Abstract
Increasing bacterial resistance combined with a steady decline in the discovery of new antibiotics has resulted in a global healthcare crisis. Overuse of antibiotics, for example, in the poultry and cattle industry, and misuse and improper prescription of antibiotics are leading causes of multidrug resistance (MDR). The increasing use of antibiotics, particularly in developing countries, is a big concern for antibiotic resistance and can cause other health threats such as increased risk of recurrent infections and increased risk of cardiovascular death with chronic use of macrolides. Carbapenems are the last line of defense in many cases of resistant infection, but trends show that resistance against these agents is also increasing. This narrative review is based on relevant literature according to the experience and expertise of the authors and presents an overview of the current knowledge on antibiotic resistance, the key driving factors, and possible strategies to tackle antibiotic resistance. Collectively, studies show that hospital-wide antibiotic stewardship programs are effective in decreasing the spread of antibacterial resistance. As resistance varies according to local patterns of use, it is essential to observe the epidemiology at both a regional and an institutional level. Furthermore, adaptation of clinical guidelines is necessary, particularly for inpatient care. Future guidelines should include a justification step for continued treatment of antibiotic treatments and criteria for selection of antibiotics at the start of treatment. Nonantibiotic prevention strategies can limit infections and should also be considered in treatment plans. Vaccines against MDR organisms have shown some efficacy in phase II trials in critical care patients. Nonimmunogenic and microbiologic treatment options such as fecal transplants may be particularly important for elderly and immune-compromised patients.
Introduction
The following narrative review is based on presentations from the second Foro en Infecciones Urinarias Recurrentes (FIUR2) symposium, a Latin American forum to discuss current trends and challenges in treating recurrent urinary tract infections. The literature herein was compiled based on nonsystematic review of the current literature and the expertise of the authors/presenters.
Urinary tract infections (UTIs) are one of the most common types of bacterial infection found in both inpatient and outpatient settings: the most frequent infections in hospitals are associated with urinary catheter use, and UTIs in the community are very common. Antibiotics are classically the method of choice to treat UTIs and other bacterial infections. The discovery and use of antibiotics in the 1900s represent one of medicine’s great success stories. Following the discovery of penicillin and sulfonamides, 14 different classes of antibiotics were identified between 1940 and 1970, and new molecules were discovered within each group. 1 However, from the 1980s onwards, the rate at which new antibiotics were discovered steadily declined. Between 2003 and 2007, only five antibiotic agents were approved by the US Food and Drug Administration, the lowest number in 10 years. To compound this issue, in recent years, antibiotic resistance has been on the rise. Several factors contribute to the global antibiotic resistance threat, including the emergence of multidrug resistant organisms, the overuse and prolonged use of antibiotics, cultural aspects that influence antibiotic prescription, and the decline in the discovery of new antibiotic agents. The US Centers for Disease Control and Prevention (CDC) reported that over 23,000 deaths were caused by antibiotic resistance in 2013. 2 Furthermore, antibiotic resistance increases mortality rates by 30–44%, increases the length of hospital stay, and raises hospital expenses.3–5 Treatment options for multidrug resistant infections are becoming increasingly limited and new solutions are urgently needed.
