Abstract
Sunitinib is a multitarget tyrosine kinase inhibitor endowed mainly by antiangiogenic effects, although an indirect inhibitory effect on tumor growth and, more recently, a complex activity on antitumor immune response has been described. From approval by the US Food and Drug Administration (FDA) in January 2006, sunitinib represents a key molecule in the treatment of metastatic renal cell carcinoma (mRCC) due to the peculiar molecular pathogenesis of this neoplasm. Over the past 10 years, clinical trials and real-world experiences helped clinicians to understand how, when and for how long to use sunitinib. Although a huge amount of data evidenced the relationship existing between sunitinib dose intensity and improved clinical outcome, the management of sunitinib-induced adverse events is often complex; thus, alternative schedules have been proposed over time which allow increased tolerability, without decreased daily sunitinib exposure, leading to improved clinical outcomes. To date, combinations of sunitinib with other approved targeted agents did not demonstrate any significant benefit over its single-agent use, mainly due to tolerability issues. Sunitinib has also been tested in the adjuvant setting, within the ASSURE and S-TRAC trials, with opposite results; indeed, equivocal risk-stratification criteria, as well as immature overall survival (OS) data prevent any definitive conclusion on this important issue. Despite being on the market for a long time, sunitinib still plays a role as the ‘comparator arm’ of a number of trials in the field of mRCC. Combinations with immune checkpoint inhibitors and vaccines look promising; once again, sunitinib can help us to optimize mRCC management.
Introduction
Sunitinib (SU011248) is a small molecule that inhibits several members of the split-kinase domain family of receptor tyrosine kinases (RTKs), including the vascular endothelial growth factor receptors (VEGFRs) types 1 and 2 (FLT1 and FLK1/KDR), the platelet-derived growth factor receptors (PDGFR-α and PDGFR-β), the stem cell factor receptor c-KIT, as well as the FLT3 and RET kinases. 1
Inhibiting these RTKs blocks downstream signal transduction, thereby affecting tumor angiogenesis and growth. Although the relative contribution of the inhibition of each of the above RTKs to sunitinib antitumor activity is still poorly understood, it is clear that its antineoangiogenic properties represent its major mechanism of action. 1
It is thus not a surprise that sunitinib has been first developed in renal cell carcinoma (RCC), an extremely angiogenesis-dependent malignancy. 2
In addition, sunitinib has been shown to have a direct inhibitory effect on tumor growth by promoting tumor apoptosis, as well as an indirect inhibitory effect on tumor growth by stimulating antitumor immune responses. 3
The aim of this review is to summarize the sunitinib clinical development program in RCC, paying particular attention to more recent data, consequently trying to hypothesize further development steps within a rapidly changing therapeutic scenario.
Sunitinib clinical development program in renal cell carcinoma
Sunitinib was studied, within different phase I studies, using various schedules, including a 3-week cycle consisting of treatment for 2 weeks followed by a 1-week rest period (schedule 2/1), a 4-week cycle comprising treatment for 2 weeks followed by a 2-week rest period (schedule 2/2), or a 6-week cycle of treatment for 4 weeks followed by a 2-week rest period (schedule 4/2). These schedules explored both daily and every other day administration, and incorporated planned rest periods due to the prolonged half-life of the drug, and evidence of accumulation with continuous daily dosing. 4 Ultimately, the fixed dose of 50 mg daily (sufficient to produce target plasma concentrations above 50 ng/ml required to inhibit the PDGFRs and VEGFR RTKs) and the 4 weeks on, 2 weeks off schedule were chosen for the subsequent development of the drug.
As far as RCC, two phase II studies were then conducted in cytokine-refractory RCC patients.5–7 These studies not only showed an extremely high rate of objective responses (40% and 39%, respectively), but also yielded an unprecedentedly long time to progression (TTP) of 8.7 months, as well as an intriguing overall survival (OS) of 16.4 months. These results warranted both an accelerated approval by the US Food and Drug Administration (FDA), as well as the subsequent design of a pivotal, phase III.
