Abstract
Background:
The aim of this study was to compare the pharmacokinetics (PK) and pharmacodynamics (PD) of two distinct formulations of leuprolide acetate (LA); subcutaneous (SC) injection and intramuscular (IM) injection.
Methods:
A total of 32 healthy men were randomized to receive a single 7.5 mg injection of SC-LA (n = 16) or IM-LA (n = 16) in this phase I, open-label, parallel-group study. PK was assessed via LA concentrations, and PD via serum luteinizing hormone (LH) and testosterone (T) concentrations.
Results:
The initial surge of LA was higher for IM-LA than SC-LA (Cmax 27 ± 4.9 versus 19 ± 8.0 ng/ml, respectively), with a shorter tmax (1.0 ± 0.4 versus 2.1 ± 0.8 h). The duration of quantifiable LA concentration was longer for SC-LA (up to 56 versus 42 days for SC-LA and IM-LA, respectively). Median LH concentrations in both groups rapidly increased, followed by gradual decrease. However, SC-LA demonstrated a longer duration of LH suppression, with median levels remaining below 1.0 IU/l through Day 56 compared with IM-LA where LH started to rise by Day 35. Consequently, serum T began to increase by Day 42 in the IM-LA group, with only four subjects maintaining levels ⩽50 ng/dl, compared with 14 subjects in the SC-LA group. By Day 56, 13 SC-LA subjects maintained serum T levels ⩽50 ng/dl. Both SC-LA and IM-LA were well tolerated.
Conclusions:
Both formulations demonstrated consistent delivery of drug over 1 month; however, SC-LA provided a longer duration of action than expected based on the dosing interval. This profile suggests that SC-LA will provide effective suppression of T over a longer period of time, permitting greater injection scheduling flexibility.
Introduction
Approximately one in seven men in the United States (US) will receive a lifetime diagnosis of prostate cancer and one in 39 will die from it. 1 Androgen deprivation therapy (ADT) decreases serum testosterone (T), a promoter of prostate cancer growth, and increases overall survival and palliative alleviation of symptoms in patients with advanced prostate cancer (PCa). 2
Historically, ADT was achieved with bilateral surgical orchiectomy. However, chronic exposure to synthetic peptides that mimic natural luteinizing hormone-releasing hormone (LH-RH) also decrease T production via a negative feedback loop. 3 T suppression equivalent to orchiectomy is the objective 4 and although a T level of <50 ng/dl has historically been the target for suppression, this was based on sensitivity of available assays at the time. 5 Assay advancements now permit detection of T as low as 0.1 ng/ml 6 and evidence suggests that lower T levels may lead to clinical benefits. Small retrospective studies have shown reducing T to <20 ng/dl may improve survival and increase time to progression.7–9 A large, prospective study demonstrated that nadir T of <20 ng/dl improves cancer-specific survival and duration of response. 10 Since 2014, the European Association of Urology has defined castration level as T <20 ng/dl in their updated PCa guidelines. 11
Early treatments using LH-RH agonists required daily injections, 12 which were inconvenient for both patients and health care providers. 13 However, multiple long-acting formulations are now available and aim to deliver stable T suppression to castrate levels over a prescribed dosing interval, utilizing different modes of delivery and absorption technologies. Although important distinctions exist between these formulations, misperceptions persist that all LH-RH therapies are equivalent, and thus interchangeable, but they are likely not.
Long-acting drugs require fewer injections per year, and improved pharmacokinetics (PK) with prolonged duration of drug release may allow for increased scheduling flexibility. For these reasons, long-acting formulations have been associated with greater patient treatment adherence, increased quality of life, and may have improved clinical outcomes, as has been demonstrated for other classes of drugs. 14
One of the synthetic LH-RH agonists is leuprolide acetate (leuprorelin acetate, LA) which has been in use for over 30 years, primarily in advanced PCa. However, its half-life is only 3 h, hence the development of long-acting formulations such as LA for intramuscular (IM) injection (LUPRON®) and LA for subcutaneous (SC) injection (ELIGARD®).15–19 These are two distinct formulations of LA, utilizing separate technologies to lengthen durations of drug delivery, and are administered via different methods. IM-LA and SC-LA are currently the most widely used LH-RH agonists on the market and although the products share the same active ingredient, their PK profiles and subsequent PD profiles are unique to their formulation technology and delivery method.
