Abstract
Objective
To provide a practical approach to pharmacists regarding the safe and effective use of nadofaragene firadenovec.
Data sources
Clinical study data and pharmacist experience in institutional settings.
Data summary
This review article discusses the role of pharmacists caring for patients with Bacillus Calmette-Guérin (BCG)–unresponsive non–muscle-invasive bladder cancer (NMIBC) receiving nadofaragene firadenovec. Pharmacist roles in a multidisciplinary care team are described and involve considerations surrounding nadofaragene firadenovec, including its mechanism of action, indication, handling, storage, administration, adverse event profile, disposal, counseling, and other practical points.
Conclusions
Pharmacists are positioned to be key contributors to the care team regarding the effective use of nadofaragene firadenovec. Further awareness and education will continue to optimize the clinical use of nadofaragene firadenovec in the high-risk BCG-unresponsive NMIBC setting.
Introduction
Bladder cancer is the ninth most common cancer globally, with an estimated 83,190 new cases in the United States in 2024.1–3 Bladder cancer that does not extend into or beyond the muscle layers of the bladder wall (staged as Ta, T1, or carcinoma in situ [CIS]) is defined as non–muscle-invasive bladder cancer (NMIBC). Cancer that has progressed and invaded the muscle layers of the bladder wall is classified as muscle-invasive bladder cancer (MIBC) and is associated with worse outcomes than NMIBC.4–6 Approximately 75% of all bladder cancer diagnoses are classified as NMIBC, which may be histologically categorized as low-grade or high-grade.4,6 Low-grade NMIBC presents with well-differentiated cells on histological examination and is less likely to progress to MIBC compared with high-grade NMIBC. 4
Guideline-recommended routine workup of NMIBC includes stratification of disease into low-, intermediate-, or high-risk groups based on grade of disease, previous therapies, tumor characteristics, and other disease features. This stratification enables better quantification of risks of recurrence and progression.4–6 Risk stratification of patients with NMIBC assists in informing therapy decisions, as 5-year risks range from 31% to 78% for risk of recurrence and 0.8% to 45% for risk of progression, depending on risk group. 7
An initial transurethral resection of the bladder tumor (TURBT) is required for diagnosis, staging, and grading. Treatment of NMIBC is then informed by risk group; select patients may undergo repeat TURBT within 6 weeks. Guideline-recommended treatment options for patients with high-risk NMIBC include intravesical Bacillus Calmette-Guérin (BCG) therapy or radical cystectomy.4–6
BCG is a live attenuated bacterium that relies on adaptive and innate immune responses for antitumor effects and has been used for several decades in the treatment of bladder cancer. 8 Intravesical BCG is generally well tolerated but has an adverse event profile notable for local and systemic side effects, including rare risks of disseminated infections, particularly in immunocompromised patients.9–11 Additionally, BCG intravesical instillations are retained for a 2-h dwell time, which may be difficult for some patients to tolerate. 12 Furthermore, a global BCG shortage has led to exploration of modified dosing schedules, with recommendations on prioritization of BCG for high-risk patients. 13
While BCG has high initial complete response rates of up to 80%, patients who fail to achieve an initial complete response are more likely to have poor outcomes. 14 Furthermore, of patients who do respond to BCG, up to half will experience recurrence and/or progression of disease during long-term follow-up; many of these patients will be classified as having BCG-unresponsive disease.14–17
Previous clinical trials have had heterogeneous definitions of BCG-unresponsive disease. The US Food and Drug Administration (FDA) has provided guidance on standardized definitions of BCG-unresponsive disease and adequate BCG therapy. 18 In the setting of BCG-unresponsive disease, radical cystectomy is definitive treatment. 5 However, radical cystectomy is a major surgery that requires removal of the bladder, prostate, and seminal vesicles in men and the bladder, uterus, fallopian tubes, and anterior vagina in women. 19 The surgery is associated with increased morbidity and mortality, and decreased quality of life outcomes. Therefore, the procedure may not be suitable for all patients, as NMIBC often presents at an advanced age where patients may have other comorbidities that increase the risks of negative outcomes. 20 Given the significant challenges associated with radical cystectomy, many patients may prefer bladder-preserving approaches. 21
Care for patients with cancer requires a multidisciplinary approach involving physicians, advanced practice providers, nurses, pharmacists, and other healthcare personnel working collaboratively to provide effective patient care. 22 As part of the healthcare team, pharmacists may be involved in the selection of appropriate therapy, assessing for drug-drug interactions, preparation and dispensing of medications, assisting with medication-related management of adverse events, risk factor modification (such as smoking cessation counseling), and patient counseling and follow-up. 23 In addition to direct clinical responsibilities, pharmacists may be responsible for developing operational processes and workflows, assisting with medication authorizations, product acquisition and storage. Pharmacists can also serve as a resource for inquiries regarding dosing, administration, medication interactions, adverse events, and medication-related supportive care to the care team.
