Abstract
Background:
To present the operative and post-operative comparison between patients who underwent tumor-bed closure with sutures compared with bovine serum albumin–glutaraldehyde (BioGlue®) tissue sealant only.
Methods:
We retrospectively analyzed data from our ongoing database of 507 eligible patients who underwent open NSS nephron-sparing surgery in our department between January 1995 and May 2014. Patients had tumor-bed closure with sealant adhesive (255 patients) or standard suture technique (252 patients). Demographic, clinical and perioperative data were compared between the two groups, by Chi-square test or by Fisher–Irwin exact test for categorical variables, and by t test for differences in means or by Wilcoxon rank sum test for continuous variables. A multivariate analysis was also done.
Results:
Patients’ baseline characteristics showed similar distribution of the analyzed parameters among both groups, with few differences: younger age in the sealant group (65.4 versus 68.4 years, p = 0.01) and slightly larger mass size in the suture group (4.0 versus 3.9 cm, p = 0.03). Ischemia time was significantly shorter in the sealant group (21.8 versus 27.0 minutes, p = 0001). Blood loss and transfusion rate (0.8% versus 11.9%, p = 0.0001) were significantly less in the sealant group. A multivariate analysis showed date of surgery and blood loss as the major parameters affecting transfusion rate.
Conclusions:
Closing the tumor bed with BioGlue® tissue adhesive is feasible, safe, can shorten ischemia time and potentially reduce transfusion rate.
Introduction
Renal cell carcinoma (RCC) represents 2–3% of all cancers with increasing incidence of about 2% over the last two decades [Lindblad, 2004]. Based on the available oncological and quality-of-life outcomes, the current evidence suggests that localized renal tumors are best managed by nephron-sparing surgery (NSS), irrespective of surgical approach [Ljungberg et al. 2015]. Hence, almost all the guidelines recommend this approach in individuals who are fit for anesthesia and surgery, and when partial nephrectomy is feasible [Campbell et al. 2009; Ljungberg et al. 2015].
Closure of the tumor bed after tumor excision remains a challenging task and is usually carried out by using various suture techniques with or without additional tissue adhesives in order to prevent hemorrhage or urine leakage. This step could be time consuming and may prolong ischemia time. It is also associated with renal parenchymal damage that is related to the compressed functioning tissue and blood vessels [Bak et al. 2004].
We have previously reported our initial experience of using tissue adhesive only for closing the parenchymal defect following NSS [Hidas et al. 2006]. Others have also used BioGlue® (Cryolife, Atlanta, US) or other sealants in laparoscopic partial nephrectomy [Breda et al. 2007; Nadler et al. 2006].
The goal of the current study was to retrospectively compare perioperative outcomes between patients who underwent NSS with suturing of the tumor bed with those in whom tissue sealant was used instead.
Methods
Patients
The study group included 548 consecutive patients who underwent open NSS for solid enhancing renal mass operated at our department between January 1995 and May 2014. Data from our database on NSS were retrieved and analyzed. The study was approved by a suitably constituted Ethics Committee of our institution and the study conforms to the provisions of the Declaration of Helsinki.
After excluding patients with missing data and those who underwent multiple surgeries, the cohort included 507 patients: 255 patients in whom the tumor bed was closed with sealant (sealant group) and 252 patients in whom the tumor bed was closed by conventional suturing (suture group).
Surgical technique
A standard supra-11th rib flank approach with the patient in full flank position was used. After complete mobilization of the kidney within Gerota’s fascia, the ureter and vascular pedicle were identified and isolated on a vascular tape. Mannitol, at 0.5 g/kg was given intravenously before clamping the renal artery with subsequent cooling of the kidney surface with ice-slush. We perform every NSS under cold ischemia, as we believe that by doing this, we could preserve maximal renal function. Enucleation of the tumor was initiated with circumferential incision followed by blunt dissection between the fibrous tumor pseudo-capsule and the renal parenchyma. Attention was taken to remove a rim of minimal normal renal tissue. The tumor bed was sampled with frozen section analysis to verify tumor-free margins. Transected blood vessels in the resection bed were hemostatically suture ligated and collecting system entries were sutured using Monocryl 4/0 sutures. Tumor bed was closed either with sutures (1/0 Vicryl with blunt-end) to approximate the edges of the parenchymal defect, or by using 2–10 ml of bovine serum albumin–glutaraldehyde sealant (BioGlue®) to fill the tumor bed. It should be emphasized that while applying the BioGlue®, the operated kidney was isolated from its surrounding tissue by surgical pads. Vessel clamps were then removed, ischemia time determined, and the kidney was inspected for bleeding or leakage. The vast majority of the initial cases were performed with standard suture technique, whereas the latter cases were mainly undertaken using the tissue adhesive. There were no standardized selection criteria for the choice of renorrhaphy technique. All operations were performed by a single surgeon (ON).
