Abstract
Background:
Percutaneous tibial nerve stimulation (PTNS) is widely used in the treatment of neurogenic detrusor overactivity (NDO) in multiple sclerosis (MS); however, controlled studies are still lacking.
Objective:
To assess effectiveness of PTNS in MS patients with NDO unresponsive to pharmacological and behavioural therapies.
Methods:
MS patients with NDO were enrolled. Inclusion criteria were NDO not responding to pharmacological and behavioural therapies. Exclusion criteria were the presence of relevant comorbidities and urinary tract infections. Patients were evaluated using 3-day bladder diaries and validated questionnaires at baseline, after 4 weeks of educational therapy and after 12 PTNS sessions. The primary outcome measure was the percentage of patients considered responders after the behavioural therapy and after the PTNS in a historical controlled fashion (definition of ‘responder’ was reduction ⩾50% of urgency episodes).
Results:
A total of 33 patients (26 women, 7 men) were enrolled. Two patients dropped out for reasons not related to the protocol. Two out of 31 patients (6.5%) and 21/29 (72.4%) were considered responders at visits 1 and 2, respectively. In PTNS responders, a statistically significant improvement in both bladder diary results and standardized questionnaire scores was recorded, compared with that obtained with behavioural therapy alone. No serious adverse events were reported.
Conclusion:
This historically controlled study suggests that PTNS may be effective in improving NDO in MS patients.
Keywords
Introduction
Patients affected by multiple sclerosis (MS) often present micturition disorders (about 65% of cases), 1 including neurogenic detrusor overactivity (NDO) and urge urinary incontinence (UUI), which represents a social discomfort.2–4 The natural course of MS can also be complicated by alterations affecting the upper urinary tract, such as vesico-ureteral reflux, hydronephrosis and loss of kidney function. 5 In patients with MS, urologic conditions can heavily impact the psychological, occupational, domestic and social aspects of quality of life (QoL), limiting liquids intake, sexual activity, and often implies the use of diapers as precautionary measure. 6
Currently, specific and validated questionnaires have been widely used in neurourology, focusing on lower urinary tract symptoms (LUTS) and on their impact on QoL. 7 Percutaneous tibial nerve stimulation (PTNS) is an effective neuromodulatory technique widely used as a third-line 8 treatment for MS patients experiencing LUTS;9–13 however, to our knowledge, controlled studies are still not available. The aim of this study was to evaluate the efficacy of PTNS in the treatment of MS patients with NDO unresponsive to pharmacological and behavioural therapies.
Materials and methods
From July 2020 to December 2020, MS patients affected by urodynamically proven NDO were enrolled in two designed centres (Tor Vergata University Hospital and IRCSS Santa Lucia). All patients underwent general, neurological and urological history collection, along with physical examination. They were checked for urinalysis, urine cytology, urodynamic studies, kidney and bladder ultrasound scan. We included all the patients diagnosed with relapsing/remitting MS in a stable phase with Expanded Disability Status Scale (EDSS) <6.5 14 presenting with NDO. Details about inclusion and exclusion criteria are listed in Table 1.
Inclusion and exclusion criteria.
