Abstract
Non-muscle-invasive bladder cancer (NMIBC), which is restricted to the mucosa (stage Ta, carcinoma in situ (CIS)) or submucosa (stage T1), comprises 75% of bladder cancer diagnoses. Intravesical bacillus Calmette-Guérin (BCG) therapy is the standard-of-care initial treatment for high-risk NMIBC; however, a significant proportion of patients have BCG-unresponsive disease. While radical cystectomy is a definitive treatment in this setting, not all patients are willing or able to undergo this complex procedure associated with morbidity, mortality, and decreased quality of life. Bladder-preserving options for patients with BCG-unresponsive NMIBC represent an unmet need in this patient population. Nadofaragene firadenovec-vncg (Adstiladrin) is a nonreplicating adenoviral vector-based gene therapy indicated for the treatment of adult patients with high-risk BCG-unresponsive NMIBC with CIS with or without papillary tumors. The antitumor efficacy of nadofaragene firadenovec is driven by its local delivery of copies of the gene encoding for interferon alpha-2b (IFNα-2b) to urothelial cells. In the phase III CS-003 study, over half of participants with CIS exhibited a complete response by month 3 after instillation, with minimal serious adverse events. The favorable efficacy and safety profile, clinical utility, novel mechanism of action, and every 3-month dosing schedule give nadofaragene firadenovec a unique role in the treatment of high-risk BCG-unresponsive NMIBC. This review provides a practical approach to the effective clinical use of nadofaragene firadenovec regarding pre-instillation visit arrangements, storage, handling, instillation procedures, and post-instillation procedures. Implementation of these recommendations will ensure efficient real-world use of nadofaragene firadenovec and the development of useful training materials and relevant standard operating procedures to help support a clinic’s treatment for patients with BCG-unresponsive NMIBC with CIS.
Video Abstract https://vimeo.com/user17898099/review/953723559/e18af7ec43
Introduction
Bladder cancer is the 10th most commonly diagnosed cancer globally, 1 with an estimated 82,290 new diagnoses in the United States in 2023. 2 Global incidence and mortality rates in men are 9.5 and 3.3 per 100,000 individuals, respectively, and rates of incidence and mortality are approximately four times higher in men than in women. 1 Approximately 75% of patients with bladder cancer have non-muscle-invasive bladder cancer (NMIBC; Ta, T1, and carcinoma in situ (CIS)), and 58% of these cases are low-grade NMIBC. 3 Low-grade NMIBC is characterized by well-differentiated cancer cells and typically exhibits an indolent natural history, making it less prone to invasion of the bladder muscle layer, which is the hallmark of muscle-invasive bladder cancer (MIBC). The remainder of NMIBC diagnoses typically involve high-grade (HG) tumors that are poorly differentiated, grow more aggressively, and are more likely to progress to MIBC or metastasize. 4 The risk of NMIBC recurrence and progression is substantial, with 5-year probabilities ranging from 31% to 78% and 1% to 45%, respectively, while overall survival for localized cancer is approximately 71% at 5 years.5,6 Unfortunately, among patients who progress from NMIBC to MIBC or metastatic disease, the mortality rate is high, ranging from 56% (for stage II disease) to 91% (for stage IV disease, including metastatic disease), emphasizing the need for preventing or delaying disease recurrence and progression to more advanced stages.7,8 In addition, a diagnosis of NMIBC leads to lower health-related quality of life (HRQoL) than the general population, and disease progression can be associated with further decreases in HRQoL with increased surveillance and surgical treatment. 9