Nadofaragene firadenovec for BCG-unresponsive NMIBC
Nadofaragene firadenovec-vncg (ADSTILADRIN®; nadofaragene or rAd-IFN/Syn3) is an intravesical nonreplicating adenoviral vector–based gene therapy that delivers copies of a gene encoding for an interferon alpha-2b (IFNα-2b) directly to urothelial cells, enabling production of the IFNα-2b protein that is responsible for multiple anticancer and immunomodulatory effects. 24 Nadofaragene firadenovec was approved by the FDA in December 2022 for the treatment of adult patients with BCG-unresponsive NMIBC with CIS with or without papillary tumors.10,25 Its effects are related to IFNα-2b signaling and include direct cytotoxicity, immune-mediated antitumor effects, and antiangiogenic effects. 24
In a single-arm, multicenter, open-label, repeat-dose, phase 3 study (CS-003; NCT02773849), eligible adults who met the FDA definition of BCG-unresponsive NMIBC were enrolled into one of two cohorts by diagnosis at enrollment: (1) participants with CIS with or without high-grade Ta or T1 NMIBC, or (2) participants with high-grade Ta or T1 tumors without concomitant CIS. 9 The primary endpoint was complete response rate in the CIS cohort at any time within 12 months after the first dose of nadofaragene firadenovec; no reinduction was allowed for participants without a complete response after the first dose. Overall, of 103 participants with CIS ± high-grade Ta/T1, 55 participants (53.4%) had a complete response at 3 months after the first dose. Among these 55 participants, response was maintained in 45.5% and 10.9% of patients at 12 months and 57 months, respectively.9,25 Of 48 participants in the high-grade Ta/T1 cohort, 35 participants (72.9%) were high-grade recurrence free at 3 months. Among these 35 participants, 60.0% and 20.0% remained high-grade recurrence free through 12 months and 57 months, respectively.9,25 For most participants, adverse events were transient and classified as either grade 1 or 2. No grade 4 or 5 treatment-related adverse events were reported, and there were no deaths assessed as related to administration of the study drug.9,25
Additional studies are planned to evaluate nadofaragene firadenovec in additional settings, including in combination with chemotherapy or immunotherapy (ABLE-22; NCT06545955) or in intermediate-risk NMIBC (ABLE-32; NCT06510374). An ongoing real-world noninterventional study (ABLE-41; NCT06026332) will explore early utilization, experiences, and outcomes of nadofaragene firadenovec-vncg in the routine care setting.
Nadofaragene firadenovec is administered as a 75-mL intravesical instillation once every 3 months. It represents a potential bladder-sparing therapeutic option with a novel mechanism of action in patients with BCG-unresponsive NMIBC. There is no defined treatment period for nadofaragene firadenovec and treatment duration is left to provider discretion. In the phase 3 CS-003 study, nadofaragene firadenovec was administered for an initial 12-month treatment period, with subsequent optional treatment for up to 5 years total. Optional treatment was at the discretion of the treating physician and in the absence of high-grade recurrence. Other options approved by the FDA for certain patients with BCG-unresponsive disease include valrubicin, pembrolizumab, and nogapendekin alfa inbakicept-pmln + BCG.26–28
Practical considerations for use of nadofaragene firadenovec for pharmacists
For nadofaragene firadenovec to be effective, specific attention must be paid to its transportation, storage, and administration requirements. Pharmacy staff play a vital role in several of these considerations due to their familiarity with the unique storage, handling, administration, and disposal requirements of various medications. In settings where nadofaragene firadenovec is administered, pharmacists may have a role in developing operational processes that include acquisition, authorization, preparation, and administration of nadofaragene firadenovec (Figure 1). While developing such processes, pharmacists may need to carefully consider staff roles (physicians, nursing administration, infusion center nursing staff, social workers, authorization personnel, infusion center pharmacy staff, patient assistance technicians, etc.) and site-specific needs and challenges (eg, determining locations for administration). Awareness of enrollment procedures, authorization plans, and patient assistance programs may also facilitate acquisition and administration of nadofaragene firadenovec. Authorization and billing personnel can assist with queries related to copays and patient assistance programs. Responsibilities related to treatment plans, multidisciplinary team communication, reimbursement procedures, product handling, administration, and follow-up procedures should be clearly outlined and communicated.