Trends in antibiotic use
The consumption of antibiotics to treat infection remained stable in high-income countries between 2000 and 2015; however, significant increases were observed in developing countries during this period (Figure 1). 6 The top three countries with fluoroquinolone resistance of Gram-negative bacteria in urine cultures found in a global 2010 study were: India 75%, Panama 70%, and Mexico 49%. 7 A systematic literature review and meta-analysis of studies conducted in primary care shows that the use of antibiotics for a respiratory or urinary infection in the past 3 months increases the chance of recurring infection by a resistant species by up to fivefold. The increased risk decreases over time but persists for up to 12 months after treatment. 8
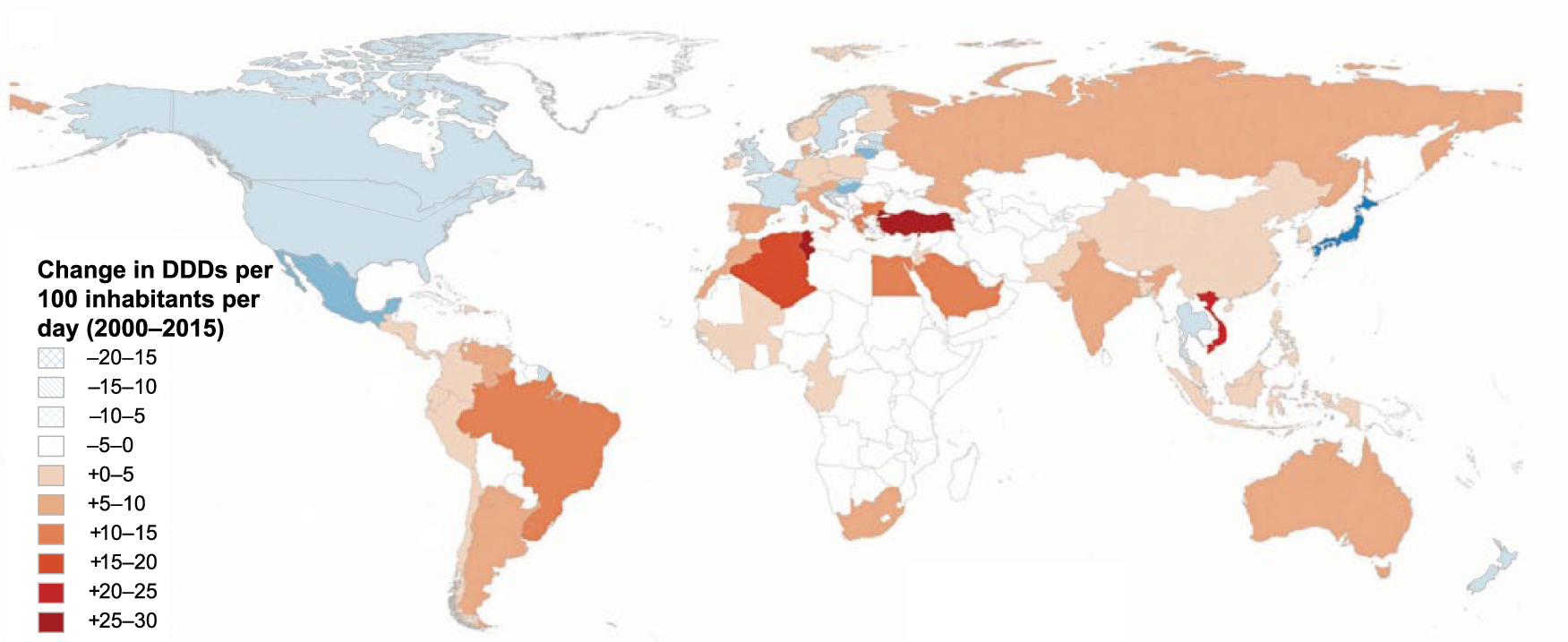
Global antibiotic use per country between 2000 and 2015. 6
Epidemiology
Escherichia coli is the most common uropathogenic species and causes >85% of UTIs.9–11 In the SENTRY antimicrobial surveillance program, cultures collected from patients with urinary tract, bloodstream, respiratory tract, and skin and soft tissue infections were collected from 1997 onwards by over 80 centers worldwide, including 10 centers located in Latin American countries (Brazil, Argentina, Chile, Uruguay, Colombia, Venezuela, and Mexico).10,12 The most common pathogenic species in urine cultures from patients with UTI between 1997 and 2000 were E. coli, Klebsiella spp., Pseudomonas aeruginosa, and Proteus mirabilis. 10 Ciprofloxacin-resistant bacteria were found in more than 20% of urine samples, while resistance to ampicillin and trimethoprim/sulfamethoxazole was above 50%. 10 Alarmingly, more recent results from the SENTRY study, analyzing isolates from bloodstream, pneumonia, and skin and soft tissue infections showed that Klebsiella pneumoniae carbapenemase-producing K. pneumoniae emerged in 2009 (10 strains) and is increasing in prevalence (44 strains in 2010; p < 0.0001). 12 Quinolone-resistant bacteria were found in 50% of hospitalized UTI patients in Asia, and the percentage of trimethoprim-/sulfamethoxazole-resistant species was up to 70% in Africa and South America (2003–2010). 13 Carbapenems represent the last line of defense in many cases of resistant infection, but general trends suggest that resistance against these agents is increasing. In 2013, the CDC reported that 11% of the enterobacteria found in the urinary tract are carbapenem resistant, 63% of acinetobacter are multidrug resistant, and 23% of klebsiella and E. coli have an extended-spectrum beta-lactamase (ESBL) phenotype. 2 Close to two thirds of infectious disease specialists have seen at least one infection without treatment option in the past year.