Nadofaragene firadenovec-vncg (Adstiladrin, also known as rAd-IFN/Syn3) is an intravesical nonreplicating adenoviral vector-based gene therapy that delivers the gene encoding for interferon alpha-2b (IFNα-2b) directly to urothelial cells, enabling secretion of IFNα-2b protein that triggers anticancer and immunomodulatory effects. Nadofaragene firadenovec was approved by the US Food and Drug Administration (FDA) in December 2022 for the treatment of adult patients with high-risk bacillus Calmette-Guérin (BCG)-unresponsive NMIBC with CIS with or without papillary tumors. 10 The mechanism of action of nadofaragene firadenovec is multifold and encompasses direct cytotoxic effects of IFNα-2b on cancer cells and induction of antitumor immunity. These actions include direct cytotoxicity,11,12 antiangiogenic effects,13,14 increased tumor cell immunogenicity,15,16 and activation of key immune cells. 16 Increased tumor cell immunogenicity is mediated by upregulation of surface tumor-associated antigen presentation via MHC-I molecules. Key immune cells modulated by IFNα-2b include cytotoxic CD8+ T cells and natural killer cells. IFNα-2b can also exert effects on immune cell proliferation, maturation, and antigen presentation. 15
Intravesical therapies for bladder cancer
The initial treatment regimen for patients with NMIBC varies based on risk group per the American Urological Association/Society of Urologic Oncology classification. Patients with low-risk or intermediate-risk disease receive transurethral resection of the bladder tumor (TURBT), with follow-up surveillance and/or intravesical chemotherapy based on risk factors and patient and/or urologist preferences. 17 For patients with high-risk NMIBC, initial treatment regimens with TURBT and intravesical BCG are intended to prevent subsequent recurrence or progression. 17
BCG is a live-attenuated bacterium used for routine immunization against tuberculosis and has been approved by the FDA for use in NMIBC treatment since 1990. 18 In clinical trials, intravesical BCG has had initial complete response (CR) rates between 70% and 75% in CIS. 19 In addition, recurrence rates of 16%–40% have been observed in patients receiving BCG following TURBT for high-risk papillary disease, which are lower than the recurrence rates of TURBT alone. 20 However, approximately one-third of patients with NMIBC will not respond to BCG. Among those who demonstrate an initial response, more than 50% will experience recurrence or progression during long-term follow-up. 21 While BCG is efficacious and has a favorable safety profile, it may have distinct adverse effects and tolerability considerations. Symptoms of bladder irritability related to the inflammatory response are reported in approximately 60% of patients receiving BCG; other adverse events (AE) include influenza-like symptoms, fever, malaise, and rare systemic BCG infections. 22 BCG infections have been reported in healthcare workers preparing BCG for administration, and nosocomial infections have been reported in patients receiving parenteral drugs prepared in the same area as BCG. BCG is capable of dissemination when administered by intravesical route, and rare serious reactions have occurred in patients receiving intravesical BCG. 22 Care should be taken not to traumatize the urinary tract or introduce contaminants into the urinary system.
