited published data to guide diagnosis or treatment. Most literature consists of isolated case reports, and the non-specific presentation often delays diagnosis. This report presents two further cases and a review of the literature with a view to expand current understanding. We report two cases of PMAC. The first involves a 67-year-old male with progressive lower urinary tract symptoms initially treated as benign prostatic hyperplasia. Histology following a transurethral resection of the prostate revealed invasive mucinous adenocarcinoma with enteric features. He underwent radical prostatectomy, chemotherapy and radiotherapy for metastatic disease. The second case is a 52-year-old female with chronic urinary symptoms and a urethral mass initially misattributed to being from gynaecological origin. However, a biopsy later confirmed mucinous adenocarcinoma of the urethra. She underwent chemotherapy and radiotherapy, with partial response, but later developed further metastatic spread and is now undergoing palliative chemotherapy. PMAC is aggressive, often presents with non-specific urinary symptoms and lacks standardised diagnostic or treatment protocols. These cases highlight the importance of early detection through biopsies, immunohistochemical analysis and a multidisciplinary approach. Broader awareness and further reporting of cases are essential to develop evidence-based diagnostic and treatment guidelines for this rare malignancy.
Introduction
Primary urethral carcinoma (PUC) is extremely rare, accounting for only 0.02% of all cancers. 1 According to Swartz et al., 2 in the early 2000s in the Unites States, the annual incidence rates for male and female PUC were 4.3 and 0.5/million, respectively. The rarest histological subtype is adenocarcinoma (AC) accounting for 10%–16% of all PUC. 3 In male patients, AC is the least common, accounting for 11.6% of all PUC. 2 In female patients, PUC accounts for less than 1% of all genitourinary tract malignancy; however, of those, AC is the commonest subtype accounting for 38%–47%. 4
Urethral AC arises in histological subtypes: clear cell adenocarcinoma, adenocarcinoma arising in urethral accessory glands and adenocarcinoma not otherwise specified (NOS), which encompasses mucinous, enteric, signet-ring and mixed subtypes. 5 This report’s focus is primarily on the subtype, primary urethral adenocarcinoma (PUAC), and within this subtype, the paper looks more closely at primary mucinous adenocarcinomas (PMAC) of the urethra. Within Case 1, the paper also touches briefly on the enteric components of PUACs. The PMAC subtype is characterised by an abundance of extracellular mucin that frequently dissects the stroma, with single or small clusters of tumour cells floating in mucin pools. 6 Recent literature has led to an improved understanding of this unique tumour and has begun to define the clinicopathological and immunohistological features; however, due to the rarity of cases, the extent of research remains limited.
This study reports two cases of PMAC of the urethra, whilst summarising a further 21 previously reported cases, to provide insight into this rare tumour and add value to the already, albeit small, pool of research.
Case presentations
The reporting of this study conforms to CARE guidelines (Supplemental Material). 7 All patient details have been de-identified.
Case 1
A 67-year-old male patient presented with severe voiding lower urinary tract symptoms (LUTS), including poor stream, hesitancy and occasional severe dysuria, thought to be due to benign prostate hyperplasia. Notably, the past medical history included exposure to asbestos. He underwent a transurethral resection of the prostate (TURP) in February 2018, and his symptoms resolved. Histology revealed a tubulovillous adenoma of the prostate. Post-TURP prostate-specific antigen (PSA) was undetectable, and computed tomography (CT) was normal.
A year later, he presented with a recurrence of the same symptoms and a PSA of 0.11 ng/mL. Flexible cystoscopy revealed a tight bulbar urethral stricture and a probable urethral lesion beyond the stricture point, possibly a recurrence of the villous adenoma. He underwent a second TURP in April 2019, and histology showed large areas of necrosis, with acute inflammation and large amounts of extracellular mucin (Figure 1(e) and (f)). In the preserved areas, the epithelial lining showed tubulo-villous architecture (Figure 1(a)), with underlying glands showing stratification of lining, moderate nuclear enlargement and scattered mitotic figures. Focally more complex cribriform architecture was seen, and in one focus, a few single cells were seen infiltrating the lamina propria. No normal urothelial lining of the prostatic urethra was seen. Very few benign prostatic glands were seen.
