Abstract
Background:
Renal cell cancer (RCC) is the tenth most common malignancy in adults. In recent years, several approaches of active and passive immunotherapy have been studied extensively in clinical trials of patients with RCC. The aim of this systematic review was to assess the clinical efficacy of various approaches of specific immunotherapy in patients with RCC.
Methods:
We searched Medline, Scopus, CENTRAL, TRIP, DART, OpenGrey and ProQuest without any language filter through to 9 October 2015. One author reviewed search results for irrelevant and duplicate studies and two other authors independently extracted data from the studies. We collated study findings and calculated a weighted treatment effect across studies using Review Manager (version 5.3. Copenhagen: The Nordic Cochrane Centre, the Cochrane Collaboration).
Results:
We identified 14 controlled studies with 4013 RCC patients after excluding irrelevant and duplicate studies from 11,319 references retrieved from a literature search. Overall, five autologous tumor cell vaccines, one peptide-based vaccine, one virus-based vaccine and one dendritic cell (DC)-based vaccine were studied in nine controlled studies of active specific immunotherapies. A total of three passive immunotherapies including autologous cytokine-induced killer (CIK) cells, auto lymphocyte therapy (ALT) and autologous lymphokine-activated killer (LAK) cells were studied in four controlled studies. The clinical efficacy of tumor lysate-pulsed DCs, with CIK cells was studied in one controlled trial concurrently. The overall quality of studies was fair. Meta-analysis of seven studies showed that patients undergoing specific immunotherapy had significantly higher overall survival (OS) than those in the control group [hazard ratio (HR) = 0.72; 95% confidence interval (CI) = 0.58–0.89, p = 0.003]. In addition, a meta-analysis of four studies showed that there was a significant difference in progression-free survival (PFS) between patients undergoing specific immunotherapy and patients in control groups (HR = 0.86; 95% CI = 0.73–1, p = 0.05).
Conclusions:
Results of this systematic review suggest that some specific immunotherapies such as Reniale, ACHN-IL-2, Newcastle disease virus (NDV) virus-infected autologous tumor cells, ALT and CIK treatment could be beneficiary for the treatment of patients with RCC.
Keywords
Introduction
Renal cell cancer (RCC) comprises a diverse group of solid tumors originating from renal parenchyma [Bodmer et al. 2002; Longo et al. 2007; Linehan and Rathmell, 2012]. RCC, with a globally rising incidence, is the seventh most common cancer in men and the ninth most common in women and constitutes approximately 2–3% of adult malignancies [Goey et al. 1996; Fishman and Seigne, 2002; Reniale, Wagstaff, 2006; Chow et al. 2010; Siegel et al. 2012]. RCC resulted in more than 140,000 deaths worldwide in 2012 [Ferlay et al. 2012]. Almost, half of patients, at the time of presentation, have locally advanced or metastatic disease. Moreover, the disease progresses to metastatic stages or recurs in approximately one third of patients with initially localized disease [Fishman and Seigne, 2002; Longo et al. 2007; Chow et al. 2010; Santos et al. 2011; Linehan and Rathmell, 2012]. Localized RCC is primarily treated with nephrectomy. However, the management of RCC in advanced stages represents a great dilemma in clinical practice [George et al. 2011]. Standard chemotherapeutic agents and targeted-therapies yielded disappointing results in advanced stages of RCC due to adaptive resistance and various side effects [Goey et al. 1996; Longo et al. 2007; Chi et al. 2010; Linehan and Rathmell, 2012; Rasmussen, 2013; Raman and Vaena, 2015]. The 5-year survival rate of patients with metastatic cancer is 11.8% [Surveillance, Epidemiology, and End Results (SEER) Program, 2006–2012].