In the pivotal randomized phase III trial, 750 previously untreated metastatic RCC patients were randomized to receive either sunitinib (administered according to the standard dose and schedule), or interferon (IFN)-α (given subcutaneously at a dose of 9 MU three times week), the primary endpoint of the study being progression-free survival (PFS), while OS was among the secondary endpoints.
The average PFS in the group of patients treated with sunitinib was significantly longer than that of patients treated with IFN-α (11 versus 5 months), corresponding to a hazard ratio (HR) of 0.42 (i.e. a reduction in the risk of progression or death of 58%). 8 Notably, the advantage in terms of PFS in favor of sunitinib was then maintained in all three prognostic groups according to the Memorial Sloan Kettering Cancer Center (MSKCC) criteria. As expected, based on the results of previous phase I and II studies, sunitinib induced objective responses in a high percentage of patients (31%).
Regarding tolerability, patients treated with sunitinib showed a higher incidence of diarrhea, vomiting, hypertension, hand–foot syndrome, and neutropenia, a safety profile consistent with what had been observed within earlier phase studies. Overall, a better quality of life was observed in sunitinib-treated patients, as compared with IFN-α8,9 Regarding OS, although it was higher in patients treated with sunitinib compared with those treated with IFN-α (26.4 versus 21.8 months, respectively), this difference did not reach statistical significance. 10 However, since the primary endpoint of the study was PFS (and not OS), and the p value for OS was close to statistical significance, it was obvious that the study was simply underpowered to show a significant benefit in terms of OS. Indeed, when patients who crossed over from IFN-α to sunitinib were censored within a nonpreplanned analysis, a statistically significant benefit was observed also in OS. 11
The activity and safety of sunitinib in an unselected, real world patient population was then confirmed by the results of its global expanded access program (EAP); furthermore, sunitinib activity in specific subpopulations of patients usually excluded, or under-represented in clinical trials (e.g. elderly, patients with brain metastases, patients with nonclear-cell histotypes, etc.) clearly emerged from this huge cohort of patients.12–14
Sunitinib clinical development is summarized in Table 1.
Sunitinib clinical development program in RCC.

Primary endpoint.
CDD, 37.5 mg/day on a continuous daily dosing schedule; HR, hazard ratio; IFN, interferon-α;
mRCC, metastatic renal cell carcinoma; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; schedule 4/2, 50 mg/day on a 4-weeks-on/2-weeks-off schedule; TTP, time to tumor progression.
As soon as sunitinib became commercially available on a global scale, it assured as the standard of care for the treatment of metastatic RCC, and as such, it remained until pazopanib joined it in that position following the presentation of the results of the COMPARZ and PISCES studies.
Sharing a leading position with pazopanib
In the COMPARZ study 15 1110 patients with clear-cell, metastatic RCC, were randomized 1:1 to receive pazopanib (at a dose of 800 mg once daily, continuous dosing) or sunitinib (standard dose and schedule), its primary endpoint being PFS; the study was powered to show the non-inferiority of pazopanib versus sunitinib.
Pazopanib proved to be non-inferior to sunitinib with respect to PFS [HR: 1.05; 95% confidence interval (CI): 0.90–1.22], meeting the predefined non-inferiority margin (upper bound of the 95% CI: <1.25); also, OS was similar (HR: 0.91; 95% CI: 0.76–1.08). Furthermore, 11 of 14 health-related quality-of-life (QoL) domains favored pazopanib, when it came to QoL. 15
On the other hand, the PISCES study 16 had as its primary endpoint patients’ preference. In this innovative study, patients with metastatic RCC were randomized to pazopanib for 10 weeks, a 2-week washout, and then sunitinib for another 10 weeks, or the reverse sequence; doses and schedules were the usual ones for both agents. The primary endpoint, patient preference for a specific treatment, was assessed by a questionnaire at the end of the two treatment periods. Other endpoints and analyses included reasons for preference, Health Related Quality of Life (HRQoL) and physician’s preference.
