Abstract
Over the past few years, the role of robotic-assisted partial nephrectomy (RPN) has exponentially grown. Multiple recognized factors contribute to postoperative renal function in patients undergoing RPN. The aim of this review is to identify these potential factors, and to evaluate strategies that may help optimize the goal of renal function preservation. A nonsystematic literature review was performed to retrieve the most recent evidence on factors contributing to renal function post-RPN. Analyzed elements include baseline factors (tumor complexity and patient characteristics), intraoperative (surgical) factors (control of the renal hilum and type of ischemia, resection technique, renorrhaphy technique), and pharmacotherapeutics. In conclusion, the advantages of robotic surgery in the setting of partial nephrectomy (PN) are becoming well established. Maximal preservation of renal function remains a priority goal of the procedure, and it is influenced by a plethora of factors. Adequate patient selection using radiomics, control of comorbidities, utilization of evidence-based intraoperative techniques/strategies, and postoperative care are key components of postoperative preservation of renal function. Further investigations regarding these factors and their effects on long-term renal function are necessary and will continue to aid in guiding appropriate patient care.
Introduction
Partial nephrectomy (PN) is the current gold standard surgical treatment for clinical T1 renal tumors, whenever technically feasible. Although more challenging than radical nephrectomy, it offers the advantage of nephron preservation and subsequent protection against long-term kidney disease and need for dialysis, without significant risk of compromising oncologic outcomes.1–3 The introduction of robotic-assisted partial nephrectomy (RPN) has offered improved perioperative outcomes compared with both open 4 and laparoscopic surgery. 5 Furthermore, robotic surgery has offered a shorter learning curve compared with the standard laparoscopic approach, which translated into a rapid implementation of this minimally invasive surgical approach.6,7 Over the past few years, indications for RPN have expanded to also include larger and more complex renal masses. 8 It has been estimated that decline in function after PN averages approximately 20% in the operated kidney. Multiple recognized factors are involved in determining ‘global renal damage’ and postoperative renal function. 9 These factors can be categorized as ‘host’ and ‘surgical’ factors.10,11
The aim of this review is to identify these potential factors, and to evaluate strategies that may help optimize the goal of renal function preservation in patients undergoing RPN.
Literature search
A nonsystematic literature review was performed by using PubMed and Scopus to retrieve publications related to RPN with a focus on factors related to renal function preservation up to July 2018. In the free-text protocol, different combinations of the following search terms were applied: functional outcomes; nephron-sparing surgery; renal function; robotic partial nephrectomy. An evidence-based critical analysis was conducted by focusing on the most recent literature.
Preoperative factors
Tumor complexity
As with any surgical procedure, patient selection is a critical component in the decision to proceed with RPN. Anatomic factors such as hilar location, vessel involvement, and large size, subsequently resulting in potentially significant parenchymal volume loss, may preclude an adequate nephron-sparing approach. In addition to the increased technical difficulty associated with such lesions, larger tumors and those near the hilum are associated with longer ischemia times and greater blood loss. 12 Therefore, careful consideration must be given to tumor location through judicious evaluation of preoperative imaging.
