Abstract
The use of artificial intelligence is changing the landscape of breast cancer surgery through enhanced precision, accuracy, and personalized treatment strategies. This narrative review assessed artificial intelligence applications across the surgical stages of care in breast cancer surgery, including preoperative planning, intraoperative assistance, and postoperative care. A structured literature search of PubMed, Scopus, and Web of Science over the past 15 years was conducted to identify studies on artificial intelligence applications across all stages of breast cancer surgery. In the preoperative phase, artificial intelligence contributes to tumor detection, segmentation, and surgical margin assessment through advanced imaging and predictive modeling. During surgery, real-time image-guided systems and robotic platforms powered by machine learning enable greater accuracy and intraoperative decision support. Postoperatively, artificial intelligence–driven tools aid in complication prediction, recurrence monitoring, and follow-up personalization, thereby improving patient outcomes and reducing variability in care. Integration of multimodal artificial intelligence approaches from imaging, robotics, and predictive analytics across the surgical continuum can help highlight translational gaps and evaluate clinical readiness, providing insights that have not been emphasized in previous reviews. Despite these advancements, many artificial intelligence applications remain in early research stages or have limited clinical use, facing challenges in data standardization, model interpretability, ethics, and integration into practice. Clinical impact depends on infrastructure, surgeon expertise, and regulations. Nevertheless, interdisciplinary collaboration allows artificial intelligence to enhance accuracy, reduce complications, and improve patient-centered care in breast cancer surgery.
Introduction
Recent advances in artificial intelligence (AI) are reshaping surgical oncology and have direct implications for breast cancer care. By enhancing image interpretation, predictive modelling, and intraoperative guidance, AI can improve clinical decision-making, surgical precision, and postoperative surveillance. Among oncologic fields, breast cancer surgery is emerging as an area where AI-driven solutions are rapidly evolving to address long-standing clinical and technical challenges. Breast cancer, the most common malignancy among women worldwide, originates from the uncontrolled proliferation of abnormal breast epithelial cells. These cells form tumors with the potential to invade adjacent tissues and metastasize if not treated promptly. It remains a leading cause of cancer-related morbidity among women, accounting for over 10% of all newly diagnosed cancers annually, and risk reduction in breast cancer can significantly decrease its morbidity. 1 Advances in breast cancer management—including earlier detection and the development of more targeted therapies—have contributed to improved survival rates in recent years. Current treatment strategies typically involve surgery, radiotherapy, chemotherapy, or immunotherapy, with the specific approach determined by tumor stage and histopathologic features. Familiarity with updated clinical guidelines is essential for accurate diagnosis, staging, and optimal patient management. 2 Surgery remains a cornerstone of breast cancer management, playing a crucial role in removing the primary tumor and assessing regional lymph nodes. In recent decades, there has been a shift toward breast-conserving surgery whenever clinically feasible, supported by trials demonstrating comparable survival outcomes and improved quality of life compared with mastectomy. Nevertheless, mastectomy and reconstructive options remain essential for many patients, depending on tumor characteristics and individual preferences. 3 Breast surgery continues to face several challenges, including intraoperative uncertainty in decision-making, the need for precise surgical technique, variability in cosmetic outcomes, and differences in clinical expertise across centers. 4 In this dynamic field, AI has emerged as a valuable tool to support breast cancer surgeons. AI systems are designed to perform tasks that typically require human intelligence. Recent AI-based tools have shown promise in enhancing preoperative planning, predicting prognosis, and providing intraoperative guidance through real-time image analysis and augmented reality applications. 5 Although these technologies are still under development and clinical integration is ongoing, early studies have demonstrated encouraging results in specific domains such as preoperative planning, intraoperative guidance, and postoperative monitoring.6–8 Advances in surgical techniques have further enabled the development of new AI applications to support decision-making and evaluation throughout the surgical process. Machine learning (ML), a core subset of AI, utilizes algorithmic models to identify complex patterns in large datasets and make predictive decisions without explicit programming. Its integration into surgical workflows, which began in the early 21st century, has led to measurable improvements such as enhanced preoperative planning accuracy, reduced intraoperative decision errors, improved postoperative assessment consistency, and better coordination among multidisciplinary teams in breast cancer surgery.9,10 Recent reviews have documented the growing integration of AI tools across oncologic surgery, from preoperative planning to intraoperative navigation and postoperative outcome prediction, highlighting a shift from experimental research toward clinical implementation. Moreover, in breast cancer care specifically, AI-assisted workflows are increasingly being explored not only for diagnostic imaging and pathology but also as support tools during surgical treatment and treatment planning.11,12 This narrative review aimed to provide a comprehensive overview of current challenges in breast cancer surgery and explore the emerging role of AI in improving decision-making, surgical accuracy, and patient-centered outcomes.