IM-LA is composed of LA encapsulated in polylactic-co-glycolic acid microspheres that may be reconstituted up to 2 h prior to injection. 20 After injection, the microspheres slowly degrade in situ, releasing drug in a controlled manner. 21
SC-LA is composed of LA suspended in ATRIGEL® Delivery System. ATRIGEL® is comprised of biodegradable polymers dissolved in a biocompatible carrier that provides consistent delivery of drug over long periods. 22 Administration must occur within 30 minutes of reconstitution. After injection, the solution solidifies into a solid in-situ implant that biodegrades over time, releasing the drug in a controlled manner.
The authors are not aware of any published head-to-head studies comparing these two currently available formulations, hence there is value in determining whether any differences exist that may impact clinical decision-making. Below we report results of the first comparison of PK/PD characteristics of 7.5 mg SC-LA versus 7.5 mg IM-LA. The primary objective of this study was to contrast the PK of SC-LA with that of IM-LA; PD comparisons were exploratory.
Methodology
Study design
This was an open-label, randomized, single-dose, analytically-blinded, parallel-group study in healthy men, conducted in accordance with Good Clinical Practice, Declaration of Helsinki (with amendments), and all local legal and regulatory requirements. Written informed consent was obtained from all subjects before screening.
Subjects
A total of 32 subjects were planned, with 16 per treatment group. No formal sample size estimation was conducted. Healthy men 18–55 years and within ±15% of ideal weight were eligible. Enrolled subjects had normal clinical laboratory test parameters (biochemistry, hematology, and urinalysis), no clinically significant findings by physical examination, and adequate venous access. Subjects with hypersensitivity to any gonadotropin-releasing hormone, clinically relevant illness, heavy smokers or drinkers, history of drug or alcohol abuse, recent immunization, or recent treatment with anabolic steroids were ineligible.
Study drugs, procedures and analytical methods
Eligible subjects were randomly assigned to receive single 7.5 mg injections of either SC-LA (ELIGARD®, Atrix Laboratories, Fort Collins, CO) or IM-LA (LUCRIN Depot, Abbott Australasia). Randomization was conducted using RANCODE version 3.6 (idv Datenalyse und Versuchsplanung, Gauting/Munich, FRG, 1997). Blood samples to assess serum leuprolide, luteinizing hormone (LH), and T concentrations were taken before drug administration and at 0.25, 0.5, 1, 2, 3, 4, 6, 8, 10, 24, 36, and 48 h after administration and on days 3, 4, 7, 14, 21, 28, 35, 42, 49, and 56.
Serum leuprolide concentrations were analyzed using high performance liquid chromatography (HPLC) with mass spectrometry. LH concentrations were measured using two-site immunochemiluminometric assay. T levels were measured using radioimmunoassay.
Both study drugs were comprised of the active ingredient leuprolide acetate; SC-LA is formulated with 50% w/w 85/15 poly(DL-lactide-co-glycolide) (PLG) and IM-LA is formulated with 50% w/w N-methyl-2-pyrrolidone (NMP) (SC-LA) or polylactic-co-glycolic acid microspheres.
Data analysis
PK parameters were determined from individual leuprolide concentration–time data by noncompartmental analysis: observed maximum concentration (Cmax); time of observed maximum concentration (tmax); apparent terminal half-life (t1/2); area under the leuprolide serum concentration–time curve (AUC) from time zero to time of last quantifiable concentration (AUClast); AUC time zero to 168 hours (AUC0–168h); AUC last extrapolated to infinity (AUCinf).
None of the PK variables were specified as primary. Individual concentration–time data were used to determine Cmax and tmax. Terminal elimination rate constant (lambda z) was estimated by linear least squares regression with logarithmic concentration data of the terminal part of the concentration–time curve using at least three data points. The AUClast was calculated by the linear trapezoidal rule. Extrapolation from AUClast to AUCinf was made by dividing the last quantifiable concentration by lambda z and adding this to AUClast.
