Abstract
Background:
The debate regarding the optimal drainage method for acute obstructive upper urinary tract infection persists, focusing on the choice between percutaneous nephrostomy (PCN) and retrograde ureteral stenting (RUS).
Aims:
This study aims to systematically examine the perioperative outcomes and safety associated with PCN and RUS in treating acute obstructive upper urinary tract infections.
Methods:
A comprehensive investigation was conducted using the Medline, Embase, Web of Science, and Cochrane databases up to December 2022, following the guidelines of the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) statement. The utilized keywords included ‘PCN’, ‘RUS’, ‘acute upper obstructive uropathy’, and ‘RCT’. Inclusion criteria encompassed studies providing accurate and analyzable data, which incorporated the total subject count, perioperative outcomes, and complication rates. The assessed perioperative outcomes included fluoroscopy time, normalization of temperature, normalization of serum creatinine, normalization of white blood cell (WBC) count, and operative time. Safety outcomes encompassed failure rate, intraoperative and postoperative hematuria, postoperative fever, postoperative pain, and postoperative nephrostomy tube or stent slippage rate. The study protocol was prospectively registered at PROSPERO (CRD42022352474).
Results:
The meta-analysis encompassed 7 trials involving 727 patients, with 412 assigned to the PCN group and 315 to the RUS group. The outcome of the meta-analysis unveiled a reduced occurrence of postoperative hematuria in the PCN group [odds ratio (OR) = 0.54, 95% confidence interval (CI) 0.30–0.99, p = 0.04], along with a decreased frequency of insertion failure (OR = 0.42, 95% CI 0.21–0.81, p = 0.01). In addition, the RUS group exhibited a shorter fluoroscopy time than the PCN group (mean difference = 0.31, 95% CI 0.14–0.48, p = 0.0004).
Conclusion:
Given the significant impact of hematuria and catheterization failure on postoperative quality of life, the preference for PCN appears more advantageous than RUS.
Plain language summary
The optimal drainage method for acute obstructive upper urinary tract infection between PCN and RUS is currently debatable. Our meta-analysis found PCN performed better than RUS in hematuria and catheterization failure rate, although PCN was associated with longer exposure time.
Keywords
Introduction
Upper urinary tract obstruction is a typical structural or functional impedance of the urinary system, which is often secondary to various diseases, such as ureterolithiasis, malignancies, and so forth. 1 Acute obstructive uropathy can cause hydronephrosis, which provides favorable conditions for the growth of bacteria in the urinary system.2–5 While there is an increased obstruction for any reason, the intrarenal pressure will suddenly increase, accompanied by blood circulation disorder, resulting in a rapid rise of bacteria.6,7 If various bacteria enter the bloodstream without immediate treatment, it will lead to severe sepsis or septic shock.8,9 Furthermore, routine antibacterial agents make it difficult to reach lesions or achieve sound curative effects.10,11
Hence, it becomes imperative for surgeons to meticulously perform a convenient surgical procedure aimed at alleviating obstruction and concurrently reducing intrarenal pressure while also orchestrating infection control and proactively addressing the broader clinical context. 12 As primary interventions, percutaneous nephrostomy (PCN) and retrograde ureteral stenting (RUS) are widely endorsed measures for initiating drainage to mitigate urinary tract obstruction. 13 Due to the limitations of various factors, both PCN and RUS exhibit different prognoses in actual clinical treatment.12,13 RUS is a commonly used adjunctive treatment in urological surgery, exhibiting excellent efficacy in maintaining the patency of the ureter. Simultaneously, it demonstrates remarkable results in facilitating the expulsion of stones post-lithotripsy and the drainage of urine and blood, thereby alleviating pressure within the bladder and renal pelvis. However, influenced by various factors, there is a varying degree of increased risk of urinary tract infections in patients. This elevated risk may even directly impact the clinical prognosis. During RUS interventions, introducing contrast material for enhanced visualization of the ureter via an angiographic sheath remains feasible. The re-establishment of patency in an obstructed ureter is generally attainable using a guidewire. Nevertheless, instances might arise where navigating a wide-bore JJ catheter through a mildly constricted ureteral obstruction proves challenging. 13 RUS also gives rise to mild hematuria and ascending urinary tract infections due to urothelial irritation. PCN is widely utilized for relieving upper urinary tract obstruction due to its advantages of minimal trauma, simplicity in surgical procedures, and reduced radiation exposure. The refinement and development of interventional and ultrasound technologies have significantly lowered the risks associated with PCN, allowing patients with the aforementioned condition to receive safe and effective treatment through minimally invasive procedures. PCN, typically conducted under the guidance of ultrasound and/or fluoroscopy, employs simple equipment comprising puncture needles, guide wires, and catheters. Notably, sepsis emerges as the most frequent and severe systemic complication following PCN, warranting escalated care and, in severe cases, bearing the potential for fatal outcomes. Concurrently, bleeding represents the most prevalent local complication associated with PCN. 14
Our study objective was to systematically review and conduct a meta-analysis that juxtaposes PCN and RUS, collating comprehensive evidence from randomized controlled trials (RCTs) concerning these two surgical procedures in the management of acute obstructive upper urinary tract infections.
