Abstract
Following approval of the oral, multitargeted tyrosine kinase inhibitor sunitinib malate for the treatment of patients with metastatic renal cell carcinoma (mRCC) in Europe and the USA in 2006, the agent has had a substantial impact on the treatment landscape in this setting. Sunitinib is now recommended in international treatment guidelines for the first-line treatment of favourable- or intermediate-risk mRCC and as an alternative option in poor-risk mRCC. In the 6 years since the approval of sunitinib, the range of agents available for the treatment of mRCC has expanded substantially, and this, together with a number of additional therapies in late-stage development, has increased the treatment options available to patients. Results from a phase III trial and a global expanded access study have provided robust data to support the efficacy of sunitinib in mRCC, including in real-world populations. Data also suggest a significant quality of life benefit with sunitinib, with superior patient-reported outcomes observed with this agent compared with interferon-α therapy. Both clinical and real-world study data also support the safety profile of sunitinib; most treatment-associated adverse events are mild to moderate in severity and can be managed effectively with close monitoring and proactive management. Clinical experience with sunitinib has demonstrated that therapy management, involving optimal dosing, maximum treatment duration and prompt and effective adverse event management, supports optimal patient outcomes with sunitinib. In this review we discuss clinical experience with sunitinib in mRCC, with an emphasis on real-world data, and utilize clinical case studies to examine the successful implementation of therapy management strategies for optimal patient outcomes. An increasing body of evidence suggests that side effects associated with sunitinib therapy, including hypertension, hand–foot syndrome and hypothyroidism, may represent effective markers of treatment response, and these will also be discussed.
Keywords
Introduction
The advent of sunitinib malate (Sutent®, Pfizer Inc., NY, USA) for the first-line treatment of metastatic renal cell carcinoma (mRCC) had a substantial impact on the treatment landscape in mRCC; sunitinib represented the first oral multitargeted tyrosine kinase inhibitor (TKI) to gain regulatory approval in this setting. By targeting receptor tyrosine kinases implicated in tumour growth and angiogenesis [including vascular endothelial growth factor receptor (VEGFR)-1, -2 and -3, platelet-derived growth factor receptor (PDGFR)-α and -β, stem cell growth factor receptor (KIT), fms-related tyrosine kinase 3 (FLT3), glial cell-line derived neurotrophic factor receptor (RET) and colony-stimulating factor receptor 1 (CSF1R)], sunitinib exerts both cytotoxic effects on tumour cells and anti-angiogenic effects on tumour vasculature, leading to tumour cell necrosis and delayed growth or regression [Faivre et al. 2007].
A randomized, phase III trial of sunitinib compared with interferon (IFN)-α in treatment-naïve mRCC patients reported median progression-free survival (PFS) of 11 months in the sunitinib treatment group and median overall survival of more than 2 years with sunitinib therapy [Motzer et al. 2007a, 2009]. This trial was associated with a substantial improvement in PFS compared with cytokine therapy, the reference standard of care at the time (median PFS was 5 months in the IFN-α group; p < 0.001). Median overall survival was 26.4 and 21.8 months respectively; p = 0.051 (the survival data were confounded by patients crossing over to sunitinib from the IFN-α arm following interim analysis) [Motzer et al. 2002, 2007a, 2009; Coppin et al. 2005]. Sunitinib was approved in 2006 as a treatment for mRCC in the USA (January 2006) and in Europe (July 2006 as second-line therapy). Sunitinib was approved for first-line treatment of mRCC in Europe in January 2007 [Pfizer Ltd., 2012; Pfizer Inc., 2012]. Sunitinib has since become a reference standard of care that is recommended in international treatment guidelines for the first-line treatment of favourable- or intermediate-risk mRCC [European Association of Urology (EAU): Patard et al. 2011; European Society of Medical Oncology (ESMO): Escudier and Kataja 2010].
In the 6 years since the introduction of sunitinib, the range of agents licensed in Europe for the treatment of mRCC has increased substantially. The VEGF-binding monoclonal antibody bevacizumab (Avastin®; Genentech Inc., CA, USA) was approved in 2007 in combination with IFN-α; the mammalian target of rapamycin (mTOR) inhibitors temsirolimus (Torisel®; Pfizer Inc.) and everolimus (Afinitor®; Novartis Pharmaceuticals, Basel, Switzerland) were approved in 2007 and 2009 respectively; and pazopanib (Votrient®; GlaxoSmithKline Group Ltd., Middlesex, UK) joined sunitinib and sorafenib (Nexavar®; Bayer HealthCare, Leverkusen, Germany/Onyx Pharmaceuticals, CA, USA, approved in 2006) in the class of multitargeted TKIs in 2010. In addition, a number of next-generation VEGFR-targeted TKIs are currently in late-stage clinical development, including axitinib (Pfizer Inc.; approved in the USA at the time of manuscript submission), tivozanib (Aveo Pharmaceuticals, MA, USA; phase III) and cediranib (AstraZeneca, London, UK; phase II) [Bhargava and Robinson, 2011].
