Abstract
Background:
Novel receptor tyrosine kinase inhibitors and immune checkpoint inhibitors have been introduced to the treatment of advanced renal cell carcinoma (aRCC) during the past decade. However, the adoption of novel treatments into clinical practice has been unknown in Finland.
Objectives:
Our aim was to evaluate the use of systemic treatments and treatment outcomes of aRCC patients in Southwest Finland during 2010–2021.
Design and Methods:
Clinical characteristics, treatments for aRCC, healthcare resource utilization, and overall survival (OS) were retrospectively obtained from electronic medical records. Patients were stratified using the International Metastatic RCC Database Consortium (IMDC) risk classification.
Results:
In total, 1112 RCC patients were identified, 336 (30%) patients presented with aRCC, and 57% of them (n = 191) had received systemic treatment. Pre-2018, sunitinib (79%) was the most common first-line treatment, and pazopanib (17%), axitinib (17%), and cabozantinib (5%) were frequently used in the second-line. Post-2018, sunitinib (52%), cabozantinib (31%), and the combination of ipilimumab and nivolumab (10%) were most commonly used in the first-line, and cabozantinib (23%) in the second-line. Median OS for patients with favorable, intermediate, and poor risk were 61.9, 28.6, and 8.1 months, respectively. A total of 73%, 74%, and 35% of the patients with favorable, intermediate, and poor risk had received second-line systemic treatment. In poor-risk patients, the number of hospital inpatient days was twofold higher compared to intermediate and fourfold higher compared to favorable-risk patients.
Conclusion:
New treatment options were readily adopted into routine clinical practice after becoming reimbursed in Finland. OS and the need for hospitalization depended significantly on the IMDC risk category. Upfront combination treatments are warranted for poor-risk patients as the proportion of patients receiving second-line treatment is low.
Registration:
Clinical trial identifier: ClinicalTrials.gov NCT05363072.
Plain Language Summary
The aim of the study was to evaluate the use of novel medical treatments for advanced kidney cancer in routine clinical practice in Southwest Finland from 2010 to 2021 and to study the impact of IMDC risk factors on patients’ survival and healthcare resource utilization.
Before 2018, sunitinib (79%) was the most common first-line treatment for advanced kidney cancer, and pazopanib (17%), axitinib (17%), and cabozantinib (5%) were frequently used in the second-line. After 2018, sunitinib (52%), cabozantinib (31%), and the combination of ipilimumab and nivolumab (10%) were most commonly used in the first-line, and cabozantinib (23%) in the second-line treatment.
The IMDC risk category predicted the patient’s prognosis accurately as the median overall survival times for patients with favorable, intermediate, and poor risk were 61.9 months, 28.6 months, and 8.1 months, respectively. 73–74% of the patients with favorable and intermediate risk had received second-line medical treatment for advanced disease, whereas only 35% of the patients with poor risk had received second-line treatment after disease progression on the first-line treatment. Among patients with poor risk, the number of hospital inpatient days was twofold higher compared to intermediate and fourfold higher compared to favorable-risk patients.
This study demonstrated that new treatment options for advanced kidney cancer were readily adopted into clinical practice and IMDC risk scoring was a valuable tool in determining patient prognosis and healthcare resource utilization.

