Abstract

Kidney cancers (including renal pelvis cancers) are expected to occur in about 81,800 patients in 2023 with an estimated mortality in 14,890. 1 The treatment of renal cell carcinoma (RCC) has rapidly evolved over the last few years and while early-stage RCC is curable with surgery, survival of patients with metastatic RCC remains poor. Clear cell RCC makes up the most common histology of all RCCs. 2 The treatment of RCC is also highly dependent on the stage of presentation and ranges from active surveillance to controversies over treatment of metastatic disease including oligometastatic treatment as well as treatment beyond frontline immunotherapy or immunotherapy and vascular endothelial growth factor inhibitor combinations. During the 2023 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, a session on multidisciplinary perspectives on challenging renal cell cancer cases encompassed topics ranging from active surveillance, treatment of oligometastatic RCC, consolidation treatment with either surgery or radiation after systemic treatment and finally, second-line systemic therapy after failure from frontline treatment of mRCC and further discussed within.
Active surveillance of localized small renal masses and RCC is a treatment approach that has evolved in the last few years, following suit with other solid tumors such as prostate cancer in which active surveillance is regarded as an increasingly viable treatment approach. This is especially relevant given known risks of treatment including perioperative morbidity and mortality and predictable decline in renal function,3,4 though this has to be balanced with potential increased risk of metastasis. 5 Several guidelines have incorporated use of active surveillance especially in patients who are infirm and otherwise not great candidates for radical nephrectomy. 6 In addition, the rates of metastatic disease in several large series of patients who underwent active surveillance showed low rates of metastatic events in about 1.8% of patients over 24–48 months,7,8 arguably similar to patients who undergo resection for small renal masses. Therefore, following a strict surveillance schedule that measures closely the growth rate of the renal mass would allow for adequate monitoring of these small renal lesions. 9 On the other hand, active surveillance in those who already developed overt metastatic RCC is also feasible in a select group of patients,10,11 especially to understand the biology and pace of the cancer.
Once treatment for metastatic RCC (mRCC) is established, several first-line therapies have been approved either in combination setting using immune-oncology (IO) drugs with IO/IO drug combination or IO with vascular endothelial growth factor/tyrosine kinase inhibitors (VEGF-TKIs). However, some patients would manifest with oligoprogression or with limited progression of metastatic disease, for whom treatment with localized surgery or radiation may be feasible. Metastasectomy as a treatment has a role in mRCC in the form of stereotactic body radiotherapy or ablative techniques, 12 especially for those who are considered to have low-volume metastatic RCC. 13 In addition, various goals of metastasectomy include palliation of symptoms, delaying the need for systemic therapy, or even improve survival for those who are able to achieve complete metastasectomy. 