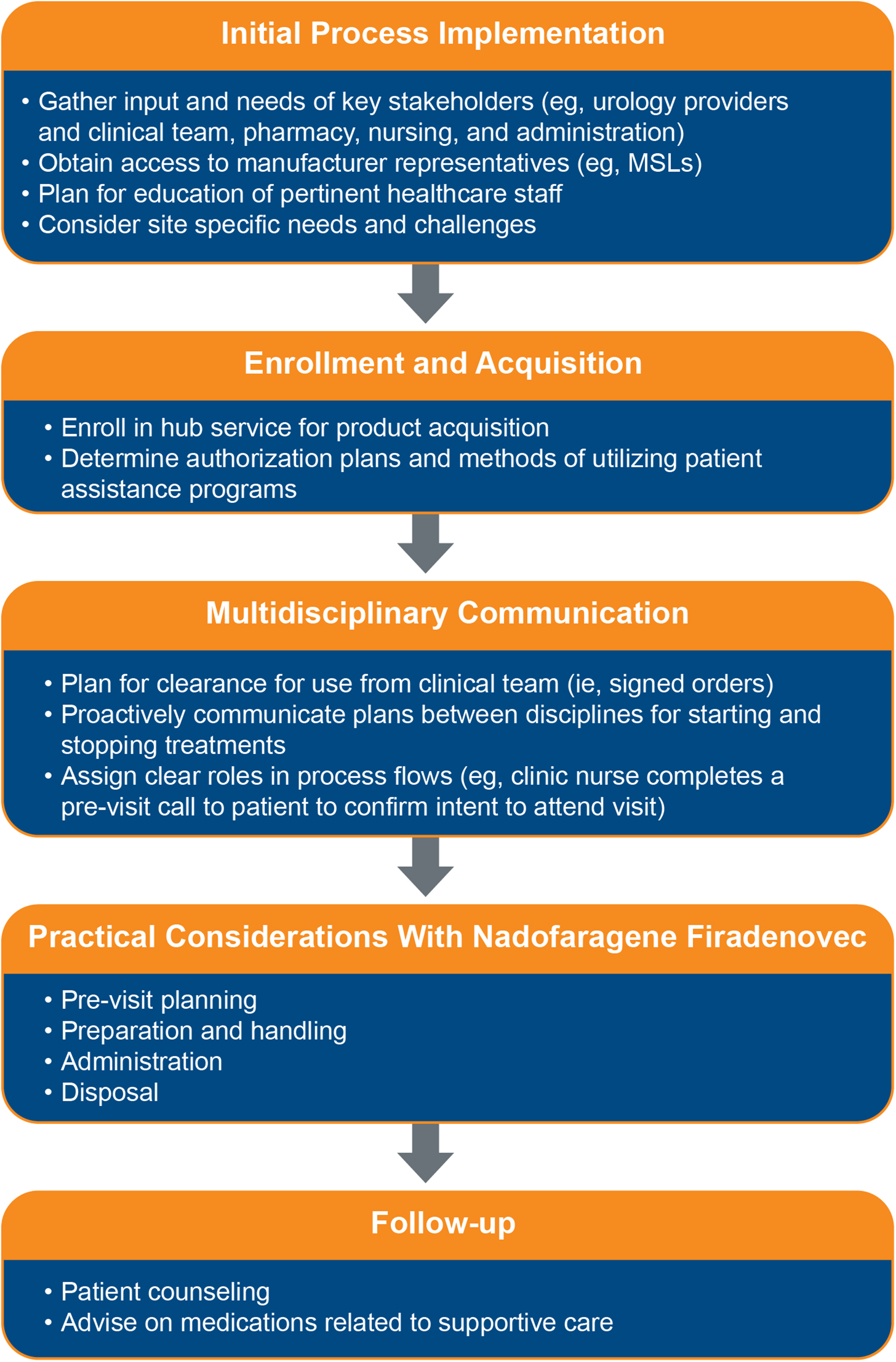
Considerations for pharmacists in acquiring and administering nadofaragene firadenovec. MSL, medical science liaison.