Importance of local epidemiology
Studies show that it is essential to observe the local epidemiology on both a regional and an institutional level, and to determine treatment plans accordingly. A Mexican study recognized E. coli, klebsiella, and pseudomonas as the most common pathogenic species in samples from patients with a multidrug resistant infection in the region. 14 A 10-year study conducted in tertiary oncology hospitals found that >50% of nosocomial isolates and >20% of community-acquired infections had an ESBL profile. Resistance to ciprofloxacin was 65%, and an increase was observed in ESBL-producing E. coli and K. pneumoniae in nosocomial infections between 2004 and 2013. 14 In a Colombian study (n = 1959), resistance levels in E. coli isolated from patients with UTIs were highest against ampicillin (61%), followed by nalidixic acid (48%), trimethoprim/sulfamethoxazole (48%), and ciprofloxacin (42%). 15 A recent small study (n = 87) from Argentina showed substantial differences between resistance in community- and healthcare-acquired UTIs. Prevalence of multidrug resistant pathogens was significantly higher in healthcare-acquired UTIs (49%) than in community-acquired UTIs (10%; p < 0.01). 16 Comparably, large differences are observed between different institutions (e.g. in private hospitals with surgical, internal medicine, gynecological, and obstetrics patients versus trauma hospitals or hospitals that only receive gynecological patients) in Mexico. In general, public institutions tend to have higher rates of resistance. 14 Elderly patients are a particularly vulnerable group; they often experience recurrent infections and reside in chronic care centers and nursing homes, where they run increased risk of exposure to resistant organisms. Patients residing in chronic care are twice as likely to contract a resistant bacterium compared with patients in acute centers (14% versus 6% in the USA). 17 Moreover, repeated admission of elderly patients to hospital and back to chronic care centers or nursing homes increases the chances of emergence and dissemination of multidrug-resistant species among this group. A 6-month study with elderly patients in 2010 found >80% resistance to ampicillin and >60% resistance to ciprofloxacin. 18 Meta-analysis shows that the percentage of bacterial resistance is high and rising among patients residing in chronic care centers or nursing homes, with multidrug resistance (MDR) levels rising from 23% in studies conducted before 2012 to 32% in those conducted after 2012. 19
Bacterial resistance: management strategies
The emergence of penicillin resistance was predicted soon after the discovery of the drug itself and had emerged before its formal introduction as a medical treatment. Exposure to antibiotics drives mechanistic adaptation of bacteria and selects for resistant species. Although different classes of antibiotics act on different pathways to eliminate bacteria, these microorganisms employ equally diverse ways to counteract the drugs.
Mechanisms of antibiotic resistance
Antibiotics typically act through five main processes: (a) inhibition of cell-wall synthesis (penicillin, cephalosporins, vancomycin); (b) disruption of cell membrane function (polymyxins such as colistin); (c) inhibition of protein synthesis (tetracycline, streptomycin, chloramphenicol); (d) inhibition of nucleic acid synthesis (rifamycin, quinolones); and (e) action as antimetabolites (trimethoprim, sulfamethoxazole; Figure 2).20,21 Agents that act on the bacterial cell wall or membrane [(a) and (b)] are often classified as bactericidal. Antibiotics that block protein synthesis, deoxyribonucleic acid (DNA) synthesis, or metabolism (c–e) inhibit bacterial growth and are classified as bacteriostatic. However, these categories may overlap depending on the bacterial species, dosing, growth conditions, etc., and the distinction offers little relevance in clinical practice.22,23 Certain antibiotics, such as nitrofurantoin, also employ a multifaceted mode of action. Nitrofurantoin is effective against common UTI pathogens and resistance is low (79–92% cure rates).24–26

Main mechanisms of antibiotic function. 21
Multidrug-resistant, extensively drug-resistant, or pandrug-resistant organisms employ several resistance mechanisms that act on different pathways simultaneously. The major resistance mechanisms of bacteria include: production of antibiotic-modifying enzymes; decreased drug permeability; synthesis of ribonucleic acid (RNA), DNA, protein, or cell-wall components that alter or bypass the drug target; and active extrusion of the drug with efflux pumps.27,28
Resistance-coding genes can be trafficked between bacterial species via plasmids or transposons. An interesting example is β-lactamase, an enzyme that recognizes and inactivates β-lactams. Carbapenemases, a type of β-lactamase, have versatile hydrolytic properties that inactivate different types of antibiotics (including penicillins, cephalosporins, monobactams, and carbapenems). 21 Resistance genes to β-lactams are sometimes found on DNA plasmids that are present in the bacterial cytoplasm. These plasmids may contain multiple resistant genes conferring resistance to different antibiotics. This mechanism is classically employed by enterobacteriaceae to pass on MDR. Moreover, DNA plasmids with resistant genes can also be transmitted between different species.