For patients with no response to BCG therapy or therapy failure, the likelihood of a positive response to more than two induction courses of BCG is low, and the risk of invasive or metastatic disease may be increased compared to those who had initial responses to BCG therapy. 21 The FDA defines BCG unresponsiveness as at least one of the following: (1) persistent or recurrent CIS alone or with recurrent Ta/T1 disease (noninvasive papillary disease/tumor invades the subepithelial connective tissue) within 12 months of completion of adequate BCG therapy, (2) recurrent HG Ta/T1 disease within 6 months of completion of adequate BCG therapy, or (3) HG T1 disease at the first evaluation following an induction BCG course. To aid in the classification of patients with recurrent disease after BCG treatment, adequate BCG therapy is defined as at least five of the six induction doses and two of the three maintenance treatments with BCG, or at least two of six instillations of a second induction course in which maintenance BCG is not given. 23 Patients with BCG-refractory disease who do not respond to initial BCG treatment are at higher risk of progression and death and are less likely to benefit from additional BCG therapy. Patients with early relapses with recurrence within 6 months of the last BCG dose for HG Ta/T1 NMIBC and within 12 months for CIS may have similar outcomes to BCG-refractory disease. However, patients with later relapses may have more favorable outcomes and respond to further courses of BCG therapy, so it is important to delineate patients who are truly BCG-unresponsive and unlikely to respond to further BCG therapy. 24
BCG therapy may ultimately fail in more than 40% of patients with high-risk NMIBC, underscoring the need for new efficacious and safe treatment options for BCG-unresponsive NMIBC. 25 According to the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology, radical cystectomy is the preferred treatment for patients with high-risk BCG-unresponsive or -intolerant NMIBC. The goals of treatment for NMIBC include reducing recurrences, preventing disease progression, and preserving the bladder. 26 Bladder preservation may be preferred in appropriate patients, as radical cystectomy is associated with a significant risk of morbidity and mortality and can lower patient HRQoL outcomes in mental and physical health domains. Patients may have comorbidities that preclude them from receiving cystectomy, and many patients prefer therapies that avoid or delay cystectomy. 9 Until recently, there were few bladder-preserving therapies available to patients with BCG-unresponsive NMIBC. For a treatment to be considered effective in this setting, a clinically meaningful initial CR rate (in CIS) or recurrence-free rate (in papillary tumors) of at least 50% at 6 months, 30% at 12 months, and 25% at 18 months is recommended. 27 Valrubicin (administered in a urology office or clinic) is the only FDA-approved chemotherapeutic agent that is indicated for intravesical therapy for BCG-refractory CIS of the urinary bladder in patients for whom immediate radical cystectomy would be associated with unacceptable morbidity or mortality. 28 Other frequently used agents for intravesical chemotherapy include gemcitabine, docetaxel, and mitomycin C, used both as monotherapy and in combination. 