Several anatomic classification systems assessing tumor complexity have emerged and have been evaluated in the context of PN outcomes, thereby highlighting a growing field of ‘radiomics’. One such system, the RENAL nephrometry score, examines the size and location of the renal tumors by assigning a complexity score, with higher score values being assigned to tumors that are larger in size, endophytic, closer to the collecting system, and cross polar lines. 13 Another complexity rating system, the PADUA score, attempts to predict the feasibility of PN using measures other than size. This scoring system accounts for polar location, medial versus lateral renal rim, endophytic versus exophytic location, renal sinus and urinary-collecting-system involvement, with higher scores assigned to middle pole location, endophytic tumors, medial renal rim and tumors with sinus and collecting-system involvement. 14
There have been several attempts to improve upon these classification systems. In further assessing endophytic versus exophytic tumors, Leslie and colleagues developed a method to quantify the so called ‘contact surface area’ of a renal tumor with adjacent parenchyma. In addition to offering a new method for preoperative patient stratification, this study also demonstrated that increasingly endophytic tumors had increased loss of renal parenchyma. 15 Another metric, the Mayo Adhesive Probability Score examined adherent perinephric fat, specifically, perinephric fat thickness and stranding, to predict the difficulty of PN. 16
Despite the perioperative utility of these scoring and classifications systems, there is considerable disagreement regarding their ability to predict renal functional outcomes, 17 and a certain degree of interobserver variability. 18 Furthermore, they generally suggest that increasingly complex tumors are associated with longer ischemia times and increased volume that must be surgically resected in order to remove the tumor with clear margins.19,20 On the other hand, characteristics of the renal mass (including nephrometry score and size) were not found to have any clinically relevant impact on baseline renal function, whereas patient age and comorbidities were confirmed to represent unmodifiable significant factors. 21
Patient characteristics
As with any surgical patient, chronic diseases may contribute to poorer perioperative outcomes. The introduction of the Enhanced Recovery after Surgery protocols highlights the importance of preoperative conditioning and optimization as part of improving postoperative outcomes. 22 Preoperative blood pressure control has been shown to affect postoperative glomerular filtration rate (GFR). Rajan and colleagues examined 1955 patients undergoing PN and found an increased risk for acute kidney injury (AKI) in patients with uncontrolled preoperative hypertension. Interestingly, the number of antihypertensives was not associated with increased risk of postoperative AKI. 23
Chronic kidney disease is present in approximately 25–30% of patients undergoing PN. 24 Preoperative estimated GFR (eGFR) has been shown as a determinant of postoperative eGFR, as patients with higher baseline eGFR seem to have less decreases in eGFR postoperatively. Furthermore, eGFR > 45 ml/min has been found to be advantageous in avoiding dialysis postoperatively, and to improve the likelihood of ⩽50% decrease in postoperative eGFR. 25 These findings suggest that medical optimization of kidney function prior to PN could lead to improved postoperative GFRs. Conversely, Mir and colleagues found that the poorly functioning kidney has the same ability to recover from ischemia as the functioning kidney, proportional to the remaining kidney parenchyma. 26 While postoperative renal function may primarily be a consequence of residual tissue volume, preoperative efforts for optimization may still yield some potential benefit.
Intra-operative factors
Management of renal hilum
Clamping of the main renal artery remains the gold standard technique for obtaining a temporary warm ischemia, allowing the resection of the renal mass in a bloodless field. 27 However, various alternative clamping methods have been explored in the effort of minimizing the ischemic damage to the renal parenchyma, including early unclamping, off-clamp resection, and segmental clamping. 12
An early-unclamping technique prevents the kidney parenchyma from prolonged ischemia by removing the arterial clamp before the resection has been completed. Although there is a theoretical advantage in renal function, George and colleagues did not find any significant difference in 6-month postoperative GFR. 28 On the other hand, early unclamping carries concerns regarding margin status and increased blood loss; however, Peyronnet and colleagues did not find an increase in the rate of transfusion or complication. 29
Kaczmarek and colleagues compared off clamping versus main-artery clamping in a multicenter analysis of tumors matched for complexity and found off-clamp RPN to be associated with higher blood loss, shorter operative time, and lower decrease in renal function. 30 In another recent multicenter matched-paired analysis, Rosen and colleagues found that the use of off-clamp RPN marginally increased blood loss without providing a renal function benefit. 31 One potential variant of this method includes ‘on-demand’ ischemia, which involves initiation of tumor resection off clamp, and any subsequent clamping is only performed when excision cannot be continued due to obscurative bleeding. 32