Significantly more patients preferred pazopanib (70%) over sunitinib (22%), whilst 8% expressed no preference (p < 0.001); less fatigue and better overall QoL were the main reasons for preferring pazopanib, with less diarrhea as the main reason for their choice in those patients who preferred sunitinib. 16 Again, adverse events were consistent with each drug’s known profile, but pazopanib proved to be superior to sunitinib in terms of QoL, thus corroborating the QoL results of the COMPARZ study.
Even though methodologically not faultless, these two important studies have put the two drugs on the same level (at least in terms of evidence-based medicine) as standard first-line treatments for metastatic RCC.
Sequential use of sunitinib and sorafenib
Sunitinib and the other multikinase inhibitor sorafenib were the very first targeted agents that reached patients’ bedsides in the field of metastatic RCC.
In retrospective studies, sequential use of sunitinib and sorafenib provided additional clinical benefit beyond the use of either agent alone.17,18 SWITCH was the first prospective, randomized, phase III study aimed at testing the hypothesis that sequential therapy with sorafenib–sunitinib is superior to sunitinib–sorafenib in prolonging total PFS (defined as time from randomization to confirmed progression or death during second-line therapy).
No significant difference in either total PFS (median 12.5 versus 14.9 months; HR: 1.01; 90% CI: 0.81–1.27; p = 0.5 for superiority) and OS (median 31.5 and 30.2 months; HR: 1.00, 90% CI: 0.77–1.30; p = 0.5 for superiority) between sorafenib–sunitinib and sunitinib–sorafenib emerged. 19 Ultimately, the results of this trial suggested a degree of noncross-resistance between sunitinib and sorafenib.
Sunitinib versus everolimus
More recently, a multicenter, randomized phase II trial (RECORD-3) was conducted to compare the standard sequence of first-line sunitinib followed by everolimus with the inverse sequence, the primary endpoint of the study being PFS non-inferiority of first-line everolimus compared with first-line sunitinib. 20 The primary endpoint of this study was not met, median PFS being 7.9 months for first-line everolimus versus 10.7 months for first-line sunitinib (HR: 1.4, 95% CI: 1.2–1.8). Among patients who discontinued first line, just 108 (45%) crossed over from everolimus to second-line sunitinib, and 99 (43%) crossed over from sunitinib to second-line everolimus, making the evaluation of the two-agents’ sequence almost impossible. However, the median combined PFS was 25.8 months for sequential sunitinib followed by everolimus versus 21.1 months for sequential everolimus followed by sunitinib (HR: 1.3, 95% CI: 0.9–1.7). As far as OS, it was 32.0 months for sequential sunitinib followed by everolimus versus 22.4 months for sequential everolimus followed by sunitinib (HR: 1.2, 95% CI: 0.9–1.6). 20
Trying to optimize treatment: dose, schedule and treatment duration
From the very beginning it was clear that at least in some patients, the antitumor activity of sunitinib can be hampered by its safety profile, which is often difficult to manage, especially for inexperienced physicians; indeed, real-world clinical data21–23 confirmed the relationship between sunitinib exposure and clinical efficacy already evident in a huge population pharmacokinetic study, 4 showing that unnecessary dose reduction and treatment interruptions do correlate with a poor treatment outcome.21,22,23
Several researchers addressed the topic of sunitinib dose and schedule, as summarized in Table 2.
Treatment optimization trials.