In the era of nonspecific immunotherapy, treatment with high-dose interleukin-2 (IL-2) and interferon alpha (IFN-α) was used as the mainstay of treatment for advanced RCC [Goey et al. 1996; George et al. 2011]. These nonspecific immunotherapies resulted in durable complete remissions in 7–8% of patients with relapsed or unresectable stage IV RCC [Rini et al. 2009]. However, they did not confer any advantage regarding overall survival (OS) or progression-free survival (PFS) and caused severe side effects in >30% of patients [Rini et al. 2009; May et al. 2010]. However, in recent years, more specific approaches of immunotherapy in the form of passive or active immunotherapy have been developed [Fishman and Seigne, 2002; van Poppel et al. 2009; George et al. 2011; Raman and Vaena, 2015]. Passive immunotherapy involves administrating components of the immune system such as immune cells or antibodies to patients to provide immunity against tumor cells [George et al. 2011; Raman and Vaena, 2015]. On the other hand, active immunotherapy stimulates host immune responses to recognize and destroy malignant cells [Fishman and Seigne, 2002; George et al. 2011; Raman and Vaena, 2015]. Unlike, the nonselective effects of conventional treatments, immunotherapy, theoretically, could selectively target and destroy malignant cells with minimal side effects [Stadler, 2000; van Poppel et al. 2009; Raman and Vaena, 2015].
In recent years, several approaches of active and passive immunotherapy have been studied extensively in clinical trials of patients with RCC [Fishman and Seigne, 2002; van Poppel et al. 2009; Raman and Vaena, 2015]. The aim of this systematic review was to review the clinical efficacy of various approaches of specific immunotherapy in patients with RCC, according to the guidelines of the Border of Immune Tolerance Education and Research Network (BITERN) and Cochrane Collaboration.
Methods
We conducted this systematic review in accordance with general methods recommended by Cochrane Collaboration and BITERN guidelines.
Eligibility criteria for inclusion of studies
In this systematic review, all controlled clinical trials (irrespective of blinding and randomization) and controlled studies with historical controls (in case of proper matching) investigating clinical efficacy of specific immunotherapy (irrespective of active or passive, modes of administration, dosage, frequency and duration) in patients with primary RCC (irrespective of age, sex or race) according to inclusion criteria stated in the protocol were included (see Appendix 1 in the Online Supplementary Material).
Search methods for identification of studies
The primary search process was conducted on 1 June 2014 in Medline, Scopus, TRIP and CENTRAL databases (as databases for journal articles) and DART, OpenGrey and ProQuest (as databases for grey literature) according to the search strategies described in the protocol (see Appendix 1 in the Online Supplementary Material) without any language filter or date restriction. We updated the search process in Medline, Scopus, CENTRAL, ProQuest, DART and OpenGrey databases up to 9 October 2015.
Citations from all databases were imported into an Endnote library (version X6, Thomson Reuters, USA) and duplicate citations were removed using the ‘find duplicates’ feature of Endnote software. Then one of the authors reviewed the title and abstract of the remainder of the search results for irrelevant studies and obvious irrelevant studies were excluded. Full texts of the remainder of the citations were retrieved for further screening and data collection.
Data collection
Full texts of articles were carefully read by two reviewers and the eligibility of the articles independently evaluated according to the inclusion criteria stated in the protocol (see Appendix 1 in the Online Supplementary Material). They excluded ineligible studies along with documenting reasons for exclusion. Data from articles were extracted independently by two reviewers to predesigned data extraction forms in Microsoft Excel spreadsheets (version 2010, Microsoft Corporation, USA).
Quality assessment
The Cochrane ‘risk of bias’ tool was used by two reviewers to assess methodological quality of studies on the following domains (see Appendix 2 in the Online Supplementary Material):
(1) Random sequence generation (selection bias for controlled trials)
(2) Allocation concealment (selection bias for controlled trials)
(3) Blinding of participants and personnel (performance bias)
(4) Blinding of outcome assessment (detection bias)
(5) Incomplete outcome data (attrition bias)
(6) Selective reporting (reporting bias)
Each domain was independently judged by two reviewers to be of ‘low risk’ of bias, ‘high risk’ of bias, or ‘unclear risk’ of bias for each article. Any disagreements between two reviewers regarding either eligibility of articles or quality of studies were resolved through discussion or consultation with a third author.
Evidence synthesis
Evidence synthesis was done using systematic approaches like tabulation and textual description. Results from each study were transformed into a common rubric using Review Manager (version 5.3. Copenhagen: The Nordic Cochrane Centre, the Cochrane Collaboration, 2014). For missing data, we considered statistics that allow calculation of missing data in article or in some cases we contacted the corresponding author.