Methods
Search strategy
The Medline, Embase, Web of Science databases, and Cochrane Controlled Trials Register databases were searched up to 20 December 2022. The keywords employed for the search included ‘PCN’, ‘RUS’, ‘acute upper obstructive uropathy’, and ‘RCT’. Only English-language articles were included. Cited references from the included literature were also searched for relevant studies. The databases were explored employing the subsequent search terms within titles and abstracts (alongside combinations with terms): [(double-J stent) OR (DJS) OR (ureteral stents) OR (double J ureteric stent) OR (bilateral JJ) OR (JJ ureteric stent) OR (JJ ureteric stent) OR (double J Ureteral Stenting) OR (DJ Stenting) OR (retrograde ureteral catheterization) OR (JJ ureteric stent) OR (JJ stent) OR (JJ stent)] AND [(percutaneous nephrostomy) OR (nephrostomies, percutaneous) OR (percutaneous nephrostomies)] AND (upper urinary tract obstruction) AND (urinary tract infection). Two authors independently screened all abstracts and full-text articles.
Inclusion and exclusion criteria
We conducted a comprehensive search and meticulously evaluated relevant studies based on their titles and abstracts. Two reviewers carried out this assessment process independently, and any discrepancies in the outcomes were resolved through their consensus. Studies published as review articles and conference abstracts were excluded from consideration. Our focus was solely on RCTs that compared the effectiveness of PCN and RUS in treating acute obstructive upper urinary tract infections. The selected studies were required to present accurate data suitable for analysis, encompassing the subject count and the outcomes of each complication rate. Studies not meeting the RCT criteria and those inaccessible in full-text form were excluded from our assessment. A visual representation of the study selection process, following PRISMA guidelines, is presented in Figure 1.
Flow diagram of the study selection process.
Data extraction
Two reviewers independently screened the literature according to the inclusion and exclusion criteria (Figure 1). The following data were recorded: author name, year of publication, type of study design, country, patient number in each group, time to normalization of temperature, time to normalization of creatinine, time to normalization of white blood cells (WBCs), insertion failure, operative time, postoperative fever, fluoroscopy time, postoperative hematuria, postoperative analgesic application, and postoperative nephrostomy tube or stent displacement.
Outcome measures
The primary outcome measures of the review were time to normalization of temperature, time to normalization of creatinine, and time to normalization of WBC. A secondary outcome was fluoroscopy time and operative time. Moreover, safety outcomes were evaluated in terms of complications, including the incidence of insertion failure, postoperative hematuria, postoperative fever, postoperative analgesic application, and postoperative nephrostomy tube or stent displacement. The normal serum creatinine level was set as less than 2 mg/dL, the normal temperature was set as less than 37.3°C in the present study. The failure rate was defined as the incidence of failure of insertion in the JJ group or PCN group.
