Abstract
Digital twins, dynamic, patient-specific virtual replicas of physical systems are gaining momentum across various medical fields. Their capacity to simulate and predict biological behavior offers unprecedented opportunities for personalized care. While their utility has been demonstrated in oncology, their application in urology remains nascent, and there is still a significant gap in literature findings. This review explores the theoretical framework, current evidence, and future potential of digital twins technology in managing urological conditions such as urolithiasis, benign prostatic hyperplasia (BPH), and nonmuscle invasive bladder cancer. A narrative literature review, guided by SANRA criteria, was conducted using PubMed and Scopus databases to identify relevant studies on the application of digital twins in healthcare, with a focus on urology and related fields. Additional sources from AI, imaging, and computational modelling were incorporated to support technological and theoretical concepts. Real-world use cases and clinical scenarios were constructed to illustrate the conceptual application of digital twins in endourology. Although limited in number, preliminary studies have shown promising results in uro-oncology, particularly for prostate and renal cancers. No models have yet been developed for benign urological diseases. Hypothetical applications in endourology include personalized procedural planning, dynamic follow-up, prediction of obstruction or recurrence, and real-time intraoperative guidance. Integration of imaging, functional data, and AI algorithms could create continuously adaptive simulations to support decision-making and improve treatment outcomes. However, barriers such as data complexity, cost, lack of validation frameworks, and clinician trust persist. Digital twins hold transformative potential for the future of urology, promising to personalize care across diagnosis, treatment, and surveillance. While the path to clinical integration is challenged by technical, ethical, and infrastructural hurdles, collaborative efforts and real-world validation studies are critical to realizing their full potential in clinical practice.
Plain language summary
Digital twins consist in a virtual computer model of a real patient, created using medical scans, test results, and clinical information. It can be updated as new data become available. In urology, digital twins could help doctors plan operations, predict whether kidney stones will return, estimate how prostate treatments will work, or determine which bladder cancer patients need more frequent monitoring. Although digital twins are increasingly used in engineering and some medical fields, they are still new in urology and require further research.
Introduction
Digital twins refer to virtual, continuously updated computational representations of a physical system, organ, or patient. Originating from aerospace engineering, the concept was formalized by NASA in 2010. 1 Since then, important progresses have been made in the healthcare system, that was finally ready to incorporate the same subject in the field of personalized medicine. By creating a digital replica with patient-like characteristics, physicians are now able to perform tests on the model before actually moving to the physical person. 2 This process translates in the ability to test drugs or protocols, or even surgical procedures, in a replicable and risk-free settings, that however goes beyond classical simulation: the digital twins is in fact adjustable and can mimic patient-specific features.3,4
Surprisingly, the application of digital twins has found fertile field in many specialties, but urology stays behind. Only few preliminary studies have been performed on the subject,5,6 and only on uro-oncological conditions, in particular prostate and renal cancer. Despite the massive implement in technological advancement seen in endourology, and the growing interest towards artificial intelligence (AI) integration into daily practice, no model of digital twins has been yet ideated.
The possible creation of digital twins model for urological conditions has many applications. 7 Several models have been proposed for oncological diseases, urological and nonurological, with obvious theoretical role on disease identification, staging, treatment planning and prediction of response and progression. However, a different discussion must be made for benign conditions such as urolithiasis and benign prostatic hyperplasia (BPH). This review aims to (1) summarize the theoretical and technological foundations of digital twins, (2) critically evaluate current evidence in urology, and (3) propose clinically meaningful conceptual applications for future development.
Methods
This narrative review followed the SANRA (Scale for the Assessment of Narrative Review Articles) guidelines. 8 A structured literature search was conducted in PubMed and Scopus covering January 2010 to January 2025. Search terms included: “digital twins,” “urology,” “prostate cancer,” “renal cancer,” “bladder cancer,” “benign prostatic hyperplasia,” “urolithiasis,” “computational modelling,” “physics-informed neural networks,” “foundation models,” “surgical simulation,” and “endourology.”