The AUCs and Cmax were analyzed with analyses of variance (factor:formulation, log-transformed data). Consistent with two-sided test procedures, 90% confidence intervals for the difference between drug product least-square means were calculated for AUClast, AUCinf, and Cmax using log-transformed data. Statistical Analysis Software version 8.2 (SAS Institute, Cary, NC, USA) was used for statistical analyses. PK parameters were determined using TopFit version 2.1 (VCH Publishers, Inc.). 23
The tmax data and PD of LH and T were evaluated descriptively using non-transformed data. Safety was assessed via adverse event (AEs) reports (coded with COSTART), clinical laboratory evaluations, and safety examinations.
Results
Subject characteristics
A total of 32 white males aged 21–52 years were enrolled in the study; 16 received SC-LA and 16 received IM-LA, and 31 subjects completed the study (Supplementary Figure 1). One subject withdrew from the IM-LA group at week 3, for reasons unrelated to study drug and his data were included in all analyses. Demographics and baseline characteristics were balanced between groups with no statistically significant differences observed (Table 1).
Subject demographic and baseline characteristics.

Data are mean ± standard deviation unless otherwise indicated.
IM, intramuscular injection; LA, leuprolide acetate; SC, subcutaneous injection.
PK/PD
During the first 36 h after dosing, mean leuprolide concentrations showed rapid increases followed by continuous decreases in both groups (Figure 1). Further PK parameters of LA are provided in Table 2. The mean Cmax of SC-LA was 30% lower compared with IM-LA (ratio of least-square mean = 0.7). The tmax of SC-LA was approximately twice that of IM-LA and the AUClast was 90% greater than IM-LA. Subjects given SC-LA had AUC levels 1.9-times higher than IM-LA subjects. The differences in Cmax, tmax and AUC between SC-LA and IM-LA groups were found to be statistically significant with p-values of <0.002, <0.0001, <0.0001, respectively (Table 2).
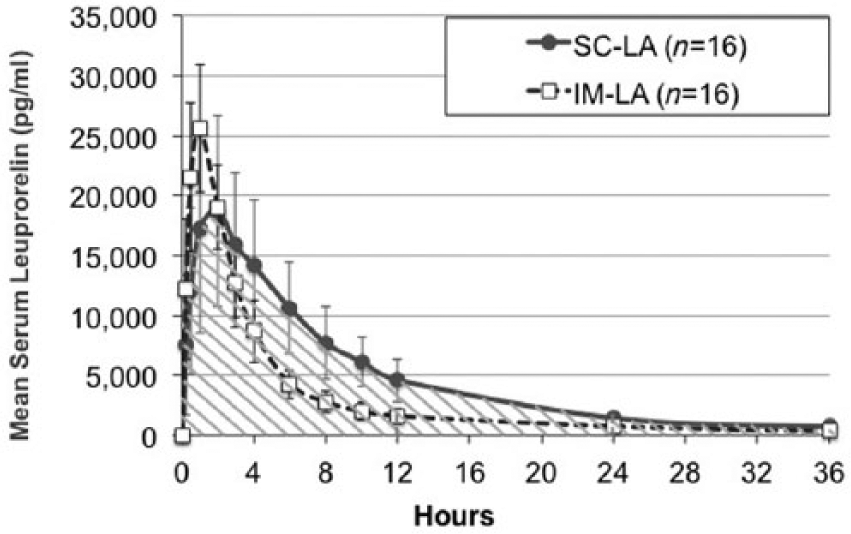
Mean leuprolide serum concentration–time curves during the first 36 h after drug administration (linear scale).
Leuprolide pharmacokinetic parameters.