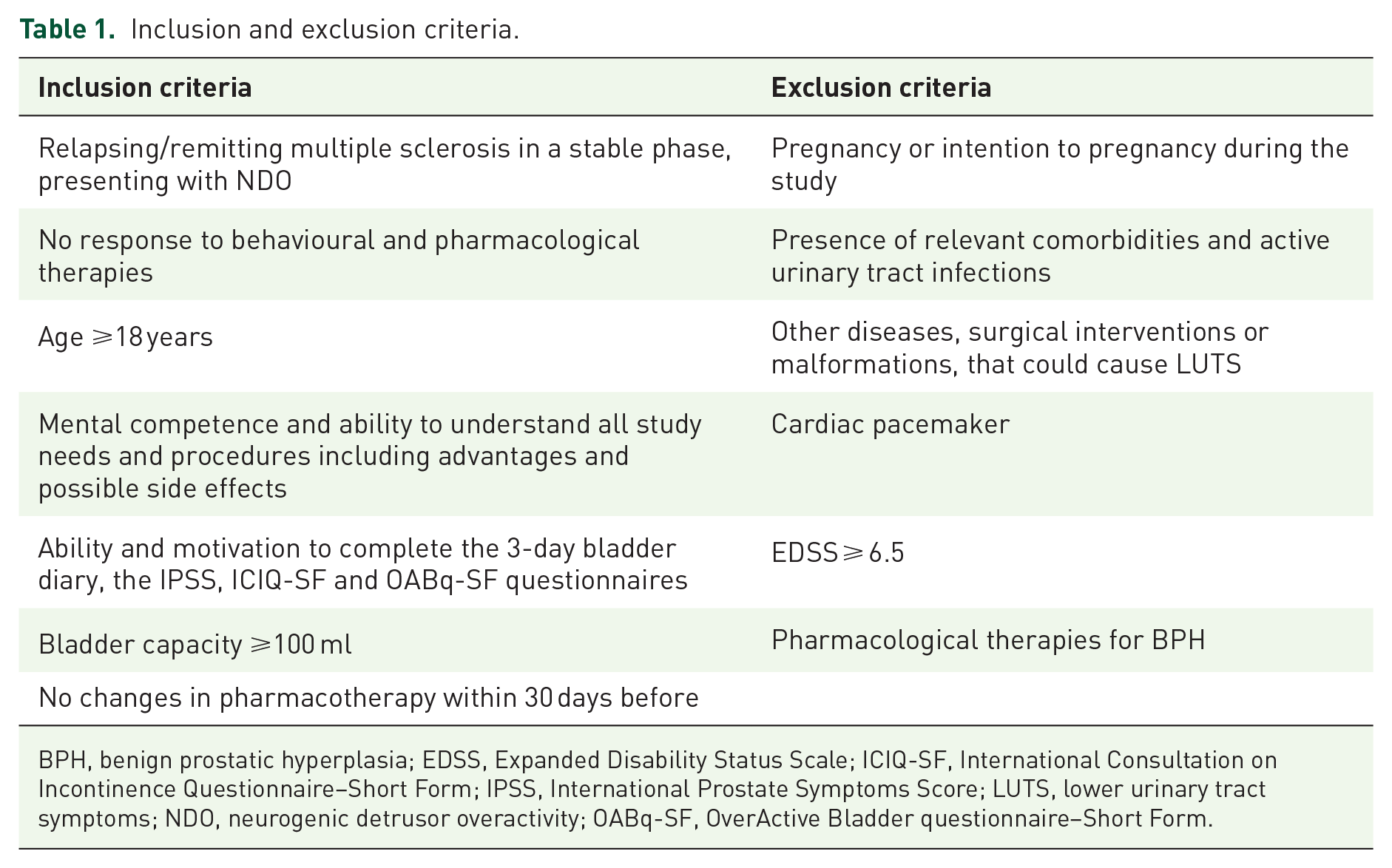
BPH, benign prostatic hyperplasia; EDSS, Expanded Disability Status Scale; ICIQ-SF, International Consultation on Incontinence Questionnaire–Short Form; IPSS, International Prostate Symptoms Score; LUTS, lower urinary tract symptoms; NDO, neurogenic detrusor overactivity; OABq-SF, OverActive Bladder questionnaire–Short Form.