Inclusion criteria comprised: (1) studies applying digital twins in healthcare; (2) urology-specific digital-twin applications; (3) biomedical engineering or AI studies relevant to organ modelling. Studies on nonbiomedical industrial applications, non-English publications, and abstracts without full text were excluded from our narrative review. Two reviewers independently screened titles and abstracts, with disagreements resolved through discussion. Given the conceptual nature of the field, additional foundational computational-science literature was included to contextualize technological enablers.
Rationale and development of digital twins
Starting from the initial role in replicating physical systems and interconnections, the rationale behind the development of digital twins has far changed. Referring to healthcare, the digital twins consist in a digital replica of human organs, with tissues, cells, and micro-environments that continuously vary according to real-time modifications and updates, in order to predict future behavior. 9
Essentially, by leveraging on available data from a specific patient’s anatomical features, comorbidities, past medical history, laboratory tests, and so on, a digital replica is created and constantly updated. 10 Available predictive models such as nomograms, algorithms or causal correlations are also integrated into the model.
The type of technologies usually crucial in the creation of the digital twins are labelled into two categories: (A) statistical and data-driven model, and (B) mechanical model integrated with multiscale knowledge. While the first model can be used to predict performance, the second is more reliable in simulation. 11
In urology, digital twins would consist of a 3D virtual replica of a specific organ or system, integrated with much needed surrounding structures or characteristics, aided with predictive AI tools and patient-specific information. The distinct advantage of these systems is their capacity to continuously incorporate new patient data, allowing for dynamic, personalized simulations that enhance adaptive treatment planning and improve the accuracy of prognostic predictions (Figure 1). The strict collaboration between expert urologists, radiologists, AI-trained engineers and data analysts is pivotal for the accurate creation of such an elaborate tool.
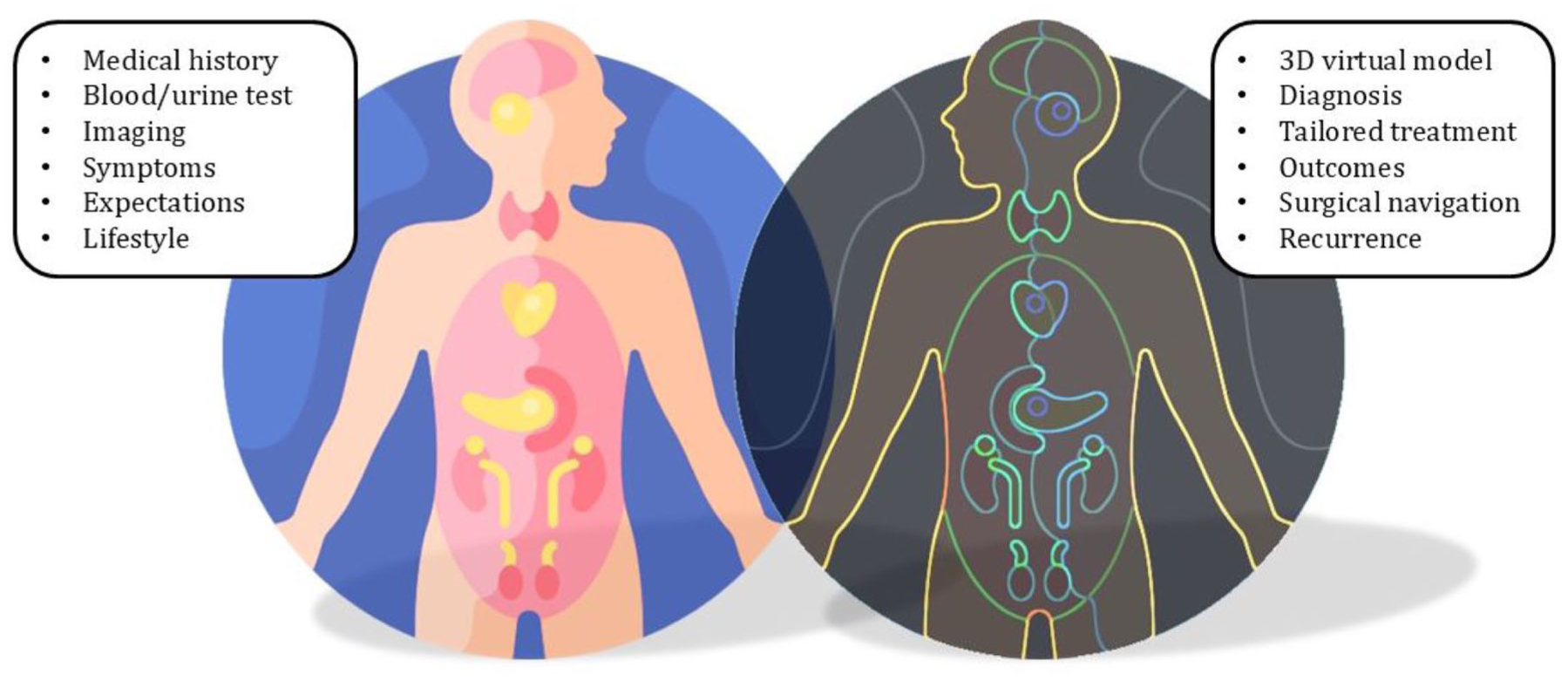
Application and creation of personalized digital twins, showing the counterpart from real-life patients and their digital twins. On the left side of the figure, the needed patient-specific information are mentioned, including past medical history, symptoms, laboratory and imaging findings, patient’s expectations and habits. On the right side, those translate into the digital replica, creating a 3D specific model and providing information on diagnosis, best treatments, possible outcomes and risk of recurrence.
Literature review
There is still a scarcity of original papers on the role of digital twins in the urological fields. Table 1 presents an overview of the findings from a literature search.
