Abstract
Background:
There are two slow-release ready-to-use forms of leuprorelin acetate (1-month and 3-month) that are available as solid, biodegradable implants for the treatment of advanced, hormone-sensitive prostate cancer. These implants have been shown to be as effective as traditional leuprorelin acetate microspheres for achieving successful testosterone suppression (⩽0.5 ng/ml) and lowering prostate-specific antigen (PSA) levels. Here we further evaluate testosterone suppression levels from four clinical trials evaluating the 3-month leuprorelin implant, including analysis below the European Association of Urology (EAU) castration level (<0.2 ng/ml).
Methods:
Studies were conducted in patients with locally advanced/metastatic prostate cancer: (1) a randomised, controlled single-dose study comparing the leuprorelin implant with leuprorelin microspheres; (2) a single-arm, single-dose study of the leuprorelin implant; (3 and 4) two long-term studies with the leuprorelin implant administered twice, 12 or 16 weeks apart. Patients received 3-month leuprorelin (5 mg) implant or 3-month leuprorelin (10.72 mg) microspheres. Testosterone levels were analysed using radioimmunoassay or ultrasensitive liquid chromatography tandem mass spectrometry.
Results:
Both the leuprorelin implant and the leuprorelin microspheres achieved mean testosterone suppression (⩽0.5 ng/ml) within 4 weeks for >3 months. In both long-term, single-arm studies with the leuprorelin implant, median values of testosterone ⩽0.2 ng/ml were achieved at Week 4 and maintained until study completion (6 and 8 months); PSA decrease was also observed versus baseline.
Conclusions:
Long-lasting steady serum levels of testosterone, comparable with orchiectomy and consistent with the EAU-recommended castration level (<0.2 ng/ml), were achieved at Week 4 and maintained up to 8 months in men with advanced prostate cancer who received the leuprorelin implant.
Introduction
Androgen deprivation therapy (ADT) is the mainstay of treatment for men with metastatic prostate cancer.1–4 Standard ADT includes bilateral orchiectomy, luteinizing hormone-releasing hormone (LHRH) agonists (with or without antiandrogens) and antagonists. Long-acting LHRH agonists are the dominant form of ADT, 1 , 2 of which leuprorelin acetate is one of the most commonly prescribed. 5
The two, unique, slow-release pharmaceutical forms of leuprorelin acetate (Leuprorelin Sandoz®, HEXAL®/Sandoz®, Germany), a 1-month 3.6 mg leuprorelin depot and a 3-month 5 mg leuprorelin depot implant, were approved for clinical use in 2007. 6 , 7 The delivery system contains a fully biodegradable polymer of poly-(lactide-co-glycolic acid) or poly-(D,L-lactide) for the 1-month and 3-month formulation, respectively. Using a syringe applicator, the rod-shaped, solid implant is injected subcutaneously into the anterior abdominal wall, where it is hydrolytically degraded to release the homogeneously dispersed active substance. These implants are solvent-free and are supplied in prefilled syringes ready to use, with no refrigeration or reconstitution required. 6 , 7
Previous clinical trials have demonstrated that both slow-release formulations of the leuprorelin implant had comparable safety and efficacy with the traditional leuprorelin microspheres (Trenantone®, Takeda, Germany) for achieving successful testosterone suppression (⩽0.5 ng/ml, 1.7 nmol/l) and normalising prostate-specific antigen (PSA) levels. 6 The two formulations do, however, differ both in practical aspects (the leuprorelin implant is ready to use, while the leuprorelin microspheres require reconstitution) and bioavailability (the leuprorelin implant has a four-fold greater bioavailability than the leuprorelin microspheres). 6 , 7
The European Association of Urology (EAU) first recommended lowering the threshold of the target castrate level from <0.5 ng/ml to <0.2 ng/ml (0.7 nmol/l) in 2015, 8 following the finding that the mean value of testosterone is 0.15 ng/ml after surgical castration. 9 Failure to reach testosterone levels <0.2 ng/ml following ADT could correlate with time to castrate-resistant prostate cancer (CRPC) and mortality. 10
The main objective of the original four clinical studies of the leuprorelin implant was to identify the proportion of patients with successful testosterone suppression (<0.5 ng/ml) within 8 weeks and continuing for the study period. 6 Here, we have reviewed unpublished data from these studies for the 3-month leuprorelin implant, in particular, testosterone suppression below 0.2 ng/ml.