This paper reviews clinical experience with sunitinib in mRCC, with emphasis on real-world data from patient case studies and from the global expanded access study in patients who received sunitinib on a compassionate use basis.
First-line treatment for metastatic renal cell carcinoma
With an increasing number of treatment options available for the management of mRCC and additional agents in development, treatment selection remains an important consideration in the first-line setting. Current treatment guidelines recommend that patients with clear-cell mRCC at favourable or intermediate prognostic risk should receive first-line therapy with sunitinib, pazopanib or bevacizumab plus IFN-α.
Sunitinib as first-line therapy for metastatic renal cell carcinoma
Efficacy
Two phase II studies of sunitinib in patients with cytokine-refractory mRCC reported an objective response rate (ORR) of 40 and 33%, median PFS of 8.7 and 8.8 months and median overall survival of 16.4 and 23.9 months respectively [Motzer et al. 2006a, b, 2007b]. A pooled analysis of the two trials (N = 168) reported an ORR of 42%, as assessed by the trial investigators, and median PFS of 8.2 months [Motzer et al. 2006b].
Based on these data, a randomized, multicentre, phase III trial was conducted in 750 treatment-naïve patients to compare the efficacy and tolerability of sunitinib (50 mg/day for 4 weeks followed by 2 weeks off-treatment; Schedule 4/2) with that of IFN-α in the first-line setting [Motzer et al. 2007a]. Patients with comorbid conditions were permitted to participate in the study if their comorbidities were adequately controlled with standard medical interventions; those with brain metastases, uncontrolled hypertension or clinically significant cardiovascular events were excluded. This relatively unselected patient population comprised predominantly patients at favourable or intermediate prognostic risk, according to the Memorial Sloan-Kettering Cancer Center (MSKCC) criteria. There were no significant differences between the two treatment arms with respect to baseline demographics or disease characteristics. Patients in the IFN-α arm were permitted to cross over to the sunitinib arm following an interim analysis.
The primary endpoint of the study was PFS; overall survival, ORR, patient-reported outcomes and safety were secondary endpoints. Median PFS was 11 months in the sunitinib treatment group [95% confidence interval (CI) 11–13 months] and 5 months in the IFN-α group (95% CI 4–6 months) [Motzer et al. 2007a]. The difference in PFS between the two treatment groups corresponded to a hazard ratio for progression of 0.42 (95% CI 0.32–0.54; p < 0.001). The investigators reported a 5-fold greater ORR with sunitinib compared with IFN-α treatment in the interim analysis (31 versus 6%; p < 0.001) and a 4-fold greater ORR with sunitinib versus IFN-α in the final analysis (47 versus 12%; p < 0.001) [Motzer et al. 2007a, 2009]. Median overall survival was 26.4 and 21.8 months in the sunitinib and IFN-α groups respectively (95% CI 23.0–32.9 and 17.9–26.9 months), resulting in a hazard ratio of borderline significance (0.821, 95% CI 0.673–1.001; p = 0.051) [Motzer et al. 2009] (Figure 1). Adjusting for pre-specified stratification factors [lactate dehydrogenase >1.5× or ≤1.5× the upper limit of normal; Eastern Cooperative Oncology Group (ECOG) performance status 0 or 1; and presence/absence of prior nephrectomy], the hazard ratio narrowly achieved statistical significance (0.818, 95% CI 0.669–0.999; p = 0.049) [Motzer et al. 2007a]. However, when patients who crossed over from IFN-α to sunitinib following the interim analysis (n = 25) were censored from the results, median overall survival was 26.4 and 20.0 months (95% CI 23.0–32.9 and 17.8–26.9 months) respectively, corresponding to a significant hazard ratio in favour of sunitinib (0.808, 95% CI 0.661–0.987; p = 0.036) [Motzer et al. 2009].

Kaplan–Meier estimates of overall survival with sunitinib versus interferon-α in the phase III trial [Motzer et al. 2009]. Reprinted with permission. © 2009 American Society of Clinical Oncology. All rights reserved. Motzer, R. et al. J Clin Oncol 2009; 27: 3584–3590.
