Abstract
Background:
High sodium intake is associated with cardiovascular and kidney disease, as well as the development of lower urinary tract symptoms (LUTS). The amount of salt intake can influence the development of symptoms, but reduction may benefit patients by lowering the presentation of LUTS. However, there is a lack of systematization of evidence between salt intake and LUTS.
Objectives:
Determine the relationship between a high salt intake and LUTS, and the impact of salt intake reduction on the development of LUTS.
Design:
Systematic review.
Data sources and methods:
We included observational studies and clinical trials that assessed any clinical manifestations of LUTS and any method of measuring salt intake to establish a comparison between high and low salt intake. A search was conducted on PubMed, Scopus, Ovid-Medline, WOS, and Embase databases in February 2024, including studies published at any time until the date of the search. Risk of bias was assessed using the Newcastle-Ottawa Scale and risk of bias in non-randomized studies of interventions. A qualitative synthesis of the included studies was performed, and a meta-analysis was not conducted due to the heterogeneity of how LUTS and salt intake were measured.
Results:
We obtained 3085 articles, from which 10 were included in this review. In regards to urinary incontinence and general LUTS, patients who had a salty diet had more frequency and severe symptoms according to the Overactive Bladder Symptom score (H: 286, 1% vs N: 356, 0.6% vs N: 225, 0.8%; p < 0.001) and International Prostate Symptom score (H: 688, 2.5% vs N: 670, 1.7% vs B: 419, 2.3%; p < 0.001) respectively, compared to patients who had neutral or bland diets.
Conclusion:
Some evidence suggests a possible association between a high salt intake and LUTS, and lowering salt intake might help to improve symptoms. We suggest that future studies improve clinical trial designs to clarify the relationship and methods to assess salt intake and measurement of LUTS.
Plain language summary
Our research systematizes all available evidence on the relationship between high sodium or salt intake and the presence of lower urinary tract symptoms. We found evidence suggesting a relationship between high sodium or salt intake and the presence of lower urinary tract symptoms. Furthermore, we found that reducing salt intake can improve symptoms. However, our findings were not generalizable due to the different methods used to measure sodium or salt intake and the wide variety of urinals evaluated, as well as some shortcomings in the studies found. Future research using better methods and in diverse populations is important to obtain conclusive results.
Introduction
High sodium intake is strongly associated with noncommunicable diseases, including cardiovascular and kidney disease.1,2 The World Health Organization (WHO) recommends a daily intake of less than 2 g of sodium. 1 The American Heart Association and the United States Department of Agriculture recommend restricting sodium intake to 2300 mg per day for the general population. The typical American diet contains between 8000 and 1000 mg of sodium. Instead, some ethnic diets include even higher sodium levels. 3
In 2017, high sodium intake was responsible for more than half of diet-related deaths and two-thirds of disability-adjusted life years (DALYs). In China, Japan, and Thailand, high sodium intake was the principal dietary risk factor for deaths and DALYs. In Beijing, the average sodium intake was 6.9% g/day in men and 5.8% g/day in women.4,5 Reducing sodium intake has beneficial effects in blood pressure, leading to a better prognosis in patients with cardiovascular and kidney disease. 6 In addition, reducing salt consumption has been associated with other beneficial effects such as improved lower urinary tract symptoms (LUTS).7,8
LUTS include all those symptoms caused by alterations in the organs and structures of the lower urinary tract or related to it. The LUTS are divided into three groups: storage symptoms, voiding symptoms, and post-micturition symptoms.9,10 However, each of these groups encompasses a series of specific symptoms with their own characteristics, the evaluation of which determines their proper assessment. For instance, urinary incontinence is included in storage symptoms, and it should be described by specifying relevant factors such as type, frequency, severity, triggering factors, social impact, effect on hygiene, quality of life, measures used to manage leakage, and whether the individual seeks or desires help. 9 An epidemiological study carried out in the USA, UK, and Sweden determined that the prevalence of LUTS in men and women over 40 years is greater than 70%. 11 The presence of LUTS negatively compromises the quality of life of those who suffer from it and is associated with higher levels of anxiety and depression. 12
The amount of salt in the diet can influence the development of LUTS 7 through several mechanisms, either indirect or direct. A larger amount of salt present in the diet can increase symptoms such as an overactive bladder. 8 Patients with high salt intake could benefit from a reduction in dietary sodium by both decreasing LUTS 13 as well as reducing the risk for cardiovascular and renal disease. However, these are not universal results, with other research finding no association between higher salt intake and nocturnal polyuria or urinary incontinence.14,15
The current evidence on the relationship between sodium intake and LUTS is limited and dispersed across studies with varying methodologies and outcomes. There is a lack of comprehensive synthesis assessing whether reducing sodium intake may improve LUTS. Therefore, the aim of this study is to conduct a systematic review to determine the association between a high salt intake and the presence of LUTS. Likewise, a summary of intervention studies related to the reduction of salt consumption and its impact on the presence of lower urinary tract symptoms will be made.