Patients and methods
Patients
Eligibility criteria were similar for the four studies. 6 Briefly, patients had histologically confirmed locally and regionally advanced prostate cancer (T3–4N0M0, T1–4N1M0), metastatic (T1–4N0–1M1) or recurrent disease after prostatectomy, radiotherapy or brachytherapy, a life expectancy of >12 months and a testosterone level ⩾2.3 ng/ml (morning value).
Study design
The four open-label, multi-centre studies assessed efficacy, tolerability and pharmacokinetics of the 3-month leuprorelin implant containing 5.25 mg leuprorelin acetate, equivalent to 5 mg leuprorelin (Leuprorelin Sandoz®, HEXAL®/Sandoz®, Germany) (Figure 1). One was a comparator-controlled, single-dose trial that compared the 3-month leuprorelin implant with traditional 3-month prolonged-release microspheres containing 11.25 mg leuprorelin acetate, equivalent to 10.72 mg leuprorelin (Trenantone®, Takeda, Germany). 6 In each study, the leuprorelin implant or leuprorelin microspheres were administered subcutaneously into the abdominal wall by an oncologist. In the long-term studies, a second administration was performed after 12 weeks or 16 weeks.

Comparator-controlled and single-arm studies of the leuprorelin 3-month implant.
Prospective patients provided informed consent. All studies complied with Good Clinical Practice and were approved by the relevant ethics committees.
The original primary endpoint of the four studies was the proportion of patients with successful testosterone suppression, defined as testosterone concentration ⩽0.5 ng/ml at ⩾2 consecutive time points within 8 weeks after first administration, continuing until Week 12 for the single-dose studies, Week 24 for the 12-week administration interval, or Week 32 for the 16-week administration interval, with the exception of escapes.
In the single-arm studies, an escape was defined as a testosterone level >0.5 ng/ml for one sample after achieving suppression, followed again by at least one value ⩽0.5 ng/ml. In the comparator-controlled study, an escape was defined by a testosterone level >0.5 ng/ml for two consecutive samples after achieving suppression during 12 weeks after administration, followed by at least one value <0.5 ng/ml.
The main objective of the current retrospective analysis was to further review testosterone suppression levels from the four clinical trials including analysis below the EAU castration level (<0.2 ng/ml).
Efficacy assessments
In the four studies, testosterone levels were measured at each visit (single-dose studies: daily measurements for the first week, then once weekly; multi-dose studies: every 2 weeks). Testosterone levels were analysed at a central laboratory using radioimmunoassay with lower limit of quantification (LOQ) of 0.1 ng/ml (single-dose studies) and by ultrasensitive liquid chromatography tandem mass spectrometry with a LOQ of 0.05 ng/ml (long-term studies). PSA levels were determined every 4–8 weeks.
An overall efficacy assessment was made at the end of the clinical phase by the investigator using a five-point rating scale (very good, good, indifferent, bad, very bad).
Safety and tolerability
The occurrence of adverse drug reactions was assessed by nondirective questioning of the patient at each visit, for example: “Have you been uncomfortable since the last visit?” For each adverse event, the investigator provided the onset and stop date, intensity, treatment required, outcome, and action taken with the investigational drug. The investigator also judged whether these were treatment-related.
Improvements in symptoms of dysuria, nycturia and bone pain were recorded by the investigator using a four-point rating scale (absent, mild, moderate, severe). An overall tolerability assessment was made by the investigator using a five-point rating scale (very good, good, indifferent, bad, very bad).