Limiting the analysis to exclude patients who received post-study cancer treatment during follow up, median overall survival in the sunitinib group was twice that in the IFN-α group (28.1 versus 14.1 months; hazard ratio 0.647, 95% CI 0.483–0.870; p = 0.003) [Motzer et al. 2009]. Further, by adjusting for baseline factors identified as independent predictors of overall survival in a multivariate analysis (ECOG performance score, serum haemoglobin, time from diagnosis to treatment, corrected calcium, alkaline phosphatase, lactate dehydrogenase and number of metastatic sites), the analysis was more strongly in favour of sunitinib (hazard ratio 0.764, 95% CI 0.623–0.936; p = 0.0096) [Motzer et al. 2009].
In the phase III study, a Cox proportional hazards model was used to evaluate efficacy data according to patient baseline clinical features and prognostic factors. In all three prognostic risk groups, sunitinib therapy was associated with longer PFS and overall survival than IFN-α therapy [Motzer et al. 2007a, 2009].
Safety
In the phase III trial, the most common treatment-related adverse events of all grades in the sunitinib group, occurring in > 50% of patients, were diarrhoea, fatigue and nausea; most toxicities were mild to moderate in severity [Motzer et al. 2009]. Across all grades, hypertension, stomatitis and hand–foot syndrome (HFS) were significantly more common in the sunitinib group than in the IFN-α group, as were leucopenia, neutropenia and thrombocytopenia. Importantly, whilst cardiotoxicity is a recognized risk with TKI therapies [Schmidinger et al. 2008; Mellor et al. 2011], evaluation of ejection fraction in the interim and final analyses suggested that long-term sunitinib therapy is not associated with significant cumulative cardiotoxicity [Oudard et al. 2011]; 13 and 3% of patients receiving sunitinib and IFN-α respectively exhibited a decline in ejection fraction and grade 3 severity was reported in 3 and 1% of patients, respectively [Motzer et al. 2009]. Using Fisher’s exact test to compare grade 3/4 adverse events between the two groups showed that diarrhoea was more common in the sunitinib group, whereas patients receiving IFN-α were more likely to develop fatigue (p < 0.05 for both) [Motzer et al. 2007a].
Quality of life
The primary quality of life (QoL) endpoint in the phase III study was the Functional Assessment of Cancer Therapy (FACT)–Kidney Symptom Index Disease-Related Symptoms (FKSI-DRS) subscale, which forms part of the FKSI 15-item (FKSI-15) questionnaire. The FACT–General questionnaire and the EuroQoL 5D utility score (EQ-5D) were also used [Cella et al. 2008]. Scores in the sunitinib group were consistently higher than in the IFN-α group, suggesting that sunitinib treatment conferred a significant additional QoL benefit with superior patient-reported outcomes [Cella, 2011]. A QoL-adjusted survival analysis (Time without symptoms of disease progression or toxicity of treatment (TWiST)) supported this finding; by adjusting time to progression for the number of toxicity-free days (based on the grade 3/4 treatment-related side effects that occurred most frequently in the study), it was demonstrated that sunitinib was associated with greater toxicity-adjusted PFS than IFN-α [Patil et al. 2012].
Real-world experience with sunitinib
Whilst the phase III study enrolled a relatively unselected patient population, patients at poor prognostic risk were not well represented. A global expanded access study, intended to provide sunitinib on a compassionate use basis to trial-ineligible patients with RCC from countries where regulatory approval had not been granted, included patients with brain metastases and those with poor ECOG performance status [Gore et al. 2009, 2011]. All histological mRCC subtypes were included, provided patients had adequate organ function, had received no prior sunitinib therapy, had no residual toxic effects from previous systemic therapy, and had the potential to derive clinical benefit from sunitinib treatment as judged by the investigator. Sunitinib was given at the recommended starting dose of 50 mg/day by Schedule 4/2, with adjustments to 37.5 and 25 mg/day permitted on the basis of tolerability [Gore et al. 2009]. Safety data were collected on days 1, 14 and 28 in the first 6-week treatment cycle, and on days 1 and 28 in subsequent cycles. Tumour assessments were performed according to protocols at each of the 246 enrolling sites; data on overall survival, PFS and ORR were collected where possible [Gore et al. 2009].
A total of 4564 sunitinib-naïve patients were enrolled in the expanded access study, with 4371 evaluable in a modified intent-to-treat population [Gore et al. 2009]. Whilst most patients (89%) had undergone prior nephrectomy, only 5% had a history of previous antiangiogenic therapy. Of the metastatic sites represented, the most common were the lungs (76%) and lymph nodes (51%), with brain metastases in 7% of patients [Gore et al. 2009]. Patients received a median of five 6-weekly treatment cycles, with 56% receiving long-term (≥6 months) sunitinib therapy. Median treatment duration and overall survival were 15.6 and 18.4 months respectively in 4349 patients evaluable for survival. Prior cytokine therapy did not confer a survival advantage over cytokine-naïve patients [Gore et al. 2009].