Methods and search strategy
This narrative review was guided by the SANRA (Scale for the Assessment of Narrative Review Articles) checklist to ensure methodological rigor and transparency. 13 A structured literature search was conducted to identify relevant studies on AI applications in breast surgery, encompassing lesion diagnosis, preoperative planning, intraoperative guidance, and postoperative follow-up. The databases searched included PubMed, Scopus, and Web of Science. Articles published within the past 15 years and written in English were included. We considered a broad range of study types, including original clinical studies, technical reports, radiologic or surgical workflow assessments, and narrative or systematic reviews related to AI applications in surgical oncology or breast surgery. Keywords used in the search included “artificial intelligence,” “machine learning,” “breast surgery,” “preoperative planning,” “intraoperative guidance,” and “postoperative care.” Studies were screened based on relevance to AI applications in surgical decision-making, imaging, robotics, or postoperative monitoring. Screening was performed independently by two reviewers, and any disagreements regarding study eligibility were resolved through discussion and consensus, or, when necessary, by consulting a third reviewer. The structured search initially identified 82 articles. After screening for relevance and excluding studies not directly related to AI applications in breast surgery, a total of 68 studies were included and synthesized narratively. The remaining references were used for background, methodology, and supporting context. Selected articles were reviewed and summarized to provide a comprehensive overview of current practices, challenges, and future perspectives.
Emerging applications of AI in breast surgery
Preoperative applications
Preoperative planning is crucial for the success of breast cancer surgery, requiring consideration of a wide range of data, including tumor characteristics, patient-specific anatomy, comorbidities, imaging findings, and therapeutic options. With the increasing complexity and volume of medical data as well as the expansion of treatment options, AI has emerged as a valuable tool to support clinicians during this critical phase.10,14,15 AI-driven clinical decision support systems (CDSS) have been developed to assist with treatment planning. For example, Bouaud et al. designed a guideline-based system to generate comprehensive care plans for breast cancer patients. The system was implemented at the multidisciplinary tumor board level rather than for individual surgeons, providing decision support to ensure consistency with established clinical guidelines. In 17% of cases, the tumor board modified its initial treatment decisions after reviewing the system’s recommendations. Notably, 75% of these changes improved adherence to evidence-based care. 16 Within the multidisciplinary framework of breast cancer management, treatment decisions are typically made collaboratively by surgeons, oncologists, radiologists, and pathologists during tumor board meetings, where adherence to evolving clinical guidelines is essential. In this setting, AI-based decision support tools serve as complementary aids rather than substitutes for expert judgment. These systems help harmonize recommendations across specialties, promote consistent guideline-based practice, and minimize interobserver variability in treatment planning. By delivering evidence-informed suggestions, CDSS platforms improve the consistency, transparency, and coordination of multidisciplinary decision-making, ultimately enhancing the overall quality of patient care.17,18 In a similar study, Xu et al. (2019) compared the recommendations of their decision support system with actual oncologists’ choices and reported a 45% nonconcordance rate. The observed differences were primarily due to individualized interpretation of patient data by physicians (21%), stricter adherence to clinical guidelines (15%), and lack of access to system-recommended treatments (5%). 19 A follow-up evaluation by Xu et al. (2020) showed that the decision support system led to changes in treatment decisions for 5% of patients, thereby improving compliance with breast cancer treatment guidelines. Among the revised decisions, 63% were driven by the system’s recommended treatment options, 23% were influenced by the system’s emphasis on specific patient characteristics, and 13% resulted from the logical structure underlying its decision-making process. 8 Moreover, ML models have enabled surgeons with lower procedural volume to achieve decisions comparable to those of high-volume experts by learning from vast datasets. 