When between-study heterogeneity allowed, we used Review Manager to do a meta-analysis and calculate a weighted treatment effect across studies. We evaluated between-study heterogeneity by the Chi-square statistic and its p value and I2 statistic. We considered a p value < 0.1 and I2 > 40% as indicating significant between-study heterogeneity. In cases of significant statistical heterogeneity or clinical heterogeneity, a random effects model was used to do the meta-analysis; otherwise a fixed-effects model was used. We expressed the results of the meta-analysis as a hazard ratio (HR) with a 95% confidence interval (CI) for time-to-event outcomes and a risk ratio (RR) with a 95% CI for dichotomous outcomes.
Results
Description of studies
We found 11,319 references in seven databases of which 2168 citations were duplicates and were removed using Endnote software (Figure 1). In primary screening of titles and abstracts of the remaining 9151 citations, 8854 articles were excluded due to obvious ineligibility. After secondary screening of 297 full texts and exclusion of 285 articles, 14 controlled studies with 4013 patients with RCC (Table 1) were included in this systematic review.

Study flow diagram.
Main characteristics of included studies.
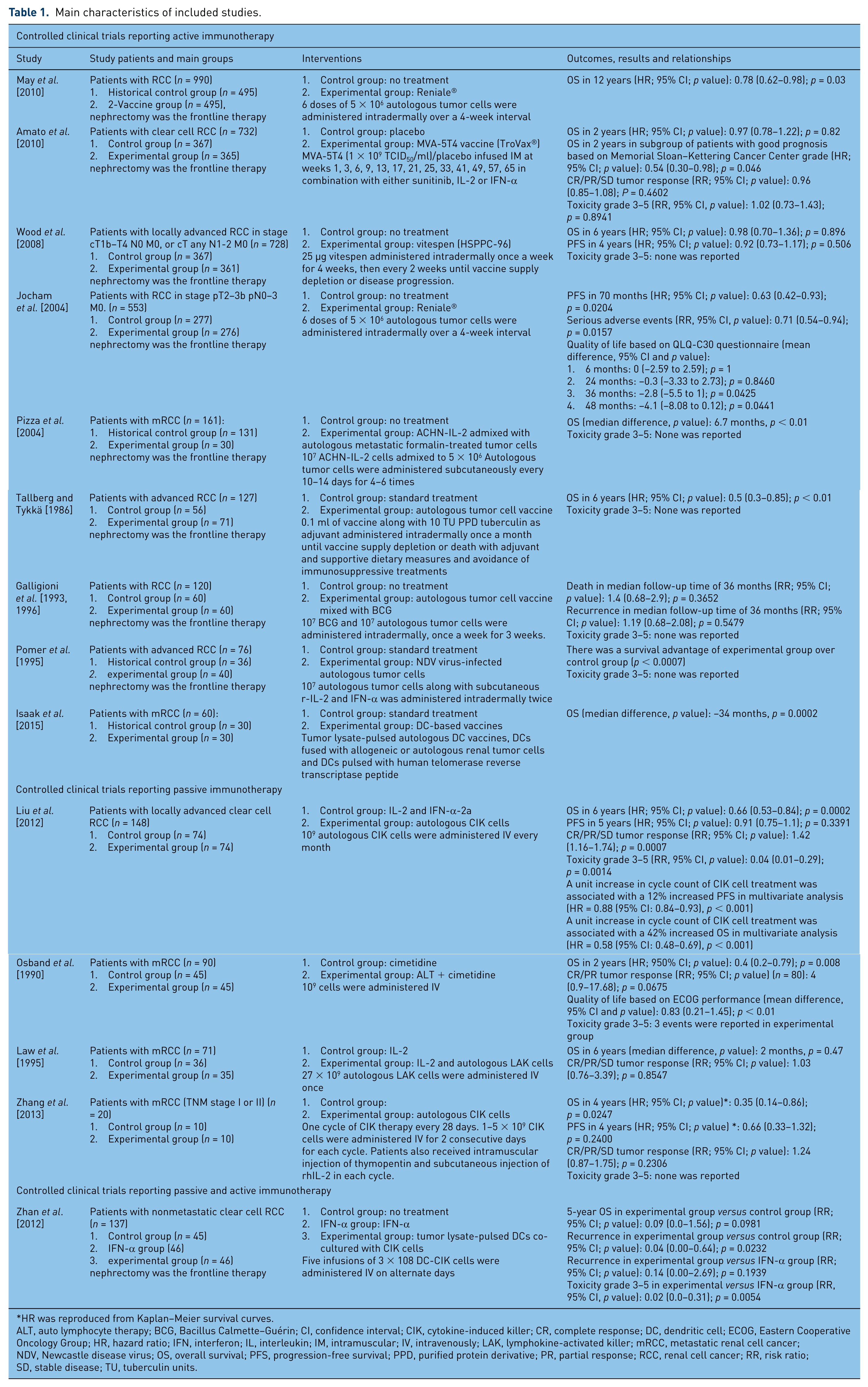
HR was reproduced from Kaplan–Meier survival curves.