Analysis
Due to the similar designs of the studies, pooled analysis of data could be performed. Results are reported for the per-protocol population.
Descriptive statistics were used to evaluate the data based on binomial distribution, stating absolute and relative frequencies and the lower bound of the 95% confidence interval.
Results
Baseline patient demographics and characteristics have been reported previously 6 and are illustrated in Figure 2. In summary, initial mean testosterone levels were similar across the studies and, in the randomised, comparator-controlled study, initial median PSA levels suggested that patients who received the leuprorelin implant had more severe disease (40.1 ng/ml) than those who received the leuprorelin microspheres (20.6 ng/ml).
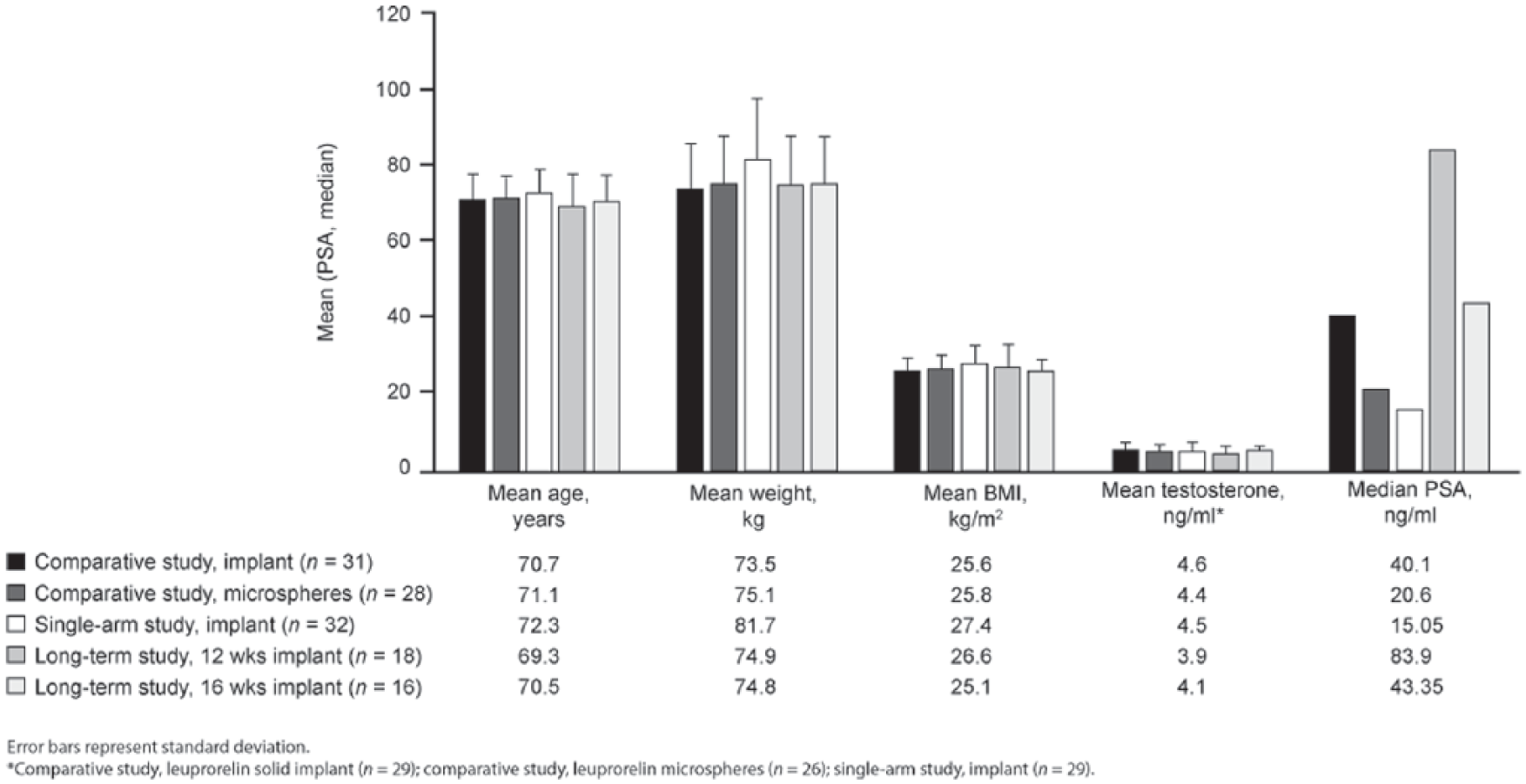
Patient demographics and baseline characteristics.
