Abstract
Primary female urethral carcinomas are uncommon and have a low morbidity rate. Most of these patients have advanced illness with high invasion and a poor prognosis. There is no standard treatment, and multimodal therapy is recommended. The use of radiotherapy and chemotherapy were mostly reported in previous studies on advanced female urethral squamous cell carcinoma. We report that chemotherapy combined with a programmed death-1 (PD-1) inhibitor was effective in treating metastatic female urethral squamous cell carcinoma. During four cycles, we used systemic chemotherapy of albumin-paclitaxel + carboplatin in combination with a PD-1 inhibitor (toripalimab 240 mg) every 3 weeks, and a complete response was achieved. We performed a genetic test on the patient who had a tumor mutation burden of 5.7 mutations/Mb, tumor proportion score of 20%, and combined positive score of 20% (22C3). No recurrence or distant metastasis was found after 20 months of follow-up. In conclusion, in patients with positive PD-1 ligand 1 expression in primary female urethral squamous cell carcinoma, chemotherapy combined with PD-1 inhibitors may be effective. Larger sample studies are required to determine PD-1 ligand 1 expression and the curative effect of PD-1 inhibitors, as well as their effect on survival.
Keywords
Introduction
Primary female urethral carcinoma is extremely uncommon, accounting for only 0.02% of all systemic cancers in women. The majority of patients with this condition have no evident symptoms in the early stages and are in the advanced stages when diagnosed, with considerable invasiveness and a poor prognosis.1–6 The incidence of primary female urethral carcinoma is 1.5/100,000 people in the United States, and it rises with age. The incidence of primary female urethral carcinoma is 0.6/100,000 people in Europe.7,8 In China, there are no epidemiological data on this condition. Urothelial carcinoma, adenocarcinoma, and squamous carcinoma are the most common findings, accounting for 45%, 29%, and 19%, respectively. 9
In an early series, treatment of primary female urethral carcinoma was mostly monotherapy with either surgery or radiation,10,11 whereas there is no standard treatment for advanced disease. To improve survival rates, multimodal therapy, including neoadjuvant chemotherapy, surgery, radiotherapy, adjuvant chemoradiotherapy, and other treatments, is recommended.10–14 Immunotherapy has been shown to be effective in a variety of tumors in recent years, but it is rarely used in primary female urethral carcinoma. To the best of our knowledge, we report for the first time that chemotherapy combined with a programmed death-1 (PD-1) inhibitor was effective in the treatment of metastatic female urethral squamous cell carcinoma.
Case presentation
Our patient was a 53-year-old Chinese woman who had a urethral orifice tumor discovered without obvious symptoms for more than 3 months, with no active medical treatment. She then had difficulties urinating, which worsened her condition. The tumor steadily grew and caused urinary retention, and a catheter was implanted in a nearby hospital. She did not have any specific therapies performed before visiting our center. Her physical health was good. She had a history of mild hearing loss, but the exact reason is unknown. She works as a teacher and has a child. She denied ever smoking or drinking alcohol. She denied any sickness or cancer in her family background.
A physical examination showed the following. In the lithotomy position, there was a dark red neoplasm protruding from the external urethral orifice and pointing to the 6 o'clock direction. The neoplasm was approximately 2.0 × 2.0 cm in size and grew to the urethra before extending inwards. The neoplasm's surface was smooth. Her neoplasm caused pain when pressed, but there was no visible bleeding when it was touched. There was no palpable inguinal lymph node. A preoperative laboratory examination showed that her blood chemistry results are within normal limits.
Preoperative imaging examinations showed the following. The results of chest and abdominal computed tomography scans were normal. Pelvic magnetic resonance imaging (MRI) showed a 4.0 × 3.1 × 3.6-cm lower urethral mass with a slightly low signal on T1-weighted imaging (T1WI), a medium and high signal on T2-weighted imaging/fat suppression, and a high signal on diffusion-weighted imaging (DWI). An enhanced scan showed obvious uneven enhancement, and the mass partially protruded from the external urethral opening and invaded the vaginal anterior wall. There was a 3.2 × 2.8-cm mass on the right side of the uterus, with a high possibility of uterine fibroids (Figure 1). The pathology of the mass was intermediate differentiation squamous cell carcinoma, P16 (+), CK7 (−), uroplakin (−), P63 (+), P53(−), and Ki-67 (90% +).