ALT, auto lymphocyte therapy; BCG, Bacillus Calmette–Guérin; CI, confidence interval; CIK, cytokine-induced killer; CR, complete response; DC, dendritic cell; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; IFN, interferon; IL, interleukin; IM, intramuscular; IV, intravenously; LAK, lymphokine-activated killer; mRCC, metastatic renal cell cancer; NDV, Newcastle disease virus; OS, overall survival; PFS, progression-free survival; PPD, purified protein derivative; PR, partial response; RCC, renal cell cancer; RR, risk ratio; SD, stable disease; TU, tuberculin units.
Overall, five autologous tumor cell vaccines [Tallberg and Tykkä, 1986; Galligioni et al. 1993; Pomer et al. 1995; Galligioni et al. 1996; Jocham et al. 2004; Pizza et al. 2004; May et al. 2010]; one peptide-based vaccine derived from heat-shock protein (glycoprotein 96) peptide complex (vitespen) [Wood et al. 2008]; a virus-based vaccine engineered to deliver the tumor antigen 5T4 (MVA-5T4 or TroVax® vaccine) [Amato et al. 2010]; and one dendritic cell (DC)-based vaccine [Isaak et al. 2015] were studied in 3547 patients in nine controlled studies of active specific immunotherapies. A total of three passive immunotherapies including autologous cytokine-induced killer (CIK) cells [Liu et al. 2012; Zhang et al. 2013], auto lymphocyte therapy (ALT) [Osband et al. 1990] and autologous lymphokine-activated killer (LAK) cells [Law et al. 1995] were studied in 329 patients in four controlled studies. The clinical efficacy of tumor lysate-pulsed DCs (an active immunotherapy) with CIK cells (a passive immunotherapy) was studied in one controlled trial with 137 patients concurrently [Zhan et al. 2012].
Clinical outcomes investigated in these studies included OS, PFS, tumor response, quality of life and toxicity.
The overall quality of studies were fair. There was a high risk of selection and performance bias in studies of May and colleagues, Tykkä and colleagues, Pomer and colleagues, Pizza and colleagues, and Isaak and colleagues (Table 2). Studies done by May and colleagues, Pomer and colleagues, Pizza and colleagues, and Isaak and colleagues were not randomized clinical trials. In these studies, the outcomes of immunotherapeutic interventions were compared with outcomes of standard treatments in demographically and oncologically-matched historical controls. The randomization method was not described in studies of Galligioni and colleagues and Zhang and colleagues. There was high risk of attrition bias for studies of May and colleagues and Wood and colleagues.
Risk of bias in included studies.

 Low risk of bias.
Low risk of bias.
 Unclear risk of bias.
Unclear risk of bias.
 High risk of bias.
High risk of bias.
NA, not applicable; OS, overall survival; PFS, progression-free survival.