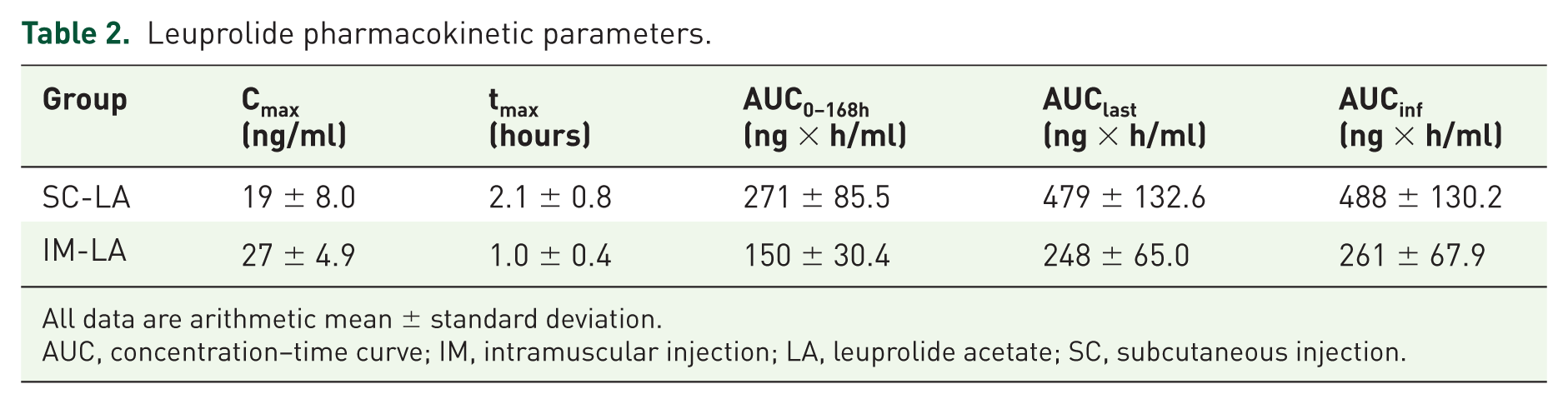
All data are arithmetic mean ± standard deviation.
AUC, concentration–time curve; IM, intramuscular injection; LA, leuprolide acetate; SC, subcutaneous injection.
The duration of serum leuprolide detection above the level of quantitation (LOQ) ranged from 14 to 35 days in IM-LA subjects compared with 42–56 days in SC-LA subjects [Figure 2(a)]. Concentrations of leuprolide below LOQ are not expected to be active.17–19 On Day 14, all IM-LA subjects had detectable levels of serum LA; by Day 21, levels were unquantifiable in one subject. By Day 28, 5/16 (31%) IM-LA subjects did not have detectable LA, by Day 35 this increased to 15/16, and by Day 42, all IM-LA subjects had undetectable levels. In contrast, all SC-LA subjects maintained quantifiable LA levels through Day 42. By Day 49, serum LA fell below quantitation in 4/16 SC-LA subjects and by Day 56 two subjects remained with detectable serum LA.

(a) Mean leuprolide serum concentration–time curves (pg/ml) over the course of 56 days after drug administration (semi-logarithmic scale, y-axis truncated). The limit of quantitation was 50.1 pg/ml. (b) Median LH serum concentration–time curves over the course of 56 days after drug administration (linear scale). (c) Median testosterone serum concentration–time curves over the course of 56 days after drug administration (linear scale).
Median LH serum concentrations were similar between treatment groups up to Day 35, after which LH increased in the IM-LA group to near baseline levels [Figure 2(b)]. At Day 35, 2/15 (1 subject did not record measurements past Day 14) IM-LA and 1/16 SC-LA subjects had LH serum concentrations above 1.0 IU/l. At Day 42, LH serum concentrations rose above 1.0 IU/l, and to near baseline levels in 13 IM-LA subjects and 1 SC-LA subject. At Day 49, all evaluated IM-LA subjects had LH concentrations >1.0 IU/l compared with no SC-LA subjects. In fact, median LH concentrations remained below 1.0 IU/l through Day 56 in the SC-LA group [Figure 2(b)].