Original studies published so far on digital twins in urology.

CTV, clinical target volume; ML, machine learning; RAPN, robotic-assisted partial nephrectomy; SBRT, stereotactic body radiation therapy.
A first study in 2024 was conducted in Italy by Amparore et al., 6 investigating the role of digital twins in the surgical navigation of robotic-assisted partial nephrectomy for renal tumors. Digital replica of anatomical features, including details on kidney’s parenchyma, vessels, tumor, and collecting system were created for four selected patients with PADUA 6 or 8 kidney cancer. One young and one experienced surgeon acted as testers for the new models, comparing them to the standard 3D nonrealistic CT scan reconstruction. The 3D digital twins models were reported as superior to the control model, both in terms of anatomy representation and usefulness of intraoperative navigation, with identification of the tumor, vascular pedicle and ureter.
In 2025, two original studies were published on the use of digital twins for prostate cancer. In Spain, Pérez-Benito et al. 12 tested an MRI based prostate model, rendering precise shape and vascularization of the gland and the tumor. The model was then integrated with PSA dynamics and the Finite Element Method: a model including information on dynamics of tumor growth, transport of oxygen, and cellular processes such as proliferation, differentiation, and apoptosis. By testing the model on six patients, they found good accuracy in predicting prostate and tumor growth and PSA dynamics, with potential to support individualized diagnosis and treatment planning. In parallel, Chang and his group 13 in the USA created a model for stereotactic body radiation therapy for prostate cancer. The digital model was based on CT scan imaging (pretreatment and corrected cone-beam), and machine learning (ML) tailored treatment plans on patient-specific clinical target volumes were tested on 10 patients. The models were found effective in determining clinical target volumes, with promising results on radiation doses involving the bladder and the rectum.
John et al. discuss the possible applications of digital twins models in the care of prostate cancer. 7 By analyzing current evidence in other oncological fields and discussing feasible applications in urology, they conclude that AI-driven digital twins signify a groundbreaking shift in personalized prostate cancer care, offering the potential to transform diagnosis, treatment, and patient management. The review emphasized key advancements, including the use of convolutional neural networks (CNNs) for medical imaging, reinforcement learning for dynamic treatment planning, and hybrid models that merge AI with physics-based simulations to enable more tailored care. Additionally, emerging technologies such as large language models and vision-language models were discussed as new opportunities to enhance digital twins by integrating multimodal data and delivering natural language insights that improve the transparency and interpretability of clinical decisions.