Median PFS was consistent with that seen in the phase III trial (10.9 versus 11.0 months), suggesting similar efficacy with sunitinib across a broad range of mRCC patient populations [Gore et al. 2009]. This was further explored in specific subgroups: the elderly (aged ≥65 years), patients with brain metastases [Gore et al. 2011] and patients with poor ECOG performance status. Median PFS and overall survival in elderly patients were 11.3 and 18.2 months respectively, consistent with data recently presented on 154 Italian patients aged ≥70 years (10.6 and 20.1 months respectively) [De Giorgi et al. 2012]. In patients with brain metastases, median PFS and overall survival with sunitinib were 5.6 and 9.2 months respectively [Gore et al. 2011], while in patients with ECOG performance status ≥2, median PFS and overall survival with sunitinib were 5.1 and 6.7 months respectively [Gore et al. 2009].
The adverse event profile of sunitinib in the expanded access programme was broadly consistent with that described in the phase III trial, despite the greater diversity of patients [Gore et al. 2009, 2011]. In patients from the expanded access programme, the safety profile of sunitinib was similar in those who had undergone prior nephrectomy and those without prior nephrectomy [Szczylik et al. 2008], and a comparison of patients who received sunitinib for ≥6 months with those treated for <6 months showed a greater number of adverse events in the long-term treatment group, as expected, but revealed no new or unexpected toxicities [Gore et al. 2009].
Therapy management to optimize outcomes with sunitinib
Sunitinib has a distinct and predictable profile of adverse events, most of which are manageable with standard medical interventions. Therapy management, involving optimal dosing, prompt and effective adverse event management and maximum treatment duration, is integral to the optimization of clinical outcomes in patients receiving sunitinib. Experience with sunitinib in clinical practice has demonstrated that the implementation of simple therapy management strategies, together with an effective patient–physician relationship that involves close patient monitoring, patient education and patient involvement in therapeutic decision-making, can promote optimal treatment efficacy [Schmidinger et al. 2010a; Négrier and Ravaud, 2007; Ravaud, 2009, 2011; Eisen et al. 2012; Négrier, 2012].
Optimal dosing
At the recommended dose of 50 mg/day on Schedule 4/2, sunitinib is associated with a median overall survival of more than 2 years [Motzer et al. 2009]. Indeed, data from a pharmacokinetic/ pharmacodynamic (PK/PD) meta-analysis of six sunitinib studies, including two in mRCC, have suggested that higher sunitinib exposure is associated with longer median overall survival [Houk et al. 2010 ], underlining the importance of maintaining patients on appropriate doses to derive clinical benefit [Ravaud, 2009].
A randomized phase II multicentre study (the Renal EFFECT trial) that compared the efficacy and safety of sunitinib 50 mg/day on Schedule 4/2 (n = 146) with 37.5 mg continuous daily dosing (CDD; n = 146) suggests that a 50 mg/day dose on Schedule 4/2 remains the treatment goal for patients with advanced RCC [Motzer et al. 2012]. The median time to tumour progression (the primary endpoint of the trial) was 9.9 months with Schedule 4/2 versus 7.1 months with CDD (hazard ratio 0.77, 95% CI 0.57–1.04; p = 0.09). The ORR for the two arms was 32% versus 28% (p = 0.444), and median overall survival was 23.1 versus 23.5 months (p = 0.615), respectively. Adverse event profiles were similar between the two schedules.
If required for the management of toxicities, dose modifications can be made in 12.5 mg increments; however, dosing should not decrease below 25 mg/day or exceed 75 mg/day on Schedule 4/2 [Pfizer Ltd., 2012; Pfizer Inc., 2012]. Whilst effective therapy management can often prevent the need for sunitinib dose adjustments, modifications may be required if a patient is taking one or more concomitant agents that inhibit cytochrome P450 enzyme activity (e.g., erythromycin, clarithromycin, ritonavir or grapefruit juice) or enhance cytochrome P450 activity (e.g. rifampicin, dexamethasone, phenytoin, carbamazepine and herbal preparations containing Hypericum perforatum [St John’s Wort]) [Pfizer Ltd., 2012; Pfizer Inc., 2012].