Key drivers of antibiotic resistance
Several key factors have been identified that drive and promote antibiotic resistance. Besides evolution-driven mutations of bacterial DNA, resistance can also be driven by environmental and epidemiological factors. 29 The main cause of environmental selection is the overuse of antibiotics. Large-scale use of antibiotics in the poultry and cattle industry is one of the leading causes of MDR. 29 The quantities of antibiotics used in this industry are staggering, and the first evidence of a Gram-negative colistin-resistant organism was found in cattle, and in waste waters originating from cattle-raising farms. 30
Misuse and improper prescription of antibiotics is the other leading cause of MDR. A large-scale study conducted in the US in 2010 and 2011 showed that, during that time, between 30% and 50% of ambulatory prescriptions were incorrect for either indication (incorrect), dose (improper or subtherapeutic), or time period for treatment. 31 Dissemination and spread of bacteria further drives antibiotic resistance (e.g. via improperly sterilized hospital beds or equipment, or by improperly sterilized hands). Other promoting factors that can potentially be modified by improved medical practices include: the purchase of antibiotics without a prescription, which often results in unindicated use or subtherapeutic doses; suboptimal use of vaccination or other preventative strategies resulting in infections or complications from infections that could have been prevented; increased intercontinental travel, which promotes the spread of resistant species between countries; and the lack of new antibiotics coming to market. 29
The underlying belief that antibiotics are benign and nontoxic has led to several misconceptions, including that inflammatory symptoms require immediate treatment, that it is more effective to treat infections with a broad-spectrum antibiotic, and that it is advisable to switch to or add another antibiotic to the treatment if the patient does not respond. Recent studies, however, have shown that prolonged antibiotic use may not be as safe as presumed. A large-scale meta-analysis in 2015 showed that chronic use of macrolides increased the risk of cardiovascular death. 32 Comparable results were found in a study including women aged 60 years and over and who were using antibiotics for ⩾60 days: an increased risk of dying from any cause [hazard ratio (HR) = 1.27; 95% confidence interval (CI) 1.07–1.49] and from cardiovascular causes (HR = 1.58; 95% CI 1.02–2.46), but not from cancer (HR = 0.86; 95% CI 0.63–1.16), was observed. 33
Cultural factors dictate how people behave toward each other, and also play a role in antibiotic resistance. 34 When the perceived distance between societal roles in a hierarchical structure (distance to power) is large (e.g. between doctors, nurses, and patients), it decreases the likelihood that patients or nurses will correct doctors when a mistake is made. In addition, uncertainty avoidance stimulates the tendency of physicians to prescribe more antibiotics for a longer time than necessary, likely due to a tendency toward risk avoidance and beliefs around the importance of convention. Profligate prescribing patterns are more common in countries that score highly for uncertainty avoidance and distance to power.35,36 Southern European countries tend to display these cultural characteristics more than those in northern Europe, and they are also typical of Latin American countries, among others. 37
Strategies to improve the use of antibiotics
The threat of antibiotic resistance is still growing, and soon there will be few treatments options left for many patients. It is imperative that we regain control over bacterial infections and implement management strategies to counteract MDR. To this end, antimicrobial stewardship programs have been developed. Hospital-wide strategies are now being employed to mitigate improper prescription and overuse of antibiotics in the medical sector.