20 AI has also been applied to predict patients’ responses to neoadjuvant systemic therapy (NAST). In early-stage breast cancer, conservative surgery followed by radiotherapy is often the preferred management.21–23 However, some patients achieve a complete pathological response after NAST and may avoid surgery altogether. Accurately identifying these patients is essential to prevent unnecessary procedures and avoid missing residual malignancies. AI models trained on magnetic resonance imaging (MRI) and pathological data have demonstrated promising performance in predicting residual disease and determining whether patients are suitable candidates for surgery.24–28 AI is further being explored in patient education prior to surgery. A randomized controlled trial is underway to evaluate whether an AI model can better inform women about expected esthetic outcomes after locoregional surgery. The goal is to enhance patient satisfaction, reduce psychological stress, and minimize the need for follow-up plastic surgery. Additionally, AI has been applied to predict the financial burden associated with surgery, primarily addressing the financial toxicity experienced by patients due to treatment-related costs and income loss. However, the generalizability of such cost analyses remains limited, as healthcare financing structures differ across health systems. Nevertheless, AI models have demonstrated high accuracy in forecasting these financial outcomes.29,30 In imaging and surgical planning, AI tools have enhanced the precision of preoperative assessments. Once a lesion is detected, AI can analyze breast volume, symmetry, and vascular anatomy. Imaging modalities such as ultrasound, MRI, and computed tomography (CT), when combined with ML techniques, assist in selecting optimal reconstructive strategies. For instance, the Faster-RCNN with Inception ResNet-v2 deep learning model has shown efficacy in analyzing ultrasound breast images for surgical planning.31,32 Mavioso et al. (2020) assessed the feasibility of AI-supported planning for DIEP flap reconstruction and developed a convolutional neural network (CNN) capable of identifying key perforators in 97.7% of patients. 33 These models enhance microsurgical planning by reducing uncertainty, shortening operative time, and lowering procedural risk. Beyond surgical planning, AI can simulate cosmetic outcomes preoperatively, providing tangible benefits for both clinicians and patients. Chartier et al. (2022) trained a neural network on real clinical images to generate preoperative visualizations that closely resembled actual postoperative results. 34 This capability improves the informed consent process, helps set realistic expectations, and supports shared decision-making between patients and surgeons. AI-assisted three-dimensional reconstructions of patient anatomy further support both immediate and delayed breast reconstruction. By improving MRI and CT interpretation, these models can more accurately identify tumors, vessels, and anatomical landmarks, facilitating safer, more precise surgical execution. 35 Collectively, AI-driven tools in the preoperative phase of breast cancer care have demonstrated significant utility in multiple domains: guiding clinical decision-making, predicting responses to systemic therapy, optimizing surgical planning, simulating cosmetic outcomes, and enhancing patient education. These innovations not only aim to streamline workflow and personalize treatment strategies but also have the potential to improve both clinical outcomes and patient satisfaction.
Intraoperative assistance and enabling technologies
The intraoperative phase of breast surgery presents particular challenges, where accurate tumor localization, margin assessment, and avoidance of injury to critical structures are crucial. AI has been suggested as a method to enable real-time decision-making and improve surgical accuracy and clinical outcomes. AI applications during the intraoperative phase include real-time imaging analysis. Specifically, deep learning models, particularly CNNs such as U-Net and its variants, are being combined with intraoperative imaging modalities to enable segmentation of tumor margins in real time, allowing precise excision. This is particularly important in breast-conserving surgeries, where achieving clear margins while preserving healthy tissue is critical to reduce recurrence and avoid re-excision. Some intraoperative imaging modalities, such as laser Raman spectroscopy (LRS), serve primarily as diagnostic tools providing real-time molecular information about tissue. LRS itself does not incorporate AI. However, ML algorithms can be applied to the spectral data obtained from LRS to enable real-time classification of tumor margins, supporting intraoperative decision-making. LRS is an optical imaging modality that detects vibrational molecular signatures, allowing differentiation between normal and abnormal tissues. It can also identify microcalcifications and malignant tissue during surgery.6,36–39 For example, Kothari et al. (2021) developed an ML model that analyzed LRS data to classify resection margins. This model was partially validated in live surgical patients and provided real-time malignancy probabilities at the surgical margin, facilitating more precise excisions and potentially reducing the need for reoperation. 