26 The mechanisms associated with intravesical chemotherapy are varied and depend on the agent selected, and may include DNA replication inhibition or cell cycle arrest and apoptosis induction. 26 Intravenous pembrolizumab, administered at an infusion center every 3–6 weeks, is another FDA-approved therapy for BCG-unresponsive NMIBC. Pembrolizumab works by binding to the PD-1 receptor, blocking both immune-suppressing ligands, PD‑L1 and PD‑L2, from interacting with PD-1 to help restore T-cell response and immune response. 29 With few agents available for high-risk BCG-unresponsive NMIBC, additional safe and effective therapies are needed. a
Nadofaragene firadenovec
Nadofaragene firadenovec is a nonreplicating and nonintegrating adenoviral vector-based gene therapy comprising recombinant adenovirus-interferon alpha-2b (rAd-IFNα2b) that delivers the gene encoding for IFNα-2b to urothelial cells plus Syn3, which enhances viral transduction in the urothelium. Bladder epithelial cells display sustained expression and secretion of IFNα-2b, which inhibits tumor cell growth and activates innate and adaptive antitumor immune responses to delay or prevent disease progression. Intravesical gene therapy using a nonreplicating recombinant adenovirus involves a reduced risk of “viral cystitis” of the bladder, a significantly reduced risk of systemic absorption and viremia, and a manageable biohazard risk. 19 Nadofaragene firadenovec administration involves a simple dosage schedule compared with intravesical chemotherapy or BCG dosing once every 3 months. 10 In a multicenter phase II trial, 40 patients with HG BCG-refractory or -relapsed NMIBC were randomly assigned to receive either low-dose or high-dose intravesical nadofaragene firadenovec; seven patients (33%) and seven patients (37%) achieved 12-month HG recurrence-free survival in the low- and high-dose groups, respectively; the higher dose was selected for use in the subsequent multicenter phase III trial. 30 In the single-arm, multicenter, open-label, repeat-dose, phase III trial, eligible patients were aged 18 years or older and had diseases that met the definition of BCG-unresponsive NMIBC according to the 2018 FDA Guidance for Industry. 31 Patients were enrolled into 1 of 2 cohorts by diagnosis at enrollment: (1) patients with persistent CIS with or without HG Ta or T1 NMIBC or (2) patients with HG Ta or T1 tumors without concomitant CIS. The primary endpoint was the CR rate in the CIS cohort at any time within 12 months after the first dose of nadofaragene firadenovec. Among the patients with CIS with or without HG Ta or T1 disease, 53.4% had a CR at 3 months after the first dose; among patients with an initial response, the response was maintained through 12 months in 45.5% of patients and 57 months in 10.9% of patients. In the cohort of patients with HG Ta or T1 disease, 72.9% were HG recurrence-free at 3 months, with 43.8% remaining recurrence-free through 12 months, and 14.6% remaining recurrence-free through 57 months. In most patients, the AEs were transient and were classified as either grade 1 or 2. No grade 4 or 5 treatment-related AEs were reported.31,32