Maximum treatment duration
Findings from the phase III study and the PK/PD meta-analysis indicated that greater exposure to sunitinib is associated with longer time to progression and prolonged overall survival in patients with mRCC [Motzer et al. 2009; Houk et al. 2010]. With longer follow-up in the phase III study, sunitinib therapy was associated with an ORR of 47% (versus 12% with IFN-α treatment; p < 0.001) and with complete responses in 3% of patients (versus 1% of patients receiving IFN-α) [Motzer et al. 2009]. Data from the sunitinib expanded access programme further suggested that sunitinib can be administered in the long term (≥6 months) without significant cumulative toxicity; adverse events were cited as a reason for sunitinib discontinuation in 13% of patients treated for <6 months and only 8% of patients treated for ≥6 months in the programme [Porta et al. 2008].
A subsequent study suggested that switching from first-line sunitinib for any reason other than disease progression may be associated with poor disease control with second-line therapy [Al-Marrawi et al. 2011]. Thus, in order to optimize treatment duration, first-line sunitinib therapy should be continued until disease progression is confirmed; lack of an objective tumour response alone should not be considered an indication of treatment failure.
Adverse event management
Strategies for the active management of treatment-related toxicities should be implemented as soon as sunitinib therapy is commenced, including prophylaxis where appropriate. Strategies should include patient education and close patient monitoring to support early identification and resolution of adverse events and to avoid the need for dose delays, dose reductions or treatment discontinuations [Schmidinger et al. 2010a]. If the toxicity fails to resolve with standard medical interventions, dose reduction should be considered in order to maintain the patient on treatment. When the severity of an adverse event requires the treatment to be stopped, treatment interruption should be attempted to enable the toxicity to subside before treatment discontinuation is considered [du Bois et al. 2009]. Brief treatment interruption may be beneficial in cases of severe (grade 3–4) diarrhoea, haematological toxicity, HFS, rash or mucositis, and uncontrolled hypertension; appropriate prophylaxis is generally sufficient to prevent grade 4 HFS. Sunitinib treatment should be discontinued promptly in cases of serious adverse events, such as pancreatitis and congestive heart failure [du Bois et al. 2009]. However, patients with cardiac adverse events may resume treatment after recovery, provided this is accompanied by close cardiac monitoring [Schmidinger et al. 2009].
Nearly one-third of sunitinib-treated patients in the phase III trial reported hypertension. This is consistent with an earlier, smaller study in which home monitoring of patients’ blood pressure showed a rapid increase in blood pressure during the on-treatment phase of each sunitinib cycle and a reduction during the off-treatment phase [Azizi et al. 2008]. Management of transient treatment-related hypertension should involve antihypertensive medication during the 4-week on-treatment phase of each cycle, with a dose reduction during the off-treatment phase to avoid hypotension [Kollmannsberger et al. 2007; Azizi et al. 2008; Di et al. 2009]. In cases of cardiotoxicity involving a decline in left ventricular ejection fraction, sunitinib treatment interruption and standard medical treatment, for example with angiotensin-converting enzyme inhibitors or diuretics, has been shown to improve cardiovascular status and enable sunitinib treatment to be reinstated [Schmidinger et al. 2009].
Case studies
The case studies that follow describe patients with mRCC who received treatment with sunitinib and in whom simple therapy management strategies enabled treatment duration to be maximized to promote optimal clinical outcomes.
Case study 1
A 53-year-old female presented in July 2006 with thoracic and sacral pain and a history of arterial hypertension. In October 2006, a computed tomography (CT) scan demonstrated a renal tumour with multiple bone metastases (ECOG 0). Radical nephrectomy was performed in November 2006 and histological analysis demonstrated clear-cell RCC (grade 3, pT3a). On the basis of the patient’s disease characteristics (ECOG 0, six bone metastases, normal laboratory values), she was classified with a favourable prognostic risk profile according to MSKCC criteria.
In January 2007, the patient experienced sciatic pain and an increase in right sacral pain, and disease progression was confirmed by CT (Figure 2). Sunitinib was commenced in January 2007 at a dose of 50 mg/day (Schedule 4/2), and the patient received two infusions of zoledronic acid. After two treatment cycles, CT demonstrated disease stabilization, while a subsequent CT scan after four treatment cycles indicated tumour shrinkage and peripheral tumour calcification (Figure 3). Following four cycles of sunitinib therapy, the patient experienced hypothyroidism, grade 2 asthenia and grade 3 HFS. Her hypothyroidism and asthenia were managed with thyroid supplementation and exercise respectively. Grade 3 HFS was managed with daily application of topical creams and rubber insoles to limit pressure on the soles of the feet. From June 2007 to October 2010, the dose of sunitinib was reduced to 37.5 mg/day (Schedule 4/2), to reduce treatment-related toxicity and enable the patient to maintain an active lifestyle; the patient reported reduced toxicities, with grade 0–1 asthenia and grade 0–1 HFS.