14 Metastasectomy can be achieved either with surgery or radiation. The use of stereotactic ablative radiation therapy (SABR) for oligometastatic renal cell carcinoma (ORCA) was evaluated in a meta-analysis of 28 studies, 15 which included 1602 mutually exclusive patients involving 3892 lesions with the potential benefit of effective delivery in lieu of systemic therapy, 16 potential delay of subsequent line of systemic treatment in true oligoprogressive disease or perhaps in combination with systemic therapy. The feasibility of a combination approach was demonstrated in a small phase I/II RAPPORT trial which enrolled 30 patients who received SABR followed by pembrolizumab for 8 cycles, 17 that resulted in complete responses in 40% of patients and one-year overall survival (OS) of 90% and progression-free survival (PFS) of 60%, suggesting feasibility of such approach. On the other hand, one trial involving 30 patients showed 1-year PFS of 64% and 22.7 months, 16 with systemic-therapy-free outcomes of 82%, suggesting the ability of local therapy to delay need for systemic treatment for a select group of patients. There have been several trials using SABR in those with oligoprogressive disease in combination with TKIs which showed median delay of next systemic therapy of up to 11.1 months 18 and 12.6 months.19,20
Another contentious issue is the role of cytoreductive nephrectomy which was established early on as beneficial in the cytokine era, 21 until more recent studies that brought into question the role of unselected front-line cytoreductive nephrectomy, 22 especially in the targeted therapy era with CARMENA 23 and SURTIME 24 trials showing no improvement in immediate versus deferred nephrectomy compared to upfront systemic therapy approach, with lack of specific guidelines in recommending delayed cytoreductive nephrectomy in patients who might derive benefit from systemic treatment. 25 Historical trials utilizing systemic therapy with VEGF-TKI and interleukin-2 included patients who have underwent prior nephrectomy rates from 67% to 100%. 26 However, more contemporary front-line therapies with IO/IO or IO/VEGF-TKI treatment often includes patients who have not undergone nephrectomy with 16.6% of the population in Keynote-426, 27 with the primary tumor in place, and 20.2% in the JAVELIN Renal 101, 28 22% in the CheckMate 214, 29 25.1% in the CLEAR trial, 30 30.1% in the CheckMate 9ER, 31 and 36% in the COSMIC-313 trial, 32 the latter of which has the most number of patients who have primary renal tumor in place and have not undergone nephrectomy. Questions regarding safety and efficacy also abound with the use of upfront nephrectomy. For instance, conflicting data show increased surgical complications, and morbidity can be seen after attempting nephrectomy in those who have undergone prior systemic therapy. In one small study including 11 patients who underwent delayed nephrectomy after first- and second-line IO therapy, 9 out of 11 patients had difficult dissection planes and a 54.6% complication rate was seen with one surgery-related mortality. 33 Another small study including five patients showed significant fibrosis and desmoplastic reaction at the time of surgical resection which necessitated increased operative time and surgical expertise. 34 On the other hand, there are other studies that showed no undue increase in complications or untoward intraoperative challenges with good pathologic response in metastatic 35 or even high-risk non-metastatic RCC. 36 In addition to the surgical techniques and feasibility, there may be a biologic rationale toward improvement in efficacy since there is upregulation of M2-like macrophages, fewer pro-inflammatory cells and more terminally exhausted CD8 cells with worsening and advanced metastatic RCC. 37 There is also a noticeably lower overall response in patients who receive systemic therapy in those who have an intact primary renal tumor compared to the overall population who have undergone nephrectomy without a primary tumor in place in both the CheckMate 214 trial, 38 and the NIVOREN trial, where there was no observed CR and only PR in the primary as best response in 6 out of 47 patients (6%). 