Statistical analysis
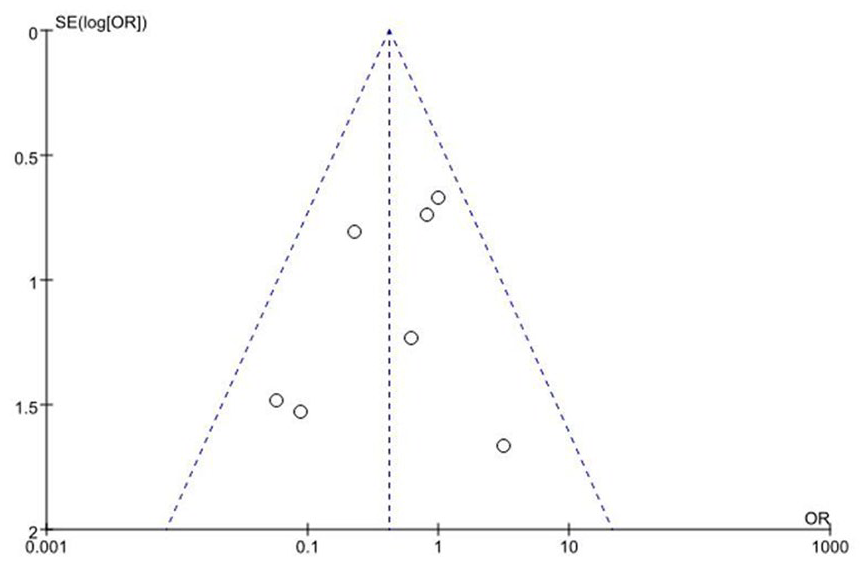
The data included was analyzed using Review Manager software (version 5.3). For continuous variables, the mean difference (MD) and 95% confidence interval (CI) were used to analyze continuous variables. We performed statistical analysis for the dichotomous variables with a pooled odds ratio (OR), and the relative effect was calculated with a 95% CI. Given the heterogeneity between studies, a fixed-effect model (Mantel–Haenszel method) was used in the absence of statistical heterogeneity (p > 0.05), and a random-effect model was used in the presence of statistically significant heterogeneity (p < 0.05). The chi-square test was employed to calculate p values. In addition, a p value < 0.05 was considered statistically significant. Funnel plot analysis has been carried out to examine potential publication biases and log OR was plotted against its standard error. A p value less than 0.05 by funnel plots was considered a statistically significant publication bias.
Assessment of risk of bias
The risk of bias assessment is shown in Figure 2(a) and (b). Begg’s funnel plot was used to evaluate the publication bias of the identified studies in this review.

(a) Risk of bias graph: authors’ judgment on each bias risk in all included studies. (b) Risk of bias assessment for studies included in the systematic review.
Results
Study identification and selection
We refrained from incorporating conference abstracts, case reports, editorials, and reviews, as their methodological appropriateness was lacking. The initial exploration of the search databases yielded 1764 articles, with 91 duplicates subsequently eliminated. Following an assessment of the abstracts and titles of the remaining 1673 articles, 1661 were excluded due to limited relevance to the scope of the review. Ultimately, for the conclusive analysis, seven RCTs were encompassed within this review (Figure 1).
Characteristics of the included studies
Eventually, 7 RCTs recruiting 727 patients were included. The characteristics of these trials are summarized in Table 1. Seven studies were selected for the review, including 412 PCN cases (56.67%) and 315 RUS cases (43.33%).
Baseline characteristics of included studies in meta-analysis.

NR, no report; PCN, percutaneous nephrostomy; RCT, randomized controlled trial; RUS, retrograde ureteral stenting; WBC, white blood cell; Fr, French size.
Risk of bias in the included studies
Out of the seven RCTs included in this review, six were conducted as single-center trials. Furthermore, in two out of the seven studies, allocation concealment was insufficiently addressed. However, blinding of both participants and personnel was implemented across all the included studies. Last, the analysis of the funnel chart exhibited no discernible evidence of publication bias (Figure 3). The plot demonstrated a high degree of symmetry, with seven squares enclosed within the larger triangle, signifying the absence of bias (Figure 3).
Risk of bias graph and summary for the included studies.
Perioperative outcomes
Fluoroscopy time
Four of the seven studies were available to evaluate data for fluoroscopy time.15–18 No significantly high statistical heterogeneity was detected in the analysis of fluoroscopy time, and a fixed-effect model was applied to process data (p = 0.19, I2 = 37%). However, the fluoroscopy time was relatively shorter in the RUS group [MD = 0.31, 95% CI 0.14–0.48, p = 0.0004; Figure 4(a)].

Forest plots showing changes in clinical resolution. (a) Fluoroscopy time in patients treated with PCN and RUS. (b) Time to normalization of temperature in patients treated with PCN and RUS. (c) Time to normalization of serum creatinine in patients treated with PCN and RUS. (d) Time to normalization of WBC in patients treated with PCN and RUS. (e) Operative time in patients treated with PCN and RUS.
Normalization of temperature
Three of the seven studies were available to evaluate data for time to normalization of temperature.17–19 There was significantly high statistical heterogeneity in the analysis of time to normalization of temperature, and a random-effect model was applied to process data (p < 0.0001, I2 = 89%). There was no significant difference between PCN and RUS in terms of time to normalization of temperature [MD = −0.77, 95% CI −1.69 to 0.14, p = 0.10; Figure 4(b)].
