Abstract
Background:
Novel androgen receptor inhibitors (ARIs) have been recommended for patients with high-risk nonmetastatic castration-resistant prostate cancer (nmCRPC).
Objective:
The present study aims to evaluate the cost-effectiveness of darolutamide, enzalutamide, apalutamide, and bicalutamide when combined with androgen deprivation therapy (ADT) for treating nmCRPC in the context of national drug price negotiations in China.
Design:
A cost-effectiveness analysis.
Methods:
A Markov model was developed to assess the cost-effectiveness of first-line therapy (darolutamide, enzalutamide, apalutamide, and bicalutamide) combined with ADT in nmCRPC patients, as well as second-line treatment options receiving chemotherapy after disease progression. The model included three health states: progression-free survival, progression survival, and death. The transfer probability per period was calculated using a Log-normal distribution. Drug costs were obtained from national price negotiations and relevant medical institutions, and health state utility values were obtained from the literature. Uncertainty was addressed through one-way sensitivity, probabilistic sensitivity, and scenario analyses.
Results:
Compared with bicalutamide plus ADT, apalutamide, darolutamide, and enzalutamide provided incremental benefits of 3.52, 4.96, and 3.86 quality-adjusted life years (QALYs), respectively. This resulted in incremental cost-effectiveness ratios (ICERs) of $117,261, $166,618, and $238,170 in nmCRPC patients. Specifically, the ICERs of apalutamide, darolutamide, and enzalutamide were $33,357/QALY, $33,600/QALY, and $61,740/QALY, respectively.
Conclusion:
Compared with bicalutamide plus ADT, apalutamide plus ADT and darolutamide plus ADT are more cost-effective under the willingness-to-pay threshold of $38,223/QALY. Nonetheless, enzalutamide plus ADT is not cost-effective compared with bicalutamide plus ADT.
Introduction
Prostate cancer (PCa) is the most common cancer of the male genitourinary system and the fifth leading cause of cancer mortality. 1 As one of the top 10 malignancies among men, PCa accounts for about 29% of all cases, posing a significant threat to men’s health worldwide and imposing a heavy economic burden on patients, their families, and society. 2 In 2020, there were over 1.414 million estimated new cases of PCa globally. 3 The incidence and mortality rates of PCa are rising rapidly in Asia, with China ranking second in the number of new cases, only behind the United States. Furthermore, the crude incidence rate (CIR) and mortality rate for cancer continue to increase in China, highlighting the need for effective prevention and control measures. 4 According to estimates from the GLOBOCAN 2022 online database, there were 4.825 million newly diagnosed cancer cases and 2.574 million cancer deaths in China in 2022, including 134,000 new cases of PCa and 56,000 deaths. 5 Moreover, both the morbidity and mortality rates of PCa are on the rise. From 2000 to 2018, the age-standardized incidence and mortality rates of PCa showed an overall upward trend in China. 6 One study reported that the inpatient expenses for cancer reached 241.9 billion Chinese yuan (CNY) in 2017. Specifically, the inpatient costs associated with PCa increased from 710 million CNY in 2008 to 3.65 billion CNY in 2017, growing at a rate significantly higher than the global average. 7
Androgen deprivation therapy (ADT) combined with novel androgen receptor inhibitors (ARIs) is currently recommended as a first-line treatment regimen for patients with high-risk nonmetastatic castration-resistant prostate cancer (nmCRPC) (class 1A evidence).8,9 The novel ARIs, including apalutamide, darolutamide, and enzalutamide, are second-generation anti-androgens that directly target the ligand-binding domain of androgen receptor (AR), thereby preventing AR nuclear translocation, DNA binding, and transcription of AR target genes. 10 Compared with bicalutamide, novel ARIs have a higher affinity for AR and are more potent inhibitors of AR activity. 11
Therefore, these ARIs provide an important treatment option for patients with nmCRPC and significantly extend average survival. The phase III SPARTAN study demonstrated that, compared with placebo, metastasis-free survival (MFS) was significantly prolonged in high-risk nmCRPC patients treated with ADT plus apalutamide (40.5 vs 16.2 months), and end-of-study data confirmed a significant overall survival (OS) benefit (73.9 vs 59.0 months). 12 In the phase III ARAMIS study, treatment with ADT plus darolutamide significantly extended MFS (40.4 vs 18.4 months), improved OS, and reduced the risk of death by 31% compared with ADT alone. In addition, ADT plus darolutamide also significantly improved progression-free survival (PFS) (36.8 vs 14.8 months) and delayed the time to prostate-specific antigen progression (33.2 vs 7.3 months). 13 Similarly, the phase III PROSPER study found that enzalutamide plus ADT significantly prolonged MFS compared with placebo (36.6 vs 14.7 months), and reduced the risk of metastasis or death by 71%. 14
Nevertheless, cost-effectiveness studies on these three novel ARIs remain limited. Therefore, a model-based economic evaluation of these agents in patients with nmCRPC is warranted. Against the backdrop of national medical insurance drug negotiations, a Markov model was constructed to assess the economic impact of ADT combined with apalutamide, darolutamide, or enzalutamide for the treatment of nmCRPC patients from the perspective of the Chinese healthcare system. The aim is to provide a reference to support clinical and patient treatment decisions.