Single-dose studies: successful testosterone suppression
In the comparator-controlled study, successful testosterone suppression (⩽0.5 ng/ml) was comparable between the two treatment groups at Week 8, 12 and 16. 6
In the single-dose, single-arm study, 100% (29/29) of patients who received the leuprorelin implant achieved successful testosterone suppression using the early criteria of ⩽0.5 ng/ml within the first 8 weeks and was maintained at 12 and 16 weeks (Figure 3a–b); median duration 19.7 weeks (range: 16.0–32.1). No patients had an escape in testosterone level.

(a) Individual testosterone serum concentrations and (b) concentration–time profile for mean serum testosterone levels following the injection of single-dose leuprorelin 3-month implant (single-arm, single-dose study).
Data from the single-dose, single-arm study and the leuprorelin implant arm in the comparator-controlled study were compared with data from the leuprorelin microspheres arm from the comparator-controlled study (per-protocol populations). For both formulations, mean time to onset of castration level (⩽0.5 ng/ml) was approximately 4 weeks and lasted for >3 months (Table 1a). Mean times of testosterone re-increase >0.5 ng/ml were also similar. Median testosterone levels remained stable at Week 4, 8 and 12 in both treatment groups (Table 1b).
(a) Mean time to testosterone suppression and duration of testosterone suppression and (b) median testosterone levels with the leuprorelin 3-month implant and the leuprorelin 3-month microcapsules.

SD, standard deviation; Tonset, time post-dose to the first of the two measurements indicating testosterone suppression; Tw, duration of testosterone suppression; Tlast, last timepoint of testosterone suppression.
Per-protocol population.
n = 24 as two patients did not achieve testosterone suppression.

CI, confidence interval.
Pooled analysis of the two single-dose studies (per-protocol populations).
The proportion of patients achieving a castration level ⩽0.2 ng/ml were similar at Week 8, 12 and 16 (leuprorelin implant: 48%, 52% and 48%, respectively; leuprorelin microspheres: 48%, 45% and 37%, respectively).
Long-term, multi-dose studies: successful testosterone suppression and other clinical findings
In both long-term studies, median serum levels of testosterone ⩽0.2 ng/ml were achieved at Week 4 and lasted up to 24 weeks and 32 weeks for the administration interval of 12 and 16 weeks, respectively (individual maximum median value of 0.21 ng/ml at Week 12 and 0.14 ng/ml at Week 16 for the administration interval of 12 weeks and 16 weeks, respectively). At the last value measured (Week 24 and Week 32 for the 12- and 16-week administration interval studies, respectively) median testosterone levels were 0.09 ng/ml in both studies.
The mean serum testosterone level when the leuprorelin implant was administered at a 12-week interval was 0.13 ng/ml [standard deviation (SD) 0.1 ng/ml] at Week 24, with an individual maximum value of 0.46 ng/ml (Figure 4a). The mean serum testosterone level when the leuprorelin implant was administered with a 16-week interval, was 0.09 ng/ml (SD 0.05 ng/ml) at Week 32, with an individual maximum value of 0.17 ng/ml (Figure 4b). There were no testosterone escapes.