BioGlue® sealant
BioGlue
Statistical analysis
For the demographic and clinical baseline categorical variables (gender, side, presentation, etc.), and for the perioperative categorical variables (opening of collecting system, transfusion, complications, etc.), numbers and percentages were calculated. Distributions for these variables were compared and analyzed by the Chi-square test or by Fisher–Irwin exact test (for small numbers).
For the demographic and clinical baseline continuous variables (age, BMI, tumor size, RENAL nephrometry score, etc.), and for the perioperative continuous variables [operation time, ischemia time, estimated blood loss (EBL)], ranges, medians, means and standard deviations were calculated. Test for normality was done by Shapiro–Wilk test for normality. Results between continuous variable were analyzed by t test for differences in means or by Wilcoxon rank sum test. In order to compare the differences between both groups regarding transfusion rate, we analyzed the significant parameters in a multivariate analysis using binary logistic regression with transfusion rate being the dependent variable.
All statistical tests were analyzed to a significance level of 0.05. Statistical analyses were performed using STATA v.12 software.
Results
The two study groups were similar in almost all baseline characteristics except for age and distribution of pathologic findings. Median patients’ age was lower in the sealant group compared with the suture group (65.4 ± 12.0 years versus 68.4 ± 14.6 years, p = 0.01). The suture group had higher benign pathology compared with the sealant group (12% versus 5.9%, p = 0.01). The stage-per-stage distribution was similar in both groups.