Methods
Perspective
Our study was conducted from the perspective of the Chinese healthcare system to assess the utilization of healthcare resources and the benefits of interventions within the context of the national medical system.
Model overview
Herein, a Markov model with three health states, PFS, progression survival, and death, was developed using TreeAge Pro Healthcare 2022 software to simulate the lifetime health outcomes of high-risk nmCRPC patients receiving first- and second-line therapy (Figure 1 and Figure S1). Patients entered the model in the progression-free state and received first-line treatment. Those who experienced disease progression subsequently received second-line therapy, as illustrated in the treatment hierarchy chart (Figure S2). The starting age of patients was set at 65 years, and the model’s time horizon was set at 20 years to capture the lifetime cost-effectiveness of treatment. 15 A cycle length of 30 days with half-cycle correction was adopted in the base case scenario. Outcomes were measured in life-years (LYs) and quality-adjusted life-years (QALYs). The discount rate(s) used for costs and QALYs were discounted at an annual rate of 5% in accordance with the Chinese pharmacoeconomic evaluation guidelines. 16 The willingness-to-pay (WTP) threshold was set at $38,223 per QALY gained, equivalent to three times China’s gross domestic product per capita in 2022 ($12,741), using an exchange rate of 1 USD = 6.7261 CNY. 17

State-transition schematic. (1) Patients enter the Markov model in the progression-free state and receive first-line therapy (bicalutamide, apalutamide, darolutamide, or enzalutamide). In this state, patients may remain progression-free, experience disease progression, or die. (2) Patients who transition to the progressed-disease state may either remain in this state or die. (3) Progressed patients can remain alive with progressed disease or die. (4) The absorbing state of the model is death.
Clinical data inputs
The clinical inputs considered in the model included OS and PFS. Survival data were reconstructed from published Kaplan–Meier curves of the SPARTAN, 12 ARAMIS, 13 PROSPER, 14 and STRIVE trials, 18 as well as other relevant clinical studies. Data points were extracted using GetData Graph Digitizer software (version 2.26). Individual patient data were then reconstructed using maximum likelihood estimation based on the extracted values and the number of surviving patients at each time interval in R version 4.0.3, based on the algorithm built by Guyot. 19 Finally, parametric survival models, including Weibull, log-logistic, log-normal, Gompertz, and exponential distributions, were fitted to the reconstructed pseudo-individual data. 20 The results showed that the Akaike information criterion (AIC) between the models was within five points, indicating no significant difference in goodness-of-fit to the observed data. The best-fitting distribution was selected based on statistical criteria, visual inspection of the survival curves, and clinical plausibility. Following consultation with clinical experts, log-normal distributions were chosen for the final model (Table S1-1, S1-2, and Figure S3). Here, the long-term background mortality was adjusted using the 2022 China life tables.
Cost and utility
In the present study, the following direct medical costs were considered from the perspective of the Chinese healthcare system: costs of ADT, first-line treatment with novel anti-androgens, second-line salvage chemotherapy, routine follow-up, supportive care, regular medical check-ups, treatment-related adverse events, and hospice care (Table 1). The prices of apalutamide, darolutamide, and enzalutamide were obtained from the Zhejiang Provincial Center for Drug & Medical Device Procurement (ZJCDDP). 21 The costs of salvage chemotherapy, routine follow-up, regular medical check-ups, and supportive care were based on the diagnosis-related groups (DRGs) payment standard for prostate cancer in Zhejiang Province. The costs of end-of-life care and treatment-related adverse events were estimated using data from published studies. 22
Input parameters.

ADT, androgen deprivation therapy; AEs, adverse events; DRG, diagnosis-related groups; ZJCDDP, Zhejiang Provincial Center for Drug & Medical Device Procurement.
Health state utility values refer to the weight assigned to different health states in relation to complete health and serve as an indicator for evaluating the quality of a particular health state. Health utility values for stable disease and progressive disease were estimated from EQ-5D-3L responses collected in previously published literature.23,24 In addition, disutility due to adverse events was also incorporated into the analysis.