Long-term testosterone suppression and decrease in mean PSA levels with the leuprorelin 3-month implant in the long-term, multi-dose studies. Long-term testosterone suppression lasting up to (a) 6 months with the 12-week administration interval study, n = 15 and (b) 8 months with the 16-week administration interval study, n = 15. Decrease in mean PSA levels over time for patients in the (c) 12-week administration interval study, n = 15 and (d) 16-week administration interval study, n = 15. Black arrows indicate time of dosing.
Testosterone suppression with the leuprorelin implant was accompanied by stable or improved clinical findings (digital rectal examination, prostatic status or diameter of the tumour lesion) in 100% of patients who completed the 12-week interval study, and in the majority (>85%) of patients who completed the 16-week interval study. In the 12 and 16-week interval studies, a PSA decrease (⩽4 ng/ml) was observed in 100% (15/15) of patients following leuprorelin administration (Figure 4c–d).
Using a five-point scale, the overall efficacy of the leuprorelin implant was judged by the investigator as either good or very good in 15/15 completers in both long-term studies (Table 2).
Investigator-rated subjective improvement of overall efficacy as well as symptoms and judgement for the long-term, multi-dose studies (12-week administration interval study and 16-week administration interval study) following injection of leuprorelin 3-month implant.
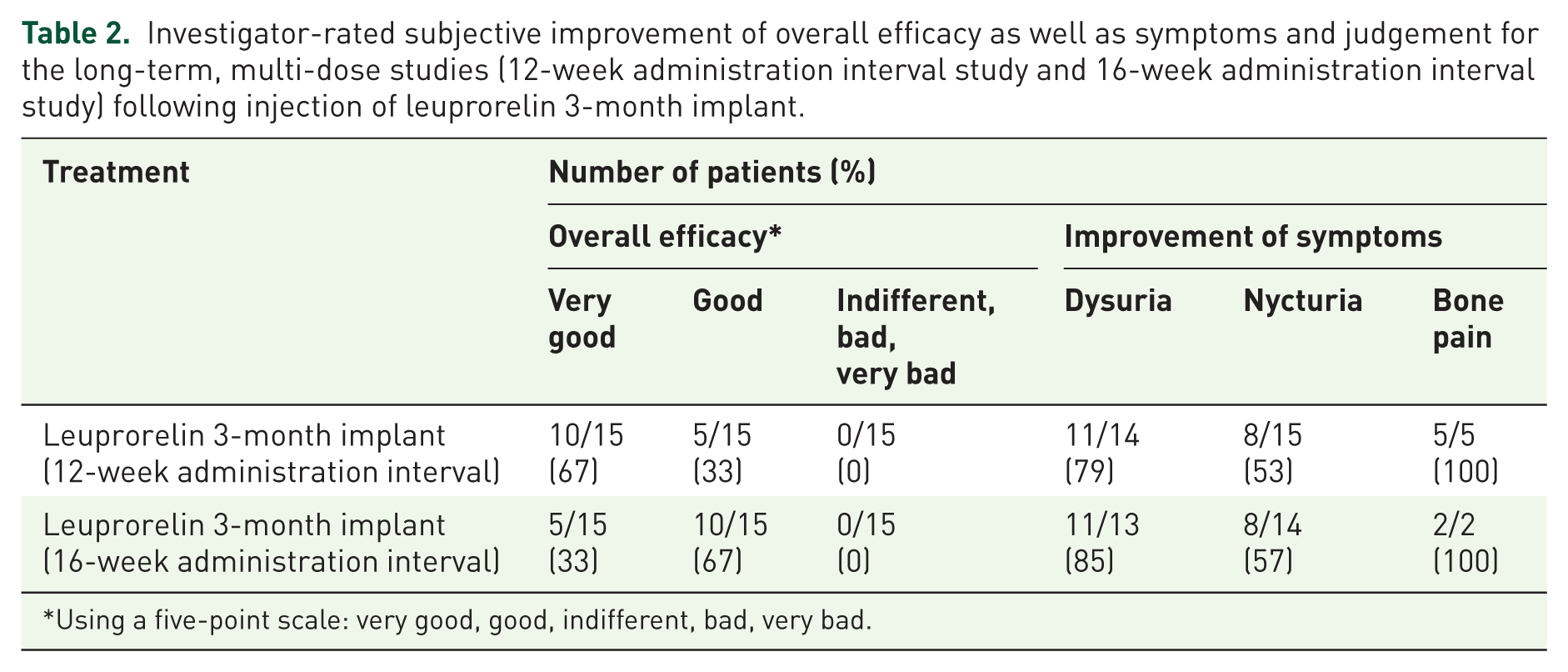
Using a five-point scale: very good, good, indifferent, bad, very bad.