Methods
Protocol and registration
The protocol followed the methodology proposed by the Preferred Items for Systematic Reviews and Meta-Analyses (PRISMA) (Table S1), and was reported on the Open Science Framework (OSF) database (https://doi.org/10.17605/OSF.IO/BS846).
Study selection data extraction
Our systematic review included observational studies and clinical trials. Studies that reported any clinical manifestations of LUTS and any method of measuring salt or sodium intake to establish a comparison between high and low salt or sodium intake were included, as well as studies whose participants were equal to or above 18 years of age, irrespectively of their sex, and available for full review. Studies such as scoping reviews, letters to the editor, conference abstracts, and others were excluded.
Database and search strategy
An exhaustive search about the development of LUTS in patients with either low or high sodium or salt intake was carried out in the following databases: PubMed, Scopus, Ovid-Medline, WOS, and Embase. The search strategy was built using the PubMed Advanced Search Builder and was then adapted and introduced in each database (Table S2). There was no time-restriction for included publications. Furthermore, a manual search of additional studies was conducted throughout the citations in the included studies. The conducted search was carried out in February 2024 and lasted 2 months. There were no limitations on language or publication date.
Data extraction
All articles obtained from the electronic search were exported to the “Rayyan QCRI” software, and posteriorly duplicates were eliminated. Four authors (RAF-L, MLG-L, FIR-V, JA-V) screened each article independently by title and abstract, and subsequently assessed by full-text review those which could be potentially included. Any discrepancies or conflicts in the decision were debated among the authors. If there were still discrepancies, a fifth author (VAB-Z) acted as a diriment for its resolution. The data from the included articles was extracted into a Google Sheets form, and was done by all reviewers. The data extracted included: author, year and country of publication, in or out-hospital setting, body mass index (BMI), comorbidities, sample size, LUTS, salt or sodium intake, and method of assessment of salt or sodium intake, symptoms severity, and effect sizes.
Risk of bias assessment
To assess the risk of bias of observational studies, we used the Newcastle-Ottawa Scale (NOS). The clinical trial was evaluated with the risk of bias in non-randomized studies of interventions (ROBINS-I) tool. The risk of bias assessment of every article was done independently by every author. Any discrepancies or conflicts were debated among authors. If there were still discrepancies, a fifth author acted as a diriment for their resolution. The cut-off point for the NOS was seven stars. We assigned a star to every item following the instructions provided per item in the NOS manual. For the ROBINS-I tool, risk of bias was determined following the tool’s manual. A high risk of bias was determined in any domain if there was at least a high-risk response option in any of the domain’s questions.
Data synthesis
We present the most relevant characteristics of the studies in tables. We decided not to conduct a meta-analysis due to the differences between the studies found regarding the way salt or sodium intake was measured and the variety of LUTS reported. Instead of meta-analysis, we conducted a qualitative synthesis of the included studies.
Results
Study selection
The study selection process is depicted in the PRISMA flow diagram (Figure 1). Across all databases, 5337 references were retrieved, and duplicates were eliminated. Posteriorly, 3085 references were screened by title and abstracts, and 22 articles remained for full-text review. After the screening, eight articles were included for qualitative synthesis. In addition, 335 references were retrieved manually from the included articles, and five studies were screened for possible inclusion. Two studies were included from the mentioned manual search, leaving a total of 10 included articles for the qualitative synthesis.7,8,14–21

PRISMA flow diagram.
Study characteristics and risk of bias
The general characteristics of the studies are depicted in Table 1. The present review included eight cross-sectional studies and two non-randomized clinical trials. Most of the studies were conducted in Asian countries (seven studies), and the majority were conducted in out-of-hospital settings (eight studies). Most prevalent comorbidities in participants were diabetes mellitus, hypertension, hyperlipidemia, and renal dysfunction. All studies were included for qualitative synthesis
General characteristics of studies.