Eligible patients were evaluated at baseline and enrolled in the study (visit 0). All the patients were refractory to a double antimuscarinic or antimuscarinic + beta-3 adrenergic therapy with a persistent condition of neurogenic overactive bladder (OAB). At baseline, patients were evaluated for potential unappropriated habits, and a lifestyle intervention was suggested regarding fluid intakes and bladder training. A 3-day bladder diary was collected, and all the patients filled in validated questionnaires: the International Prostate Symptoms Score (IPSS) questionnaire and QoL score, 15 the International Consultation on Incontinence Questionnaire–Short Form (ICIQ-SF), 16 and the Overactive Bladder questionnaire–Short Form (OABq-SF) bother score. 17
Patients enrolled discontinued OAB pharmacological therapies at least 10 days before starting behavioural therapy, which included lifestyle and bladder training intervention. After 4 weeks of behavioural therapy (visit 1), patients underwent clinical re-evaluation and completed the same questionnaires again. Patients with a reduction of at least 50% of urgency episodes on 3-day bladder diary were considered ‘responders’ and excluded from the study.
In nonresponders, a 6-week PTNS protocol was initiated with 12 stimulation sessions of 30 min each, performed twice weekly. PTNS was performed according to Stoller’s method approved in 2000 by FDA (Food and Drug Administration). 18 The patient was asked to lie down with their leg bent, and a 34-gauge needle was placed 3–4 cm cranially to the medial malleolus between the posterior margin of the tibia and the soleus muscle. Subsequently, a transcutaneous contact electrode was placed on the same leg posteriorly to the medial malleolus. Both needle and electrode were connected to a low voltage (9 V) electrical stimulator (UrgentPC®, Uroplasty, Minnetonka, MN, USA). With a fixed frequency of 20 Hz and a pulse width of 200 ms, the stimulation current (0–10 mA) was increased to obtain a motor response coincident with the big toe’s plantar flexion or with the ‘fan-shaped’ opening of all toes. During the session, the current intensity could be increased basing on patient’s tolerability towards a tingling sensation, that from the stimulation zone, radiated to the sole of the foot’. If no clear motor response was shown, the needle was removed, and the insertion procedure was repeated. In most patients, the motor response was accompanied by a sensitive tingling response in the sole of the foot. At the end of treatment (visit 2), patients were asked to complete a 3-day bladder diary and the validated questionnaires. A CONSORT flow diagram 19 is outlined in Figure 1.

CONSORT flow diagram.
Continuous variables were summarized using mean and standard deviation (SD); frequencies and proportions were used to report categorical variables. Student’s t test for unpaired data was performed to compare results of bladder diaries and questionnaires scores over time. Fisher’s exact test was used for categorical variables. Data analysis was conducted using Stata 16.1 software (StataCorp LLC, College Station, TX, USA). Statistical significance was defined as p value <0.05.
Results
A total of 40 consecutive patients were screened. Seven patients were excluded because did not meet inclusion criteria. Thirty-three patients were enrolled, 26 females and 7 males. Two male patients dropped out for reasons not related to the protocol.
Table 2 shows baseline demographic characteristics. All patients were able to void spontaneously. All male patients did not receive any benign prostatic hyperplasia (BPH) therapy. Thirty-one patients completed baseline assessment; two patients (6.5%) were considered responders at visit 1 (one male and one female).
Clinical and demographic baseline characteristics.