Keywords
Background
Renal cell carcinoma (RCC) is the third most common urogenital cancer after prostate and bladder cancer with an annual incidence of 18 per 100,000 people and accounting for nearly 3% of all cancer-related deaths in Finland in 2020. 1 The course of advanced RCC is heterogeneous ranging from asymptomatic, small lung metastases to widespread disease with, for example, symptomatic bone or brain metastases. The International Metastatic RCC Database Consortium (IMDC) risk classification is still the only prognostic algorithm available in clinical practice. 2 Favorable-risk patients (0 risk factors) have long survival with antiangiogenic receptor tyrosine kinase inhibitors [median overall survival (OS) 43.2 months], whereas intermediate- (1–2 risk factors) and poor-risk (⩾3 risk factors) patients have shorter survival with the median OS of 22.5 and 7.8 months, respectively. 3
The treatment of advanced RCC (aRCC) has evolved from antiangiogenic receptor tyrosine kinase inhibitor (TKI) monotherapy to combination therapies involving immune checkpoint inhibitors (ICI) and TKIs during the last 10 years. 4 In clinical trials, combination therapies have prolonged OS compared to sunitinib especially among patients with intermediate- and poor-risk factors. 4 In Finland, IMDC criteria did not guide the choice of systemic treatment before 2018, as TKIs were available for all patients regardless of the IMDC risk category. The first-line reimbursement of cabozantinib in 2018 and ipilimumab–nivolumab in 2019 has changed the treatment of intermediate and poor-risk aRCC in routine clinical practice in Finland. TKI monotherapy with sunitinib or pazopanib still remains the standard first-line treatment option for favorable-risk patients. In Finland, first-line ICI-TKI treatment has been available in routine clinical practice only after 2021 for intermediate- and poor-risk patients.
The aim of the study was to utilize electronic medical records (EMRs) from the Hospital District of Southwest Finland (HDSF) to evaluate the use of systemic treatments for aRCC in routine clinical practice from 2010 to 2021. Treatment outcomes including OS and time to next treatment (TTNT), as well as healthcare resource utilization (HCRU), were analyzed according to IMDC risk categories.
Materials and methods
All patients with the diagnosis of RCC (The International Classification of Diseases, 10th Revision; ICD-10; code C64.88) were retrospectively identified from the EMRs of HDSF from 1 January 2003 to 31 December 2021. The HDSF is serving about 500,000 patients each year. The HDSF data lake provides a centralized database enabling the linking of structured and unstructured healthcare information using a unique 11-digit national identification (ID) code. Using the ID code, the modern data lake systems set up in hospitals can combine and harmonize various patient record systems used in clinical practice to one continuously or daily updated data source, which can be utilized for scientific studies.
This study focused on patients with aRCC from 1 January 2010 to 31 December 2021. Advanced disease was determined based on any of the following criteria: ICD-10-diagnosis code for metastasis (C77*−C79*), American Joint Committee on Cancer stage 4, a recorded visit to an oncologist (specialty code 65 Y), radiation therapy for metastasis (procedure code WF049), or the initiation of systemic treatment for aRCC (sunitinib, pazopanib, sorafenib, axitinib, cabozantinib, everolimus, ipilimumab, or nivolumab). The patient selection flowchart is described in Supplemental Figure 1. The study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
Patient stratification according to IMDC risk category
Patients with aRCC were stratified into risk categories [favorable (0 risk factors), intermediate (1–2 risk factors), and poor (3–6 risk factors)] according to IMDC criteria. 5 As the IMDC risk category was not structurally available in the EMRs for all patients, we calculated the IMDC risk category based on the criteria described below and in Table 1. Each fulfilled component increased the patient’s IMDC score by one point. The IMDC risk factors were as follows:
Less than 1 year from the initial RCC diagnosis to the initiation of treatment for aRCC.
Performance status* <80% (Karnofsky) (i.e. ECOG performance status >1).
Hemoglobin* below lower limit normal (women < 117 g/l; men < 134 g/l).
Serum calcium* above upper limit normal (>2.51 mmol/l).
Neutrophils* above upper limit normal (>6.3 × 109/l).
Thrombocytes* above upper limit normal (>350 × 109/l).
*For Eastern Cooperative Oncology Group (ECOG) performance status, data from ±3 months around the treatment initiation and for laboratory values data from 3 months before the treatment initiation were utilized. If multiple records were available, the record closest to the treatment initiation was utilized.
IMDC risk categories and scoring utilized in this study.

IMDC, International Metastatic RCC Database Consortium; RCC, Renal cell carcinoma.
Demographic and clinical characteristics
Baseline demographic and clinical characteristics including patient’s age, sex, performance status, Charlson comorbidity index (CCI), and laboratory test results were obtained from EMRs at the time of the initiation of first-line treatment for aRCC. Histological subtype (clear cell, papillary, chromophobe) was reported without time limitations. CCI was computed from baseline ICD-10 codes except the codes C64*, C77*, C78*, C79*, and C80*. 6 All systemic treatments available in Finland for advanced RCC in routine clinical practice during the study period were collected (sunitinib, pazopanib, sorafenib, axitinib, cabozantinib, everolimus, nivolumab, and ipilimumab combined with nivolumab). Descriptive methods were used in reporting demographical and clinical characteristics: median and range were reported for continuous variables and frequencies and proportions for categorical variables.