Sensitivity analyses
To assess the robustness of the results, one-way sensitivity analysis (OWSA) and probabilistic sensitivity analysis were conducted to examine the uncertainty of the model. In the OWSA, parameters were adjusted independently within a reasonable range based on published data or 95% confidence intervals (CIs). If the confidence was unapplicable, values were modified by ±10% to ±20% of the corresponding base case value.
In addition, a probabilistic sensitivity analysis (PSA) was conducted using second-order sensitivity analysis with 10,000 iterations (Monte Carlo simulation) to account for uncertainties in the model parameters. Gamma distributions were applied for costs, while beta distributions were used for utility and time-dependent transition probabilities in our study. Moreover, several scenario analyses were conducted to explore methodological and structural uncertainties. These included alternative discount rates, gamma extrapolation, Weibull distributions, log-logistic distributions, time horizons (20 years), and starting ages.
Results
Base case analysis
The results of the base case analysis are presented in Table 2. Over a 10-year lifetime horizon, 99.32%, 90.82%, 76.01%, and 85.69% of patients who were treated with bicalutamide, apalutamide, darolutamide, or enzalutamide, respectively, died during the first-line treatment phase, while 0.68%, 4.72%, 0.20%, and 2.15% of patients received second-line chemotherapy, respectively. Furthermore, 0%, 4.46%, 23.79%, and 12.16% of patients treated with bicalutamide, apalutamide, darolutamide, or enzalutamide, respectively, with PFS. The cumulative costs were $89,131, $206,392, $255,749, and $327,302 for patients treated with bicalutamide, apalutamide, darolutamide, or enzalutamide, respectively. Meanwhile, the cumulative effects were 2.40 QALYs, 5.91 QALYs, 7.35 QALYs, and 6.25 QALYs for the respective treatments. By year 10, compared with bicalutamide, the incremental benefit of apalutamide, darolutamide, and enzalutamide was 3.52 QALYs, 4.96 QALYs, and 3.86 QALYs in nmCRPC patients with incremental costs of $117,261, $166,618, and $238,170, respectively. In addition, the incremental cost-effectiveness ratios (ICERs) were $33,357/QALY, $33,600/QALY, and $61,740/QALY, respectively (Figure 2 and Table 2).
Summary base case results.
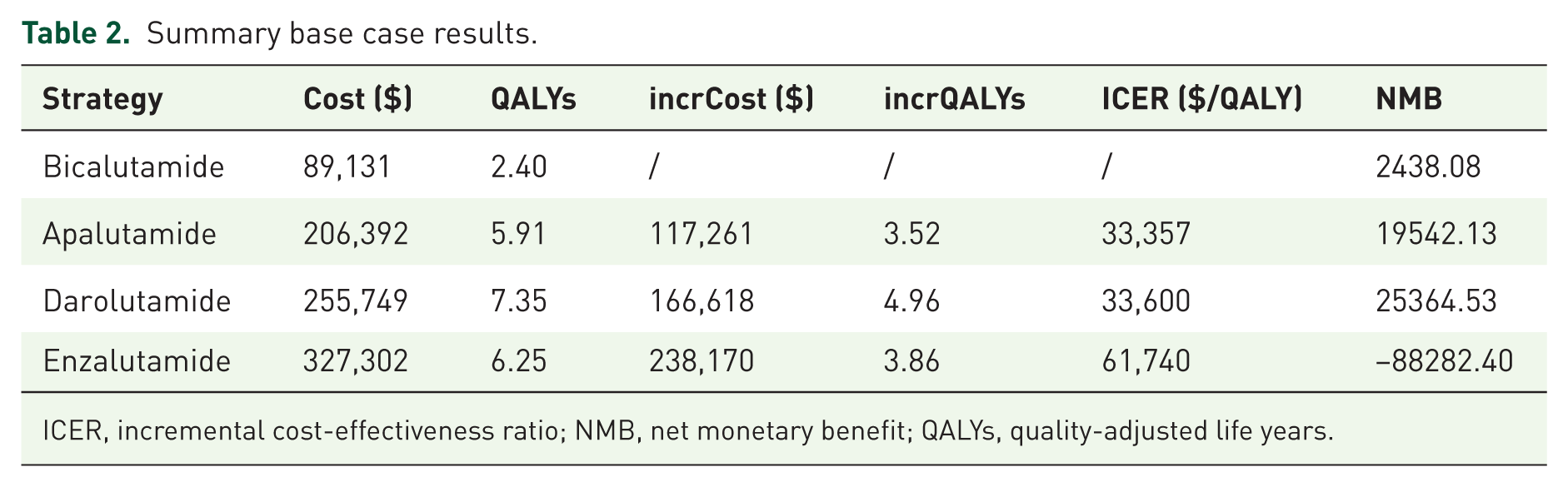
ICER, incremental cost-effectiveness ratio; NMB, net monetary benefit; QALYs, quality-adjusted life years.