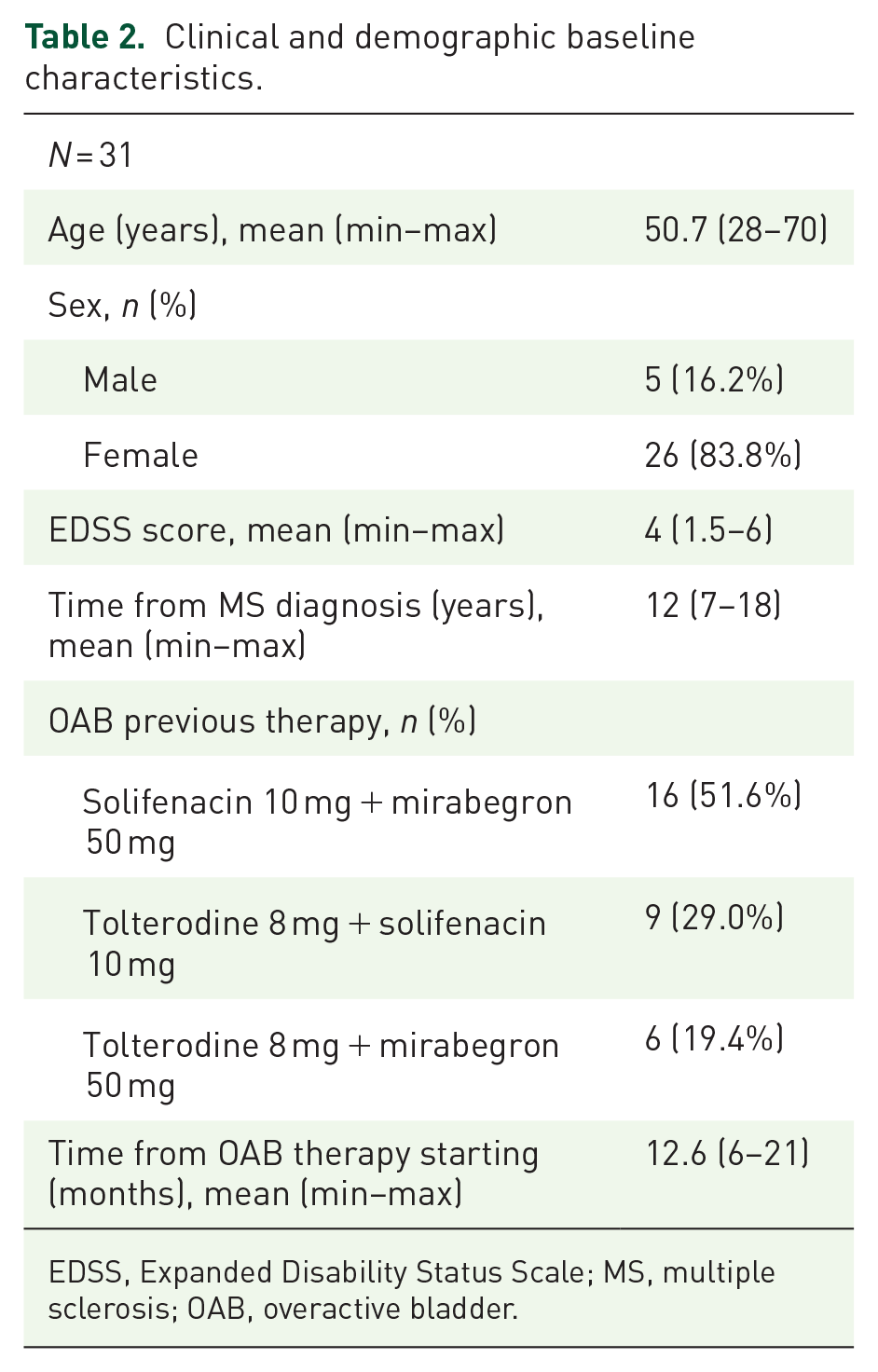
EDSS, Expanded Disability Status Scale; MS, multiple sclerosis; OAB, overactive bladder.
The remaining 29 patients (25 females and 4 males) underwent PTNS according to the study protocol. At visit 2, 21/29 patients (72.4%) were finally considered responders. Overall, the number of urgency episodes per day in the cohort study decreased from 6.89 ± 3.51 at visit 1 to 3.14 ± 2.66 at visit 2 (p < 0.001).
Data from bladder diaries and standardized questionnaires (IPSS, IPSS-QoL, ICIQ-SF and OABq-SF) were recorded at visits 0, 1 and 2, as shown in Tables 3 and 4 for PTNS responders and nonresponders, respectively. In PTNS responders, there was a 72% reduction in urgency episodes per day between visits 1 and 2, while in PTNS nonresponders, this reduction was 22% during the same time span. Nevertheless, the reduction of urgency episodes was statistically significant in both groups. Sex differences in PTNS response were not significant (p = 0.3).
Results of bladder diaries and questionnaires scores and in PTNS responders.