As expected, both groups showed transient increases in median serum T following injection, followed by a drop to similar nadirs [Figure 2(c)]. After Day 35, median T concentrations began to rise in the IM-LA group, but remained low past Day 49 in the SC-LA group. At Day 21, 8/15 and 5/16 subjects had T levels ⩽50 ng/dl in the IM-LA and SC-LA groups, respectively. At Day 28, 14 IM-LA subjects and 12 SC-LA subjects had T⩽50 ng/dl and by Day 35, 14 subjects in both groups had T ⩽50 ng/dl. By Day 42, serum T had begun to rise in the IM-LA group, with only 4 subjects remaining ⩽50 ng/dl, compared with 14 SC-LA subjects. By Day 49, all IM-LA-treated subjects had T levels >50 ng/dl, whereas 14 in the SC-LA group maintained castrate serum T levels. At Day 56, 13 SC-LA subjects continued to demonstrate T levels ⩽50 ng/dl compared with none in the IM-LA treated group (Table 3). Of interest, T levels in 14/15 IM-LA subjects were higher at study completion than at baseline, with a median T level for IM-LA subjects of 768 ng/dl at the end of study, compared with their baseline of 562 ng/dl.
Number of subjects with serum testosterone level below 50 ng/dl.

n = 15 for this time point in both SC-LA and IM-LA.
IM, intramuscular injection; LA, leuprolide acetate; SC, subcutaneous injection.
Safety
A total of 86 AEs were reported for SC-LA subjects and 37 AEs for IM-LA subjects. No serious AEs or AE-related subject withdrawal occurred. Most AEs were classified as possibly or probably related to the study medication (81/86 in the SC-LA group and 27/37 in the IM-LA group).
The most frequently reported AEs were injection-site disorders and known effects of T suppression such as hot flashes (coded as vasodilation), increased sweating, and decreased libido (Supplementary Table 1). Hot flashes or increased sweating were reported by 13 subjects in the SC-LA group and 11 subjects in the IM-LA group. Decreased libido was reported by 9 subjects in the SC-LA group and 11 subjects in the IM-LA group.
Injection-site disorders were more common among SC-LA subjects than IM-LA subjects. Overall, 15 subjects reported injection-site pain, all classified as mild (5/15) or moderate (10/15). A total of 14 subjects reported injection-site erythema, all of which were mild (10/14) or moderate (4/14). Overall, two SC-LA subjects reported an injection-site mass: one mild and one moderate in severity. Injection-site exanthema, itching, hemorrhage, and hypersensitivity were each reported by one subject; all cases were mild. A total of two IM-LA subjects reported three cases of injection-site events. Among all events, most cases of injection-site pain persisted for a few minutes after the injection, erythema for a few hours. Neither the subjects nor the investigator deemed any of the injection-site disorders as beyond acceptability for the injection.
Clinical laboratory data revealed that 12 subjects had increased potassium values at final examination, compared with 1 at screening. A total of 13 subjects had elevated alanine aminotransferase levels at final examination, compared with 5 at screening, and 8 had decreased erythrocyte counts at final examination, compared with 2 at screening. Only two individual deviations were considered clinically relevant: increased blood glucose at final examination in one and decreased T in another (at follow up, T level had returned to normal). The number of subjects with abnormal lab values was similar between the two treatment groups. There were no clinically relevant abnormalities in body temperature, blood pressure, pulse rate, respiratory rate, electrocardiogram, or muscle strength test.
Discussion
This is the first published, head-to-head study comparing PK/PD of SC-LA with IM-LA. SC-LA had significantly different PK/PD profiles when compared with IM-LA, with a duration of action that was longer than 1 month. Specifically, leuprolide AUC was higher and Cmax lower with SC-LA. The duration of leuprolide levels remaining above LOQ (i.e. duration of clinically relevant systemic exposure) for SC-LA was 10–20 days longer than for IM-LA and at 28 days after IM-LA administration, 31% of subjects had undetectable levels of serum LA. These PK differences between the two extended-release formulations are likely responsible for resultant PD profiles. While the magnitudes of initial T suppression were similar, SC-LA suppressed LH and T for longer.
These results align with previous trial data showing that both formulations produce castrate levels of T within 4-weeks.19,24–30 However, differences in PK/PD profiles seen in this head-to-head comparison may have product-specific clinical implications. For example, continuous levels of quantifiable leuprolide may decrease the likelihood of T escapes above castrate levels. It has been reported that T escapes may be hazardous to patients and should be avoided. 31 If monitoring T levels on one formulation shows a rise in T at the end of the dosing period, it may be beneficial to try another formulation to achieve appropriate T suppression levels, before switching to more advanced or expensive PCa therapies.