Nadofaragene firadenovec provides a novel mechanism of action as the first FDA-approved intravesical gene therapy for high-risk BCG-unresponsive NMIBC. With an every 3-month administration schedule that may coincide with other routine workup schedules such as cystoscopy and cytology, it may be easier to ensure patient adherence compared to therapies with more frequent administration schedules (such as weekly intravesical chemotherapy or pembrolizumab every 3–6 weeks).10,26 Comparison of efficacy and safety across studies is difficult due to the lack of standardization, reinduction at progression in some studies, and other differences in methodology; however, nadofaragene firadenovec has demonstrated a favorable risk–benefit profile across phase II and phase III clinical studies.30,31
In addition to efficacy and safety, the cost of NMIBC treatment is an important factor in clinical decision-making as it affects accessibility and affordability for patients, impacts equitable access to effective and safe therapies like nadofaragene firadenovec, and ultimately impacts healthcare outcomes and resource allocation. While additional research assessing the cost-effectiveness and budget impact of nadofaragene firadenovec is required, it is anticipated that detailed cost analyses will be conducted in the near future once pricing, plan coverage, and long-term outcomes become available.
Practical considerations for nadofaragene firadenovec handling and administration
Effective administration of nadofaragene firadenovec requires specific considerations for handling and administration due to its deep freeze cold-chain requirements and intravesical instillation administration procedures. While urology offices and clinics have extensive clinical and research experience with BCG and/or nadofaragene firadenovec, there is an increasing reliance on infusion centers and a need for patient-level personalized counseling by a physician or nurse. Logistical and practical considerations associated with nadofaragene firadenovec administration are broadly consistent with other intravesical agents. Some considerations include assessment and counseling prior to the first visit (including providing pre-visit patient instructions), performing urinalysis (if the patient is symptomatic for a urinary tract infection per the treating physician’s discretion), managing patient interaction, thawing the product, updating electronic health records and documentation, administering treatment, allowing for dwell time, and providing written post-visit patient instructions (Figure 1). The clinic staff must ensure that they are equipped with the necessary administration and disposal tools not supplied with nadofaragene firadenovec, including four vented vial adaptors suitable for a standard 20-mL vial, two standard Luer lock adaptors, two standard 50- or 60-mL Luer lock syringes, a urinary catheter, lubricating jelly, personal protective equipment, a virucidal agent, and a biohazard waste container. 10
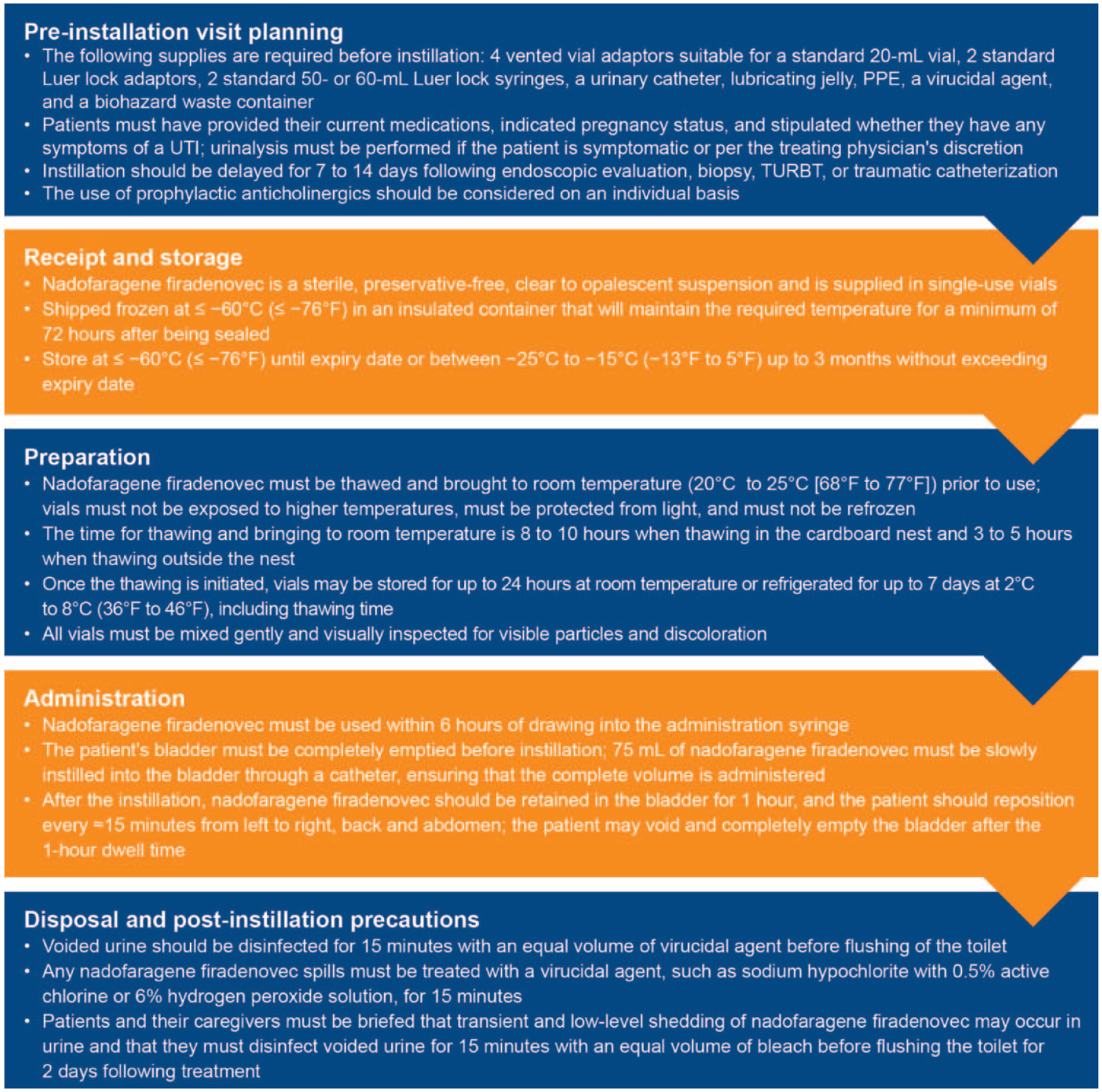
Overview of the nadofaragene firadenovec administration visit planning.
Instillation should be delayed for 7–14 days following endoscopic evaluation, biopsy, TURBT, or traumatic catheterization to avoid potential adverse effects. Patients should be informed that intravesical therapies, including BCG and nadofaragene firadenovec treatment, can increase the risk of bladder and urinary tract infections. The use of prophylactic anticholinergics should be considered on an individual basis to suppress involuntary bladder contractions caused by various factors. Other medications may also be considered on an individual basis, including urinary tract analgesics such as phenazopyridine (Pyridium).