ICIQ-SF, International Consultation on Incontinence Questionnaire–Short Form; IPSS, International Prostate Symptoms Score questionnaire; OABq-SF, OverActive Bladder questionnaire–Short Form; QoL, quality of life; SD, standard deviation; UUI, urgency urinary incontinence.
p value between visit 1 and visit 0.
p value between visit 2 and visit 1.
Results of bladder diaries and questionnaires scores and in PTNS nonresponders.
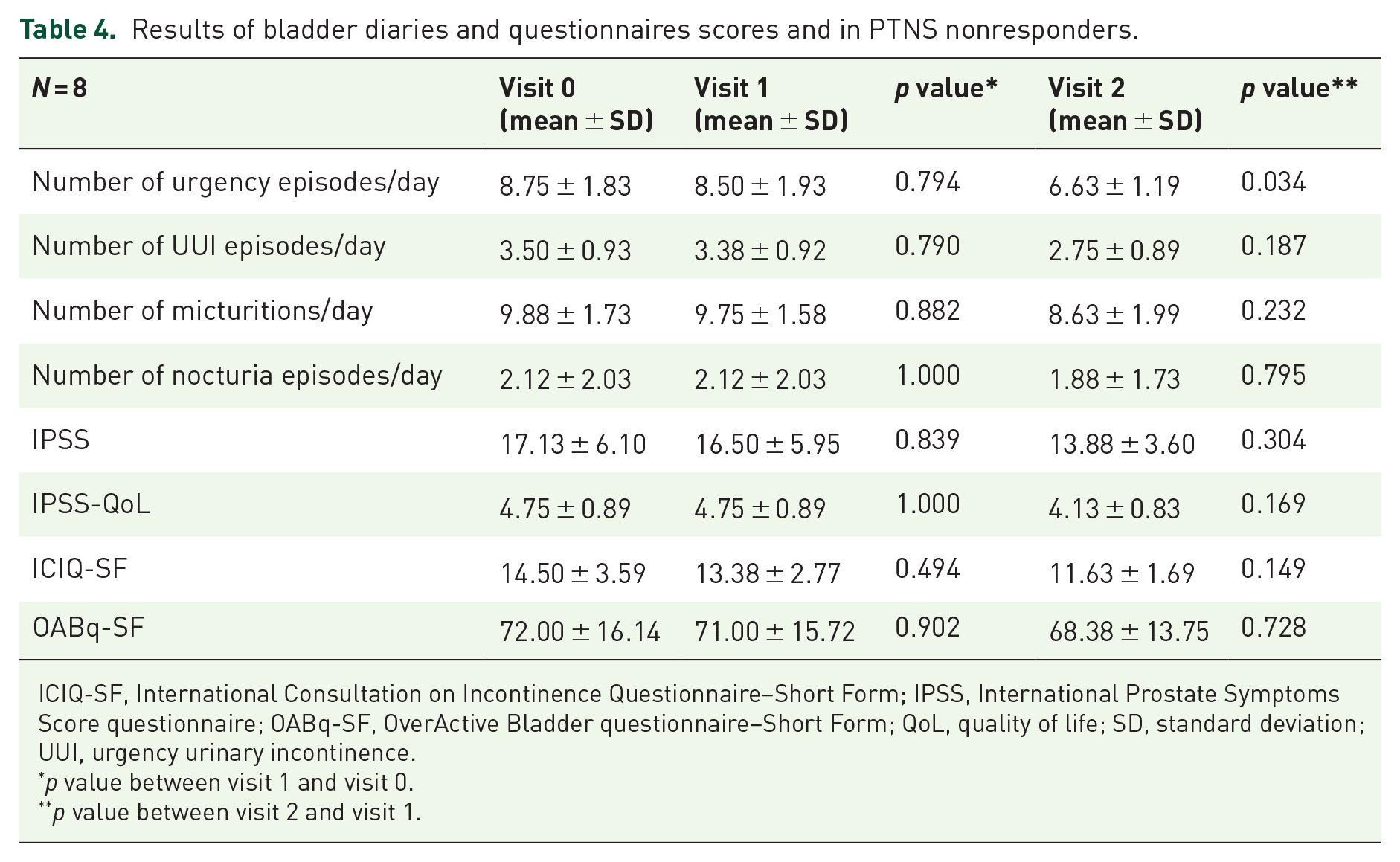
ICIQ-SF, International Consultation on Incontinence Questionnaire–Short Form; IPSS, International Prostate Symptoms Score questionnaire; OABq-SF, OverActive Bladder questionnaire–Short Form; QoL, quality of life; SD, standard deviation; UUI, urgency urinary incontinence.