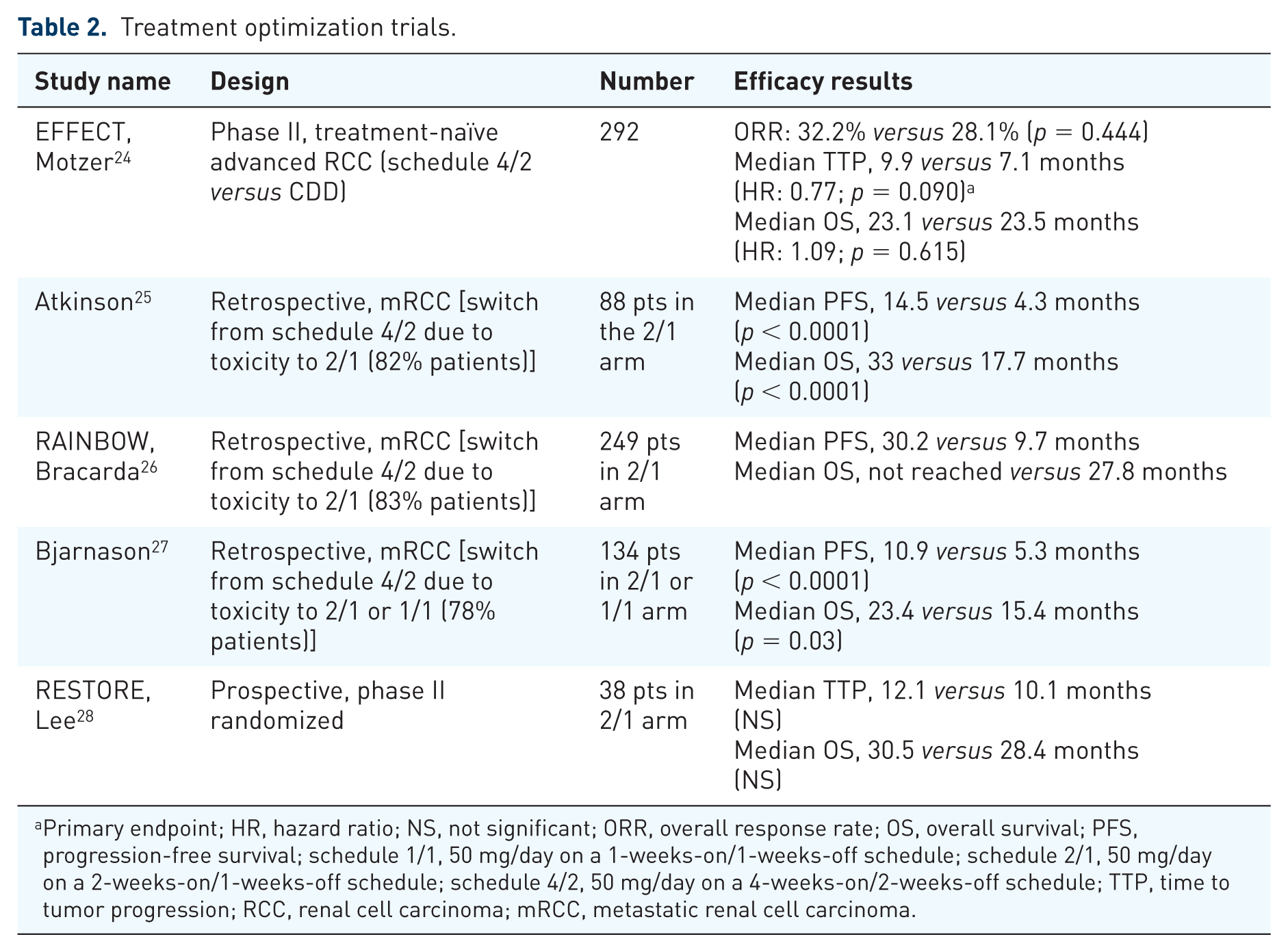
Primary endpoint; HR, hazard ratio; NS, not significant; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; schedule 1/1, 50 mg/day on a 1-weeks-on/1-weeks-off schedule; schedule 2/1, 50 mg/day on a 2-weeks-on/1-weeks-off schedule; schedule 4/2, 50 mg/day on a 4-weeks-on/2-weeks-off schedule; TTP, time to tumor progression; RCC, renal cell carcinoma; mRCC, metastatic renal cell carcinoma.
A first attempt to optimize sunitinib dose and schedule and make it more tolerable without losing efficacy came with the renal EFFECT trial. 24
In this randomized phase II trial, the traditional sunitinib schedule (schedule 4/2, 50 mg/day on a 4-weeks-on/2-weeks-off schedule) was compared with a reduced (37.5 mg/day), but continuous daily dosing (CDD) schedule. The median TTP (primary endpoint) was numerically longer with schedule 4/2 than with CDD. In addition, schedule 4/2 was superior to the CDD schedule according to a composite endpoint of time to deterioration, which included death, disease progression, and progression of disease-related symptoms.