CT scan in January 2007 of a patient with favourable-risk, clear-cell mRCC, demonstrating disease progression prior to commencing targeted therapy (Case 1).

CT scan in June 2007 of a patient with favourable-risk, clear-cell mRCC demonstrating tumour shrinkage and peripheral tumour calcification following four cycles of sunitinib 50 mg/day, Schedule 4/2 (Case 1).
To manage symptomatic bone metastases, the patient underwent vertebroplasty to the thoracic vertebrae (T10–T11) in September 2010, with subsequent palliative radiotherapy (L4–L5, S2) in October 2010. Sunitinib 37.5 mg/day was continued until April 2011, when diarrhoea and grade 3 colitis resulted in treatment interruption after 52 months of treatment. Sunitinib 37.5 mg/day was restarted in July 2011 with no evidence of disease progression according to CT. Subsequently, treatment was discontinued because of colitis; at the time of writing, the patient continues to exhibit no evidence of disease progression.
In this patient with favourable-risk clear-cell mRCC, sunitinib therapy was associated with a partial treatment response and prolonged disease control for a total of 55 months. The toxicities reported by the patient were consistent with the known safety profile of sunitinib and were addressed with effective therapy management strategies to optimize the patient’s quality of life.
Case study 2
A 63-year-old female with no relevant medical history presented in March 2009 with an abdominal mass and intermittent blurring of her vision. The patient had a haemoglobin level of 20 g/dL and performance status of 1, and her serum lactate dehydrogenase and calcium levels were normal. CT of the head, thorax, abdomen and pelvis indicated a large mass in the right kidney with splenic and lung metastases (Figure 4). Biopsy confirmed the primary tumour to be clear-cell RCC, deemed to be inoperable, and the patient was classified as intermediate prognostic risk according to MSKCC criteria.
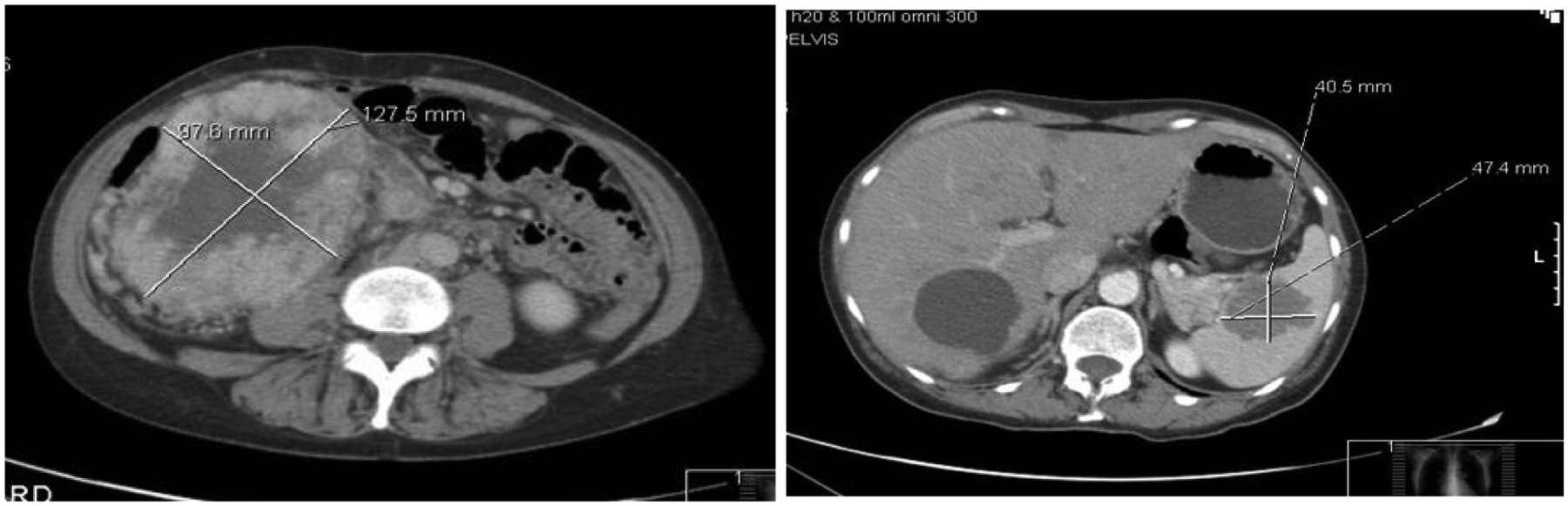
Baseline CT scan in March 2009 of a patient with clear-cell mRCC showing a large inoperable tumour in the right kidney and a mass in the spleen (Case 2).