COPD, chronic obstructive pulmonary disease; DM, Diabetes Mellitus; NR, No report.
Risk of bias
Among the eight cross-sectional studies evaluated, four were classified as low risk of bias, while the remaining four were considered to have high risk of bias. The main concerns were in the selection domain, specifically in terms of representativeness and justification of the sample size, as well as confounding control (Table 2). Regarding the two non-randomized clinical trials, one study was rated as moderate risk, whereas the other one was considered high risk of bias due to risk in the domains of confounding, measurement of the outcomes, and selection of the reported results (Table 3).
Newcastle-Ottawa scale risk of bias assessment.

The symbols “*” and “**” correspond to the Newcastle–Ottawa Scale (NOS) scoring system. Each asterisk represents one awarded point (star) for the respective item/domain. Stars are summed across items to obtain the total NOS score for each study, which is then used to appraise overall methodological quality and risk of bias.
ROBINS-1 risk of bias assessment.

Measurement of salt intake
Regarding the methodology used to measure sodium or salt intake in the studies, it is observed that three different techniques were used. Two studies7,16 used a preference scale in which the preference for sodium or salt consumption was self-evaluated, and it was grouped into tasteless, neutral, and salty. On the other hand, another two studies14,17 carried out a more objective measurement of sodium consumption, by measuring sodium in urine, which, after applying a formula adjusted to age, height, and weight, helped to calculate the real intake of sodium in the diet. Alternatively, four studies15,18–20 used questionnaires and food frequency tables, which contain the amount of salt present in each food. By applying these, the total salt consumption was estimated by analyzing the diet of each participant (Table 4).
Characteristics of cross-sectional studies.