Safety and tolerability
The leuprorelin implant has a similar safety and tolerability profile to the leuprorelin microspheres. 6 In the two long-term studies, only three mild adverse events in four patients were suspected to be related to the leuprorelin implant, but resolved completely (arterial hypertension, n = 1; hot flushes, n = 2; headache, n = 1). Overall, there were three deaths during the study periods but none were classified as related to the treatment. No other serious adverse events were reported and no clinically significant deviations from normal were registered with regard to laboratory findings.
In the two long-term studies, subjective clinical symptoms attributable to prostate cancer (dysuria, nycturia and bone pain) were improved in the majority of patients (Table 2). Using a five-point scale, for all patients who completed the study, investigators rated the overall tolerability as good or very good (data not shown).
Discussion
The development of pharmaceutical formu-lations of LHRH agonists have focused on extended-release, more convenient depot forms, which effectively suppress serum testosterone to castrate levels, while meeting physician and patient needs. From an efficacy perspective, the most important parameters are time of onset and duration of testosterone suppression to castrate levels. The four clinical studies have previously shown the 3-month depot formulation of the leuprorelin implant to be an effective extended-release drug that achieves and maintains castrate serum testosterone levels (⩽0.5 ng/ml). 6
The retrospective analysis of unpublished data presented here, further confirms that the 3-month leuprorelin implant and the traditional 3-month leuprorelin microspheres are comparable in terms of achieving mean testosterone suppression to levels ⩽0.5 ng/ml. In addition to comparable times to, and duration of, testosterone suppression, the concentration–time profile of serum testosterone levels from the single-dose study showed a similar profile to the comparator-controlled study. 6 These pharmacodynamic results are in agreement with previous phase I study results. 11
It is also important to evaluate testosterone levels below the EAU-revised level of castration (<0.2 ng/ml). 2 , 8 The long-term, repeated-administration study of the 3-month leuprorelin implant showed mean and median testosterone values ⩽0.2 ng/ml to study completion (6 months) with a 12-week administration interval. This deep testosterone suppression was also observed to study completion (8 months) with a potentially longer administration interval (16 weeks). In both studies, this testosterone suppression below 0.2 ng/ml was accompanied by stable or improved clinical findings and a PSA decrease compared with baseline.