Treatment lines – Sankey plots
Treatment lines visualized in Sankey plots present the order of the first three treatments given. Treatment lines were constructed based on prescriptions for the systemic treatment of aRCC. If there was a difference of >2 years between the same medication, they were considered as two separate treatment lines. Otherwise, multiple prescriptions for the same medication were combined into one treatment line. Treatment line visualization was stratified based on patients’ index year [defined as pre-2018 (2010–2017) and post-2018 (2018–2021)] because of changes in the treatment practices.
Treatment outcomes
Treatment outcomes were assessed from the initiation of systemic treatment to all-cause death (OS) or to the initiation of the next systemic treatment or death (TTNT) using the time-to-event Kaplan–Meier method with 95% confidence intervals (CI). The patients who reached the end of follow-up before the event were censored at the end of follow-up (31 December 2021). Treatment outcomes were stratified according to the IMDC risk categories. The patients with unknown IMDC risk categories were left out of survival analyses.
HCRU calculations
HCRU was computed based on the contacts at specialized healthcare during the study follow-up period. The number of contacts was divided by the sum of patients’ follow-up length to report HCRU as the number of contacts per patient-year (PPY). 95% CIs were computed using bootstrapping with 10,000 samples. The HCRU calculations were stratified according to IMDC risk category and contact type. The patients with unknown IMDC risk category were left out of the HCRU analyses.
Statistical analyses
Statistical analyses and visualizations were performed using R version 4.0.37 and GraphPad Prism version 8.0.0 for Windows (GraphPad Software, Inc., California, USA). Only existing data were used, with no imputation of missing values. The proportion of missing values was reported where applicable. Small patient groups (frequencies below 5) have not been reported in detail because of the possibility of identifying individuals. Thus, they were replaced with ‘<5’, and the corresponding proportions were not reported except survival estimates. P-values <0.05 are considered significant.
Results
Clinical characteristics
We identified 1112 patients with newly diagnosed RCC between 1 January 2010 and 31 December 2021 at the HDSF. A total of 336 patients (30%) had locally advanced or metastatic disease (aRCC). Of the aRCC patients, 191 (57%) had received systemic treatment for aRCC. The patients who had not received systemic treatment for aRCC were on average 7.9 years older (the median age 76.9 versus 69.0 years) and had more commonly impaired performance status (ECOG performance status 3–4: 28.6% versus 7.1%) compared to patients with systemic treatment for aRCC. The baseline clinical characteristics of the patients with systemic treatment for aRCC are described in Table 2. The patients in this study population were predominantly male (64.4%) and the median age at the initiation of systemic treatment was 69 years. A total of 91% of the patients with the information on histological subtype available had clear cell RCC, although histological subtype could not be confirmed for 34% of the patients. The IMDC risk category could be calculated for 89% of the patients, with most patients having either intermediate (47.6%) or poor (43.5%) risk. The first line of systemic treatment was initiated with sunitinib in 67.5%, cabozantinib in 13.1%, pazopanib in 12.6%, and ipilimumab–nivolumab in 4.2% of the patients during the whole study period.
Baseline clinical characteristics of the 191 aRCC patients who received systemic treatment.

aRCC, advanced renal cell carcinoma; ECOG, Eastern Cooperative Oncology Group; IMDC, International Metastatic RCC Database Consortium; RCC, renal cell carcinoma.
Systemic treatments for advanced RCC pre- and post-2018
Pre-2018, most patients received sunitinib (78.9%, n = 86) or pazopanib (17.4%, n = 19) as the first-line treatment. Second- and third-line treatments included axitinib (second-line 16.5%, n = 18; third-line 39.1%, n = 6), everolimus (second-line 11.0%, n = 12; third-line 18.2%, n = 12), pazopanib (second-line 16.5%, n = 18; third-line 13.6%, n = 9), cabozantinib (second-line 4.6%, n = 5; third-line 0%), and sorafenib (second-line 8.3%, n = 9; third-line 0%) [Figure 1(a)].