From a certain viewpoint, surprisingly, the CDD schedule not only proved to be less active, but also was not better tolerated, as initially expected.
Some retrospective studies25–29 suggested that patients who experience intolerable toxicities on a 4/2 schedule could switch to a 2/1 schedule (50 mg/day on a 2-weeks-on/1-week-off schedule) to improve tolerability without compromising efficacy. The prospective randomized phase II RESTORE trial evaluated the efficacy and safety of the sunitinib 2/1 schedule versus the standard 4/2 schedule in 74 Asian patients with clear-cell metastatic RCC (mRCC). The 2/1 schedule resulted in a higher 6-month failure-free survival (the primary endpoint, 63% versus 44%) and median time to treatment failure (7.6 versus 6.0 months, HR: 0.57, p = 0.029). The 2/1 schedule was associated with lower toxicity in terms of neutropenia (all grades 61% versus 37%, p = 0.037) and fatigue (all grades 83% versus 58%, p = 0.017) and time-to-first-dose reduction (HR: 0.35, p = 0.014). 28 Additional prospective studies with larger sample size will eventually confirm the efficacy of the 2/1 schedule.
Furthermore, we do not yet know if a successful sunitinib treatment leading to a complete response or a long-lasting disease stabilization may be discontinued. Several small studies suggest that intermittent dosing of sunitinib (‘drug holidays’) may be better tolerated than standard fixed schedule without compromising efficacy.30–32 A randomized multicenter phase III study is ongoing (STAR trial) that compares the conventional schedule of sunitinib 50 mg daily 4/2 weeks with a drug-free interval strategy, namely a treatment break after completion of at least four cycles and maximal radiological response until progressive disease (PD) and then a restart of sunitinib for a minimum of four cycles and maximal radiological response. 33
Sunitinib in nonclear-cell renal cell carcinoma
The evidence for sunitinib treatment in nonclear-cell RCC is available from several retrospective and prospective phase II trials, as well as from a subgroup analyses of the large EAP.13,34
Only two randomized prospective phase II trials compared everolimus and sunitinib as first-line treatment in nonclear-cell RCC (Table 3). The results of the first trial (ESPN trial) showed no benefit for everolimus in terms of median PFS (sunitinib 6.1 months, everolimus 4.1 months) and median OS (sunitinib 16.2 months, everolimus 14.9 months), both agents showing only modest efficacy. 35 The second trial (ASPEN) showed a significant increase in the median PFS with sunitinib (8.3 months) versus everolimus (8.3 versus 5.6 months; HR: 1.41; p = 0.16), while the median OS was not different between the two groups. 36
Randomized controlled trials of sunitinib in nonclear-cell histologies.

Primary endpoint. HR, hazard ratio; OS, overall survival; PFS, progression-free survival, HR, hazard ratio; CI, confidence interval.
The results of a recent systematic review and meta-analysis showed a trend towards favoring vascular endothelial growth factor-targeted therapy for PFS and OS compared with mammalian target of rapamycin inhibitors, although statistical significance was not reached. 37 The relative benefits and harms of these treatments remain uncertain. A randomized controlled trial with sufficient power to detect potential differences between treatments, is needed, although one should acknowledge that for rare cancers, specific trial designs are badly needed. 38
Given available data, sunitinib is still recommended as an option for first-line treatment in nonclear-cell RCC in current guidelines, especially for patients with good MSKCC risk scores.39,40
Neoadjuvant sunitinib therapy
Presurgical targeted molecular therapy may be considered for a subset of patients with bulky/central renal tumors for whom partial nephrectomy would not be readily feasible and in patients with metastatic RCC having a low relative volume of disease in the primary tumor. The integration of surgery and targeted therapy is relatively safe, with low overall morbidity in most patients. 41
The favorable outcomes reported in several retrospective reviews and case studies have provided sufficient rationale for ongoing prospective phase II clinical trials evaluating the efficacy of sunitinib in the neoadjuvant setting. Preliminary results from one such study (56 patients with metastatic RCC) show a 5% response in the primary tumor according to RECIST standards, with no local progression. Nearly, 20% of patients, however, showed progression of systemic disease prior to surgery. Overall, 71% of patients underwent nephrectomy with 20% experiencing surgical complications including one death. 42 However, randomized, controlled, long-term studies are desperately needed to provide substantial evidence of the neoadjuvant effectiveness of sunitinib.