Selective arterial clamping is composed of multiple techniques used to achieve regional ischemia rather than ‘global’ kidney ischemia. These methods have included clamping of polar arteries, tumor-feeding arteries, anterior or posterior division arteries, or tertiary arteries (using a super-selective microdissection). 33 Near-infrared technology has been employed in attempts to increase the accuracy of selective arterial clamping and in tumor identification.34,35 These selective clamping techniques aim to create a ‘zero ischemia’ effect on the kidney parenchyma that remains after resection. Li and colleagues observed shorter ischemia time and reduced decrease in immediate postoperative GFR with segmental clamping than with main-artery clamping. 36
In a very comprehensive and contemporary meta-analysis evaluating surgical factors on RPN outcomes, Cacciamani and colleagues found reduced percentage decrease of latest eGFR in the off-clamp group versus the on-clamp group, but subgroup analysis showed no difference between selective/super-selective clamp and main-artery groups in regard to transfusion rates, length of stay, positive margins, complication rates, and percentage change of latest eGFR. However, further subgroup analysis comparing super-selective and main-artery clamping did reveal superior renal function in the former group. 11
Ischemia
Ischemia time has been historically considered a significant determinant in postoperative eGFR. 37 A warm ischemia time (WIT) of less than 25–30 min is the widely recommended standard at which any acute kidney injury is considered reversible. Porpiglia and colleagues found that renal function on scintigraphy significantly decreased with WIT > 30 min despite a lack of difference in GFR. 38 Additionally, Lane and colleagues observed that WIT of <20 min was associated with a median GFR decrease of 11%, which significantly increased to 15% with WIT 21–30 min at mean follow up of 1.5 years. 39 Furthermore, multiple studies have demonstrated worsening functional outcomes associated with WIT > 25 min and 10-min longer ischemia time.40–42
Cold ischemia was developed in the setting of open PN and used to allow prolonging the ischemia time without altering the postoperative GFR. Funahasi and colleagues found that cold ischemia permitted longer ischemia times of nearly 1 h, with preservation of renal function on MAG3 scan. 41 Cold ischemia has been shown as technically feasible and safe in RPN, 43 and possibly associated with better functional preservation in the short-term follow up, without a clear advantage in the long term. 44 The main indication for the use of ice slush during RPN would be in those cases where, given the complexity of the mass, a longer (above 30 min) ischemia time is expected. Despite the importance of keeping the ischemia time below 25–30 min to prevent excessive GFR loss, more recently, it has been advocated that a more important factor might be the quality and quantity of the remaining kidney.9,39,40
Resection technique
Standard PN techniques include wedge resection, polar resection, and heminephrectomy. This technique typically involves removal of the tumor with a traditional, yet arbitrary, margin of up to 1 cm; however, acceptable margin rates have been reported with 1–5 mm. 45 Multiple studies have recently examined the technique of robotic renal tumor ‘enucleation’, which consists of removing only the tumor and no surrounding kidney parenchyma by bluntly dissecting the tumor along the plane that arises from the ‘pseudocapsule’. 46 This technique does not often require renorrhaphy and can lead to decreased WIT and decreased devitalized parenchyma from renorrhaphy. 47 Studies suggested that tumor enucleation has potential for maximum parenchymal preservation optimized functional recovery. 48 A recent meta-analysis showed tumor enucleation is non-inferior to standard PN regarding positive margin rate, and tumor recurrence rates. 49 Minervini and colleagues compared oncologic outcomes for patients undergoing enucleation versus traditional PN and found similar progression-free survival rates. 50
Other techniques are halfway between a standard resection and a tumor enucleation, allowing the removal of the tumor with a ‘minimal margin’ (typically 1 mm) of normal kidney parenchyma. 51 This can be used for tumors that lack a pseudocapsule to allow safe oncologic resection while still minimizing normal renal parenchyma that is removed.
Regardless of the technique, surgical planning can be facilitated by recent advances in three-dimensional (3D) imaging and reconstruction (Figure 1). Moreover, integration of augmented reality images in the robotic platform could further aid the robotic surgeon in tumor identification and resection (Figure 2). 52

3D reconstruction from CT imaging showing a 3 cm partially exophytic lower pole left renal mass.

Use of augmented reality during a robotic partial nephrectomy procedure for a totally endophytic upper pole left renal mass.