39 A National Cancer Database (NCDB) study also showed improvement in outcomes with patients undergoing cytoreductive nephrectomy even in the contemporary IO era. 40 These trials therefore lends support to a nuanced approach of increased use of neoadjuvant systemic therapy followed by nephrectomy to help define the role of combined multimodality treatment for mRCC, of which several studies are underway (Table 1). However, surgeons should be alerted to the possibility of increased fibrosis or post-operative morbidity in those who receive pre-operative IO or combined IO/VEGF-TKI therapy.
Select phase III trials utilizing the neoadjuvant trials followed by nephrectomy.
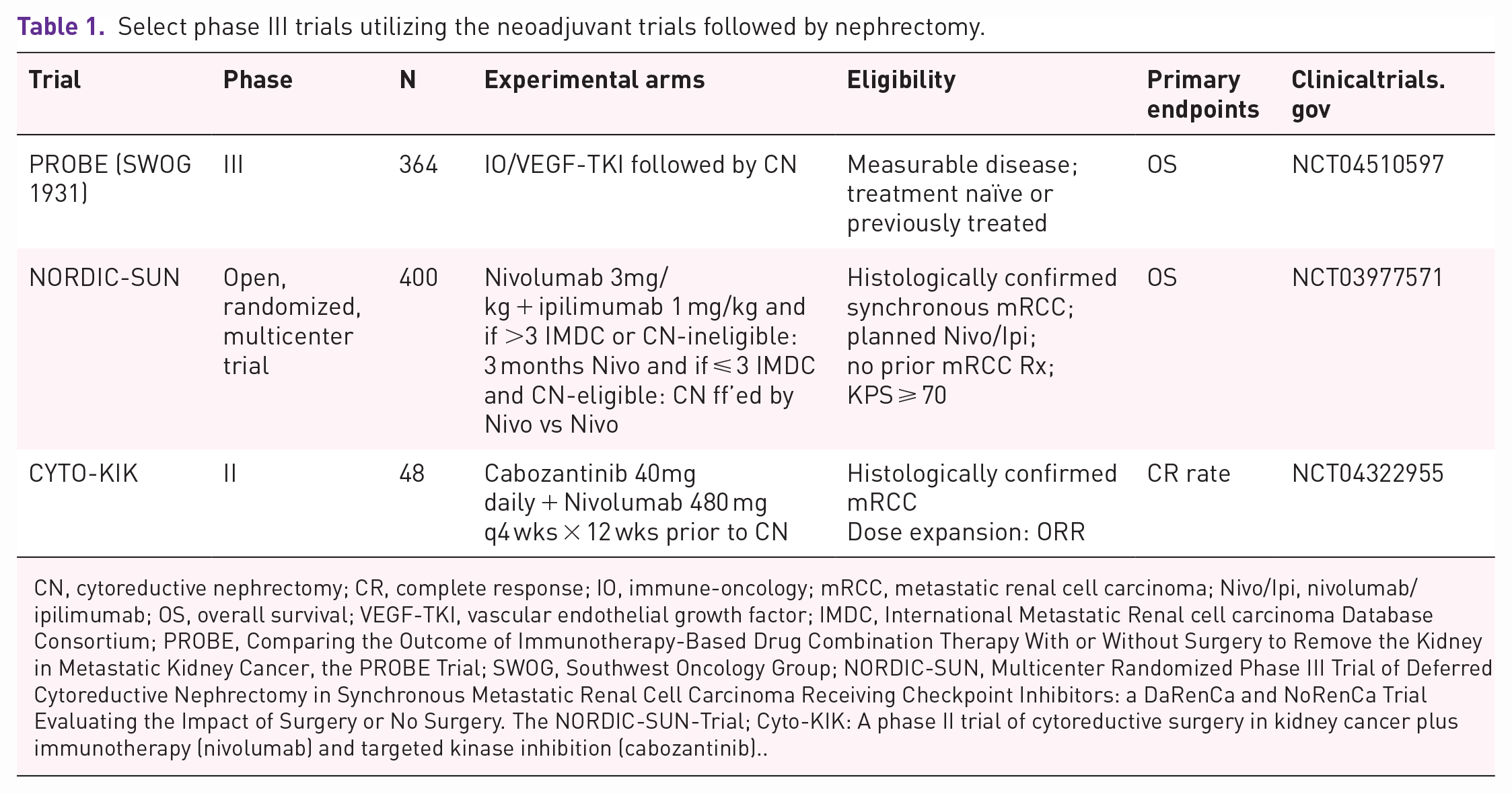
CN, cytoreductive nephrectomy; CR, complete response; IO, immune-oncology; mRCC, metastatic renal cell carcinoma; Nivo/Ipi, nivolumab/ipilimumab; OS, overall survival; VEGF-TKI, vascular endothelial growth factor; IMDC, International Metastatic Renal cell carcinoma Database Consortium; PROBE, Comparing the Outcome of Immunotherapy-Based Drug Combination Therapy With or Without Surgery to Remove the Kidney in Metastatic Kidney Cancer, the PROBE Trial; SWOG, Southwest Oncology Group; NORDIC-SUN, Multicenter Randomized Phase III Trial of Deferred Cytoreductive Nephrectomy in Synchronous Metastatic Renal Cell Carcinoma Receiving Checkpoint Inhibitors: a DaRenCa and NoRenCa Trial Evaluating the Impact of Surgery or No Surgery. The NORDIC-SUN-Trial; Cyto-KIK: A phase II trial of cytoreductive surgery in kidney cancer plus immunotherapy (nivolumab) and targeted kinase inhibition (cabozantinib)..
The other area of discussion in mRCC treatment involves consolidation therapy after frontline immunotherapy treatment. While most of the historical second-line therapies were conducted in the VEGF-TKI monotherapy setting, several trials were conducted after failure from contemporary IO/IO or IO/VEGF-TKI combination setting. Historical second-line therapies included cabozantinib based on the METEOR 41 trial and nivolumab based on the CheckMate 025 42 trial, both of which yielded an overall survival benefit over the standard of care which was everolimus at the time. Over the last several years, certain second-line therapy concepts have become increasingly clear, adaptive response-based immunotherapy with salvage ipilimumab does not yield robust effects, as shown in the HCRN-GU16-260 43 trial or TITAN-RCC 44 or OMNIVORE-RCC 45 where achievement of CR rates remain poor for all 3 trials. Recent reports of negative results with CONTACT-03 46 showed lack of robust responses in continuation of an IO, with results from a similar phase III trial TiNivo-2 (NCT04987203) that has completed accrual and eagerly awaited. On the contrary, VEGF-TKI after prior IO-based regimen or prior IO/VEGF-TKI regimen have varying response rates. Of all VEGF-TKI studied, responses appear to range from 20% to 54%, with PFS or time to treatment failure at 6 to 13 months. 47 Therefore, the treatment sequence for mRCC remains to be VEGF-TKI after prior treatment with IO/IO combination or even IO/TKI combination (Figure 1 48 ). However, there are still additional relevant questions to consider especially in the era of adjuvant pembrolizumab approval, 49 such as the next treatment options after failure from adjuvant pembrolizumab, and what would be the considered acceptable timelines or definition of resistance from IO therapy.

Metastatic renal cell carcinoma (mRCC) sequence of treatment.
In summary, the treatment of mRCC encompasses a wide variety of experts in different disciplines requiring true multidisciplinary care. Every effort should be made to offer multidisciplinary care to each patient facing a diagnosis of RCC across all stages which would serve to improve treatment and outcomes.