Sunitinib in the adjuvant setting: two-faced Janus
The adjuvant treatment of RCC has traditionally been a significant unmet need: several adjuvant strategies, including cytokine therapy, radiotherapy, and hormone therapy, have been explored to decrease the rate of relapse in RCC, but none were successful. 43
As always happens in oncology, sunitinib activity in the metastatic setting prompted many researchers to move it in the adjuvant setting, in the attempt of reducing the risk of relapse in radically resected patients at high risk of disease recurrence.
To date, the results of two large trials have been published, which yielded conflicting results; indeed, while the ECOG ASSURE trial 44 failed, the S-TRAC trial 45 succeeded, (although OS data of this study are not mature enough).
ASSURE was a double-blind, placebo-controlled, randomized, phase III trial in which 1943 patients from the United States and Canada were randomized (1:1:1) to receive 54 weeks of sunitinib 50 mg per day, 4 weeks on and 2 weeks off, sorafenib 400 mg twice per day CDD or placebo.
Unfortunately, no significant differences in disease-free survival (DFS), the primary endpoint of the study, were observed between the three treatment arms: median DFS was 5.8 years for sunitinib (HR: 1.02; 97.5% CI: 0.85–1.23; p = 0.8038), 6.1 years for sorafenib (HR: 0.97; 97.5% CI: 0.80–1.17; p = 0.7184), and 6.6 years for placebo. The most common adverse events leading to dose reduction were hand–foot syndrome (grade ⩾ 3: sunitinib arm 15%, sorafenib arm 33%, placebo arm 1%), hypertension (grade ⩾ 3: sunitinib arm 17%, sorafenib arm 16%, placebo arm 4%) and fatigue (grade ⩾ 3: sunitinib arm 18%, sorafenib arm 7%, placebo arm 3%). Despite the reduction in starting dose, the proportion of grade ⩾ 3 adverse events still exceeded 55% in both the sunitinib and sorafenib groups. 44
Given these negative results (which indeed were coupled with a not-so-strong biologic rationale), somewhat surprisingly, the S-TRAC, which compared 1 year of sunitinib versus 1 year of placebo, yielded positive results; indeed, the median duration of DFS (the primary endpoint of the study) was 6.8 years (95% CI: 5.8 to not reached) in the sunitinib group and 5.6 years (95% CI: 3.8–6.6) in the placebo group (HR: 0.76; 95% CI: 0.59–0.98; p = 0.03). 45
The selection of patients with different degrees of risk of relapse due to the use of different and equivocal (Table 4) risk-stratification criteria, has been advocated to justify the different results of these two trials. 46
Key inclusion criteria of adjuvant trials of sunitinib.

AJCC, American Joint Committee on Cancer; TNM classification for Renal Cell Carcinoma: T (primary tumors), N (regional lymph node), M (distant metastasis); N0: no regional lymph node metastasis, NX: regional lymph nodes cannot be assessed, N+ or N1: metastasis in regional lymph node(s); G: nuclear grading; UISS, UCLA Integrated Staging System.
None of the recently completed or ongoing adjuvant trials with targeted agents has been already prospectively implemented with genetic analyses such as the genetic recurrence score. 47 Thus, one could speculate if this score could help us to understand why sunitinib, as an adjuvant treatment failed in the ECOG ASSURE trial, while succeeding in the S-TRAC one.