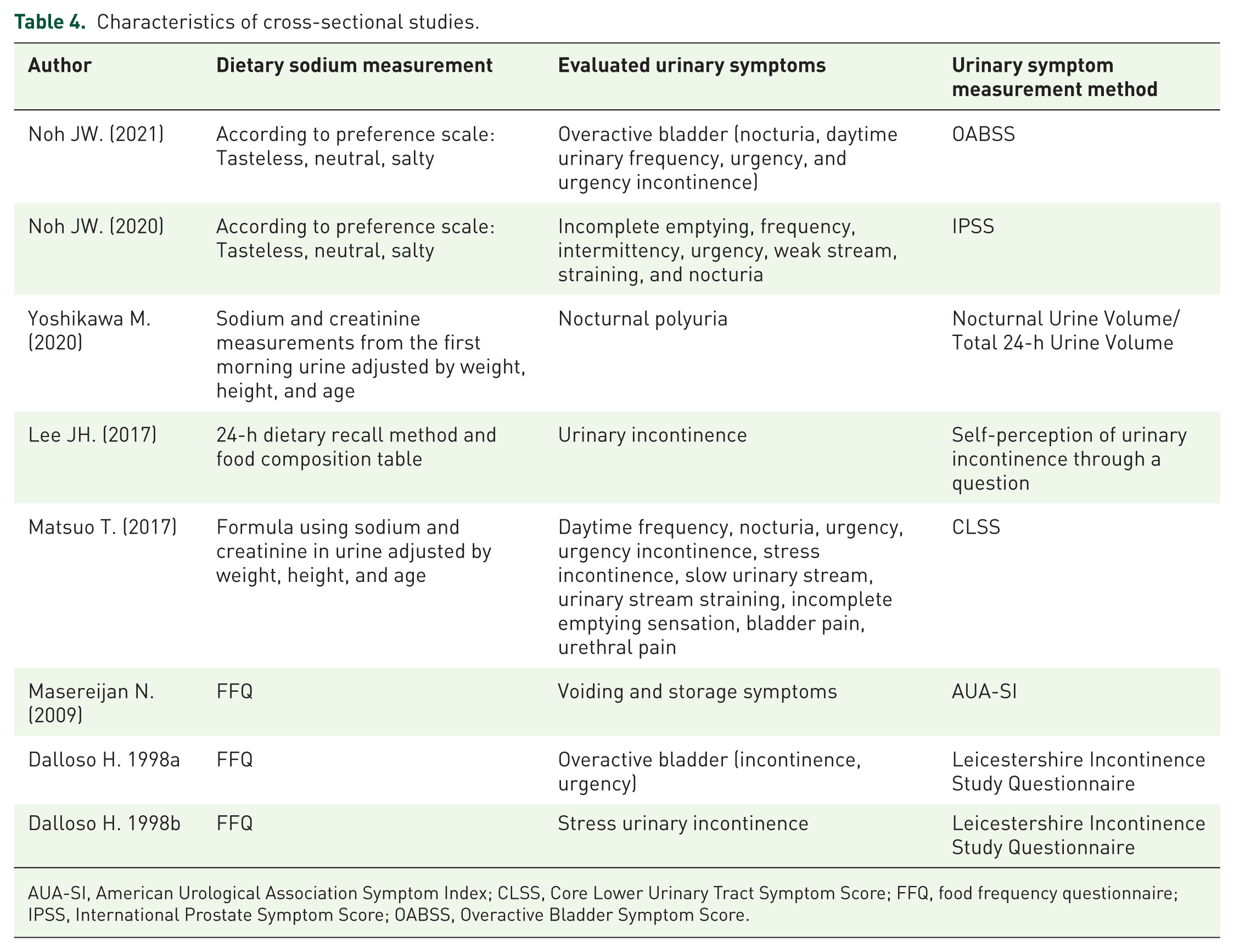
AUA-SI, American Urological Association Symptom Index; CLSS, Core Lower Urinary Tract Symptom Score; FFQ, food frequency questionnaire; IPSS, International Prostate Symptom Score; OABSS, Overactive Bladder Symptom Score.
Measurement of LUTS
Each of the studies included in our review evaluated different LUTS and therefore used different methods for their measurement. Two studies14,15 analyzed a particular symptom and did not use questionnaires to measure them, while the other studies analyzed two or more symptoms, which, in turn, were included in the standardized questionnaires used in those respective studies (Table 4). Thus, two studies8,16 used the Overactive Bladder Symptom Score (OABSS) to analyze those symptoms (Table 5). A single study 7 evaluates prostate symptoms using the International Prostate Symptom Score (IPSS), while, on the other hand, another study 18 uses the American Urological Association Symptom Index (AUA-SI) to analyze the same symptoms (Table 4). Two studies17,21 used the Core Lower Urinary Tract Symptom Score (CLSS) to analyze various LUTS (Table 5). In contrast, two studies19,20 use specific questions from the questionnaire developed for the Leicestershire Incontinence Study to assess the specific symptoms of each of their studies (Table 4).
Characteristics of non-randomized clinical trials.

CLSS, Core Lower Urinary Tract Symptom Score; OABSS, Overactive Bladder Symptom Score.
OABSS score and salt intake
In one study 16 where daytime urinary frequency, nocturia, urgency and urgency incontinence are assessed, evidence that patients who had a salty diet had more frequency of symptoms as well as higher percentage of severe symptoms according to the OABSS score in comparison to patients who had neutral or bland diets (Salty: 285, 1% vs Neutral: 359, 0.6% vs Bland: 225, 0.8%; p < 0.001; Table 6).
Results of cross-sectional studies according to salt intake self-perception.