p value between visit 1 and visit 0.
p value between visit 2 and visit 1.
The treatment was well-tolerated by all participants, and none reported pain after it.
Discussion
The role of PTNS in the treatment of OAB has been investigated in several studies.20–22 Despite some bias (including heterogeneity of populations, selection of clinical and urodynamic parameters and definitions of therapeutic success), PTNS is widely considered as a feasible and effective treatment for OAB. 12 A recent systematic review and meta-analysis confirmed the efficacy of PTNS in treating LUTS in patients with MS. 13 To our knowledge, however, controlled studies are missing.
Owing to ethical reasons, we were not allowed to enrol patients for a ‘true’ control group. Indeed, comparing ‘PTNS’ versus ‘no treatment’ in patients was considered inappropriate by our ethics committee. Thus, we opted for a historically controlled study, in which nonresponders to pharmacological and behavioural treatments (historical group) were used as the ‘control’ group for PTNS results. 23
As stated in the inclusion criteria, a bladder capacity ⩾100 ml (recorded by bladder diaries) was arbitrarily considered sufficient, in order to offer a possible advantage for patients undergoing PTNS. Indeed, previous studies highlighted that OAB patients with very low baseline cystometric capacity are more prone to be unresponsive to PTNS. 20
At baseline, enrolled patients completed the wash-out period (at least 10 days) before starting behavioural therapy. In our opinion, the duration of wash-out period was sufficient in order to get a fair baseline values, given the antimuscarinic and beta-3 adrenergic pharmacokinetics.24,25
This study seems to confirm the efficacy of peripheric neuromodulation in patients with MS-related NDO. After 12 sessions of PTNS, most of the patients (72.4%) reported a significant reduction of frequency, urgency and urge incontinence episodes in bladder diaries. IPSS, IPSS-QoL, ICIQ-SF and OABq-SF scores had all been affected positively by treatment, with a significant reduction of scores compared with that obtained with behavioural therapy alone. Of note, patients classified as nonresponders at visit 2 (i.e. reduction of less than 50% of urgency episodes after PTNS) showed a statistically significant reduction in urgency episodes.
In this cohort study, female patients were prevalent (about 84%). This finding is in line with the literature, in which major evidence is available for the treatment of urinary and sexual dysfunction in females.26,27 In comparison, there is limited evidence for the effectiveness of PTNS in the male population, and there is no evidence that PTNS cures UUI in this setting. 28 Nevertheless, male patients treated with PTNS in this study showed conflicting results (50% responders versus 50% nonresponders), and no statistically significant differences were observed between male and female success rates (p = 0.3). These findings should be interpreted with caution, given the small sample size.
This study is aligned to prior findings. In a prospective, open-label trial published by Gobbi et al., 10 18 patients with MS and refractory LUTS were treated with PTNS. Ten patients were affected by relapsing–remitting multiple sclerosis (RRMS), seven by secondary progressive multiple sclerosis (SPMS) and one by primary progressive multiple sclerosis. None of the patients enrolled in the trial had symptoms of peripheral neuropathy. At 3-month follow-up, 89% of patients, evaluated with the Patient Perception of Bladder Condition (PPBC) questionnaire, were satisfied by the treatment results. No statistically significant reduction in urinary leakage was observed, however. One patient with advanced disability and detrusor sphincter dyssynergia (DSD) SPMS-related and another patient without disability but also with DSD did not show any improvement.