Transportation and storage
Nadofaragene firadenovec is a sterile, preservative-free, and clear to opalescent suspension for intravesical instillation and is supplied in single-use vials; a single-use vial contains an extractable volume of 20 mL. Nadofaragene firadenovec is shipped frozen at ⩽ −60°C (⩽ −76°F) in an insulated shipping box that will maintain the required temperature for a minimum of 72 h after being sealed. Each carton contains a removable cardboard nest of four clear glass vials of nadofaragene firadenovec. On receipt, the cartons can be stored in a freezer at ⩽ −60°C (⩽ −76°F) until the expiration date or from −25°C to −15°C (−13°F to 5°F) for up to 3 months without exceeding the expiry date. When the product is stored between −25°C and −15°C (−13°F and 5°F), the date of placement in the freezer should be noted. In addition, the date for when the carton should be discarded if not used must be written on the outer carton. These dates should be 3 months apart but should not pass the original expiry date; the discard date supersedes the original expiry date. 10
Dosage
The dose of nadofaragene firadenovec is 75 mL at a concentration of 3 × 1011 viral particles/mL instilled once every 3 months into the bladder via a urinary catheter. The instillation procedure integrates with urologists’ established approaches to treating and managing the care of patients with NMIBC, including regular cystoscopy every 3 months. 10
Preparation and handling
Universal biosafety precautions should be followed during the handling of nadofaragene firadenovec, and individuals with immunosuppression or immunodeficiency should not prepare, administer, or come into contact with it. Nadofaragene firadenovec must be thawed and brought to room temperature (20°C–25°C (68°F–77°F)) prior to use; vials must not be exposed to higher temperatures, must be protected from light, and must not be refrozen. The time for thawing and bringing nadofaragene firadenovec to room temperature is 8–10 h when thawing in the cardboard nest and 3–5 h when thawing outside the nest. It is not necessary to await the patient’s arrival to begin the thawing process; thawing can begin several hours prior to the patient’s visit to minimize the amount of time the patient waits prior to administration. Once the thawing is initiated, vials may be stored for up to 24 h at room temperature or refrigerated at 2°C–8°C (36°F–46°F) for up to 7 days (including thawing time). All vials must be mixed gently and visually inspected for visible particles and discoloration. The suspension should be clear to slightly opalescent and may contain opalescent flecks. The vials must not be used if visible particles or discoloration are observed. 10
Bladder instillation
An aseptic technique should be followed when removing the nadofaragene firadenovec vial cap and attaching a vented vial adaptor to the vial per the manufacturer’s instructions. Once the vial adaptor has been attached, a syringe should be connected and the vial contents withdrawn. These steps must be repeated using the remaining three vials until 75 mL has been withdrawn into one or two syringes. Nadofaragene firadenovec must be used within 6 h of being drawn into the syringe; any remaining volume must be discarded according to universal precautions.
Only catheters made of vinyl and/or polyvinyl chloride, red rubber latex, or silicone must be used, and those coated or embedded with silver or antibiotics must be avoided. The patient’s bladder must be completely emptied using this catheter before nadofaragene firadenovec instillation; the catheter must remain in place thereafter. Then, 75 mL of nadofaragene firadenovec must be slowly instilled into the bladder through the catheter, ensuring that the complete volume is administered. After the instillation, nadofaragene firadenovec should be retained in the bladder for 1 h. The patient should reposition approximately every 15 min from left to right and from back to abdomen to maximize bladder surface exposure during this 1-h dwell period. If the patient exhibits bladder cramping or premature voiding during this time, repositioning of the patient may be adjusted or discontinued. Nadofaragene firadenovec must then be evacuated as part of routine emptying of the bladder, or the patient may void and completely empty the bladder after 1 h has elapsed. Voided urine should be disinfected for 15 min with an equal volume of virucidal agent before flushing the toilet. 10
AEs and management
Nadofaragene firadenovec has shown a favorable safety profile throughout its clinical study program, with a low risk of systemic toxicity and immune-mediated AEs. In the phase III CS-003 study, 110 of 157 (70.1%) participants had studied drug-related AEs, after 5 years of follow-up, with the majority of events being mild, transient, and localized in nature. 32 The most frequently reported study drug-related AEs were discharge around the catheter during instillation, fatigue, bladder spasms, and micturition urgency. Only 3 of 157 (1.9%) participants experienced a serious study drug-related AE, and there were no treatment-related deaths. 32 Risk of discharge from around the instillation site can be mitigated with appropriate administration techniques and ensuring that vials are thawed to room temperature. Similarly, bladder spasm risk can also be managed on an individual basis with premedication with anticholinergic medications. Decreasing the occurrence of bladder spasms will also prevent the occurrence of leakage of fluid around the catheter. Bladder pain was an uncommon AE, occurring in less than 10% of participants in the study. The localized delivery also has avoided significant systemic side effects or immune-related AEs.