40 In addition to margin detection, AI-powered augmented reality (AR) systems have emerged as valuable tools in operating rooms. These systems overlay preoperative imaging data processed via ML algorithms onto the surgical field, providing real-time visualization of anatomical landmarks such as blood vessels, lymphatic structures, and tumor foci. Technologies such as Microsoft HoloLens have demonstrated the ability to project three-dimensional reconstructions, including computed tomography angiography (CTA) images, directly into the surgeon's field of view, improving spatial awareness and surgical precision.41–43 In breast surgery, such tools can help define avascular planes and connective tissue boundaries during flap harvest or lymph node dissection. Robotic platforms, including the Da Vinci system, are not inherently AI systems. They enhance surgical performance by improving dexterity, minimizing tremor, and allowing precise motion scaling, which is particularly advantageous for tasks such as vascular anastomosis. AI-driven decision support can be applied to specific tasks on these platforms, rather than the robots functioning as autonomous AI models. Advanced robotic systems have been used in breast surgery for anchoring acellular dermal matrices, facilitating flap harvesting, and performing microsurgical tasks with high precision.44–46 Furthermore, intraoperative pathology is being accelerated through AI algorithms that analyze whole-slide images in real time, enabling rapid assessment of tumor margins and lymph node involvement while the patient is still in the operating room. Despite these advancements, the use of AI in breast reconstruction surgery during the intraoperative phase remains mostly exploratory. Although robotic systems have been clinically implemented in orthopedic and neurosurgical procedures,47,48 their application in the soft, deformable tissues of the breast is still in preclinical stages and operates at low autonomy levels. 49 Current technologies are typically limited to isolated functions, such as instrument tracking or robot control, rather than complex, multi-step surgical interventions. Overall, developments in deep learning, augmented reality, computer vision, and robotic assistance suggest that AI will continue to enhance intraoperative decision-making, reduce operative time, and improve both oncological safety and cosmetic outcomes in breast surgery.
Postoperative monitoring and patient care
Postoperative management of breast surgery involves several clinical challenges, including proper wound healing, effective pain control, and early identification of complications such as seroma, infection, or hematoma. Long-term surveillance for cancer recurrence is also an essential component of postoperative care. AI-assisted monitoring systems are primarily applied in outpatient settings, where continuous remote tracking reduces unnecessary clinic visits and supports the early detection of complications. In inpatient settings, where daily wound inspections are routine, AI serves as a complementary tool to enhance the speed and accuracy of complication recognition. AI has recently emerged as a transformative tool for optimizing postoperative outcomes by enabling continuous monitoring, early identification of adverse events, and personalized recovery planning.7,50 ML algorithms are increasingly applied to data collected from wearable sensors that track physiological parameters such as vital signs, movement, and wound conditions in real time. These systems facilitate early recognition of complications such as delayed healing or infection, allowing timely intervention and potentially preventing serious outcomes.51,52 For instance, AI-enabled smart bandages with integrated biosensors can monitor wound moisture, pH, and temperature in real time, triggering alerts when infection is suspected.53,54 Although pilot studies show promise, widespread adoption is limited by factors such as production cost, the need for single-use materials, and challenges in integrating these devices into existing clinical workflows. Ongoing efforts aim to develop reusable and cost-effective sensor systems, making these technologies more feasible for routine postoperative monitoring. 55 Similarly, mHealth platforms using natural language processing (NLP) and image analysis track recovery progress remotely by evaluating patient-reported symptoms and images of surgical sites. This approach reduces unnecessary clinical visits and is particularly beneficial in resource-limited or rural settings.56,57 Certain applications have employed AI to predict and reduce complications such as lymphedema, which may occur immediately after axillary procedures or even decades later. In 2018, Fu et al. developed an ML model that successfully predicted lymphedema based on patient-reported symptoms, demonstrating its potential for early intervention. 7 Furthermore, although large language models such as ChatGPT are being explored for postoperative counseling, studies by Lukac et al. suggest that current versions lack sufficient clinical sophistication to provide accurate recommendations in primary breast cancer care. 52 To complement these counseling applications, AI has also advanced in the field of postoperative pain prediction. Recent tools aim to forecast pain trajectories and opioid requirements, enabling more personalized pain-control strategies while reducing the risk of overprescription. Nair et al. developed an ML model that successfully predicted individualized opioid needs after ambulatory surgery, and Juwara et al. demonstrated that AI-based analytics more accurately identified predictors of neuropathic pain following breast cancer surgery compared with conventional assessment methods.58,59 In the context of reconstructive surgery, AI has proven valuable for predicting complications and enhancing cosmetic outcomes. Myung et al. applied ML models to predict donor-site complications in patients undergoing muscle-sparing TRAM flap reconstruction, with neural networks achieving an accuracy of 81%. 