Abstract

Keywords
Dear Editor,
Recently, several phase III studies showed that the combination of antiangiogenic tyrosine kinase inhibitors (TKIs) and immunotherapies had shown greater efficacy than TKI monotherapy for the primary treatment of metastatic renal cell carcinoma (mRCC).1–4 It is known that all- and high-grade hypothyroidism is a relevant adverse event during TKI therapy. 5 Similarly, immune-checkpoint inhibitors (ICIs) are also associated with endocrine dysfunctions, mainly hypothyroidism. Based on these data, the risk of hypothyroidism is potentially increased with a combination between TKIs and ICIs. With this hypothesis, we performed a pooled analysis from pivotal phase III studies investigating the combination of an ICI and a TKI against sunitinib for treatment-naïve mRCC patients, evaluating the pooled risk of hypothyroidism of the combination therapy when compared with monotherapy. In particular, we considered data from the Keynote-426, Javelin Renal-101, CLEAR and CheckMate-9ER trials.1–4 The CheckMate-214 and IMmotion-151 6 trials were excluded because they omitted TKIs in the experimental arm with ICI.
The pooled analysis with a random-effects model revealed that the incidence of any grade hypothyroidism among subjects with TKIs in combination with ICI was higher compared with that of subjects treated with sunitinib monotherapy [risk ratio (RR) = 1.42, 95% confidence interval (CI): 1.10–1.83; p = <0.01 I2 = 81%; Figure 1(a)]. The pooled analysis with a random-effects model revealed that the incidence of grade ⩾3 hypothyroidism was not statistically significantly higher in patients treated with TKIs in combination with ICI [RR = 1.74, 95% CI: 0.43–7.05; p = 0.44; I2 = 0; Figure 1(b)].
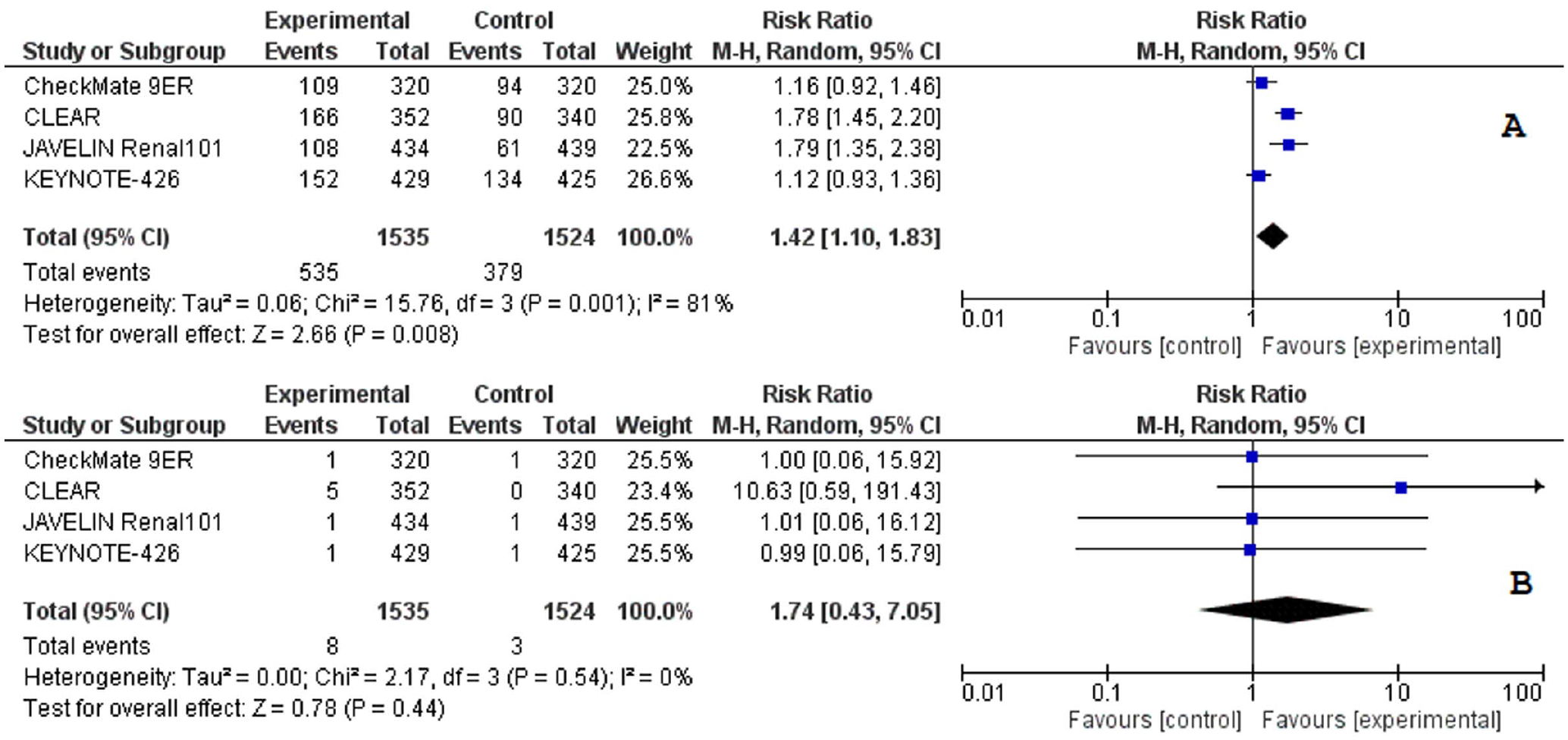
Forest plots of risk ratio for any grade hypothyroidism comparing experimental with sunitinib (a), for grade ⩾3 hypothyroidism comparing experimental to sunitinib (b).
Additionally, knowing that ipilimumab + nivolumab and atezolizumab + bevacizumab are also treatment options for mRCC and that both studies used sunitinib in the control arm, we added Check-Mate 214 and IMmotion 151 data in a subsequent analysis.6,7 However, in line with a previous report, 8 the data confirmed no increasing incidence in the RR for any grade and grade ⩾3 hypothyroidism with ICI combinations for mRCC (Supplemental material online).
Significant limitations of our evaluation should be disclosed, namely the nature of the meta-analysis based on literature data rather than on individual patients’ data and substantial heterogeneity between different experimental arm combinations, therefore definitive conclusions are precluded. Nonetheless, we showed an increased incidence of any grade hypothyroidism, without a statistically significant increase in severe cases, with ICI–TKI combinations for mRCC. Our findings suggest that the risk of hypothyroidism with combinations versus monotherapy in this setting can be relevant but manageable, preventing concerns about the synergy between the two mechanisms of endocrine toxicity.
Supplemental Material
sj-pdf-1-tau-10.1177_17562872211029784 – Supplemental material for Antiangiogenic tyrosine kinase inhibitors and immunotherapy as first-line treatment for metastatic renal cell carcinoma: is there an increased risk of hypothyroidism?
Supplemental material, sj-pdf-1-tau-10.1177_17562872211029784 for Antiangiogenic tyrosine kinase inhibitors and immunotherapy as first-line treatment for metastatic renal cell carcinoma: is there an increased risk of hypothyroidism? by Giandomenico Roviello, Sebastiano Buti, Carlo Cattrini, Alessia Mennitto, Carlo Messina, Chiara Airoldi, Gianmarco Vannini and Melissa Bersanelli in Therapeutic Advances in Urology
Footnotes
Conflict of interest statement
The author(s) declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