An editorial published in 2024 by Checcucci et al. 5 explored the transformative potential of digital twins in the context of urological cancers, particularly in enhancing precision medicine. The authors discussed that digital twins offer a powerful, emerging tool for individualized cancer care, with applications ranging from surgical planning to long-term disease management. Despite current challenges, such as model complexity, data limitations, and implementation barriers, the technology holds immense promise for transforming oncology through more precise, predictive, and participatory care. The authors advocated for multidisciplinary collaboration to overcome these hurdles and realize the full potential of digital twin’s technology in clinical practice.
Nonmuscle invasive bladder cancer
Current evidence
At present, no validated digital-twin models exist for bladder cancer. A small number of exploratory imaging-based bladder models have been described, but these remain early prototypes, particularly MRI-based bladder wall reconstructions, which are still limited by motion artefact, variable bladder filling, and segmentation inconsistency. Nevertheless, insights from digital twins in radiotherapy and systemic oncology demonstrate the feasibility of modelling tumor growth, response trajectories, and treatment-induced tissue changes.
Conceptual applications
Digital twins have already been proposed for other oncological diseases. In the context of nonmuscle invasive bladder cancer (NMIBC), digital twins would serve as a valuable tool in different steps of a patient care (Figure 2). From the first diagnosis of NMIBC, digital twins could store information on tumor location and characteristics, creating a 3D digital replica of the bladder wall based on CT or MRI reconstructions. These models could then be updated with histopathological findings and tumor location or depth of infiltration to predict progression and response to treatments.

Role of different digital twins in endourological treatment: NMIBC, BPH and urolithiasis, with specific and shared applications. Each box provides the areas of digital twin’s conceptual application in the relative field, and the lower box shows the shared applications among the three diseases.
Digital twins and AI-tools such as CNN represent a valuable tool in the pathology workflow. In the context of prostate cancer, CNN-based models have been developed to support pathologists by automating the detection and classification of Gleason patterns in biopsy slides. These systems often perform at a level comparable to, or even surpassing, that of human experts, especially for routine pattern recognition tasks. By handling these time-intensive components of diagnosis, digital replicas of the pathologist’s workflow can greatly reduce the time needed for slide analysis. This, in turn can speed up the diagnostic process, enabling pathologists to concentrate on more complex or ambiguous cases. Similarly, CNN models could increase pathologist evaluation and report of TURBTs samples, to increase accuracy and streamline the workflow. Digitalized data from TURBT samples would be stored in the model to create a patient-specific model of tumor characteristics and evolution in time.
Moreover, the 3D digital replica of the bladder, updated constantly with new radiological findings would be also modified according to cystoscopy findings and new resection site, to create a pattern of tumor implantation and recurrence. These models, when compared to real-time findings, could help in better identifying locations of recurrence and primary sites for re-resections and biopsies. Moreover, the models would provide a perception of tissue thickness to increase in-depth resection while reducing risk of perforation.
As already proposed in different setting, 14 digital twins could also provide a patient-specific risk of recurrence, that when integrated with guidelines risk stratification, would improve tailorized treatments and follow-up protocols. In instance, the model could provide a personalized risk of progression and response to intravesical therapies like BCG. By creating these specific models, patient follow-up would be personalized and cystoscopies planned accordingly. In case of disease progression with the need of radical treatments, the digital replica could also provide useful information for surgical planning and real-life consultation during the procedure. Beyond these, digital replica can also serve as models for prediction of response to surgical or systemic treatments, for example, in selected cases for bladder-sparing treatments. Several studies have already been investigating the role of digital twins for prediction of response to chemotherapy or radiotherapy, and that could be applied to bladder cancer as well. 13
Illustrative conceptual example
A 68-year-old male with a newly diagnosed papillary bladder tumor undergoes CT and cystoscopy. A digital twin’s model of his bladder is created, integrating 3D anatomy, tumor size, and histopathology. The model simulates the optimal resection path for TURBT, avoiding the bladder dome where wall thinning is noted. Postresection, the twin runs a predictive model showing a 60% chance of recurrence within 6 months unless BCG therapy is initiated. The digital twins also model the expected immune response to BCG, showing a favorable profile. The patient agrees to treatment, and follow-up cystoscopies are scheduled based on recurrence risk.