Cost-effectiveness analyses.
Sensitivity analyses
In the OWSA, the key drivers of the model outcomes were the utility values for PFS and progression survival, the prices of the four antiandrogens, the costs of ADT and chemotherapy drugs, the discount rate, and the costs associated with adverse drug reactions (Figure 3). In the PSA, compared with bicalutamide, the cost-effectiveness probabilities of apalutamide and darolutamide were both 100%, while that of enzalutamide increased to 96.5% at a WTP threshold of $76,446/QALY (Figure 4 and Figure S4). In the threshold analysis, at a WTP threshold of $38,223/QALY, the cost-effectiveness probabilities of apalutamide, darolutamide, and enzalutamide compared with bicalutamide were 87.80%, 90.7% and 0%, respectively (Figure 5). Our study demonstrated that variations in the discount rate, the use of alternative parametric distributions for transition probabilities (including Gamma extrapolation, Weibull, and log-logistic), changes in the starting age to 35 years, or a 20-year time horizon had minimal impact on the ICER results. Importantly, these findings support the robustness of our sensitivity analyses. Scenario analyses indicated that the use of exponential extrapolation for the three novel anti-androgens had the most significant effect on ICER outcomes, with apalutamide plus ADT and darolutamide plus ADT emerging as the cost-effective treatment regimens (Table 3).

One-way sensitivity analysis (Tornado diagrams of ICER, the pink bars represent low parameter value, and the purple bars represent low parameter value). (a) Apalutamide plus ADT versus bicalutamide plus ADT, (b) darolutamide plus ADT versus bicalutamide plus ADT, and (c) enzalutamide plus ADT versus bicalutamide plus ADT.

Cost-effectiveness acceptance curves.

Scatter plot of the ICER (each dot demonstrates one iteration in the Monte Carlo simulation). The green circle represents the 95% confidence interval. Red dots show iterations that are not cost-effective at the WTP threshold. Green dots show iterations that are cost-effective at the WTP threshold). (a) Apalutamide plus ADT versus bicalutamide plus ADT, (b) darolutamide plus ADT versus bicalutamide plus ADT, and (c) enzalutamide plus ADT versus bicalutamide plus ADT.
Scenario analysis results.

ICER, incremental cost-effectiveness ratio.
Discussion
Novel androgen receptor inhibitors have been incorporated into the National Medical Insurance Drug List for first-line treatment of nmCRPC through healthcare negotiations. Given these changes, a pharmacoeconomic evaluation of the guideline-recommended anti-androgens is warranted. The present study aimed to provide evidence-based data support for clinical decision-making reference for clinicians in the treatment of nmCRPC.
Herein, a Markov model was constructed with a 20-year time horizon. The results indicated that, compared with bicalutamide plus ADT, the additional costs associated with apalutamide plus ADT, darolutamide plus ADT, and enzalutamide plus ADT for patients with nmCRPC were $117,261, $166,618, and $238,170, respectively, yielding incremental gains of 3.52 QALYs, 4.96 QALYs, and 3.86 QALYs. The corresponding ICERs were $33,357/QALY, $33,600/QALY, and $61,740/QALY, respectively. These results indicated that apalutamide plus ADT and darolutamide plus ADT were more cost-effective than bicalutamide plus ADT under the WTP threshold of $38,223/QALY. By contrast, the ICER of enzalutamide plus ADT exceeded this threshold, suggesting that enzalutamide was not cost-effective compared with bicalutamide. Furthermore, the stability and reliability of the Markov model were confirmed through sensitivity and probabilistic sensitivity analyses.
In addition, our study demonstrated that darolutamide plus ADT was associated with the highest QALYs and the lowest mortality among all treatment options. After 20 years of model simulation, the QALYs for darolutamide plus ADT, enzalutamide plus ADT, apalutamide plus ADT, and bicalutamide plus ADT were 7.35 QALYs, 6.25 QALYs, 5.91 QALYs, and 2.40 QALYs, respectively. The corresponding mortality rates were 76.01%, 85.69%, 90.82%, and 99.32%, respectively. These findings are consistent with those of a previous study. Wang et al. 27 conducted a matching-adjusted indirect comparison and network meta-analysis to evaluate the efficacy and safety of different anti-androgen drugs in nmCRPC patients and found that darolutamide was associated with the lowest hazard of death, followed by enzalutamide and apalutamide. Regarding safety, darolutamide also demonstrated the lowest odds of serious adverse events, followed by enzalutamide and apalutamide.