The mean tumor size was slightly larger in the sealant group (4.2 ± 1.6 versus 3.9 ± 1.5 cm, p = 0.03) while the median size of the lesions revealed larger tumors in the suture group (4.0 versus 3.8 cm). However, tumor complexity as expressed by RENAL nephrometry score was similar in both groups. Table 1 summarizes the demographic and the clinical data of both groups.
Patients’ demographic and clinical baseline characteristics in both study groups.
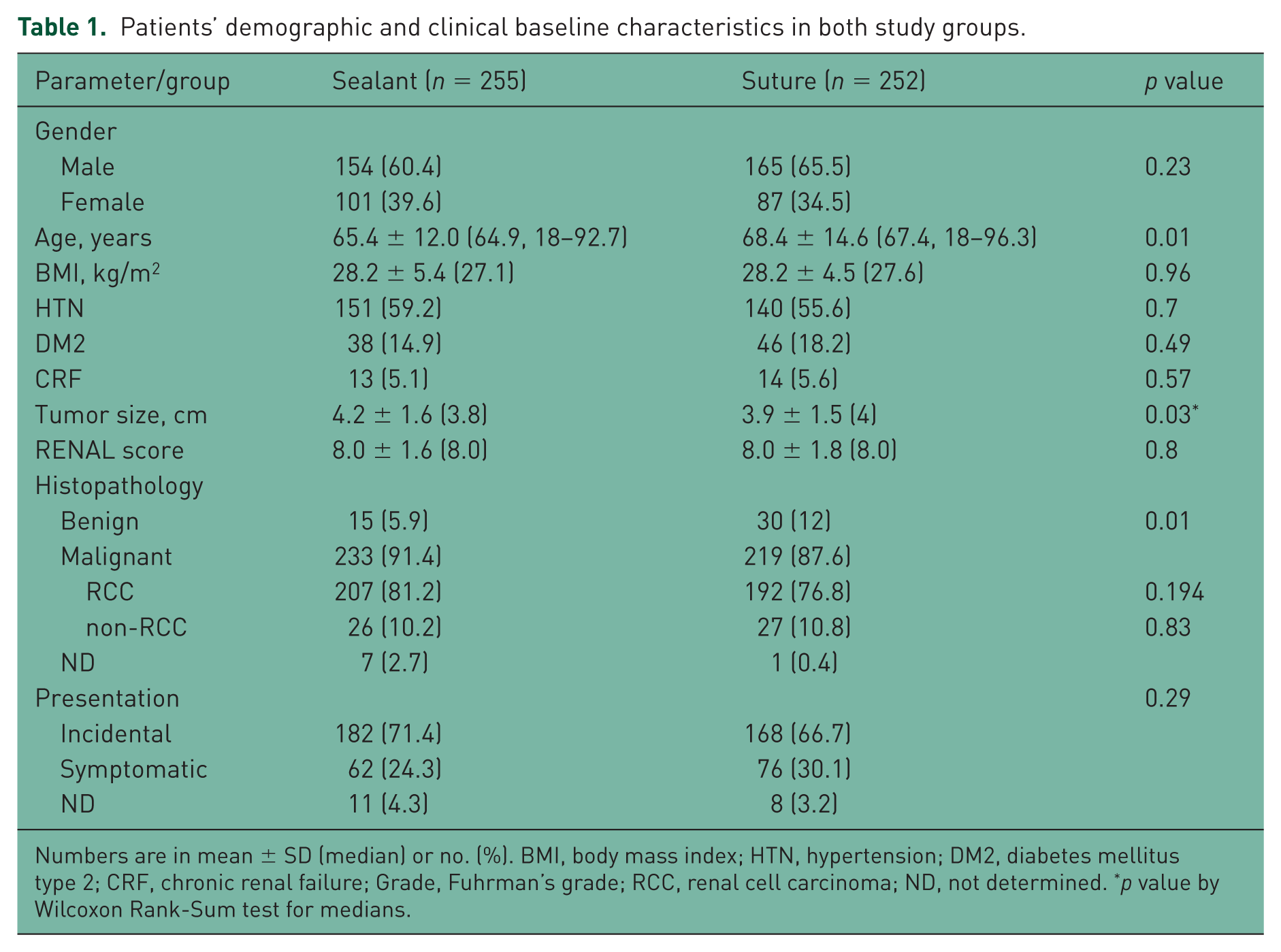
Numbers are in mean ± SD (median) or no. (%). BMI, body mass index; HTN, hypertension; DM2, diabetes mellitus type 2; CRF, chronic renal failure; Grade, Fuhrman’s grade; RCC, renal cell carcinoma; ND, not determined. *p value by Wilcoxon Rank-Sum test for medians.
The rate of intraoperative and postoperative complications was not significantly different between both groups. However, patients in the sealant group demonstrated advantages in several aspects including shorter ischemia time (21.8 ± 7.6 minutes versus 27.0 ± 9.4 minutes, p = 0.0001), decreased operative time by median of 10 minutes and reduced mean EBL (178 ± 283 versus 42 ± 82, p = 0.0001) that was translated into significantly less transfusion rate (0.8% versus 11.9%, p = 0.0001) (Table 2).
Perioperative variables in both study groups.