60 Furthermore, Kenig et al. developed a neural network for automated evaluation of breast symmetry using patient photographs, achieving a 97.7% success rate in identifying key breast landmarks within milliseconds. This automated workflow provides a rapid and standardized method for assessing postoperative esthetic outcomes. 61 Beyond physiological recovery, AI contributes to the psychological dimension of postoperative care. NLP tools can screen patient narratives and digital diaries for signs of depression, anxiety, or body image concerns, facilitating earlier mental-health referrals when needed. 60 AI-driven imaging analysis has further enhanced surgical safety. By examining preoperative scans, these models can assess the proximity of tumors to critical neurovascular structures, potentially reducing the risk of injury to nerves such as the long thoracic or thoracodorsal.62,63 During long-term follow-up, AI-assisted image analysis of mammograms or MRIs allows early detection of recurrences or new lesions that may elude human observers. Integration with electronic health records (EHRs) supports automatic reminders for follow-up imaging and laboratory tests, thereby improving patient adherence to surveillance protocols.64,65 Moreover, AI-based tools are being evaluated for their ability to guide flap monitoring postoperatively. Kim et al. proposed an automated technique for assessing flap perfusion from images, reducing reliance on continuous clinical monitoring and easing the burden on healthcare staff. A DenseNet121-based model was used to assess flap perfusion, demonstrating a sensitivity of 97.5% for venous insufficiency and 92.8% for arterial insufficiency. The area under the receiver operating characteristic curve (AUC) was 0.960 (95% confidence interval (CI): 0.951–0.969), indicating excellent diagnostic performance. The system was tested in a real-world clinical setting, successfully completing 143 automated monitoring sessions without significant issues, highlighting its readiness for clinical implementation. 66 Overall, these findings suggest that AI-based flap monitoring can serve as a reliable complementary tool in postoperative care by enabling earlier detection of complications and reducing staff workload. In general, AI technologies in postoperative breast surgery offer substantial potential for improving clinical outcomes. From early complication detection and optimized pain management to enhanced esthetic evaluation and psychological screening, AI provides a more holistic framework for supporting patient recovery. These innovations not only decrease readmissions and surgical complications but also enhance patient satisfaction and overall quality of life. However, although AI tools have demonstrated promise in controlled settings, their integration into routine clinical practice presents several challenges. The performance of AI algorithms can vary across different clinical settings due to factors such as data quality, patient demographics, and healthcare infrastructure, highlighting the need for validation across diverse patient populations and real-world scenarios. Successful implementation also requires seamless integration with existing EHRs and healthcare IT systems, which is critical for ensuring the effectiveness, usability, and sustainability of AI solutions in clinical practice.67,68
As comprehensively reviewed in this section, AI provides seamless support across all surgical phases. Figure 1 illustrates these applications in preoperative planning, intraoperative guidance, and postoperative care.

AI applications across preoperative, intraoperative, and postoperative phases of breast surgery.
Integration of multimodal AI models in breast surgery and broader breast cancer management
Multimodal AI models represent a significant advancement in breast surgery, as they integrate heterogeneous data types, including imaging modalities, histopathological data, genomic data (referring to tumor-specific molecular and genomic features rather than whole-genome sequencing), and clinical data, to enhance diagnostic outcomes, surgical planning, and postoperative care.10,69 Figure 2 provides an illustrative overview of this integration, showing how different data streams are processed by AI models to support surgical decision-making. For instance, a recent meta-analysis demonstrated that deep learning models trained on breast MRI data exhibited a pooled sensitivity of 76% and specificity of 81% in predicting axillary lymph node metastasis (ALNM) in breast cancer patients. When both the primary tumor and axillary lymph nodes were included as input targets, sensitivity increased to 81% and specificity to 86%. The overall diagnostic performance, as measured by the summary receiver operating characteristic, yielded an area under the curve (AUC) of 0.79, confirming the value of such models in supporting preoperative decision-making and reducing unnecessary surgical interventions.