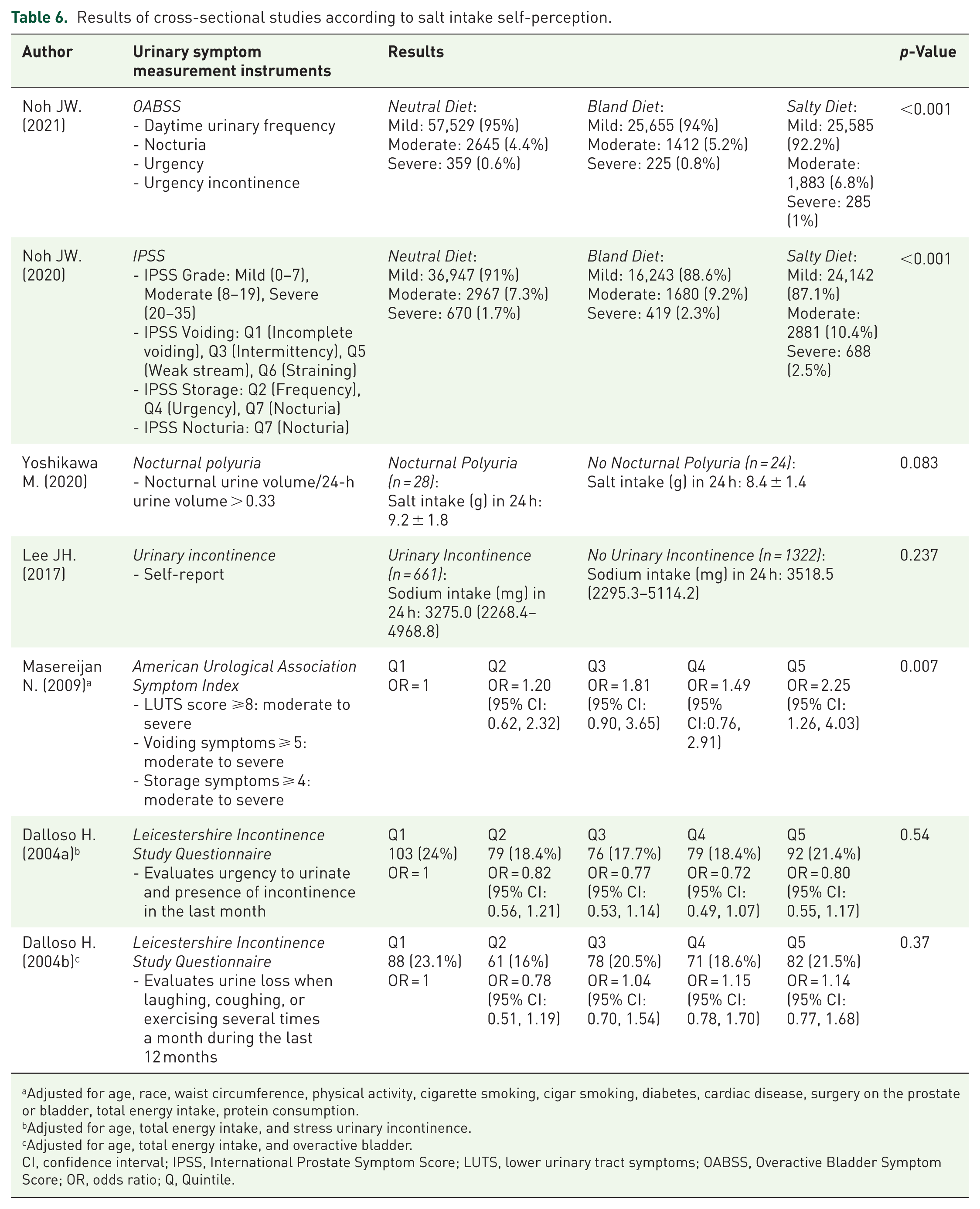
Adjusted for age, race, waist circumference, physical activity, cigarette smoking, cigar smoking, diabetes, cardiac disease, surgery on the prostate or bladder, total energy intake, protein consumption.
Adjusted for age, total energy intake, and stress urinary incontinence.
Adjusted for age, total energy intake, and overactive bladder.
CI, confidence interval; IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms; OABSS, Overactive Bladder Symptom Score; OR, odds ratio; Q, Quintile.
Urinary incontinence and salt intake
In regards to urinary incontinence, one study 16 evidence that patients who had a salty diet had more frequency of symptoms as well as higher percentage of severe symptoms according to the OABSS score in comparison to patients who had neutral or bland diets (Salty: 285, 1% vs Neutral: 359, 0.6% vs Bland: 225, 0.8%; p < 0.001). On the other hand, another study 15 showed that patients with urinary incontinence consumed less daily sodium (3275.0 mg (2268.4–4968.8)) compared to patients without urinary incontinence (3518.5 mg (2295.3–5114.2), but the difference was not significant (p = 0.237). In addition, in two studies,19,20 patients who were in higher quintiles of sodium consumption were more likelihood to present urinary incontinence, but results were not statistically significant (OR 0.80 (95% CI: 0.55, 1.17); p = 0.54, OR 1.14 (95% CI: 0.77, 1.68); p = 0.37; Table 6).