Sankey plot for the first three treatment lines of aRCC patients for (a) the initiation of systemic treatment between 2010–2017 (pre-2018) and (b) 2018–2021 (post-2018).
Post-2018, sunitinib remained the most common first-line treatment (52.4%, n = 43). Cabozantinib was used as the first-line treatment for 30.5% (n = 25) and as the second-line treatment for 23.2% (n = 19) of the patients. Post-2018, ICI were introduced into the first-line treatment with the ipilimumab–nivolumab combination (9.8%, n = 8). Nivolumab monotherapy was used in the second-line after failure on TKI therapy (12.2%, n = 10) [Figure 1(b)].
During the study period, 52.7% of poor-risk patients died before initiating second-line treatment and only 35.1% (n = 26) received subsequent systemic treatment after first-line treatment. Of the intermediate-risk patients, 16.0% died after first-line treatment and 74.1% of patients received subsequent systemic treatment after first-line treatment (Supplemental Figure 2).
Treatment outcomes
After the median follow-up of 64.8 (95% CI 51.0–90.3) months, the median OS for all aRCC patients who had received systemic treatment was 21.6 (95% CI 17.5–25.2) months. The median OS for patients with favorable, intermediate, and poor risk was 61.9 (95% CI 33.9–N/A) months, 28.6 (95% CI 21.8–40.3) months, and 8.1 (95% CI 5.5–10.4) months, respectively (Figure 2). The median OS of patients with poor risk was significantly shorter compared to patients with favorable or intermediate risk (Log-rank p < 0.0001).

Overall survival according to the IMDC risk category.
The median TTNT after initiating the first-line treatment was 24.6, 11.8, and 5.7 months, for patients with favorable, intermediate, and poor risk, respectively (Figure 3). The TTNT of patients with poor risk was significantly shorter compared to favorable-risk and intermediate-risk patients (Log-rank p < 0.0001).

TTNT according to the IMDC risk category.
Healthcare resource utilization
HCRU for patients with aRCC is presented in Figure 4 as contacts PPY stratified according to the IMDC risk category. The average number of all healthcare contacts was similar for patients with favorable, intermediate, and poor risk, 67.7, 60.6, and 79.7 PPY, respectively. Patients with poor risk had the most contacts for all categories except emergency room (ER) visits and surgeries, which were similar regardless of IMDC risk category. Notably, patients with poor risk had a twofold higher number of hospital inpatient days (12.0 PPY) compared to intermediate-risk patients (5.8 PPY) and a fourfold higher number of hospital inpatient days compared to favorable risk patients (3.1 PPY). The largest share of the total contacts originated from outpatient contacts and laboratory visits. Over half of the outpatient contacts, hospital days, and ER visits were disease specific (RCC diagnosis recorded during contact), 60.4% for favorable-, 64.2% for intermediate-, and 67.1% for poor-risk patients.