The difference may also have practical implications for healthcare providers and direct patient care. It is relevant to note that regulatory agencies approved the monthly formulation of LA based on 28-day dosing efficacy data. At least some of the Centers for Medicare and Medicaid Services regional providers and other insurers mandate a 30 day or longer period between doses for reimbursement purposes. SC-LA thus provides more dosing flexibility and confidence of T suppression. There could be less concern of T escape if the patient was unable to receive their next injection on the exact day payers permit the next dose, which, for a 1-month formulation, must be no less than 1-month from the previous dose.
Both SC-LA and IM-LA were well tolerated in this study; however, there were more reported mild/moderate AEs in the SC-LA group compared with IM-LA, most often injection-site reactions. No individuals discontinued due to an AE and no severe AEs occurred. All subjects experienced at least one AE, most of which were transient and injection-site related. There were no trends in abnormalities of laboratory results. Despite the more frequent occurrence of injection-site reactions with SC-LA in this phase I study, it is generally accepted that SC injection may be preferable to IM due to a number of factors such as shorter needles, increased scope for injection sites and less likelihood of piercing a blood vessel or damaging a nerve. 32 Furthermore, a study comparing the same formulation of triptorelin delivered via SC versus IM found that SC was equally effective as a mode of administration as IM, with a favorable safety profile. 33
There were several limitations to this study. Healthy subjects were enrolled and T suppression may be more difficult than in PCa patients. However, healthy volunteers have stable baselines, maximal capacity for PD response (hormone-producing cells unimpaired by previous chemotherapy, radiation, advanced age etc.), and they have normal subcutaneous and muscle tissue. Furthermore, subject variability is minimal. The sample size was small, although the results confirm the adequacy of the number of subjects for most objectives. Tolerability of injection-site reactions can vary widely between subjects and therefore, a larger subject number could have reduced between-subject variability. However, these limitations are typical for a phase I study.
Conclusion
The results of this comparison of single doses of 7.5 mg monthly formulations of LA demonstrated different PK/PD profiles, indicating a longer duration of release of LA and suppression of T for SC-LA, despite equal amounts of active drug being injected. This confirms that not all LA formulations can be considered equivalent and interchangeable. This may allow for easier injection scheduling and less concern about T escapes in the event that a patient cannot receive their injection within 1 month from their prior dose.
Footnotes
Acknowledgements
The authors thank Jocelyn Hybiske, PhD, a consultant at Xelay Acumen, for editorial support.
All authors had access to the full data and approved the paper in its current form. SdM was principal investigator of the trial, and led the study and statistical analysis for the paper. DS, NDS, JM, FC, RC, JAM, SA, AY, and EDC contributed to the overall concept of the analyses for the paper, and participated in writing and editing of the manuscript
Funding
The study was sponsored and funded by Mayne Pharma Pty Ltd., Australia. The design, collection and analysis of data, and writing of the report were funded by Mayne Pharma Pty Ltd. Article preparation was funded by TOLMAR, Inc.
Conflict of interest statement
JWM has been an advisor for TOLMAR Pharmaceuticals, Inc. SA and AY are employees of Tolmar Pharmaceuticals Inc. NDS has served in a consulting or advisory role for AbbVie, Astellas, Bayer, Dendreon, Ferring, Janssen, Medivation, Sanofi, and Tolmar; FC has served in a consulting or advisory role for Astellas, Bayer, Lilly, Ferring, Dendreon, Janssen, Medivation, Sanofi, and GSK; RC has served in a consulting or advisory role for Integra Connect, CUSP, Astra Zeneca, and Genomic Health, as well as participated in speaker’s bureaus for Dendreon, Astellas/Pfizer, Janssen, and Amgen; DS has served in a consulting or advisory role for Tolmar; JAM is an employee of Tolmar, Inc.; EDC has served in a consulting or advisory role for Bayer, MDx, Genomic Health, Janssen, Dendreon, Ferring, and Tolmar. SdM is an employee of SynteractHCR.
Supplementary Material
Supplementary Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