Role of the pharmacist prior to the patient visit
Prior to the patient visit, pharmacists should complete a chart review to proactively identify potential issues, particularly as they relate to medications that may be used for supportive care during and after instillation procedures. NMIBC often presents in older adults who are at higher risk for polypharmacy, other comorbidities, and hospitalization.29,30 The pharmacist can help ensure all prerequisites for instillation are met, such as avoiding instillation within 2 weeks of a surgical bladder procedure.
Transportation and storage considerations
As pharmacists may be responsible for medication acquisition (or advising in the process), they should be familiar with practical aspects related to procurement and delivery. For example, acquisition of medication may involve creating an account on the manufacturer website, which may take several days for initial setup. Pharmacists should ensure patients have been enrolled (via a patient enrollment form) with benefits secured. Confirm whether patients will have the medication supplied through an assistance program prior to ordering to avoid surplus inventory. Pharmacists should also note whether nadofaragene firadenovec will only be delivered on certain days of the week; they should develop quality control measures and train staff to appropriately maintain temperature requirements at all times.
Nadofaragene firadenovec is a sterile, preservative-free, clear to opalescent suspension. It is supplied in single-use vials, with each vial containing an extractable volume of not less than 20 mL. Each shipping container contains four clear glass vials of nadofaragene firadenovec, with all four vials comprising one dose. Nadofaragene firadenovec is transported in an insulated shipping container that maintains required temperatures of ≤−60°C (≤−76°F) for at least 72 h following sealing. After vials are received, they should be inspected for damage, and the cartons can then be stored in a freezer at ≤−60°C (≤−76°F) until the expiration date or from −25°C to −15°C (−13°F to 5°F) for up to 3 months without exceeding the expiration date. If storing at −25°C to −15°C (−13°F to 5°F), staff should mark the date when the product was placed in the freezer as well as a “discard by” date 3 months after the product was placed in the freezer; the discard by date supersedes the expiry date if it is earlier. 10 Not all institutions will have access to a freezer that provides appropriate storage temperatures, making this an important implementation consideration when developing nadofaragene firadenovec workflows.
Preparation and handling of nadofaragene firadenovec
Nadofaragene firadenovec should be fully thawed and brought to room temperature (20°C to 25°C [68°F to 77°F]) prior to instillation. Exposure of nadofaragene firadenovec to higher temperatures or to light should be avoided. Once vials have been thawed, they cannot be refrozen. The logistics of thaw timing in relation to patient arrival should be considered within the workflow of preparation and instillation. When thawing at room temperature, it will take approximately 8 to 10 h to thaw nadofaragene firadenovec in the cardboard nest, or approximately 3 to 5 h outside the nest. If thawing in the refrigerator (up to 8°C [46°F]), frozen vials will thaw in about 4 to 5 h outside the cardboard nest or in about 11 to 13 h inside the nest. It will subsequently take approximately 2.5 h to bring the thawed vials to room temperature outside the cardboard nest (or 6 h inside the nest). Open and proactive communication with other members of the healthcare team regarding expected instillation times is vital to reduce the overall time a patient spends waiting to receive therapy. Furthermore, clear communication reduces the risk of wasting medication because if a vial is inadvertently thawed, it cannot be refrozen. Members of clinic staff should contact pharmacy staff the day before patient arrival to confirm thawing can take place. Once thawing is initiated, vials may be stored for up to 24 h at room temperature (20°C to 25°C [68°F to 77°F]) or for up to 7 days refrigerated at 2°C to 8°C (36°F to 46°F), including thawing time. Vials should be mixed gently (without shaking) and examined for visible particles and discoloration.