Several international studies have evaluated the cost-effectiveness of novel antiandrogens in patients with nmCRPC. Riaz et al. 28 developed a Markov model incorporating three health states to assess the cost-effectiveness of new antiandrogens from the perspective of the U.S. healthcare system. They showed that compared with ADT alone, the ICER of darolutamide plus ADT was $119,670, and that of apalutamide plus ADT and enzalutamide plus ADT was $145,900 and $13,568,800, respectively. These findings suggested that darolutamide plus ADT was the most cost-effective option based on the $150,000/QALY threshold, followed by apalutamide plus ADT and enzalutamide plus ADT or ADT alone. Conversely, a study conducted in Greece assessed the cost-effectiveness of apalutamide plus ADT versus enzalutamide plus ADT using a partitioned survival model with three health states (nmCRPC, mCRPC, and death). 29 The results indicated that apalutamide plus ADT provided additional life years (6.2 vs 5.4) and QALYs (4.3 vs 3.8) compared to enzalutamide plus ADT. The lifetime cost per patient for apalutamide plus ADT ranged from €131,760 to €146,223, while the cost for enzalutamide plus ADT was €128,121. The incremental cost-utility ratios (ICURs) ranged from €6,998 to €34,814. The probability of apalutamide being cost-effective ranged between 56% and 68% at a WTP threshold of €50,000. These results suggest that apalutamide is a cost-effective treatment for nmCRPC patients when compared to enzalutamide in Greece.
Furthermore, a partitioned survival model study showed that, compared with apalutamide plus ADT and enzalutamide plus ADT, darolutamide plus ADT was either a dominant or cost-effective treatment option for high-risk nmCRPC patients in China. 25 The results indicated that the cost per QALY for darolutamide plus ADT was 202,897 CNY/QALY, compared to 228,998 CNY/QALY for apalutamide plus ADT and 221,409 CNY/QALY for enzalutamide plus ADT. However, this cost-utility analysis assumed that these three novel ARIs had identical prices, as the post-national reimbursement drug list prices were commercially confidential, which may limit the applicability of the results in clinical practice. Therefore, our study fills this defect and provides a more scientific reference for clinicians and patients in making informed treatment decisions. Furthermore, from a budget impact perspective, our study can estimate the total medication demand for new drugs, treatment-related complication costs, and subsequent medical resource consumption within a specific time period by simulating the transition probabilities of patients between different disease states (such as PFS, progression survival, and death). In terms of equity, the model can conduct subgroup analysis by incorporating characteristics of different populations. Regarding accessibility in the context of drug price negotiations, the results generated by the Markov model provide valuable evidence for negotiation strategies and serve as an essential tool for assessing affordability and access.
Nevertheless, our study has several limitations in our study. First, the patients in the clinical trials (ARAMIS, SPARTAN, and PROSPER) were not exclusively Asian, which may result in differences in population-based responses and clinical outcomes. Second, the utility values used in our study were estimated from foreign studies, which could introduce bias in the results. The data in our study were derived from phase III clinical trials, and caution should be exercised when generalizing these results to the real-world setting. Third, only the management costs of grade 3 and 4 AEs were incorporated in our study, as grade 1 and 2 events are generally mild and unlikely to have a meaningful impact on overall model outcomes. Lastly, although adjustment techniques were applied to align the modeled OS with outcomes reported in clinical trials, the classification of patient subgroups relied on key assumptions that may not fully reflect real-world clinical practice.
Conclusion
In conclusion, our study evaluated the economic value of three novel antiandrogen drugs for patients with nmCRPC in China. Using a Markov model, we uncovered that compared with bicalutamide plus ADT, both apalutamide plus ADT and darolutamide plus ADT were more cost-effective from the perspective of the Chinese healthcare system. Overall, our findings provide a reliable reference for clinical decision-making and reimbursement policies in China.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251414928 – Supplemental material for Cost-effectiveness of novel antiandrogens for the treatment of nmCRPC patients in the Chinese healthcare system
Supplemental material, sj-docx-1-tau-10.1177_17562872251414928 for Cost-effectiveness of novel antiandrogens for the treatment of nmCRPC patients in the Chinese healthcare system by Wenjuan Yang, Bei Zheng, Mengyue Zhou, Ying Li, Lulu Zheng, Jun Zhu and Meiling Zhang in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