Nocturnal polyuria and salt intake
One study 14 showed that patients with nocturnal polyuria had a higher dietary sodium intake (9.2 ± 1.8 g) compared to the group that did not have nocturnal polyuria (8.4 ± 1.4 g), however, the difference was not significant (p = 0.083; Table 6).
General LUTS and salt intake
The presence of LUTs in general was evaluated in one study 18 where it was found that those men evaluated with American Urological Association Symptoms Score who were in the highest quintile of sodium consumption were up to two times more likely to present LUTS in relation to those who were in the lowest quintile, with statistically significant differences (OR 2.25; p = 0.007). In addition, another study evaluated 7 patients with the IPSS score who had a salty diet had a higher frequency of symptoms as well as a higher percentage of severe symptoms compared to neutral and bland diets (Salty: 688, 2.5% vs Neutral: 670, 1.7% vs Bland: 419, 2.3%; p < 0.001; Table 6). Finally, another study 17 where CLSS was used, patients with lower salt intake had lower scores in daytime frequency (Q1) (0.6 ± 0.8 vs 1.1 ± 0.9; p < 0.001) and nocturia (Q2) (1.3 ± 1.0; p < 0.001) compared to patients with high salt intake ((Q1): 1.1 ± 0.9, (Q2): 1.8 ± 0.9)). In addition, quality of life (QOL) scores were worse in patients with high salt intake (3.8 ± 1.6) compared with patients with low salt intake (3.6 ± 1.6, p = 0.045; Table 7).
Results of cross-sectional studies according to cut-off point of salt intake.

CLSS, Core Lower Urinary Tract Symptom Score.
Reduction of salt intake and LUTS
In other two studies where reduction in salt intake was assessed,8,21 patients evaluated with CLSS who reduced their salt intake after 12 weeks had a significant reduction in daytime frequency (Q1) (p < 0.001), nocturia (Q2) (p < 0.001), urgency (Q3) (p = 0.001), urgency incontinence (Q4) (p < 0.001), slow stream (Q6) (p = 0.039), as well as an improvement in QOL (p < 0.001), compared to patients who did not reduced their salt intake. Patients evaluated with OABSS who reduced their salt intake after 12 weeks had a significant reduction of diurnal frequency (Q1) (p < 0.001), nocturia (Q2) (p < 0.001), urgency (Q3) (p < 0.001), urgency incontinence (Q4) (p = 0.003) and total score (p < 0.001; Table 8).
Results of non-randomized clinical trials.

CLSS, Core Lower Urinary Tract Symptom Score; OABSS, Overactive Bladder Symptom Score.
Discussion
Main findings
The main results of our study show that there is evidence suggesting an association between sodium or salt intake and LUTS. However, it is not possible to generalize this statement due to the heterogeneity of the symptoms evaluated, the methods used for their assessment, and the ways in which salt consumption was measured. Likewise, there is evidence suggesting that interventions to reduce salt intake could decrease LUTS in certain populations.
LUTS are defined from the individual’s perspective, who is generally, but not necessarily, a patient within the healthcare system. Symptoms are voluntarily expressed or triggered by the individual or may be described by their caregiver. 9 In this regard, the instruments used in the studies included in this analysis cover aspects as general as symptoms associated with overactive bladder or prostate-related symptoms (Table 6), which include symptoms evaluated separately with other instruments. 22 Thus, the overactive bladder syndrome may include urinary incontinence or nocturia, making it difficult to determine which symptom exactly improves when a patient has low salt intake. If we consider negative results when symptoms are assessed separately, such as urinary incontinence, the outcomes are negative.19,20 This could suggest that other symptoms of the overactive bladder syndrome improve with reduced salt intake rather than incontinence itself.
Another aspect to consider in interpreting our results is the heterogeneous methods used to assess salt intake. Some authors suggest that methods for evaluating salt consumption can be categorized into pre-ingestion measurement methods (controlled intake), peri-ingestion measurement methods (concurrent), and post-ingestion measurement methods. 23 In this regard, validated 24-h recall methods are a reasonable approach to estimating sodium intake in large epidemiological studies and individual clinical counseling sessions. 23 In contrast, although methods such as single 24-h urine collection, spot urine collection, or 24-h recall methods are valuable for estimating mean population sodium intake, they are less suitable for individual-level assessment. 23 Consequently, few studies included in this analysis employed an appropriate method for evaluating salt consumption (Table 4), which, combined with the heterogeneous assessment of LUTS, limits the generalization of the results showing this association7,17 and the interpretation of sodium levels above which symptom improvement is observed.