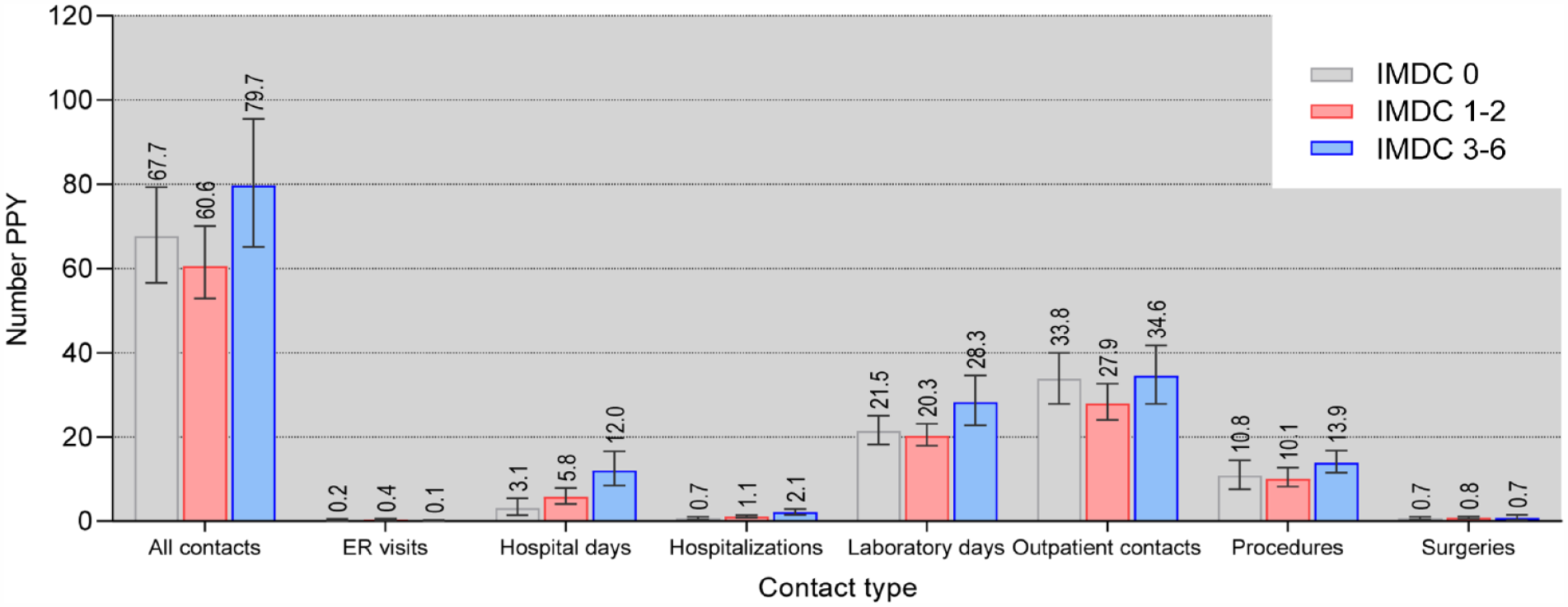
Number of aRCC patients’ healthcare contacts PPY stratified according to IMDC risk category and contact type. Average contact numbers are reported with 95% CIs.
Discussion
This Real-World Evidence (RWE) study provides insights into the demographical and clinical characteristics, treatment practices, outcomes, and HCRU of the aRCC patient population in Southwest Finland with about 500,000 inhabitants. In the present study population, clinical characteristics with a median age of 69 years, 64% of male patients, and clear cell RCC as the predominant histological subtype were similar to recent RWE studies conducted in Estonia, Denmark, and Belgium.8–10 We discovered that 43% of all patients with advanced RCC had not received any systemic treatment which is close to 46% observed in a Finnish nationwide cohort of metastatic RCC patients from 2005 to 2010. 11 These patients have likely received only palliative care due to old age, impaired performance status, or significant comorbidities.
Treatment practices for aRCC have developed rapidly within the past decade due to novel TKIs and ICI. Hence, we explored the use of systemic treatment pre- and post-2018 in the first three systemic treatment lines. The proportion of sunitinib as the first-line treatment decreased over time, while the use of cabozantinib increased in the first three treatment lines post-2018. In Finland, ICI were introduced into routine clinical practice for aRCC patients post-2018. However, only 10% of the patients had received first-line treatment with ipilimumab and nivolumab and 12.2% of the patients had received nivolumab as the second-line treatment. The current strategy to initiate upfront combination therapy with ipilimumab and nivolumab or ICI + TKI is warranted to improve treatment outcomes, as only one-third of poor-risk patients received second-line systemic treatment in our study cohort.