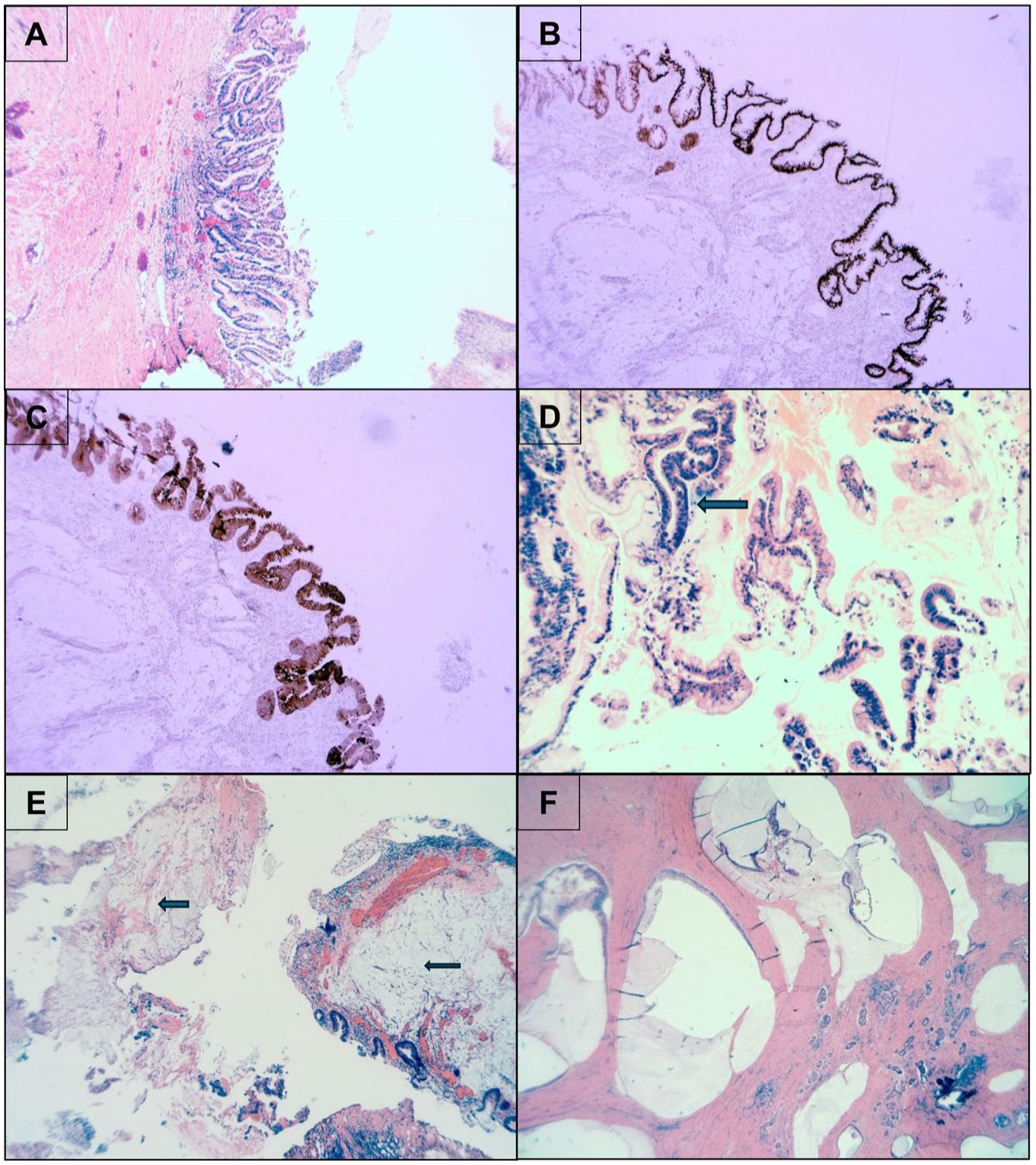
(a) Prostate TURP with tubulo-villous architecture H&E 4×. (b) Prostate TURP with few glands infiltrating stroma, CDX-2 positive – 4×. (c) Prostate TURP, CK7 positive – 4×. (d) Prostate TURP with adenocarcinoma in situ (darker areas – mitotic figure at arrow) H&E 10×. (e) Prostate with dysplastic glands and areas of extracellular mucin (arrows) H&E 4×. (f) Prostatectomy with tumour and extracellular mucin H&E 2×.