Renorrhaphy
Devitalization of renal parenchyma may occur following renorrhaphy due to tissue damage caused by the repair itself. Therefore, reduction in the volume of devitalized renal parenchyma represents an avenue to improve the quality of remaining kidney parenchyma. Careful consideration should be given to the technique employed. In a retrospective review of 56 patients undergoing PN, Bahler and colleagues found significantly greater volume loss (on radiographic analysis) and greater glomerular filtration loss among those undergoing two-layer closure of the renal base and cortex compared with those undergoing base-layer-only closure. 47 Additionally, the two-layer closure was found to be an independent predictor of volume loss on propensity-adjusted multivariable regression analysis. Interestingly, they did not find an increased rate of urine leak or bleeding between the two approaches. Porpiglia et al. developed a sliding loop technique for a single-layer renorrhaphy which was compared with a double-layer technique and revealed a greater reduction in GFR in the ipsilateral kidney managed with the double-layer repair. 53
Renorrhaphy techniques using tissue adhesive sealant have also been described and have demonstrated favorable reductions in warm ischemia time and blood loss with use of bovine serum albumin-glutaraldehyde (BioGlue®, CryoLife, Inc, Kennesaw, GA, USA). 54 A subsequent study assessing renal function via DMSA scan among patients undergoing PN revealed decreased volume loss by approximately 50% with use of the tissue adhesive compared with standard suture renorrhaphy. 55 Takagi and colleagues compared monopolar coagulation of hemorrhagic areas and a tissue-sealing sheet (TachoSil®, Baxter Healthcare, Deerfield, IL, USA) versus standard renorrhaphy. Examination of postoperative volumetric imaging studies failed to show any differences in volume loss or renal function between the two populations. 56 The use of bioadhesives and sealants represents viable alternatives for renorrhaphy that may offer potential decreases in volume loss and consequent improvements in renal function but without significantly increased risk of complications.
Pharmacotherapeutics
The use of medical therapies in modulation of postoperative renal function has been considerably investigated. Mannitol, an osmotic diuretic traditionally considered a renal-protective agent, is commonly used during partial nephrectomies, particularly during clamping. 57 However, multiple observational studies have failed to demonstrate any improvement in renal function with mannitol administration.58–60 Recently, Spaliviero and colleagues performed the first and only double-blind randomized trial investigating the use of this agent among a cohort of nearly 200 patients undergoing partial nephrectomy with normal preoperative renal function. They found that the use of mannitol did not demonstrate a significant change in renal function at 6-month follow up. Although this study suggests that the use of mannitol should be discontinued in the setting of normal preoperative renal function, it failed to address its utility or lack thereof in patients with renal dysfunction at baseline. 61
The renal-protective properties of phosphodiesterase type 5 inhibitors observed in animal models led to a randomized, double-blind, placebo-controlled trial examining the use of sildenafil in human subjects. Krane and colleagues administered 100 mg of sildenafil or placebo immediately preoperatively among 30 patients undergoing RPN. Although not the primary outcome, there was no significant difference in postoperative GFR between treatment groups. 62 Further evaluation of pharmacotherapeutic agents is warranted.
Postoperative factors
Renal function may already reach its permanent new setpoint or plateau by the immediate postoperative period. Antonelli and colleagues found an initial immediate median reduction of 11.4% (from preoperative baseline) in renal function in consecutive patients undergoing PN. This renal function appeared to remain stable as evidenced by the similar median reduction of 9.6% in eGFR at subsequent 24-month follow up. 63 Therefore perioperative minimization of the expectant decline in renal function through proper perioperative optimization is vital. Despite the suggested stabilization of long-term decreased renal function immediately following RPN, sufficient postoperative management remains necessary. Worsening or persistent acute kidney function has been shown as a risk factor for chronic kidney disease. 64 Consequently, strategies such as appropriate volume resuscitation, limitation of nephrotoxic agents, and adequate drainage of the renal unit may help decrease further AKI and consequent deterioration of kidney function.
Conclusion
The advantages of robotic surgery in the setting of PN are becoming well established. Maximal preservation of renal function remains a priority goal of the procedure, and it is influenced by several factors (Figure 3). Adequate patient selection using radiomics, control of comorbidities, utilization of tailored intraoperative techniques/strategies, and appropriate perioperative care are key components in determining postoperative preservation of renal function. Further investigations regarding these factors and their effects on long-term renal function are necessary and will continue to aid in guiding appropriate patient care.