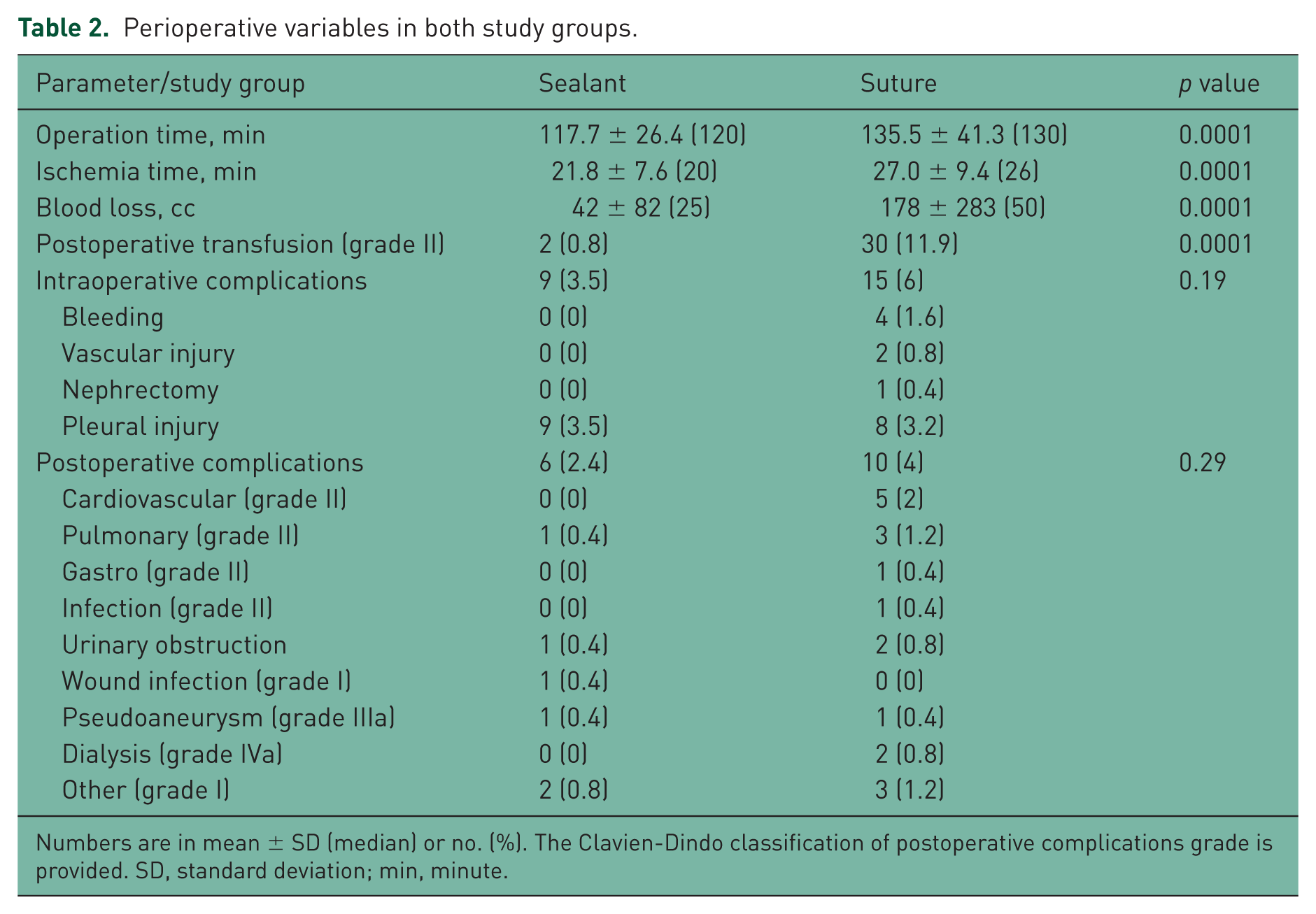
Numbers are in mean ± SD (median) or no. (%). The Clavien-Dindo classification of postoperative complications grade is provided. SD, standard deviation; min, minute.
A multivariate analysis using binary logistic regression with transfusion rate being the dependent variable showed a statistically significant correlation between EBL and transfusion rate (p = 0.0001) and date of surgeries (p = 0.0001). All other variables did not reach statistical significance.
Discussion
NSS entails complete local resection of the renal tumor while leaving the largest possible amount of normal functioning parenchyma in the involved kidney [Kenney et al. 2012].
The standard technique to close the renal defect following tumor resection is by approximating the transected margins with various suture techniques, sometimes together with hemostatic substances [Novick, 1993].
Suturing the tumor bed is time consuming [Orvieto et al. 2004], and is potentially harmful for the sutured parenchyma because of its ischemic effect [Hidas et al. 2007]. The tension of the sutures on the frail renal parenchyma and the distortion of the normal renal anatomy may result in ischemic loss of viable tissue [Hidas et al. 2007].
The impact of ischemia time on functional recovery is well established as several studies showed that when the renal hilum is clamped, ‘every minute counts’ [Becker et al. 2009; Nguyen and Gill, 2008; Thompson et al. 2010]. Thus, every effort should be done to minimize the clamping time as much as possible.