Benign prostatic hyperplasia
Current evidence
No digital-twin frameworks currently exist for benign prostate disease. MRI-based prostate models developed for prostate cancer have demonstrated the feasibility of representing glandular anatomy, zonal boundaries, and tissue deformation; however, these are limited by segmentation variability and inconsistent reproducibility across scanners and radiologists.
Conceptual applications
BPH is a complex disease that often impacts quality of life. By integrating patient’s characteristic and urological findings on prostate health, digital twins could create a personalized model to improve treatment. The model could be integrated with past medical history, comorbidities, specialist scoring systems like IPSS and IIEF-F for recording lower urinary tract symptoms (LUTS) and erectile function trends. It could also be fed with information from uroflowmetry or urodynamics, hence creating a physical model for detrusor activity and bladder obstruction. With such a model, urologists could test different drug combinations to predict response to treatments, hence, tailoring the approach on the need for timely surgical interventions. These models have the potential to enhance clinical decision-making by reducing overtreatment and facilitating timely surgical intervention (Figure 2).
New surgical treatments for BPH are now challenging guidelines by moving toward personalized treatments that take into consideration patient’s expectations and impact on sexual life. While minimally invasive surgical treatments (MISTs), such as Rezum, iTIND, and UroLift are now been proposed for small sized prostate, TURP and prostate laser enucleation remain the most effective treatments for medium and large prostates. Different enucleation or resection techniques are often challenged by the diversity in shape and volumes that can be found once in operating room (OR).
Digital twins in BPH could provide a 3D digital replica of the prostate, based primarily on multiparametric MRI findings, and would provide a visual representation of the whole gland, but also of the adenoma. These models could be easily used to plan the procedure, whether this is an enucleation or a MIST procedure. Moreover, by leveraging on ML models for outcomes prediction, different techniques could be tested for various outcomes (improvement of LUTS, postoperative incontinence, residual sexual function, risk of bladder neck stenosis etc.) to meet at best patient expectations. 15 When planning a treatment for patients with comorbidities or history of use of anticoagulants, the digital replica could even test risk of bleeding for different procedure to calculate the patient-specific risk of postoperative bleeding.
Once the surgery is performed, histopathological samples could again benefit from AI-models such as CNN to reduce reliance on manual visual assessment. An initial AI-driven screening could flag “hot samples,” those with potential abnormalities, for further evaluation by a secondary AI model or human pathologist, thereby enhancing the detection of malignancies and streamlining the diagnostic workflow.
During follow-up, the digital model could be continuously updated with changes in patient symptoms and test results. This dynamic tracking would allow for monitoring symptom progression and predicting potential recurrence of obstruction, thereby supporting timely reintroduction of medical therapy, guiding further investigations, or indicating the need for retreatment.
Illustrative conceptual example
A 72-year-old man with severe LUTS undergoes prostate MRI and urodynamic studies. A digital twin of his prostate is generated, incorporating tissue density, bladder outlet resistance, and flow dynamics. Simulations show that a standard TURP would significantly improve flow but could also carry a high risk of damaging the external sphincter due to an unusual anterior lobe configuration. The surgeon adjusts the resection plan using the twin’s 3D guidance. After surgery, wearable bladder sensors feed postvoid residual data into the twin, which confirms normal recovery. The patient avoids incontinence and reports marked symptom relief.
Urolithiasis
Current evidence
Digital-twin research in stone disease is currently nonexistent. However, extensive work in computational fluid dynamics (CFD) and urinary flow modelling provides strong precedent. CFD-based twins have been used in cardiovascular and pulmonary medicine to simulate flow, obstruction, and particulate movement—principles directly applicable to stone migration and obstruction.