The results of the two long-term, repeated-administration studies are in agreement with a large, non-interventional survey (n = 818), which showed a decrease in testosterone was accompanied by a significant decrease in PSA levels (⩽4 ng/ml), versus baseline, after two injections of leuprorelin implant in men with advanced, hormone-sensitive prostate cancer. 7 More recently, in an open-label, non-interventional, nonrandomised, multi-centre cohort study, treatment with the leuprorelin implant achieved a reduction in median testosterone levels from 3.8 ng/ml (baseline) to <0.2 ng/ml (Month 3). 12
The clinical significance of reducing testosterone levels to <0.2 ng/ml has been demonstrated in a large prospective study, where failure to reach deep testosterone suppression after first-line ADT may predict poor outcomes in men with hormone-sensitive nonmetastatic prostate cancer. 10 Support for a castration threshold <0.5 ng/ml has also been demonstrated in smaller retrospective and prospective studies, where failure to reach this level correlated with a higher risk of disease progression, developing CRPC and death from prostate cancer. These studies also discussed the importance of routinely evaluating testosterone levels.13–16
The EAU 2015/2016 guidelines state that there is no evidence for choosing between LHRH agonists for the first-line treatment of men with metastatic prostate cancer with no impending spinal cord compression, 2 , 8 but do highlight that practical differences between products should be considered in everyday practice, 2 , 8 including storage, preparation and mode of administration (e.g. subcutaneous/intramuscular versus oral administration). 17 , 18 However, it has recently been hypothesised that achieving and maintaining the lowered EAU castration level may impact on the efficacy of LHRH analogues and could be a differentiating factor for these treatments. 19
Practical differences between the leuprorelin implant and other comparable formulations of its class are that it is in a ready-to-use formulation, with no requirement for refrigeration or reconstitution. 6 , 7 The 3-month leuprorelin implant has also demonstrated four-fold greater bioavailability compared with the 3-month leuprorelin microspheres, 20 reflecting an improved galenic formulation and a lower dose of leuprorelin needed to exhibit a similar therapeutic effect to the leuprorelin microspheres (5 mg versus 10.72 mg, respectively). Furthermore, a system of objectified judgement analysis reported the leuprorelin implant to have a maximum score for user-friendliness and noted it to be less expensive than other LHRH agonists in several European countries. 17 A hypothetical cost analysis of the leuprorelin implant compared with alternative formulations of leuprorelin, including the leuprorelin microspheres, has also shown that use of the leuprorelin implant could be associated with substantial time and cost savings. 21
The safety and tolerability profile of the leuprorelin implant was comparable with the leuprorelin microspheres and other LHRH agonists within its class. 6 , 7 , 22 In the two long-term studies, for all patients who completed the study, their investigator rated the overall tolerability as good or very good, in agreement with the single-dose study 6 and a non-interventional, observational study of 1148 German men with advanced prostate cancer. 22
Limitations of the presented data include the low number of patients in the two long-term, repeated-administration studies. Despite this, serum testosterone profiles were comparable with the two single-dose studies, as well as with an observational study of 818 patients with advanced hormone-sensitive prostate cancer. 7 Further long-term studies to monitor testosterone suppression below the EAU-revised level of castration in large numbers of patients treated with the leuprorelin implant should be performed to confirm the results described here.
Conclusion
These data further confirm that the 3-month leuprorelin implant has comparable efficacy and safety with the 3-month leuprorelin microspheres in terms of achieving testosterone suppression ⩽0.5 ng/ml. In the long-term, multi-dose studies, the 3-month leuprorelin implant demonstrated excellent and long-lasting testosterone suppression at <0.2 ng/ml, in line with the EAU 2015 and 2016 guidelines.
Footnotes
Acknowledgements
Medical writing assistance was provided by Holly Kramer of Spirit Medical Communications Ltd, Manchester, UK funded by Sandoz International GmbH, Holzkirchen, Germany.
Funding
This study was funded by Sandoz International GmbH, Holzkirchen, Germany.
Conflict of interest statement
Mladen Solarić is a consultant of regional offices for and has received honoraria from Astellas Pharma, Surrey, UK; Sanofi, Paris, France; Janssen, Hrvatska, Croatia; Sandoz International GmbH, Holzkirchen, Germany; Bayer-Pharma, Berlin, Germany and Amgen, California, USA. He has also received honoraria from Abbot Nutrition, Kent, UK. Anders Bjartell has received speaker’s honoraria from Sandoz International GmbH, Holzkirchen, Germany. Ursula Thyroff-Friesinger and Davide Meani are employees of Hexal AG, Holzkirchen, Germany.