Immunostains showed positivity for CDX-2 (Figure 1(b)), CK7 (Figure 1(c)), CK20 with cytoplasmic (not nuclear) positivity for Beta-catenin. PSA, Prostatic Specific Acic Phosphatase (PSAP) (Figure 1(a)) and p63 were negative. Mib-1 showed a high proliferative index of 50–90% in different foci, with foci showing abundant intracellular mucin (Figure 1(e) and (f)), showing a low positivity of 1%. This was diagnosed as a villous adenoma arising presumably from the prostatic urethra, with foci of adenocarcinoma in situ (Figure 1(d)) and invasive adenocarcinoma of enteric and mucinous type, which is at least pT1 in the tissue.
Subsequent robotic radical prostatectomy in June 2019 (Figure 2) confirmed invasive urethral moderately differentiated adenocarcinoma, stage pT3a, with extracapsular spread and positive apical urethral and circumferential margins, but no lymph node involvement.

Prostatectomy with mucinous urethral adenocarcinoma, infiltrating stroma – H&E, 4×. Extracellular mucin is seen at the arrow.
A post-surgical CT showed small-volume mediastinal lymphadenopathy and pleural plaques. The case was reviewed in the urology multidisciplinary team (MDT) meeting. The asbestos-related changes and a small pulmonary nodule were considered stable, and, therefore, there was no radiological concern regarding metastatic disease.
In the subsequent 2 months, the patient underwent salvage radiotherapy to the prostate bed, at a dose of 66 Grays (Gy), over 8 weeks. The post-radiotherapy PSA was < 0.01. PSA remained low, and a year later, the patient had a PSA of 0.01. A repeat CT scan 3 years after radiotherapy showed an increase of 1.5 mm in the previously identified lung nodule. Positron emission tomography (PET) scan identified uptake in the left pelvic side wall, sigmoid mesentery, left external iliac nodes, peritoneal metastases, and small lung lesions too small to characterise on a PET scan. He underwent chemotherapy, and by mid-2023, he had completed eight cycles of capecitabine (1000 mg/m2) and oxaliplatin (130 mg/m2)
A CT scan in August 2024 showed an increase in size of the previously noted left upper lobe (LUL) nodule to 11 mm. In November 2024, the patient had a short course of radiotherapy, at a dose of 20 (Gy) of radiation over five sessions. A post-treatment CT scan in early 2025 showed a favourable response with a reduction in the LUL pulmonary nodule, measuring 5 mm, and the rest of the scan findings remained stable. The patient is currently under surveillance. Their surveillance CT in May 2025 showed stable appearances.
Case 2
A 52-year-old female patient presented with a history of difficulty voiding and recurrent urinary tract infections. She had been seen previously by the gynaecology team for post-coital bleeding 18 months prior. Initially, it was thought she had perimenopausal urethral narrowing. Transvaginal ultrasound was normal, and she had undergone various biopsies of her gynaecological tract, all of which were benign. Following an MDT discussion, she was referred for an urgent urology opinion.
A catheter was placed, and an urgent cystoscopy and urethral dilation were arranged. Cystoscopy showed no abnormalities in the urethra or the bladder, apart from trabeculation of the bladder walls. The catheter was removed, and her care continued under the urology nurse specialist. She was voiding well, and her post-void residual improved.
Two months post cystoscopy, difficulty voiding returned, and intermittent self-catheterisation was suggested, which she was unable to do. She chose to monitor her voiding and declined another cystoscopy. A normal CT urogram and repeat transvaginal ultrasound scan were undertaken. A month later, she went into urinary retention, and a catheter was unable to be inserted urethrally, necessitating a suprapubic catheter (SPC). The patient underwent an urgent cystoscopy and examination under anaesthetic with a gynaecologist present. A 1 cm mass adjacent to the urethra in the vaginal wall was detected, and a biopsy was taken.