Preoperative magnetic resonance imaging (MRI). (a) T1-weighted imaging (T1WI) in preoperative MRI; (b) T1W2 in preoperative MRI; (c) diffusion-weighted imaging in preoperative MRI; and (d) T1WI in preoperative MRI. MRI shows a 4.0 × 3.1 × 3.6-cm lower urethral mass.
She chose to have a relatively aggressive treatment plan, which involved a laparoscopic cystectomy with urinary diversion using an ileal conduit and pelvic lymph node dissection. The surgery went well.
Postoperative pathology showed intermediate differentiation of squamous cell carcinoma with high-grade intraepithelial neoplasia, with a maximum diameter of approximately 3.5 cm. The mass invaded the whole layer of the vagina and urethra, and involved the trigone mucosa of the bladder. There was no involvement of the cervix, uterine body, or fundus. There was also no vascular tumor thrombus or nerve invasion, and no cancer at the anterior margin, left ureteral margin, or right ureteral margin. Metastatic carcinoma was observed in the lymph nodes (2/33) without involvement of outer capsule tissue. In detail, the following was observed in the lymph nodes: left superficial inguinal lymph node, 1/10; right superficial inguinal lymph node, 1/8; right deep inguinal lymph node, 0/1; right pelvic lymph node, 0/6; and left pelvic lymph node, 0/8 (Figure 2).

Postoperative pathological examination showing a low-power view of a tumor in the urethra. (a) Hematoxylin and eosin staining at a magnification of ×100); and (b) hematoxylin and eosin staining at a magnification of ×400.
A pelvic MRI examination was performed at 2 months postoperatively After abdominal and pelvic surgery, no abnormal soft tissue mass was observed in the urethra or genito-urinary area, but multiple, zonal, nodular, abnormal signal areas were observed in the running area of the right iliac vessel, with a maximum cross-section of 3.7 × 2.8 cm. T1WI showed a slightly higher signal, there was no clear signal reduction in the reverse position, T2WI/fat suppression had a slightly high signal, and DWI did not have limited diffusion. An enhanced scan showed marginal enhancement, peripheral growth, a regular margin, and lymph node metastasis. The accumulation of postoperative fluid in the left iliac vascular area and the right inguinal area was altered compared with preoperatively (Figure 3).

Magnetic resonance imaging (MRI) before and after chemotherapy combined with immunotherapy. (a) T1-weighted imaging (T1WI) at pre-treatment MRI; (b) T1WI at post-treatment MRI; (c) T2WI at pre-treatment MRI; and (d) T2WI at post-treatment MRI. MRI shows a 3.7 × 2.8-cm in the running area of the right iliac vessel.
We performed a genetic test on the patient. She showed a tumor mutation burden (TMB) of 5.7 mutations/Mb, a tumor proportion score of 20%, and a combined positive score of 20% (22C3) (Figure 4). Using immunohistochemistry, we observed programmed death-1 ligand 1 (PD-L1) protein expression (Figure 5). We decided that our patient may be suitable for PD-1 inhibitor treatment. During four cycles, we used systemic chemotherapy of albumin-paclitaxel 300 mg (175 mg/m2) + carboplatin 400 mg (area under the curve = 4) in combination with a PD-1 inhibitor (toripalimab 240 mg) every 3 weeks. A complete response was observed. The patient had a grade 2 liver function injury that improved after symptomatic treatment with no other side effects. Her quality of life greatly improved after these treatments. The Short Form-36 questionnaire showed that she had favorable total physical health (actual score, 62.4; percentage scale, 75.9) and total mental health (actual score, 57.5; percentage scale, 76.0) when evaluated at 4 months. She was very satisfied with her treatment, and no recurrence or distant metastasis was found after 20 months of follow-up.