De Sèze et al. 29 performed a multicenter prospective study enrolling 70 patients with MS and refractory OAB, free from disease relapses in the previous 3 months, treated with transcutaneous posterior tibial nerve stimulation. 82.6% of patients reported an improvement in urgency, with a complete resolution of this symptom in 51.3% of cases; 66.7% of cases reported a reduction in frequency, in 62% of cases there was an improvement in continence and 44.9% of patients were completely dry. Patients had a reduction from two to six times of daily voiding, from two to three times of nocturnal voiding and of 2.7 times of weekly urine loss, respectively. Those findings were correlated with an increased voided volume (+43 to 89 ml) and a reduced post-void residual (PVR) (−16 to 55 ml).
Results from a prospective, open-label study by Zecca et al. 30 on all-subtypes MS patients treated with PTNS (51% with RRMS) suggest that on long-term follow-up (up to 24 months), there is sustained effectiveness of PTNS after an initial positive response.
In this study, we performed a treatment protocol of 12 sessions in a 6-week period, each session lasting for 30 min, as originally proposed by Stoller in the late 1990s. 18 In our experience, we reported a statistically significant improvement not only in urgency episodes per day but also in urgency urinary incontinence episodes, number of micturitions and nocturia episodes per day, as shown by bladder diaries. Furthermore, IPSS, IPSS-QoL, ICIQ-SF and OABq-SF showed similar results at visit 0 and visit 1, while they were significantly improved at visit 2 after PTNS. Interestingly, the number of urgency episodes per day in PTNS responders decreased from 6.67 (visit 0) to 1.76 (visit 2) (–76.3%).
We previously reported a comparison between the results of PTNS performed weekly versus PTNS performed three times per week in patients with OAB. The frequency of stimulation did not affect the results of treatment, except for the advantage of an earlier achievement of a clinical improvement (4 weeks instead of 10–12). 31
Long-term data are limited but suggest the need of a maintenance treatment. In 2013, Peters et al. 32 proposed an average of 1.3 treatments per month to ensure good symptoms control over time. Maintenance therapy is not easily feasible, considering the 33% dropout rate, or loss of efficacy for PTNS sessions performed with pauses longer than 2 weeks of about 30%. In a more recent study, Kabay et al. 33 evaluated the efficacy and treatment intervals for PTNS in neurogenic OAB with MS, offering periodic additional treatments during 1 year in patients who finished an initial course of 12 consecutive weekly sessions. PTNS was applied at 14-day intervals for 3 months, 21-day intervals for 3 months and 28-day intervals for 3 months. This study demonstrated a significant improvement in daytime frequency, nocturia, urgency episodes, voided volume, UUI episodes and the durability through to 12 months of prolonged therapy protocol. Alternatively, Van der Pal et al. 34 proposed the subcutaneous implantation of the electrostimulator, activated by the patient at home. Our group proposed to perform a home maintenance therapy by transdermal electrostimulation in the perimalleolar area. Unfortunately, this home protocol is affected by a high dropout rate, also considering the costs of disposable electrodes.
In agreement with the literature, in this study, we did not find any short- or long-term PTNS-related adverse events.
This study has several limitations, such as the small sample size. Moreover, due to the ethical reasons explained above, we designed a historically controlled study, where the same patients received two different treatments at two different times. Probably, PTNS results may have benefitted from the previous drugs, lifestyle and bladder training leading to a possible selection bias. Therefore, more powered studies are needed to confirm our results.
Conclusion
In this historically controlled study, we investigated the role of PTNS in patients with MS-related NDO refractory to pharmacological and behavioural therapy. After PTNS, we reported significant improvement in subjective perception of urinary dysfunction and in urinary symptoms. Although further studies are needed, according to our experience PTNS is a well-tolerated, minimally invasive technique showing good results in MS-related, refractory NDO.