Vaginal wall biopsy showed fibrous tissue infiltrated by atypical glands with irregular and angulated shape, lined by cells showing cytological atypia and suggestion of mucinous cytoplasm (Figure 3(a)). The glands were positive for CK7 (Figure 3(b)), CDX-2 (focally, Figure 3(c)), p16 and p53 (mutated). GATA-3 (Figure 3(d)), PAX-8 (Figure 3(e)), CK20 and ER were negative. Mucin stains showed positivity within the cytoplasm of the tumour cells (Figure 3(f)), in keeping with a mucinous adenocarcinoma. On review, this was diagnosed as a well-differentiated mucinous adenocarcinoma of the urethra, rather than a gynaecological primary. In situ hybridisation for HPV was also negative. Urgent magnetic resonance imaging (MRI) of the pelvis showed an altered signal in the right ischial tuberosity and a possible 4 mm urethral diverticulum. The 1 cm mass felt in the theatre, however, was not visible on MRI. PET confirmed activity in the right ischial tuberosity, L4 vertebra and para-aortic nodes.

(a) Vaginal wall biopsy, glands lined by atypical cells with mitosis – H&E, 20×. (b) Vaginal wall CK7 positive, 10×. (c) Vaginal wall CDX-2 positive, 10×. (d) Vaginal wall GATA3 negative, 10×. (e) Vaginal wall PAX8 negative, 10×. (f) Vaginal wall mucin in glandular lumina, Alcian blue, 10×.
In June 2023, the patient underwent six cycles of cisplatin (75 mg/m2) and capecitabine (1000 mg/m2) chemotherapy, and a post-treatment CT showed a favourable response in the primary tumour and pre-existing bone metastases; however, a new lesion at L2 was identified on bone scan. In August 2023, she received radiotherapy, 8 Gy single fraction, to the lumbar spine and right pelvis.
Post-radiotherapy, a bone scan identified a new lesion in the left acetabulum. After an MDT discussion, she received chemotherapy: capecitabine (1000 mg/m2), oxaliplatin (130 mg/m2) and zometa (4 mg), completing eight cycles by September 2024.
The patient had ongoing progression of disease and worsening urinary symptoms. She developed numerous acute kidney injuries and had bilateral hydronephrosis, leading to bilateral nephrostomies in November 2024. MRI pelvis post-nephrostomies showed focal disease relapse in the urethra and an extension into mid-vagina, with progressive bone metastases. After further MDT discussions, the patient started palliative chemotherapy: gemcitabine (1000 mg/m2), carboplatin (4 mg), zoledronic acid (4 mg) with granulocyte colony-stimulating factor support (300 mcg), in January 2025. She completed a total of six cycles. In March 2025, she underwent a robotic cutaneous ureterostomy. The chemotherapy was on hold to allow recovery from surgery, but she has since restarted it in June 2025. At the time of writing, she is awaiting a CT scan to assess response.
Discussion
PUC is an uncommon cancer, with PMAC being even rarer. It was first described in literature in 1995. 6 PUC in general is more common in men, with a male-to-female ratio of 3:1. 8 However, PMAC of the urethra affects women more commonly than men, making up nearly 50% of all PUC in women. 4 The rate of incidence increases with age, with the majority in women over 65. 8 An analysis of 23 published case reports on PMAC of the urethra is shown in Table 1. The reports detailed in this paper are included. The demographic shows a 10:13 male-to-female ratio. Indicating from this study, a much more even rate of cases between genders. The median age of the patients at presentation was 58 in women and 59 for men.
Analysis of case reports on primary mucinous adenocarcinoma of the urethra.

Systematic reviews were not included to avoid duplication of data.
LUTS, lower urinary tract symptoms; UTIs, urinary tract infections.
PMAC does not have known risk factors, unlike other PUC subtypes; transitional cell carcinoma (TCC) and squamous cell carcinoma (SCC). However, a subset of mucinous adenocarcinomas of the urethra may be associated with prior prostate brachytherapy. 25 The known risk factors for TCC and SCC included recurrent urinary tract infections (UTIs), prior radiotherapy, urethral diverticulum and sexually transmitted diseases. It is suggested that there may be a congenital origin in clear cell adenocarcinoma. 26
Poor prognostic factors for the PUC, and thus seemingly encompassing the PMAC, include age >65 years, higher stage and grade, lymph node involvement, distant metastasis, large tumour size, proximal tumour location, specific histological types and concomitant bladder cancer. Recurrence sites can be urethral or non-urethral. 27
Emerging molecular studies of primary urethral adenocarcinoma further characterise the pathogenesis. A recent study of eight patients demonstrated all tumours were diffusely positive for CK20, CDX2 and AMACR, and lacked nuclear beta-catenin expression, with the majority expressing CK7. 28 KRAS mutations (p.Gly12Val, p.Gly13Asp) were identified in both mucinous and NOS subtypes, including metastatic disease, while an EGFR p.Ser784Phe mutation was observed in one enteric tumour. The study concluded that urethral adenocarcinoma harbours KRAS and EGFR mutations independent of prior radiotherapy, and that radiation-independent tumours may be associated with a worse prognosis than brachytherapy-associated tumours.