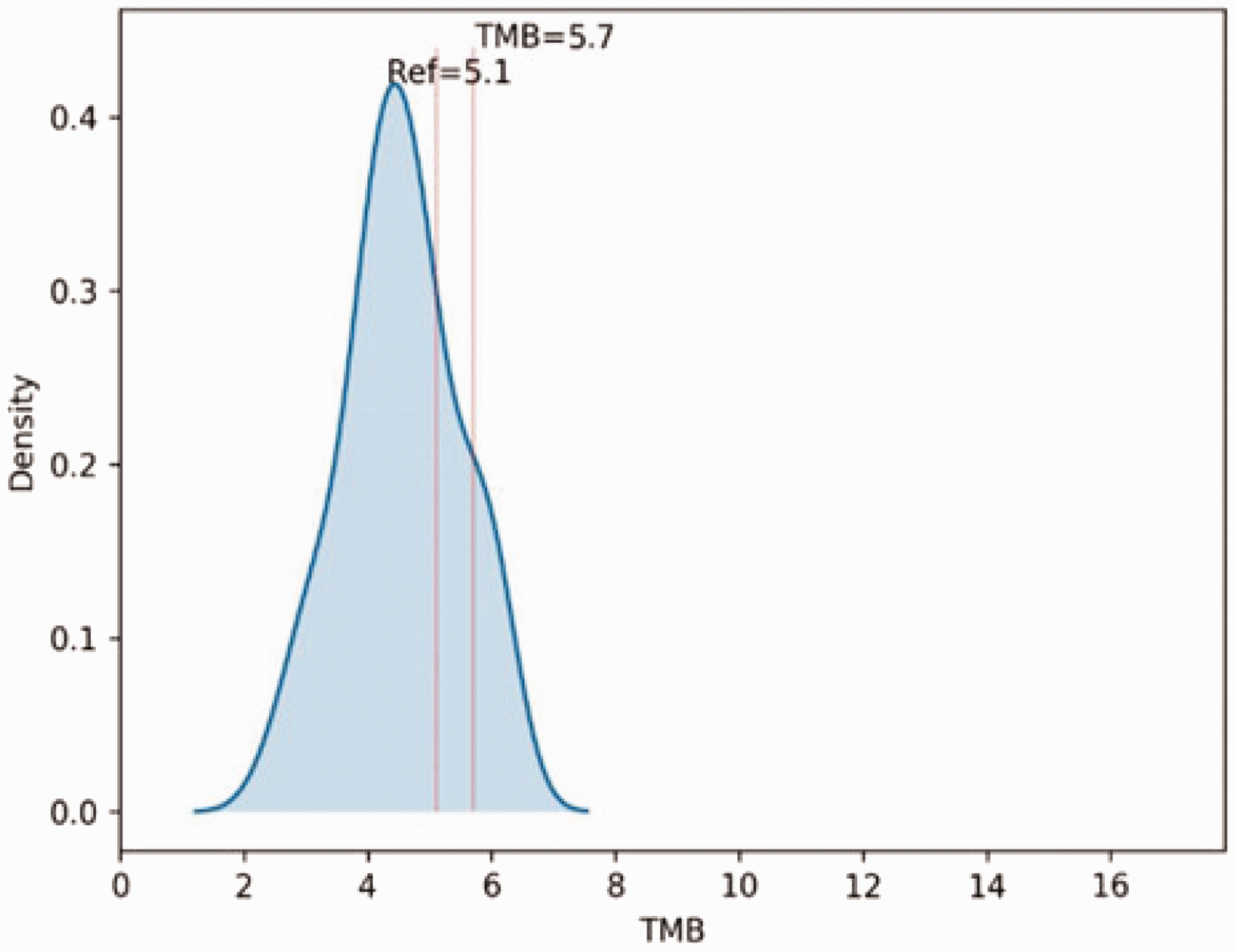
TMB schematic. The TMB was 5.7 mutations/Mb.

Immunohistochemistry of programmed death-1 ligand 1 protein expression levels. (a) Hematoxylin and eosin staining (magnification, ×400); and (b) programmed death-1 ligand 1 expression (magnification, ×400).
The reporting of this study conforms to the CARE guidelines. 15
Discussion
The most common causes of primary female urethral cancer include urethral stricture, urethral diverticulum, urinary tract infection, and human papillomavirus infection.6,16 The majority of these patients are detected in the middle and late stages, and have an aggressive natural history. The 5-year survival rate of primary female urethral cancer ranges from 32% to 54%.6,8,16–18 In advanced disease, multimodal therapy is advised, but there is no standard treatment and the application rate is low. As a result, optimizing treatment of advanced disease has been the focus of study.