70
Another compelling example of multimodal AI integration is BioFusionNet, a deep learning framework designed for survival risk stratification in estrogen receptor–positive (ER-positive) breast cancer patients. This model combines histopathological image features with genomic and clinical data to generate a comprehensive patient profile. By leveraging self-supervised vision transformers (DINO and MoCoV3), variational autoencoders, and a co-dual-cross-attention mechanism, BioFusionNet effectively captures complex interactions between multiple data modalities. It achieved a mean concordance index of 0.77 and a time-dependent AUC of 0.84, outperforming existing models. Additionally, BioFusionNet demonstrated prognostic value in both univariate (hazard ratio (HR) = 2.99, 95% CI: 1.88–4.78,
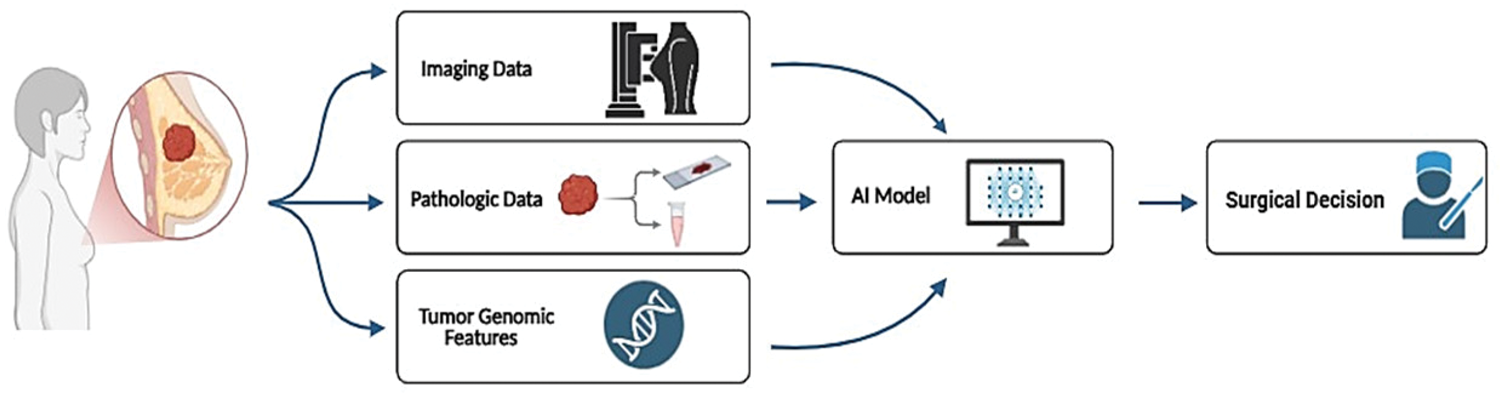
Multimodal data fusion in AI models for breast cancer surgical planning. The term “genomic data” refers specifically to tumor-specific molecular and genomic features, not whole-genome sequencing.