Normalization of serum creatinine
Two of the seven studies were available to evaluate data for time to normalization of serum creatinine.15,16 No significantly high statistical heterogeneity was detected in the analysis of time to normalization of temperature and a fixed-effect model was applied to process data (p = 0.88, I2 = 0%). There was no significant difference between PCN and RUS in terms of time to normalization of serum creatinine [MD = 0.03, 95% CI −0.23 to 0.28, p = 0.84; Figure 4(c)].
Normalization of WBC
Two of the seven studies were used to evaluate data for the time to normalization of WBCs.17,18 No significantly high statistical heterogeneity was detected in the analysis of time to normalization of temperature, and a fixed-effect model was applied to process data (p = 0.19, I2 = 41%). There was no significant difference between PCN and RUS in terms of time to normalization of WBC [MD = −0.03, 95% CI −0.39 to 0.32, p = 0.85; Figure 4(d)].
Operative time
Four of the seven studies were available to evaluate data for operative time.15–18 There was significantly high statistical heterogeneity in the analysis of operative time, and a random-effect model was applied to process data (p < 0.00001, I2 = 98%). The operative time was not found to vary between PCN and RUS [MD = 2.97, 95% CI −4.39 to 10.33, p = 0.43; Figure 4(e)].
Safety
Failure rate
Six of the seven studies were available to evaluate data for catheterization failure.15–21 No significantly high statistical heterogeneity was detected in the analysis of catheterization failure, and a fixed-effect model was applied to process data (p = 0.19, I2 = 32%). In the general analyses, PCN provided a lower rate of catheterization failure [OR = 0.42, 95% CI 0.21–0.81, p = 0.10; Figure 5(a)].

Forest plot showing safety outcomes evaluated in terms of complications: (a) Comparison between PCN and RUS for catheterization failure; (b) comparison between PCN and RUS for intraoperative and postoperative hematuria; (c) comparison between PCN and RUS for postoperative fever; (d) comparison between PCN and RUS for postoperative analgesic application; and (e) comparison between PCN and RUS for displacement.
Intraoperative and postoperative hematuria
Three of the seven studies were available to evaluate data for intraoperative and postoperative hematuria.15–21 No significantly high statistical heterogeneity was detected in the analysis of catheterization failure, and a fixed-effect model was applied to process data (p = 0.30, I2 = 17%). The incidence of intraoperative and postoperative hematuria was relatively lower in the PCN group [OR = 0.54, 95% CI 0.30–0.99, p = 0.04; Figure 5(b)].
Postoperative fever
Two of the seven studies were available to evaluate data for postoperative fever.18,20 No significantly high statistical heterogeneity was detected in the analysis of postoperative fever (p = 0.86, I2 = 0%), and a fixed-effect model was applied to process data. There was no significant difference between PCN and RUS in terms of postoperative fever [OR = 0.45, 95% CI 0.19–1.06, p = 0.07; Figure 5(c)].
Postoperative pain
Three of the seven studies were available to evaluate data for postoperative pain.15,16,21 No significantly high statistical heterogeneity was detected in the analysis of postoperative pain, and a fixed-effect model was applied to process data (p = 0.02, I2 = 76%). There was no significant difference between PCN and RUS in terms of postoperative pain [OR = 1.27, 95% CI 0.12–13.30, p = 0.84; Figure 5(d)].
Postoperative nephrostomy tube or stent slippage rate
Four of the seven studies were available to evaluate data for postoperative nephrostomy tube or stent slippage rate.15–21 No significant statistical heterogeneity was detected in the analysis of postoperative nephrostomy tube or stent slippage rate (p = 0.73, I2 = 0%), and a fixed-effect model was applied to process data. There was no significant difference between PCN and RUS regarding the postoperative nephrostomy tube or stent slippage rate [OR = 2.88, 95% CI 0.95–8.75, p = 0.06; Figure 5(e)].