She was treated with venesection and commenced therapy with sunitinib 50 mg/day (Schedule 4/2), in April 2009. After 14 days of treatment, she developed haematuria that required bladder irrigation and she experienced grade 2 HFS. The HFS was managed through appropriate footwear and the use of topical creams, and the patient was able to remain on sunitinib therapy at a dose of 50 mg/day. A repeat CT scan in June 2009, 12 weeks after the initiation of treatment, demonstrated a good response. In September 2009, 24 weeks after commencing sunitinib therapy, CT indicated a further minor response and the patient underwent splenectomy and right nephrectomy. Subsequently, she experienced acute left-sided weakness and a soft-tissue spinal metastasis was diagnosed. C7 laminectomy was performed in November 2009 and 10 doses of radiotherapy (30 Gy) were administered. The sunitinib dose was reduced to 37.5 mg/day (Schedule 4/2) and further disease control was achieved from January until July 2010, when the patient reported weakness in her right arm and a CT scan revealed brain metastases (Figure 5). Sunitinib therapy was stopped and gamma knife radiosurgery was performed. She commenced steroid therapy but experienced refractory seizures and developed insulin-dependent diabetes mellitus and proximal myopathy. In November 2010, she underwent surgery to remove the epileptogenic tumour and steroid therapy was stopped, with associated improvements in her diabetes and activity levels. Restaging of the brain metastases 3 months after surgery, in February 2011, identified two new, small lesions and gamma knife radiosurgery was repeated. A CT scan of the head, thorax, abdomen and pelvis in April 2011 demonstrated slow disease progression during the 9-month period following the cessation of sunitinib therapy. In April 2011, the patient commenced treatment with everolimus, which was well tolerated and was associated with tumour shrinkage in June 2011. She currently remains on treatment with disease control.

CT scan in July 2010 of a patient with bulky, intermediate-risk, clear-cell mRCC showing the development of brain metastases 15 months after commencing treatment with sunitinib (Case 2).
In this patient with bulky, intermediate-risk disease, sunitinib therapy enabled a large, previously unresectable, primary tumour to be resected successfully. Despite substantial toxicity, the patient was able to continue sunitinib therapy for 15 months, and achieved overall survival of more than 2 years, suggesting that targeted therapy, with effective adverse event management, significantly altered the natural history of disease in this patient.
Future developments
Biomarkers
A major focus of recent research with targeted therapies has been on the identification of clinical, imaging and molecular biomarkers to predict which patients are most likely to benefit from treatment. The utility of sunitinib-associated side effects, including hypertension, hypothyroidism, HFS and more recently neutropenia, thrombocytopenia and asthenia/fatigue, as potential predictive markers of clinical outcomes is currently under investigation.
A retrospective analysis of pooled data from four trials of sunitinib 50 mg/day (Schedule 4/2) in patients with mRCC (n = 544) suggested that sunitinib-associated hypertension is associated with improved clinical outcomes without clinically significant increases in hypertension-related adverse events [Rini et al. 2011]. Patients with treatment-induced hypertension, defined by both systolic and diastolic blood pressure, demonstrated better outcomes than those without treatment-induced hypertension in terms of PFS, overall survival and ORR (p < 0.001); rates of adverse events were similar between patients with and without hypertension [Rini et al. 2011].
The onset of subclinical hypothyroidism within the first 3 months of sorafenib or sunitinib therapy has been identified in a multivariate analysis as an independent predictor of survival (hazard ratio 0.31; p = 0.014) [Schmidinger et al. 2010b]. In this study, the occurrence of subclinical hypothyroidism during treatment was significantly correlated with both the rate of objective remission (p < 0.001) and the median duration of survival (p = 0.014).
A retrospective analysis evaluated treatment-related HFS as a potential biomarker for sunitinib efficacy using pooled data from 1186 patients in nine clinical trials, including 770 patients with mRCC. Patients who developed HFS exhibited significantly better clinical outcomes than those who did not [Puzanov et al. 2011]. On the basis of multivariate analyses, sunitinib-associated HFS remained a significant independent predictor for survival benefit in terms of both PFS and overall survival (p ≤ 0.005). Similar retrospective analyses were performed on the pooled data (n = 770) to evaluate the potential of treatment-related neutropenia and thrombocytopenia and asthenia/fatigue as biomarkers of treatment efficacy in mRCC patients receiving sunitinib. In both Kaplan–Meier and multivariate analyses, sunitinib-associated neutropenia grade ≥2 and thrombocytopenia grade >1 were associated with significantly longer time to tumour progression, PFS and overall survival. Sunitinib-associated neutropenia grade ≥2 was significantly associated with all three efficacy endpoints, while there was a trend towards improvement in the endpoints with thrombocytopenia grade >1 [Donskov et al. 2011]. Asthenia and fatigue were significantly and independently associated with improved time to tumour progression, PFS and overall survival by both Kaplan–Meier and multivariate analyses (p < 0.0001) [Davis et al. 2011].