Current limitations and future directions
Despite the remarkable progress of AI in breast surgery, its clinical adoption remains limited by several challenges. Although AI has demonstrated considerable promise in diagnostics, decision support, and intraoperative guidance, its transition from controlled research settings to routine clinical practice is not yet fully realized. A key obstacle is data heterogeneity, which encompasses variability in imaging acquisition protocols, scanner types, histopathological staining techniques, and clinical documentation across institutions.73–75 These inconsistencies significantly hinder the generalizability of AI models, as algorithms trained on single-center datasets often fail to maintain performance during external validation. 76 Another major challenge is the scarcity of high-performance AI models that are also interpretable. Many current algorithms function as opaque decision-making systems, reducing clinical trust, limiting transparency in surgical decisions, and complicating regulatory approval. Integrating AI systems in real time within the surgical environment introduces additional technical concerns, including inference delays, maintaining equipment sterility, network infrastructure requirements, and data security. 77 Handling large-scale, multimodal datasets that include imaging, molecular, and clinical information also poses challenges for governance, patient privacy, and informed consent. There remains a theoretical risk of patient re-identification when imaging-omics repositories are not fully anonymized. Differences in international data-sharing regulations further complicate efforts toward global collaboration. Despite these challenges, several emerging strategies offer promising solutions. International research consortia are increasingly developing standardized, curated multimodal datasets with harmonized imaging protocols and unified clinical annotations. These efforts facilitate robust external validation and reproducibility. At the same time, advances in explainable AI (XAI) are enabling interpretable outputs, such as concept-based attribution maps in digital pathology or saliency overlays in breast MRI, which can be incorporated into intraoperative interfaces to support surgical decision-making.78,79 Techniques such as federated learning and differential privacy are being implemented to enable collaborative model training across institutions without sharing raw data, thereby preserving patient confidentiality while enhancing algorithm robustness and generalizability. 80 In contrast to previous reviews that primarily focused on diagnostic radiology or individual surgical stages, this work emphasizes how AI can bridge preoperative, intraoperative, and postoperative workflows, highlighting the unmet need for interoperable multimodal systems. Such cross-phase integration represents a novel conceptual framework for understanding AI’s role in surgical oncology. At present, AI lacks the capacity for full independence and cannot replace the expertise of human surgical teams. However, its integration into various components of surgical practice holds significant potential to enhance overall performance and outcomes. Each AI model discussed in this review exhibits unique limitations and potential: BioFusionNet and MFF-HistoNet, although highly accurate in research settings, have not yet been fully integrated into clinical workflows; Faster-RCNN and U-Net models provide precise preoperative and intraoperative imaging analysis, but their generalizability across diverse patient populations and imaging systems remains limited; DenseNet121-based models for postoperative flap monitoring demonstrate excellent diagnostic performance in controlled studies but require broader real-world validation and workflow integration. Addressing these model-specific challenges is essential for transforming experimental AI systems into dependable, multimodal tools suitable for routine breast surgery. Embedding AI within interdisciplinary workflows is equally important to ensure collaboration and clinical validation. Multidisciplinary tumor boards and expert panels play a vital role in validating AI outputs, contextualizing algorithmic recommendations, and ensuring that clinical decisions remain patient-centered. Integrating AI tools within these established collaborative frameworks will facilitate safer, evidence-based adoption and enhance clinical trust in real-world settings.81,82 Taken together, by linking model-specific limitations with solutions such as standardized multimodal datasets, explainable interfaces, privacy-preserving collaborative training, and integration within multidisciplinary clinical pathways, these insights clarify how model-specific considerations collectively support the transition of experimental AI systems into practical, interoperable multimodal applications in breast surgery. This transition ultimately relies on coordinated efforts to enhance data quality, ensure transparency, integrate workflows seamlessly, and safeguard patient privacy.
Conclusion
The use of AI in breast cancer surgery represents a promising advancement in modern surgical oncology. AI has the potential to enhance preoperative planning, intraoperative guidance, and postoperative care, supporting more personalized and precise surgical strategies. Although current technologies remain in development or early clinical phases, initial studies suggest that they may improve surgical accuracy, reduce complications, and optimize patient outcomes. However, robust evidence demonstrating survival or patient-centered benefits is still limited, underscoring the need for further research. Clinical adoption of AI will require continued collaboration among clinicians, data scientists, and engineers to validate these tools, address ethical considerations, and ensure safe and equitable implementation. Prospective, outcome-driven studies are essential to establish the real-world impact of AI and confirm its benefits for patients.
Footnotes
Acknowledgments
We thank all participants for their time and cooperation, without which this research would not have been possible, and acknowledge the use of AI-powered tools to check grammar and enhance the academic quality of the manuscript, which was primarily written by the authors.
Author contributions
Conceptualization, M.Kh; Contributed to all experimental work, M.Kh and MA.Sh; validation, P.F and E.R; investigation, E.R and P.A; resources, all authors; writing—original draft, M.Kh and MA.Sh; writing—review and editing, P.F, E.R, and P.A; project administration, MA.Sh and M.Kh.
Consent to participate
Not applicable.
Data availability statement
Not applicable.
Declaration of competing interest
The authors declare that they have no conflicts of interest.
Ethics approval
Not applicable.
Funding
This research did not receive any external funding.