The site administering nadofaragene firadenovec should ensure they are equipped with the equipment required for instillation, including four thawed vials of nadofaragene firadenovec, four vented vial adaptors suitable for a standard 20 mL vial, two standard 50 or 60 mL polypropylene Luer lock syringes or one Luer lock syringe equal to or greater than 75 mL (max 100 mL), two Luer lock adaptors, a urinary catheter, personal protective equipment, a virucidal agent, and a biohazard waste container. Only vial adapters recommended per manufacturer's instructions should be used, as alternative attachments may result in potential leakage and exposure to nadofaragene firadenovec during preparation or administration. Closed system transfer devices are not required for the preparation and administration of nadofaragene firadenovec. Aseptic technique should be used when removing the vial cap and attaching to a vented vial adaptor. Once the vial adaptor has been attached, a syringe should be connected to withdraw vial contents. The contents of the remaining three vials should also be withdrawn until a total of 75 mL has been extracted. While BCG requires a negative pressure hood for preparation due to its nature as a live virus, the preparation of nadofaragene firadenovec does not require the use of a negative-pressure hood or biological safety cabinet. Institutions may choose to utilize positive pressure hoods such as compounded aseptic isolators (CAIs) or compounded aseptic containment isolators (CACIs), or other safety controls used for nonhazardous medication preparation as available. Since there are no specific requirements for decontamination of safety controls before or after nadofaragene firadenovec preparation, institution-specific decontamination protocols may be followed.
If nadofaragene firadenovec has been drawn into two syringes, each syringe should be clearly labeled with, for example, “Dose 1 of 2,” to avoid potential underdosing. Syringe volumes do not need to be equal. Nadofaragene firadenovec can be stored in syringes for up to 6 h at room temperature (20°C to 25°C [68°F to 77°F]), protected from light. Discard any remaining volume according to universal biosafety precautions. Healthcare staff who are immunosuppressed or immune-deficient should not come into contact with nadofaragene firadenovec (including preparation and instillation) due to the potential risk for low levels of replication-competent adenovirus in the solution. Ensure appropriate training and accommodations among care team staff to allow for safe preparation and dispensing of nadofaragene firadenovec.
Product verification and medication safety for pharmacists
The dose of nadofaragene firadenovec is 75 mL at a concentration of 3 × 1011 viral particles/mL instilled once every 3 months into the bladder via urinary catheter. There are no dose adjustments recommended for renal or hepatic insufficiency, and no known medication interactions. Nadofaragene firadenovec is contraindicated in patients with prior hypersensitivity reactions to IFNα or to any component of the product; all components of the formulation can be found in the FDA package insert. 10 The product should be clear to opalescent and some opalescent flecks may be visible. Avoid using the product if discoloration or visible particles are observed. Pharmacy staff should ensure that the product is clearly labeled prior to dispensing and attach relevant auxiliary labels (eg, for intravesical use only; not for intravenous, topical, or oral use). Clinic staff should also follow medication safety best practices (eg, verification of patient, dose, route, and time) to ensure safe dispensing and instillation of nadofaragene firadenovec. 31
Administration and instillation considerations for pharmacists
Premedication with an anticholinergic before each instillation of nadofaragene firadenovec is recommended to help control urinary voiding symptoms and assist in retaining the instillation for the 1-h dwell time. 10 Dosing and use of specific anticholinergics was not standardized during the phase 3 CS-003 study, and anticholinergics were omitted per provider preference or if contraindicated. 9 Pharmacists should assist in identifying potential contraindications and suggesting appropriate medications based on patient comorbidities and concomitant medications. Follow institution-specific guidelines for management of refractory bladder spasms or irritative voiding symptoms.
Nadofaragene firadenovec should be instilled manually from the syringe into the catheter and not administered via gravity drainage or any other means of administration. Ensure that the product has been fully brought to room temperature prior to instillation as cold fluids may precipitate bladder spasms or urinary urgency. Clinic staff should confirm that any medications that may be used during and after the administration process will be readily available. While there are no hold parameters indicated in nadofaragene firadenovec labeling, individual institutions may choose to specify hold parameters; follow site-specific practices in these cases.