Case 1 may represent a similar adenoma-carcinoma sequence, well-documented in colorectal tumours. Whereby the villous adenoma can be considered premalignant and either through malignant transformation or when coexistent with adenocarcinoma have a greater risk of recurrence and metastasis as seen in this case. 29 This case supports the current literature advising that more aggressive treatment with adjuvant radiotherapy and/or chemotherapy should be considered in cases of villous adenoma with concurrent adenocarcinoma.
These two cases are good examples of how PMAC presents with non-specific signs and symptoms. Both presented with voiding LUTS, whilst the female patient also had a history of recurrent UTIs. Table 1 gives further details of symptoms at presentation. It highlights the high likelihood of nonspecific symptoms at presentation; pain, irritation and LUTS being the most common, with at least one occurring in 57% of the 23 cases. Due to the lack of diagnostic signs and symptoms, there should be a low threshold for early assessment. From these cases and the literature, early evaluation of symptoms with a physical examination, urine cytology, cystoscopy and biopsy is crucial for timely diagnosis of these tumours. 9 Imaging used to assess the tumour extent and to recognise metastasis in the case reports presented here included MRI, CT of the thorax and abdomen, CT urogram and PET scans. The literature states MRI of the pelvis should be the radiological investigation of choice for assessing local extent and should be used in conjunction with CT thorax and abdomen for staging.
Case 2 demonstrates that when urethral pathology is suspected in women with persistent voiding dysfunction, early consideration of pelvic MRI or transperineal ultrasound, and targeted biopsy of the urethral wall when feasible, may reduce diagnostic delay and consequently severe sequelae.
A study of female PUC showed 46% of cases had advanced disease at the time of diagnosis. 30 This is likely because PUC often presents with non-specific symptoms, coupled with its aggressive nature. PMAC has also been shown to be the most severe subtype, often with lymph node metastasis and with the lowest 5-year survival rate of 31%, 30 compared to squamous cell carcinoma (SCC) survival at 64% and urothelial cell carcinoma (UCC) at 61%. 30
PMAC currently carries a very poor prognosis, and as Table 1 shows, due to the aggressive nature, it often leads to metastatic spread both locally, in 52% of cases, and distally, in 43%. As outlined in Table 1, the treatment for PMAC can involve a combination of surgery and adjunctive radiotherapy and chemotherapy, particularly in those with locally advanced disease. The literature suggests that surgical options are considered irrespective of histology type. 21 These range from local excision, radical urethrectomy or prostatectomy, to anterior pelvic exenteration. This variation in treatment and follow-up highlighted in Table 1 is likely due to a lack of standardised protocols and follow-up strategies.
Conclusion
Primary mucinous adenocarcinoma of the urethra is an extremely rare and aggressive malignancy, with a poor prognosis due to delayed diagnosis as well as its propensity for metastasis and recurrence. This report highlights the importance of early detection and the need for a low threshold for investigation in patients presenting with non-specific lower urinary tract symptoms. While treatment strategies vary, a multimodal approach including surgery, chemotherapy and radiotherapy appears to be the most effective, though standardised guidelines remain lacking. Further research is necessary to generate diagnostic protocols, establish more definitive treatment guidelines and, in turn, improve patient outcomes.
Supplemental Material
sj-docx-1-tau-10.1177_17562872261424118 – Supplemental material for Primary mucinous adenocarcinoma of the urethra: a diagnostic conundrum – two case reports and literature review
Supplemental material, sj-docx-1-tau-10.1177_17562872261424118 for Primary mucinous adenocarcinoma of the urethra: a diagnostic conundrum – two case reports and literature review by Elspeth Jamieson, Amit Goel, Nicola Lancaster, Amanda Clarke and Sanjeev Madaan in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