Given this discrepancy between the two studies, the possibility that in S-TRAC, sunitinib has just delayed relapse (in the absence of OS data) and the issue of toxicity (with potentially cured patients less prone, as compared with metastatic ones, to tolerate treatment-related adverse events), it appears clear that the amount of uncertainty regarding the present place, if any, of sunitinib as an adjuvant to radical surgery remains quite high, and our judgement should be postoponed at least till the availability of S-TRAC OS data. 46
Sunitinib-based combinations
Several studies have investigated sunitinib in combinations with other targeted agents. 48 The combinations sunitinib plus INF-α, sunitinib plus bevacizumab, sunitinib plus everolimus, sunitinib plus temsirolimus, were tested in phase I studies in patients with advanced RCC. These trials were terminated early due to dose-limiting toxicity.
In a single-arm phase II study (39 patients), the combination of sunitinib with the cytotoxic chemotherapy gemcitabine appeared to be more efficacious than either therapy alone in patients with sarcomatoid or poor-risk mRCC (ORR: 26% and 24% for patients with sarcomatoid RCC and poor- risk RCC, respectively). 49
RCC is a potentially immunogenic tumor: the immunologic dysfunctions described in this tumor are numerous, being evident even in the presence of localized and not-yet metastatic disease. 50 Given this feature, attempts to combine sunitinib with different forms of immunotherapy (from cytokines to novel checkpoint inhibitors and vaccines) have been tested over the years, with somewhat conflicting results. 51
Two different types of vaccines have been tested in both cases in combination with sunitinib. In a recently published phase II trial, AGS-003, an autologous dendritic cell-based immunotherapy was tested in combination with sunitinib in patients with intermediate and poor-risk mRCC. 52 Given the promising results of this trial (ORR was 43%, the median PFS was 11.2 months), an international phase III randomized trial ADAPT [ClinicalTrials.gov identifier: NCT01582672] of AGS-003 plus standard treatment in advanced RCC is ongoing.
The cancer vaccine IMA901, comprises 10 synthetic tumor-associated peptides expressed by the majority of RCCs with demonstrated antigenicity. IMA901 with sunitinib had already proved able to lower both myeloid suppressor cells, as well as Treg lymphocytes. 53 However, a consequent phase III study of IMA901 plus sunitinib versus sunitinib alone showed a detrimental effect of the vaccine-plus-sunitinib combination on the survival of treated patients, particularly evident for patients with an intermediate prognosis. 54 Indeed, although median OS did not differ significantly between the groups [33.17 months (95% CI: 27.81–41.36) in the sunitinib plus IMA901 group versus not reached (33.67 to not reached) in the sunitinib monotherapy group], the resulting HR was 1.34 (0.96–1.86; p = 0.087).
More recently, preliminary results from a phase I trial, Checkmate 016 [CA209-016, ClinicalTrials. gov identifier: NCT01472081], using sunitinib or pazopanib and nivolumab [anti-programmed-death 1 (anti-PD-1) antibody, starting dose: 2 mg/kg intravenously every 3 weeks, with the expectation of increasing to 5 mg/kg] in pretreated mRCC have been published and show evidence of activity for the combination (ORR 52% for nivolumab plus sunitinib and 45% for nivolumab plus pazopanib) with a prominent safety profile. 55 The most common grade 3 or 4 adverse events were an elevation of alanine aminotransferase (ALT) (18%) and hypertension and hyponatremia (15% each); in 8 of 33 patients (24%), treatment-related adverse events led to permanent discontinuation of the treatment.
Sunitinib rechallenge
Treatment guidelines recommend sequential treatment of mRCC in order to offer patients the longest survival possible; after disease progression on one agent, treatment with another targeted agent generally controls the disease, giving additional PFS. Recent retrospective analysis of the International mRCC Database Consortium (IMDC) database found that patients who received multiple targeted-therapy lines lived substantially longer than those treated with only one. 56
Several retrospective studies and case reports have shown that reintroduction of sunitinib after progression with other treatments can result in additional clinical benefit,22,23 indicating further research is warranted to improve our understanding of sunitinib rechallenge therapy.