Pathophysiology
Despite these limitations, two non-randomized clinical trials8,21 in patients with overactive bladder suggested that reducing salt intake improves LUTS. Although the mechanisms explaining this association are not fully understood, it is suggested that a high salt diet affects the hyperactivation of the autonomic nervous system, particularly the innervation of the prostate and bladder. 24 In addition, sodium-induced hypertension may reduce the efficacy of alpha-blocker treatment, 25 which is used for managing prostate symptoms. 26 In patients with overactive bladder, studies in animal models have demonstrated that excessive salt intake enhances the expression of oxidative stress and neurotransmitter-related genes and induces bladder ischemia. 8 Moreover, there is a mechanical effect of increased urine volume with higher salt intake. In salt-sensitive Dahl rats, a high salt load induces urinary storage dysfunction by upregulating the epithelial Na channel in the bladder epithelium. 27 In spontaneously hypertensive, salt-sensitive rats, excessive salt intake was also found to decrease voided volume while increasing urinary frequency after salt overload.27,28 Another study showed that symptoms associated with urine storage could be induced by elevated adenosine triphosphate and prostaglandin E2 levels, caused by suppression of bladder blood flow in salt-sensitive rats fed a high salt diet. 28
Implications
We believe that our findings may support the hypothesis that an intervention to reduce salt intake could decrease LUTS, due to the biological plausibility of this association and the results of some clinical trials. Furthermore, this assessment should be conducted with standardized methods, such as sodium measurement in urine, which could support the cause of the presence of symptoms in case there is a high quantity of salt in urine. Indeed, some studies have shown that anxiety, 29 caffeine consumption, 30 and self-management 31 can affect LUTS. It is important to emphasize that the recommendation to reduce salt intake to improve LUTS could be an adjunct measure rather than a replacement for standard treatment, which includes not only medications but also non-invasive surgical options.32–34 Second, public health practitioners may take salt intake reduction into account, as it may play a harmful role in the development of lower urinary tract symptoms. It is important to explore the harmful effects of sodium in the urinary tract, besides the well-known effects on the cardiovascular system, such as the arterial pressure, or the development of kidney disease.
Limitations
This systematic review has several limitations, although they are related to the individual studies included. First, there is significant heterogeneity regarding the measurement of LUTS and intake of salt. There are various methods by which salt consumption was assessed, usually through sodium evaluation, though not always using the recommended method for individual assessments. 23 These methods include, for example, self-perception according to salt preference, morning urinary sodium measurement, or the 24-h dietary recall method using food composition tables or food frequency questionnaires. Also, LUTS measurement was done utilizing various questionnaires, such as the OABBS, the IPSS or even self-perception of urinary incontinence. The methodological heterogeneity of how LUTS and salt consumption were measured difficulted the generalization of results for specific symptoms. Second, biases and the small number of clinical trials prevented conducting subgroup analyses, publication bias, and a meta-analysis to evaluate the intervention’s effectiveness. Finally, not all studies assessed confounders of their results, such as caffeine consumption. More studies are needed that control for possible confounders. Moreover, those studies that assessed confounders such as anxiety, 29 caffeine consumption, 30 or self-management 31 did not explore other important variables such as total water intake.
Conclusion
In conclusion, there is some evidence suggesting that increased salt intake is associated with a higher prevalence of LUTS and that interventions to reduce salt consumption may improve symptoms. However, the heterogeneity and low quality among studies limit the ability to draw definitive conclusions. Future studies should include clinical trials with larger sample sizes and standardized measurement of key variables, such as 24 h sodium urinary excretion to assess salt intake, and validated questionnaires individualized for each symptom to evaluate LUTS.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251408905 – Supplemental material for Association between sodium or salt intake and lower urinary tract symptoms: a systematic review
Supplemental material, sj-docx-1-tau-10.1177_17562872251408905 for Association between sodium or salt intake and lower urinary tract symptoms: a systematic review by Rodrigo Alonso Fribourg-Liendo, Percy Herrera-Añazco, Mariana Lucia Garcia-Lopez, Jheram Abarca-Velarde, Fatima Ramos-Vallejos and Vicente Aleixandre Benites-Zapata in Therapeutic Advances in Urology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