Discussion
Upper urinary tract obstruction, whether caused by stones or other factors, presents an emergent scenario necessitating prompt decompression of the collecting system. 22 The effectiveness of both PCN and retrograde ureteral catheterization in achieving such decompression has been unequivocally established. 13 In a retrospective study encompassing 1712 patients experiencing sepsis due to stone obstruction, 78% underwent PCN or RUS. The ultimate findings indicated a notably elevated mortality rate among patients who did not receive decompression compared to the control group during their hospitalization. 23
In this meta-analysis, we have synthesized a total of seven pieces of literature. Within the encompassed RCTs, certain statistical measures are presented in terms of medians and quartiles, while others employ estimated means and SDs. We made concerted efforts to obtain more granular raw data about specific clinical outcome indicators, with the intent of presenting them in the format of estimated means and SDs. Unfortunately, in certain trials, detailed original information remained elusive. It is one of our intention to compare mortality rates in analysis. However, no relevant analysis of mortality outcomes following both treatment modalities was identified in all the studies included. Only in the included study of Rammohan et al. was PCN shown to decrease mortality associated with Gram-negative sepsis due to obstruction of the urinary tract (7.4% mortality) compared to treatment with antibiotics and steroids alone (40%) or surgical decompression.
Compared to RUS, PCN demands a lengthier radiation exposure time. However, concerning urinary tract-related symptom outcomes, the findings favor PCN. Specifically, postoperative hematuria and catheterization failure incidence were higher within the RUS group. With an escalating degree of hematuria, a noticeable augmentation in diagnostic yield for potentially life-threatening lesions becomes evident. Whether characterized as gross or microscopic, hematuria stands as a significant diagnostic indicator warranting assessment from the perspectives of risk-benefit analysis and cost-effectiveness. However, within typical clinical practice, establishing a safe lower threshold for hematuria proves challenging due to numerous variables that are often intricate to control. The lack of comprehensive supplementary information impedes our ability to gain a nuanced understanding of hematuria extent or severity. Consequently, a heightened degree of clinical suspicion is crucial for the accurate evaluation of hematuria. 24
In clinical practice, the inability to catheterize successfully necessitates either reoperation or a change in the chosen procedural approach, thereby imposing an augmented psychological and economic burden upon patients. 17 Such outcomes have the potential to significantly impact the decision-making process in regard to selecting appropriate treatment avenues.
In terms of essential postoperative factors, such as temperature recovery time, creatinine recovery time, postoperative pain, and stent displacement, no noteworthy distinctions emerged between the two groups. This observation, naturally, could be correlated with the patient’s initial body temperature and biochemical profiles. It is worth noting that a few of the included trials lack precise original data to definitively identify patients who were ultimately included based on their primary symptoms. This non-standardized approach introduces complexities that can influence the precision and interpretability of the analysis, especially in cases of variables such as the normalization of temperature and postoperative fever. Nevertheless, these outcomes are crucial and warrant consideration, given that the elevation of inflammatory markers and renal damage indices can significantly impede patient well-being.
It has been reported that in patients with obvious hydronephrosis, the success rate of PCN is close to 99%. 25 The success rate of RUS is 98%. 26 There was no difference in the overall incidence of complications between RUS and PCN. RUS achieves the decompression of obstructed collecting systems through the body’s natural cavities, offering patients enhanced comfort and reduced risk of mechanical injury compared to PCN. 27 Furthermore, RUS could hold promise as an adjunct to subsequent lithotripsy treatment for individuals with ureteral strictures and urinary calculi. However, within the context of clinical practice, the complexity of implementing RUS intensifies if patients present with multiple or larger stones or if exogenous compression leads to hydronephrosis, exacerbating the challenges associated with RUS. 28 Urinary tumors, notably bladder and prostate cancer, represent risk factors contributing to the failure of RUS catheterization. As a result, the resultant obstructions and drainage disorders present intricate challenges that prove resistant to resolution through RUS. 29 In addition, in cases where acute upper urinary tract obstruction results from multiple ureteral strictures and distortions due to tumor invasion and retroperitoneal fibrosis, the failure rate of RUS escalates. In such scenarios, PCN should be prioritized as the preferred initial choice. Since only Ahmad’s experiment analyzed the two drainage methods according to the etiology of upper urinary tract obstruction, there is insufficient research to analyze the differences in both drainage methods according to the causes of obstructive uropathy.