A real-world, non-interventional study, known as the ADSTILADRIN in BLadder CancEr U.S. Real World Evidence Study (ABLE-41; ClinicalTrials.gov Identifier: NCT06026332), is ongoing. This study is exploring early utilization, HCP and patient experiences, and outcomes of nadofaragene firadenovec in the routine care setting.
Disposal and accidental spillage and exposure
Any nadofaragene firadenovec spills must be treated with a virucidal agent, such as sodium hypochlorite with a 0.5% active chlorine or 6% hydrogen peroxide solution, for 15 min. Disposable materials that were in contact with nadofaragene firadenovec should be placed in biohazard containers for destruction, and nondisposable equipment may be decontaminated according to the facility’s standard operating procedures. 10
Precautions and considerations during patient follow-up
Patients with BCG-unresponsive CIS must be informed that delaying cystectomy could lead to the development of muscle-invasive or metastatic bladder cancer and that this risk increases the longer cystectomy is delayed in the presence of persisting CIS. Five (12.5%) of 40 patients undergoing cystectomy were upstaged to muscle-invasive or extravesical disease at cystectomy during the phase III CS-003 study. 31 Adhere to careful follow-up monitoring schedule, and counsel patients on the importance of adherence to follow-up visits. Patients and caregivers must be made aware that treatment or contact with nadofaragene firadenovec in those who are immunocompromised, including those receiving immunosuppressive therapy, may increase the risk for disseminated adenovirus infection. Patients and their caregivers must be informed that transient and low-level shedding of nadofaragene firadenovec may occur in urine and that they must disinfect voided urine for 15 min with an equal volume of bleach before flushing the toilet for 2 days following treatment. 10
Summary
Nadofaragene firadenovec is a novel nonreplicating adenoviral vector-based gene therapy indicated for the treatment of adult patients with high-risk BCG-unresponsive NMIBC with CIS with or without papillary tumors. Efficacy and safety reporting and publication led to its FDA approval in December 2022. 10 As a bladder-preserving intravesical therapy administered once every 3 months, nadofaragene firadenovec is well positioned for use in clinical urology practice. Although its transportation, storage, handling, and administration involve careful attention, evidence from clinical trials and real-world settings has demonstrated that treatment with nadofaragene firadenovec can be effectively managed when site-specific pathways such as instillation procedures, fulfillment of infrastructure requirements, allocation of nurse resources, and regulatory guidelines are implemented. The approval and use of nadofaragene firadenovec provide an important new intravesical therapy for patients with BCG-unresponsive NMIBC with CIS with or without papillary disease.
Footnotes
Acknowledgements
Medical writing support and manuscript submission, under the guidance of the authors, was provided by Paul Sobol, PhD (ApotheCom, Yardley, PA, USA), and was funded by Ferring Pharmaceuticals, Parsippany, NJ, USA. The authors authorized the third-party submission and approved any statements/declarations being made by the submitting party.