A large international, multicenter cohort trial (124 patients, 38 women) showed that patients with locally advanced cancer who were treated with neoadjuvant chemotherapy who received surgery with or without adjuvant chemotherapy had higher recurrence-free and overall survival rates. 12 Furthermore, Peyton et al. demonstrated that multimodal therapy improved survival in 39 patients with primary female urethral cancer (5 patients had squamous cell carcinoma). 13 However, other studies have demonstrated that surgery coupled with radiation is more beneficial in patients with urothelial carcinoma and locally advanced or regional lymph node metastases, but not in those with squamous cell carcinoma.14,19
As a result, the standard treatment for primary female urethral squamous cell carcinoma remains unknown. Although multimodal therapy is desirable, its total application rate is low. From 1984 to 2014, a single-center study at MD Anderson Hospital showed that only 27% (33 women) of 106 patients with primary urethral carcinoma received combined treatment after surgery. 6 The remaining women received chemotherapy (14%), chemoradiotherapy (9.4%), or radiotherapy alone (3.8%). Another multicenter study showed that only 31% (45 women) of 154 patients received perioperative therapy. 16
The application rate of multimodal therapy has grown in recent years as a result of continual reports of multimodal therapy. Mano et al. 20 studied 165 patients with primary urethral carcinoma from 1956 to 2017, and 91 of them were women and 74 were men. A total of 72% of patients had invasion of surrounding tissues or organs, and 24% had metastases to local-regional lymph nodes. The proportions of monotherapy (surgery only or radiation only), dual therapy (surgery and radiation, surgery and chemotherapy, or chemotherapy and radiation), and triple therapy (surgery, radiation, and chemotherapy) were 57%, 21%, and 10%, respectively. Monotherapy use decreased with time, whereas dual therapy rates remained stable, and triple therapy rates increased. Furthermore, after controlling for other factors, patients who received monotherapy had a poorer recurrence-free survival than those who had dual or triple therapy. After controlling for the pathological stage, histology, sex, and year of treatment, patients who received monotherapy had a poorer recurrence-free survival than patients who had dual or triple therapy (P = 0.017), and all-cause mortality was greater. 20
The use of multimodal therapy, comprising surgery, radiation, and chemotherapy is recommended by the European Association of Urology guidelines and the National Comprehensive Cancer Network guidelines for patients with primary urethral cancer.11,12 However, the particular scheme, application sequence, and application timing remain unclear. A search of the literature showed that there are few data on the detection of PD-L1 or the use of PD-L1 inhibitors in primary urethral cancer.21,22
Antigen-presenting cells, such as human monocytes, as well as activated human and murine dendritic cells, express PD-L1. 23 PD-1 inhibitors mainly act on PD-1 inhibitor molecules on the T cell membrane. These inhibitors block the binding of PD-L1 and PD-L2, promote specific T cell proliferation and cytokine release, and thus enhance anti-tumor activity. PD-L1 inhibitors are effective in a variety of tumors, including head and neck squamous cell carcinoma and esophageal squamous cell carcinoma.24–25 We performed a genetic test on our patient who had a TMB of 5.7 mutations/Mb, tumor proportion score of 20%, and combined positive score of 20% (22C3). Findings in previous studies on TMB, PD-L1, and other biomarkers, 26 suggested that PD-1 inhibitor treatment may be effective in our case. Therefore, we used systemic chemotherapy of albumin-paclitaxel + carboplatin combined with a PD-1 inhibitor for four cycles. The results were as expected and good, and a complete response was achieved. The patient had a grade 2 liver function injury that improved after symptomatic treatment with no other side effects. There was no recurrence or distant metastasis after 20 months of follow-up. Our findings suggest that PD-1 inhibitors are effective in primary female urethral squamous cell carcinoma with tolerable toxicity, but this possibility needs to be confirmed by larger sample studies.
Conclusions
Our findings showed positive expression of PD-L1 in primary female urethral squamous cell carcinoma, and chemotherapy combined with a PD-1 inhibitor showed good efficacy. Large-sample studies are required in the future to determine PD-L1 expression in primary female urethral squamous cell carcinoma, as well as the efficacy and effect of PD-1 inhibitors on survival.
Footnotes
Acknowledgement
The authors would like to thank the patient for her permission to share her case information in this report.
Author contributions
All authors participated in designing various parts of the study and in discussion and interpretation of the results. HSB and HZS wrote and revised the manuscript. FW and HFH participated in writing the part of imaging diagnosis in the manuscript and helped revise the manuscript. HZS provided the concept for the study and revised the manuscript. All authors read and approved the final manuscript.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request (e-mail:
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement and consent to participate
Approval for this study was granted by the Ethics Committee of the National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China. Written informed consent was obtained from patient before administering treatment. The patient also provided verbal informed consent for her images and other clinical information to be published.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