In our previous preliminary report, we showed that sutureless NSS with the use of sealant tissue adhesive (BioGlue®) is feasible, safe, does not increase intraoperative or postoperative complications, and has excellent oncological and functional outcomes [Hidas et al. 2006].
In the current study, we were able to demonstrate that the use of sealant agent for tumor bed closure was associated with reduced ischemia time by an average of >5 minutes and median difference of 10 minutes (21.8 minutes versus 27.0 minutes) decreasing the perioperative complications rate.
Another potential advantage of the sealant agent as observed in our study is the reduced transfusion rate in the sealant group compared with the suture group (0.8% versus 11.9%, p = 0.0001). Such a favorable outcome could potentially be attributed, at least in part, for the use of sealant instead of sutures. However, several biases exist (such as surgeon prior experience and intraoperative bleeding, excluding the use of the BioGlue®); as such, no conclusion could be drawn in this retrospective comparison, especially with a multivariate analysis showing statistically insignificant differences between both groups. Moreover, the difference between the two groups regarding the estimated intraoperative blood loss was small, and though statistically significant, its clinical significant is of limited value.
The used sealant agent in our study (BioGlue®) is a liquid gel that fills up the cavity in the enucleation site and adequately prevents urine leak and bleeding. This can prevent distortion of the renal parenchymal anatomy, which may potentially lead to better functional preservation [Hidas et al. 2006].
Tissue sealants and glues can be also applied in minimally invasive partial nephrectomy [Bak et al. 2004; Breda et al. 2007; Nadler et al. 2006; Richter et al. 2003; Simmons and Gill, 2007]. Bak and colleagues reported decreased blood loss and none of the six patients studied needed blood transfusion [Bak et al. 2004]. Ischemia time was short (10–14 minutes) compared with the standard laparoscopic suturing technique [Bak et al. 2004]. Richter and colleagues reported similar results with 25 patients and follow up of up to 1 year [Richter et al. 2003]. Simmons and Gill reported their large experience with the use of FloSeal in addition to standard suturing in laparoscopic partial nephrectomy [Simmons and Gill, 2007]. Nadler and colleagues reported the use of BioGlue® in eight laparoscopic partial nephrectomies and found it highly effective in stopping surgical oozing; they also reported decreased blood loss in these patients [Nadler et al. 2006]. From the above studies, we can conclude that the use of tissue sealant in laparoscopic procedures is also feasible.
It should be mentioned and emphasized that long before BioGlue® was used in kidney surgeries, it was used in the most complicated cardiac and vascular surgeries [Chao and Torchiana, 2003] with good results. We should also mention that before its clinical use in patients, it was preclinically studied and was shown to be safe and highly effective [Gundry et al. 2000].
The disadvantages of using BioGlue® would be the lack of prospective randomized data to support its wide use and its price (about $300), which is higher than the several sutures used for standard renorrhaphy. Another concern could be the local effect of the BioGlue® if there is an indication of re-operation on the same kidney. We have performed several re-operations on previously operated kidneys with BioGlue®, and although the stiff tissue surrounding the glue makes the operation more challenging, it is still feasible, as we managed to do a second partial nephrectomy in most cases.
The major limitations of our study were the nonprospective nature and the lack of randomization. We provide only a single-center experience with a relatively small group. Moreover, the decision to use BioGlue® was subjective and depended merely on the surgeon’s discretion. These limitations could result in selection bias. Moreover, the vast majority of the surgeries where BioGlue® was used were more recent, which could lead to a bias because of a learning curve. This is shown in the significant difference between the older and recent surgeries. However, the high number of surgeries done by the same surgeon could potentially eliminate this bias.
The long-term functional and oncological impact of the aforementioned technique deserve separate analysis, which we hope to provide in the near future.
We are aware that a randomized, multi-institutional prospective controlled trial should be conducted before we could recommend the use of tissue sealant as an alternative to standard suture renorrhaphy after NSS.
In conclusion, closing the tumor bed during NSS with BioGlue® tissue adhesive is safe and feasible and can potentially reduce blood loss and transfusion rate, and significantly shorten the ischemia time.
Footnotes
Acknowledgements
We thank GAN statistics for thorough statistical analysis.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