Conceptual applications
In the treatment of urinary tract stones, digital twins could have a pivotal role in personalized treatment and prediction of clinical behavior (Figure 2). By integrating patient-specific data, the digital twins could generate a detailed 3D model of the urinary tract, encompassing renal anatomy, including vessels, calyces, and pelvis, along with precise stone locations and morphology. The model could further extend to the ureter, incorporating data on anatomical variations such as areas of relative narrowing or dilation. With the integration of computational fluid dynamics (CFD) into the AI framework, it would be possible to perform predictive simulations of stone movement. This physics-based modelling could forecast the likelihood of spontaneous stone passage or, conversely, predict the risk of migration and obstruction.
As per the other conditions, these digital twins would tailor information on patient-specific data, such as biometric and laboratory findings (blood and urine), past medical history, family history, and symptoms. Digital twins, integrated with CNN or ML simulations and predictive modelling, could then help planning treatments and predicting outcomes. In shock wave lithotripsy, they could be used for prediction of response to treatment, fragmentation, movement, and clearance. In endoscopic treatment the velocity of fragmentation during lithotripsy could be simulated to predict an approximate time of surgery and prompt correct interventions (i.e., in choosing between a standard endoscopic procedure and a combined intrarenal surgery). Moreover, by leveraging on ML algorithms, digital twins could help predict outcomes of the procedures, 16 including stone clearance, residual fragments, risk of infectious complications, but also the need for a postoperative stent insertion or the risk of retreatment due to incomplete procedure.
Another important role of digital twins would be with regards to the personalized simulation. Relying on a 3D specific and interactive model, the internal and external anatomy of the kidney could be better studied and understood. When planning a percutaneous approach, the model could be used to simulate renal puncture and predict difficulty in accessing the calyces. In another example, an urologist could benefit from the model in complex cases with skeletal abnormalities, when a 3D reconstruction could be used to simulate the percutaneous approach in different positions (supine, prone, lateral) or in different calyces. During ureteroscopy, the system would possibly prove particularly beneficial in navigating the urinary tract in patients with anatomical variants such as horseshoe kidneys, reimplanted ureters, or urinary diversions (i.e., from cystectomy). Digital twins could also interface with real-time surgical procedures, functioning as navigation systems or providing intraoperative feedback to the urologist on key parameters such as intrarenal pressure and temperature. Additionally, by capturing and learning from endoscopic visual data during procedures, these digital replicas could continuously refine and enhance their predictive models.
In the postoperative setting, the digital twins could function as a sentinel, monitoring subtle changes in laboratory values, clinical signs, or patient-reported symptoms to anticipate adverse events and prompt timely interventions. Moreover, it could tailor dietary advice and lifestyle modifications to reduce the risk of recurrence, while generating predictive models that are both reliable and reassuring for patients.
Illustrative conceptual example
A patient with recurrent calcium oxalate stones undergoes a CT scan. A digital twin is generated, modelling their kidney anatomy and metabolic profile. The twin predicts that without dietary sodium reduction, the stone will recur in 18 months. The patient sees a simulation of their current versus optimized diet and chooses to change behavior.
Discussion
Digital twins represent a transformative paradigm for personalized urological care; however, their translation into practice requires a critical evaluation of both scientific feasibility and system-level constraints. Current-specific evidence remains limited to early applications in prostate and renal cancer. 10 Lessons from cardiology, liver modelling, and musculoskeletal digital twins demonstrate that maturation of this technology requires integration of multimodal data, rigorous validation, and alignment between computational modelling and clinical workflows.17,18
Despite the intriguing possibilities and applications of digital twins, its integration in urology faces significant challenges that may hinder its growth. First, the deployment of digital twins involves expensive investments in technology, infrastructure, maintenance, and data security. These include sensors, platforms, data storage, encryption, and backups, leading to high operational costs and complexity. Moreover, collecting hospital data (i.e., imaging, blood tests) is costly and time-consuming. Quality issues in imaging, especially for diagnostics like high-quality CT scans or multiparametric MRI depend heavily on the expertise of radiologists, further complicating data acquisition. This limitation will indeed make digital twins an expensive tool, available only in selective hospitals. 19 Second, digital twins rely heavily on vast, high-quality datasets. However, healthcare data often exist in unstructured formats within heterogeneous systems, requiring manual effort or advanced tools to extract useful information. This limits the efficiency and scalability of digital twins models. 20
Key technological enablers, such as physics-informed neural networks (PINNs), multimodal foundation models, hybrid AI–mechanistic modelling, and high-fidelity imaging, are rapidly advancing. 21 These methods provide a foundation for urological digital twins capable of simulating dynamic physiological processes, predicting treatment response, and guiding intraoperative decisions. Our clinical scenarios are conceptual illustrations rather than validated clinical tools.