The prognosis after aRCC diagnosis depends on the IMDC risk category.2,5 In our study population, there were less patients with favorable risk (9%) and more patients with poor risk (44%) category as observed in Heng et al.’s original reports (favorable risk 18–23% and poor risk 26–30%).3,5 Despite these differences, it was confirmed that the mOS was highly dependent on the IMDC risk score. In the present study, the IMDC risk category could not be defined for 11% of the patients, while a previous RWE study conducted in the United States could not define the risk category for 40% of the patients. 12 RWE studies in the UK 13 and Norway 14 have highlighted improvements in the OS of aRCC patients with the introduction of targeted therapies. However, the absence of established prognostic factors, such as the IMDC risk category, in RWE studies does not provide details on OS according to risk factors. In line with the OS results, our study revealed significantly shorter TTNT for patients with poor risk compared to patients with favorable or intermediate risk. Interestingly, a recent meta-analysis revealed a potential need for an improved prognostic model after ICI have become the new treatment standard. 15
A previous study on the expenditure of aRCC management in Finland was conducted in 2010. 16 However, treatment has developed since, and new data on HCRU of the patients with aRCC are required. Our study demonstrated a high demand for healthcare resources for patients with aRCC after treatment initiation. Previously, most of the aRCC management costs in Germany resulted from outpatient pharmacy followed by inpatient costs. 17 Our study shows that management of aRCC generates a significant HCRU burden, even without including medication or indirect costs. Our study showed that the poorer risk category was associated with increased contact frequency and, especially, longer hospital stays. This finding adds evidence that poor-risk patients are more likely to have symptomatic disease progression leading to hospitalization. Interestingly, Hall et al. reported no association of IMDC risk category with hospitalization frequency – although their window of a 6-month data capture prior to treatment start may have affected this result. 12
Study strengths and limitations
One strength of the real-world data (RWD) setting is the access to diagnoses, procedures, and visits from one data source, also utilized in this study. While RWE studies have several advantages, they also have limitations including selection bias, confounding factors, generalizability, and the lack of control groups that should be considered when interpreting the results. Furthermore, some information might have been inconsistently recorded or not available at all because it has not been recorded in a structured format. As an example, the histological subtype was missing for 34% of the patients, including both patients that had no histological samples available (n = 30) or missing information on the histological subtype (n = 35). In this study, the distribution of clear cell and non-clear cell RCC subtypes among patients with structured information on histological subtypes was comparable with previously published real-world studies.8–10 Therefore, patients with missing information on histological subtypes were not excluded from further analyses. A further limitation of this study is that it consists of regional data generated during routine clinical practice at specialized healthcare facilities and stored in the HDSF data lake. Thus, patients’ contacts with primary healthcare and other hospital district facilities are missing from this study and may decrease the presented HCRU. These limitations have been addressed in the study using quality control measures to ensure that the data are complete and accurate, with the proportion of missing values reported and appropriate statistical analyses used to account for potential confounding factors or bias. These limitations are expected in studies using RWD and are unlikely to change the study conclusions. Comparing demographic and clinical characteristics to other RWE studies provides external validity for the described cohort.
Conclusion
This study demonstrated that new treatment options for aRCC were readily adopted into clinical practice and IMDC risk scoring was a valuable tool in determining the prognosis and healthcare resource utilization of patients receiving systemic treatment for aRCC. The upfront use of combination treatments is warranted for patients with poor-risk aRCC as the proportion of patients receiving second-line treatment decreased after the first-line. There is also a significant proportion of patients who are not fit enough for systemic treatment options. In addition to novel systemic treatments, it is important to integrate palliative care into the treatment of aRCC patients.
Supplemental Material
sj-docx-1-tau-10.1177_17562872231206243 – Supplemental material for Observational study on the evolution of systemic treatments for advanced renal cell carcinoma in Southwest Finland between 2010 and 2021
Supplemental material, sj-docx-1-tau-10.1177_17562872231206243 for Observational study on the evolution of systemic treatments for advanced renal cell carcinoma in Southwest Finland between 2010 and 2021 by Olivia Hölsä, Kaisa Teittinen, Anna Anttalainen, Liisa Ukkola-Vuoti, Milla Summanen and Kalle E Mattila in Therapeutic Advances in Urology
Footnotes
Acknowledgements
The authors wish to thank Mariann I. Lassenius and Johanna Vikkula for their significant input in the design and initiation of this study as well as for their support in its execution, and Kai Kysenius from Medaffcon for medical writing.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