After instillation, nadofaragene firadenovec should be retained in the bladder for 1 h. Stress the importance of remaining at the clinic or site of administration and retaining nadofaragene firadenovec for the full 1-h dwell time. Consider repositioning every 15 min during the 1-h dwell time to maximize bladder surface exposure. If the patient exhibits bladder cramping or premature voiding, consider adjusting or discontinuing repositioning. Patients may receive supportive care for adverse events (such as pain around the instillation site, chills, and fatigue); pharmacists should ensure all prescribed supportive care medication therapy is safe and appropriate. After 1 h has elapsed, the patient should void and completely empty the bladder.
Disposal
Voided urine should be disinfected for 15 min with an equal volume of virucidal agent (eg, sodium hypochlorite with a 0.5% active chlorine or 6% hydrogen peroxide solution) for 2 days following instillation. Any nadofaragene firadenovec spills should be treated with a virucidal agent for 15 min. Disposable materials that were in contact with nadofaragene firadenovec should be placed in biohazard containers for destruction, and nondisposable equipment can be decontaminated according to an institution's standard operating procedures.
Role of the pharmacist in follow-up and patient counseling
Pharmacists may be responsible for counseling patients regarding their therapy or educating other members of the healthcare team that will provide counseling to patients. Common adverse events include leakage of fluid around the instillation site, fatigue, urinary urgency, and dysuria. Pharmacists can assist in counseling on these potential adverse events and aid in management with medication therapy and recommendations.
Patients and caregivers should be informed that treatment or contact with nadofaragene firadenovec in those who are immunocompromised or receiving immunosuppressive therapy may increase the risk for disseminated adenovirus infection. Advise on transient low-level shedding of nadofaragene firadenovec in the urine and techniques for appropriate disinfection of voided urine with a virucidal agent for 2 days following instillation. Finally, provide counseling to female patients of reproductive potential on the use of contraception during treatment and for 6 months following the last dose. Counsel male patients with female partners of reproductive potential to use contraception during treatment and for 3 months following the last dose. Emphasize the importance of therapy adherence and continued follow-up to observe for recurrence and/or progression of disease.
Summary
Nadofaragene firadenovec is the first intravesical gene therapy approved by the FDA for the treatment of adult patients with high-risk BCG-unresponsive NMIBC with CIS with or without papillary tumors. Nadofaragene firadenovec administered once every 3 months has demonstrated efficacy and a well-tolerated safety profile and allows for a potential bladder-preserving therapy option in patients who are not candidates for radical cystectomy. Pharmacists should be aware of key considerations surrounding its use, including mechanism of action, indication, handling, storage, administration, monitoring parameters, supportive care medications, disposal, and counseling. Some pharmacists may be involved in the initial operational process and workflow setup; pharmacists in these settings should also be aware of acquisition procedure and logistical considerations. Overall, implementation of these practical recommendations will allow pharmacists to confidently dispense nadofaragene firadenovec and ensure that treatment is safe and effective.
Footnotes
Acknowledgments
Medical writing support and manuscript submission, under the guidance of the authors, was provided Robin Isaac, PharmD (ApotheCom, Yardley, PA, USA), and was funded by Ferring Pharmaceuticals, Parsippany, NJ, USA. The authors authorized the third-party submission and approved any statements/declarations being made by the submitting party.
Author contributions
SDH and SMS drafted the manuscript and reviewed and approved the final version.
Consent to participate
No human participants were involved in the development of this manuscript and informed consent to participate was not applicable to obtain.
Consent for publication
This manuscript does not contain data from any individual person (including individual details, images, or videos), and informed consent to publish was not applicable.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SDH: Has served on an advisory board for Bristol Myers Squibb. SMS: Has served on a speaker’s bureau for Karyopharm and Regeneron.
Ethical considerations
Not applicable. This review article does not involve human participants, human data, or human tissue. Approval from an Ethics Committee or Institutional Review Board was not obtained.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ferring Pharmaceuticals Inc.