The mixed retrospective and prospective REchallenge with SUnitinib in MEtastatic RCC (RESUME) Study has evaluated the efficacy, as measured by PFS, and safety of sunitinib rechallenge in mRCC patients who received first-line sunitinib, then one or more lines of different targeted treatments, followed by further sunitinib. 57 A total of 28 patients (54% of 52 patients eveluated) had a complete (n = 1) or partial response (PR) with first-line sunitinib, and 8 patients (15%) achieved a PR upon sunitinib rechallenge. Median PFS with first-line sunitinib was 18.4 months (95% CI: 12.5–23.7) and 7.9 months (95% CI: 5.4–13.2) with sunitinib rechallenge.
Two ongoing prospective phase II studies (an Italian study, RETRY, EudraCT 2012-000473-23 and a Dutch study, NTR3711) will provide further information on this topic.
Sunitinib and radiotherapy
Preclinical studies show the feasibility of combining sunitinib with radiotherapy (RTx) for RCC treatment. This strategy involves different mechanisms, including vascular normalization, modulation of cell growth and apoptosis, as well as the alterations of the immune response. 58
Instigated by the promising results of preclinical research, several phase I and II clinical studies have been performed to assess the feasibility of combining sunitinib with radiotherapy in cancer patients. A phase II trial in olimetastatic RCC patients showed a local control rate of 75%, and distant control of 52% with the sunitinib–radiotherapy combination. The median time until progression was 9.5 months, and at the end of the study, 18 patients were alive, 11 of whom without disease. 59 By several case reports, encouraging results were also observed in patients with mRCC who received either sunitinib combined with single-fraction stereotactic radiosurgery 60 or high-dose hypofractionated RTx. 61 Overall, the toxicities of the concurrent combination of RTx and sunitinib appear to depend on the duration and dose of sunitinib treatment, on the concurrent dose of RTx, but also on previous treatments and type of metastases.
To translate preclinical findings into clinically relevant treatment, protocols are needed to establish the clinical benefit of combination with additional studies.
Future perspectives
mRCC is a highly angiogenic-dependent neoplasm, as well as a potentially immunogenic one: combining molecularly targeted agents with different forms of immunotherapy thus makes a lot of sense.
Novel immune checkpoint inhibitors (i.e. anti-PD1 or anti-programmed-death ligand-1 agents) have just started to reach patients’ bedsides and their combination with molecularly targeted agents is presently under clinical evaluation, although the only trial presented in detail (ASCO 2014), but not yet published in extenso, is the phase I CA209-016 study, discussed above.
Several phase III trials are presently comparing immunotherapy combinations against sunitinib, as the standard first-line treatment for metastatic RCC (Table 5).
Combination of vascular endothelial growth factor receptor inhibitors with immune checkpoint inhibitors: main ongoing studies.

PD-L1, programmed death ligand 1.
Combinations of sunitinib together with immune checkpoint inhibitors are definitely promising, but cumulative toxicity remains a potential issue. Toxicity aside, doubts also remain over the effect of different immunologic targeted agents and therefore the ability to rationally combine any of these agents with different immunotherapeutics. 51
Concluding remarks
Sunitinib has revolutionized the treatment of mRCC over the past 10 years so far. Large-scale studies have demonstrated sound and reproducible benefits (across all activity and efficacy endpoints) assigning to sunitinib a leadership position in this setting.
Available data also support an evolving role of sunitinib in the adjuvant setting. However, all the knowledge acquired in the advanced stage setting may not be enough to select the ideal patient for adjuvant therapy. Indeed, more accurate and reproducible clinical and, hopefully, also molecular tools would be needed to identify patients with radically resected RCC who are at high risk for recurrence following potentially curative nephrectomy.
Combinations of sunitinib with novel immunotherapeutics (either vaccine or immune checkpoint inhibitors) hold promise, but cumulative toxicity seems to remain an issue.
Finally, biomarker-driven clinical trials are needed to stratify patients and to evaluate novel immunotherapy-based combination regimens in selected advanced RCC patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