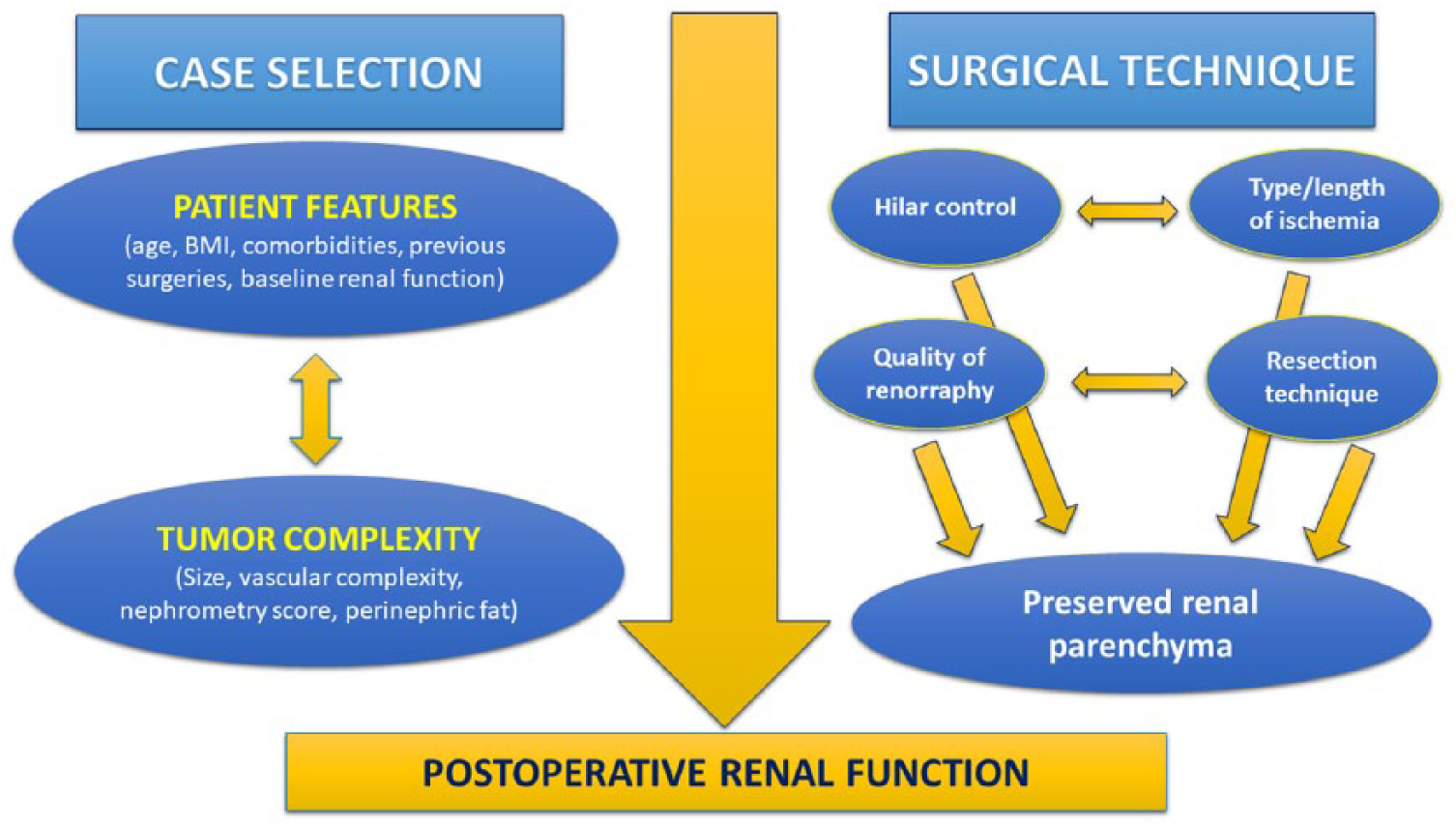
Factors playing a role in determining postoperative renal function in patients undergoing a partial nephrectomy procedure.
Footnotes
Acknowledgements
Natalie Swavely and Uzoma Anele contributed equally to this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