Hospital-wide strategies to stop bacterial resistance
Vigilance programs are being established to stop the emergence and spread of multidrug-resistant organisms. These programs include several basic and complementary measures to achieve effective surveillance of antibiotic resistance, prevent emergence and spread of MDR, and help develop alternative treatments (Figure 3). 38 One of the basic measures, hand hygiene, is a key factor to prevent the spread of both nonresistant and resistant bacteria alike. In addition, isolation policies are essential to contain the spread of MDR in inpatient care.
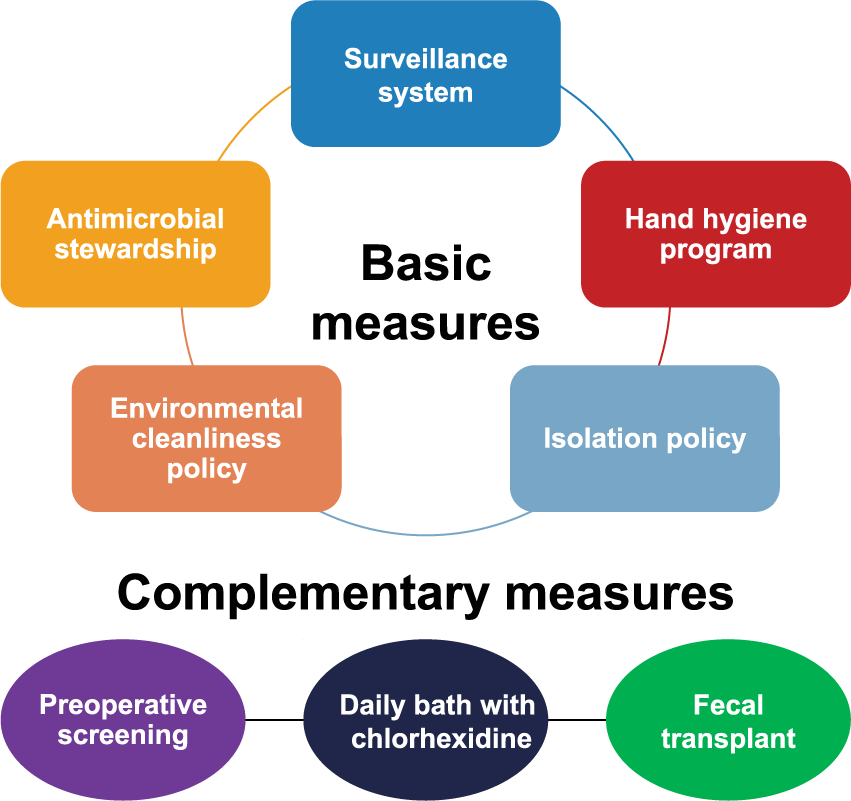
Strategies to prevent the emergence and transmission of resistant microorganisms in nosocomial setting. 35
Antimicrobial stewardship
In 2007, the Infectious Disease Society of America and Society for Healthcare Epidemiology of America published guidelines on how to develop institutional programs to enhance antimicrobial stewardship.3,4,39 They defined three main objectives for stewardship programs: (a) to obtain the best clinical result in patients undergoing antibiotic treatment in a cost-effective manner; (b) to minimize the risk of adverse events and drug–drug interactions associated with antibiotic use; and (c) to prolong the lifespan of currently available antibiotics by reducing the selection pressure and the resulting emergence of resistance. 3 Several examples show the benefits of antimicrobial stewardship. A randomized controlled trial at the Hospital of the University of Pennsylvania showed that antimicrobial stewardship programs resulted in more appropriate use of antibiotics and a better cure rate when compared with usual practice [appropriate antibiotic use: 90% versus 32% (relative risk (RR) = 2.8; 95% CI 2.1–3.8); cure rate: 91% versus 55% (RR = 1.7; 95% CI 1.3–2.1); failure rate: 5% versus 31% (RR = 0.2; 95% CI 0.1–0.4)]. 40 Similarly, hospitals that employed pharmacist-managed prescription of aminoglycosides and vancomycin significantly reduced adverse events such as hearing loss (46% reduction), kidney damage (34% reduction), extended hospital-stay length (12% reduction), and death from any cause (7% reduction). 41 A study conducted in Argentina showed that progressive hospital-wide implementation of an antibiotic stewardship program was able to significantly reduce the emergence of resistant species and overall costs by $913,236 within 3 years [P. aeruginosa from 19% to 0% (p = 0.02); methicillin-resistant Staphylococcus aureus from 47% to 30% (p = 0.04); P. mirabilis from 40% to 27% (p = 0.05); Enterobacter cloacae from 50% to 10% (p = 0.03)]. 42
To implement appropriate intervention strategies, clinical guidelines must be adapted. Each hospital should implement surgical prophylaxis guidelines, and guidelines for the management of frequent infections such as pneumonia, UTI, abdominal infections, and skin and soft tissue infections. It is advised that guidelines include a justification step for continued antibiotic treatment of patients and a plan to assess treatment via multidisciplinary teams.43,44 In addition, guidelines should include criteria for selection of an antibiotic at the start of treatment. Patient-related factors should influence the choice of antibiotic, something that is rarely considered in current guidelines. Finally, awareness among patients must be increased, and physicians have the responsibility to inform patients of the increased mortality risks associated with antibiotic use.32,34
Strategies for outpatient care