Still, verification, validation, and uncertainty quantification (VVUQ) represent a major barrier. Strict VVUQ pipelines used in engineering and include code verification, clinical dataset validation, and Monte Carlo or Bayesian uncertainty propagation. 22 However, these VVUQ have not yet been widely implemented in clinical digital-twin research. Emerging FDA digital health guidance and ISO quality frameworks emphasise transparency, traceability, and safety, all of which are essential for future urological twins. 23 Interoperability and data governance further constrain real-world deployment. Digital twins require secure, standardized data pipelines integrating radiology, electronic health records, functional studies, and intraoperative metrics. Fast Healthcare Interoperability Resources (FHIR)-based architectures, encrypted real-time data flows, and federated learning will be essential for scalability. 24
Due to the lack of standardized approach to validate digital twins models, and the fact that their introduction in the healthcare system is very recent, there is also an issue of trust. Physicians often distrust algorithm-based decisions due to lack of transparency and risk of misdiagnosis, fearing potential job displacement and questioning model accuracy. 25 Similarly, patients and their families may feel inadequately informed or hesitant to rely on innovative and experimental tools such as digital twins. To build trust and ensure clinical reliability, external validation of digital twin’s models is essential. However, validation is inherently complex due to the high degree of individual variability in human health. While sensitivity analyses are critical to assess model robustness, they can be computationally demanding, particularly when numerous parameters are involved.
There are also some concerns about applicability and communication. Current digital twin’s software lacks user-friendly interfaces that facilitate communication between clinicians, patients, and the system. 26 Successful implementation requires interdisciplinary knowledge from biomedicine to computer science, which is often lacking. 27 Ethical and economic considerations include cost barriers, equitable access, data ownership, informed consent, and algorithmic opacity. Adoption of this new technology will depend on transparent validation, robust error reporting, and user-friendly interfaces. Finally, privacy and regulatory systems are currently lacking and this represents a major concern on the development of digital twins. The use of personal health data in digital twins raises ethical concerns around informed consent, data ownership, and privacy. Clear regulatory frameworks and ethical guidelines are urgently needed to ensure responsible usage. Advancing digital twins technology requires robust collaboration among researchers, healthcare providers, and tech developers. Interdisciplinary partnerships and data-sharing initiatives are vital for overcoming these challenges and enhancing model accuracy and trust. 26
In summary, while digital twins offer transformative potential, especially in healthcare, their widespread adoption is constrained by technical, ethical, financial, and institutional hurdles that demand coordinated efforts to address. Early adoption of digital twins in urology is likely to occur within robotic and image-guided interventions. Existing platforms such as da Vinci or HUGO RAS already generate rich kinematic and imaging data. Digital twins could enable preoperative simulation, real-time navigation, and postoperative analytics.
This narrative review may be subject to selection bias and does not provide a systematic assessment of study quality. Evidence from other specialties and computational sciences was included to contextualize emerging technologies, which may limit direct applicability to urology. The field is rapidly evolving, and publications may have appeared after the search cut-off date (January 2025). The conceptual examples presented are hypothetical and not clinically validated.
Conclusion
While facing considerate challenges in development and application, the introduction of digital twins in the context of endourological procedures and associated diseases (urolithiasis, BPH, NMIBC), would possibly represent a milestone toward digitalization and personalized medicine. Digital twins hold the potential to significantly enhance personalized care across the field of urology, from diagnosis and treatment to follow-up and recurrence prevention, ultimately improving the quality of care. While current evidence is limited, advances in AI and computational modelling provide a strong foundation for development. Real-world translation will require rigorous VVUQ frameworks, multi-institutional validation, interoperable data infrastructure, and clear regulatory pathways.