In our previous attempts, we tried to conduct subgroup analyses by stratifying the included literature based on age and gender to reduce heterogeneity in the results. However, in a significant proportion of the included literature, specifically the study by Ahmad, only the gender ratio for the total population was provided. The study did not stratify age and gender according to the PCN and RUS groups. The remaining included studies also did not provide age and gender stratification for different outcomes. In this study, we included a randomized controlled study conducted by Elsheemy et al. 16 in pediatric patients. The study reported that patients treated with PCN returned to normal serum creatinine levels faster than those treated with RUS and that the placement of PCN required general anesthesia in children, which affected other indicators, such as operation time. Therefore, more and higher quality special population control studies are needed to confirm this conclusion. Goldsmith et al. reported that PCN and RUS seem to be effective in drainage in patients with obstructive urinary calculi and infection. 30 Pearle et al. obtained the same findings in a cohort of 42 patients with ureteral stone obstruction and infection. They concluded that the choice of drainage might depend on the surgeon’s preference and the characteristics of the stone. 17
In addition, a systematic review of six trials compared the role of PCN and RUS in the treatment of ureteral obstruction caused by benign and malignant causes. 31 The results showed that there was no significant difference in clinical improvement between PCN and RUS. However, PCN is associated with a higher risk of postoperative bacterial colonization. At the same time, the hospitalization time of patients receiving RUS was significantly shorter than that of patients receiving PCN, but PCN was better than RUS in terms of postoperative hematuria and dysuria. Our meta-analysis was consistent with some of the literature results.
PCN can be conveniently carried out at the bedside and administered under local anesthesia. By contrast, RUS presents several drawbacks. Individuals with severe ureteral obstructions or strictures are susceptible to treatment failure and may necessitate a transition to PCN. Second, RUS has the potential to elevate renal pelvic pressure, exacerbate inflammation, and augment the likelihood of bacterial colonization, except for patients afflicted by coagulation disorders. Notably, PCN serves as an emergent intervention for Gram-negative urosepsis, with RUS being considered more suitable as an alternative procedure. 32 Consequently, our recommendation leans toward PCN over RUS in cases involving uremic patients with obstructive infections.
There are some limitations in this meta-analysis. First, we conducted a thorough and extensive database search, including seven experiments comparing PCN and RUS. In the included trials, stone diseases constituted the predominant causative factor behind the observed obstructions. Consequently, this particular observation may not be universally applicable to the broader spectrum of obstructive urinary tract conditions, which encompass alternative etiologies such as exogenous obstruction arising from malignant tumors or bladder-related issues resulting from secondary neurogenic bladder complications. Second, certain outcomes within this analysis are constrained by the availability of data sourced exclusively from two studies, including creatinine recovery time, leukocyte recovery time, and postoperative fever.
Insufficient intervention allocation had a significant influence on the potential for bias. The incorporation of Mokhmalji’s research, 21 which employed the unconventional grouping of patients based on odd or even birth years, introduces an added element of potential selection bias. Furthermore, it is important to acknowledge that our analysis involving the included 90 pediatric patients might inadvertently obscure the distinctive anatomical and metabolic characteristics specific to children. As a result, the comparability of Elsheemy’s research across studies within our analysis may be compromised. Moreover, the meta-analysis encompassing normalization of WBCs, normalization of creatinine, and postoperative fever is based on just two available studies. Due to the scarcity of data in the literature, these findings warrant cautious consideration.
It is worth noting that the results are influenced by factors such as limitations in medical equipment and operator experience, contributing to some degree of heterogeneity and introducing a certain level of bias in this study. In light of these considerations, it is imperative to emphasize that a more comprehensive understanding of the perioperative outcomes and safety associated with PCN and retrograde ureteral stent implantation in the management of acute obstructive upper urinary tract infections requires additional long-term prospective RCTs. For different primary diseases, the ICU admission rates vary between the two treatment methods. Currently, there is a lack of large-scale epidemiological studies discussing the ICU admission rates for both. However, in a prospective study cohort of 76 patients with urosepsis, 33 one-third of the patients were admitted to ICU with ventilatory support. There is no difference in outcomes for RUS or PCN or whether the patient was admitted to the ICU or the ward.
In summary, our analysis revealed no substantial disparities in most prognostic indicators between PCN and RUS. Both approaches exhibit safety and commendable success rates. In practical medical contexts, our recommendation leans toward prioritizing PCN over RUS, attributed to its limited contraindications and relatively straightforward implementation.34,35 The enduring negative impact on the patient’s quality of life stemming from hematuria and implantation failure underscores the importance of this preference.
Conclusion
There was no significant difference between PCN and RUS in most clinical outcomes. However, considering the influence of hematuria and catheterization failure on postoperative quality of life, opting for PCN appears more favorable than opting for RUS.