The majority of antibiotic prescriptions are made in primary care. Guidelines for antibiotic stewardship for ambulatory patients are already available, and aimed at primary care physicians, specialists, and dentists, and at the emergency room, chronic facilities, and healthcare systems. 45 These guidelines include a list of situations in which antibiotic use can be optimized. For example, upper respiratory tract infections are not indicated for empirical antibiotic treatment, and proper diagnosis of streptococcal infections must be attained before treating with antibiotics (pharyngitis in children). More effort should be made to explain the reasons why antibiotics are not indicated, and to meet patients’ needs when giving an alternative treatment.
Nonantibiotic prophylaxis
Nonantibiotic prevention is a strategy to limit infection and therefore to control resistance, and includes immunogenic, nonimmunogenic, and microbiologic options. Immunogenic options are vaccines against multidrug-resistant organisms and immunostimulants. Phase I and II trials of vaccines against multidrug-resistant organisms have shown some efficacy in critical care patients.46–48 Data from meta-analyses show nonspecific immunomodulators have efficacy in recurrent UTIs.49,50 Nonimmunogenic and microbiologic treatment options may be particularly important for elderly and immune-compromised patients. Fecal transplantations, for example, have been performed successfully in the US, and have been suggested as an alternative preparation for liver transplant patients with multidrug-resistant bacteria in their cultures. 51 Nonimmunogenic options for UTI include vaginal estrogen for postmenopausal women, methenamine salts, and D-mannose.52–54 There is currently insufficient evidence to support the use of oral estrogens, probiotics, cranberry and ascorbic acid supplements, and nonantibiotic intravesical instillation to prevent UTIs.55–63 A thorough understanding of antibacterial-resistant mechanisms and the immune response are essential to further investigate and incorporate nonantibiotic measures.
Conclusion
Antibiotic resistance is a serious global health issue because of its impact on morbidity and mortality of patients, as well as on healthcare costs. The growing consumption of antibiotics has fostered the emergence of multidrug resistant organisms, and encounters with resistant microorganisms are increasingly frequent. Due to the rise of antibiotic resistance and the declining number of newly discovered antibiotics, treatment options are becoming increasingly limited, particularly for vulnerable and severely ill patient groups. Contrary to previous assumptions, studies have shown that chronic use of antibiotics (e.g. in prophylaxis) increases the risk of sudden cardiovascular death. Further exploration of alternative, nonantibiotic substances to prevent infections, and the implementation of antibiotic stewardship at an ambulatory and hospital-wide level, may offer strategies to combat the further emergence of resistant organisms.
Footnotes
Acknowledgements
Writing assistance was provided by Mariella Franker, PhD, Franker Medical Communications, Netherlands, and Ewen Legg, PhD, Halcyon Medical Writing Ltd., UK.
Funding
The symposium was organized and funded by Vifor Pharma Group, Switzerland. Writing assistance was funded by OM-Pharma, a company of Vifor Pharma Group. This supplement was supported by an educational grant from OM-Pharma.
Conflict of interest statement
Dr Quirós has worked as a lecturer and consultant for 3M, MSD, Phoenix-GSK, and Vifor Pharma Group. Dr López Romo is a lecturer for Pfizer and Vifor Pharma Group