At the molecular level, it has been suggested that levels of circulating endothelial cells during early treatment cycles may be a useful biomarker for sunitinib response [Gruenwald et al. 2010]. Genomic variability may also explain some of the variability in efficacy and toxicity in patients with mRCC treated with sunitinib, though this remains to be shown prospectively. A recent study showed a correlation between the presence of two single-nucleotide polymorphisms (SNPs) of the VEGFR3 gene (rs307821 and rs307826) and improved PFS in sunitinib-treated patients [Garcia-Donas et al. 2011]. The same study suggested that a CYP3A5 SNP genotype may be associated with poorer sunitinib tolerability.
The use of imaging techniques as noninvasive biomarkers of sunitinib response has also been investigated. VEGF positron emission tomography (VEGF-PET) results have been shown to correspond with immunohistochemical markers and tumour growth [Nagengast et al. 2011], while 18F-fluorodeoxyglucose PET-CT may also have some prognostic significance [Vercellino et al. 2009; Kayani et al. 2011].
Combination and sequential regimens with sunitinib
A number of studies have investigated sunitinib in combination and sequential regimens in mRCC, with the aim of optimizing clinical responses and prolonging treatment duration.
Sunitinib in combination with bevacizumab has yielded mixed results; poor tolerability and high incidences of hypertension (including grades 3 and 4) were reported in a phase I study of mRCC [Feldman et al. 2009]. With chronic treatment, additional toxicities were observed, including grade 3 microangiopathic haemolytic anaemia and grade 3 reversible posterior leucoencephalopathy syndrome. A phase I study of sunitinib in combination with temsirolimus was terminated because of dose-limiting toxicities occurring at low starting doses of both agents [Patel et al. 2009].
The ongoing phase II RECORD-3 (Renal Cell Cancer Treatment with Oral RAD001 Given Daily) study is evaluating sequential therapies, comparing sunitinib followed by everolimus versus the opposite sequence in treatment-naïve mRCC [Knox et al. 2010]. Also in progress, the phase III SWITCH study is investigating sunitinib followed by sorafenib versus sorafenib followed by sunitinib first-line in mRCC [ClinicalTrials.gov identifier: NCT00732914]. Data from these trials will provide further information about optimal sequencing of therapies in mRCC to prolong survival.
Conclusion
Robust clinical trial data and a wealth of clinical experience support the use of sunitinib in the treatment of mRCC, and sunitinib remains a reference standard of care for patients with favourable- and intermediate-risk mRCC. Sunitinib has a well-defined safety profile; most side effects can be managed effectively using appropriate therapy management strategies, with the aim of optimizing treatment dose and duration in order to prolong patient survival and maximize QoL. The case studies outlined in this review illustrate the successful implementation of therapy management strategies, enabling clinical outcomes to be optimized in these patients. An increasing body of evidence is becoming available to suggest that side effects associated with sunitinib therapy, including hypertension, HFS and hypothyroidism, may represent effective markers of treatment response. Ongoing investigations are also evaluating molecular, genomic and imaging factors as potential biomarkers, whilst trials are in progress to determine the optimal sequence of therapies to prolong survival and maximize patient QoL in this setting.
Footnotes
Acknowledgements
Medical writing support was provided by Caroline Patrick and Rachel Mason at ACUMED® (Tytherington, UK) and funded by Pfizer Inc.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Manuela Schmidinger has received honoraria for lectures from Astellas, GlaxoSmithKline (GSK), Novartis, Pfizer and Roche and has acted as a consultant for Astellas, GSK, Novartis, Pfizer and Roche. James Larkin is a member of advisory board panels for Novartis, Pfizer, GSK and Aveo Pharmaceuticals. He has also received research funding from Novartis and Pfizer. Alain Ravaud is a member of global, European and French advisory board panels on renal cell carcinoma for Astellas, Bayer Schering, GSK, Novartis and Pfizer. He has also received institutional grant support for clinical studies from GSK, Novartis, Pfizer and Roche.