OS
OS and mortality were reported in 13 studies [Tallberg and Tykkä, 1986; Osband et al. 1990; Galligioni et al. 1993; Law et al. 1995; Pomer et al. 1995; Galligioni et al. 1996; Pizza et al. 2004; Wood et al. 2008; Amato et al. 2010; May et al. 2010; Liu et al. 2012; Zhan et al. 2012; Zhang et al. 2013; Isaak et al. 2015]. Of these, four studies on active immunotherapy with 2577 patients [Tallberg and Tykkä, 1986; Wood et al. 2008; Amato et al. 2010; May et al. 2010] and three studies on passive immunotherapy with 258 patients [Osband et al. 1990; Liu et al. 2012; Zhang et al. 2013] reported data on HR of death between experimental and control groups.
Considering the presence of moderate heterogeneity between the study results (Tau² = 0.05; p = 0.01; I² = 63%), we employed subgroup analysis and a random effects model to perform a meta-analysis on the results of these studies. Overall, patients undergoing specific immunotherapy had significantly higher OS than those in the control group (HR = 0.72; 95% CI = 0.58–0.89, p = 0.003) (Figure 2).

Comparison of OS between patients in specific immunotherapy group and control group.
In a subgroup meta-analysis of studies investigating active immunotherapy, there was no significant difference in OS between groups (HR = 0.83; 95% CI = 0.66–1.04, p = 0.1).
Pizza and colleagues and Pomer and colleagues reported a significant survival advantage in patients treated with autologous tumor cell vaccines compared with controls (Table 1) [Pomer et al. 1995; Pizza et al. 2004]. But Isaak and colleagues reported that a DC-based vaccine had a significant negative effect on survival [Isaak et al. 2015].
In a subgroup meta-analysis of studies investigating passive immunotherapy, patients in the passive immunotherapy group had a significant survival advantage over patients in the control group (HR = 0.53; 95% CI = 0.35–0.8, p = 0.002). Liu and colleagues, in their study on CIK treatment, reported that the cycle count of CIK cell treatment significantly improved the OS of patients when analyzed as a continuous variable in the multivariate analysis [Liu et al. 2012].
Hai-Lun and colleagues investigated clinical efficacy of a combined immunotherapeutic approach (tumor lysate-pulsed DCs co-cultured with CIK cells) in patients with nonmetastatic clear cell RCC [Zhan et al. 2012]. Although patients in the experimental group had a higher 5-year survival than those in control group, this difference was not significant.
PFS
PFS was reported in six studies. There were two studies on active immunotherapy with 1281 patients and two studies on passive immunotherapy with 168 patients reporting data on HR of progression between experimental and control groups. Considering the presence of clinical heterogeneity between the study results (although there was not any statistical heterogeneity), we employed subgroup analysis and a random effects model to perform a meta-analysis on the results of these studies. Overall, there was a significant difference in PFS between patients undergoing specific immunotherapy and patients in control groups (HR = 0.86; 95% CI = 0.73–1, p = 0.05) (Figure 3).

Comparison of PFS between patients in specific immunotherapy group and control group.
In a subgroup meta-analysis of studies investigating either active immunotherapy or passive immunotherapy, there was no significant difference in PFS between groups (HR = 0.79; 95% CI = 0.55–1.14, p = 0.21; HR = 0.89; 95% CI = 0.74–1.07, p = 0.22 respectively). However, Liu and colleagues in their study on CIK treatment reported that the cycle count of CIK cell treatment significantly improved the PFS of patients when analyzed as a continuous variable in the multivariate analysis [Liu et al. 2012].
Hai-Lun and colleagues reported significantly lower rate of recurrence in patients treated with tumor lysate-pulsed DCs co-cultured with CIK cells compared with controls (Table 1) [Zhan et al. 2012].
Tumor response
Tumor response was reported in one study investigating active immunotherapy and four studies investigating passive immunotherapy [Osband et al. 1990; Law et al. 1995; Amato et al. 2010; Liu et al. 2012; Zhang et al. 2013]. Employing a random effects model, our meta-analysis showed that overall, there was not any significant difference in tumor response between patients in a specific immunotherapy group and the control group (RR = 1.14; 95% CI = 0.91–1.42, p = 0.25) (Figure 4); however, subgroup meta-analysis showed that patients in the passive specific immunotherapy group had higher rates of tumor response compared with the control group (RR = 1.25; 95% CI = 1.03–1.52, p = 0.02).
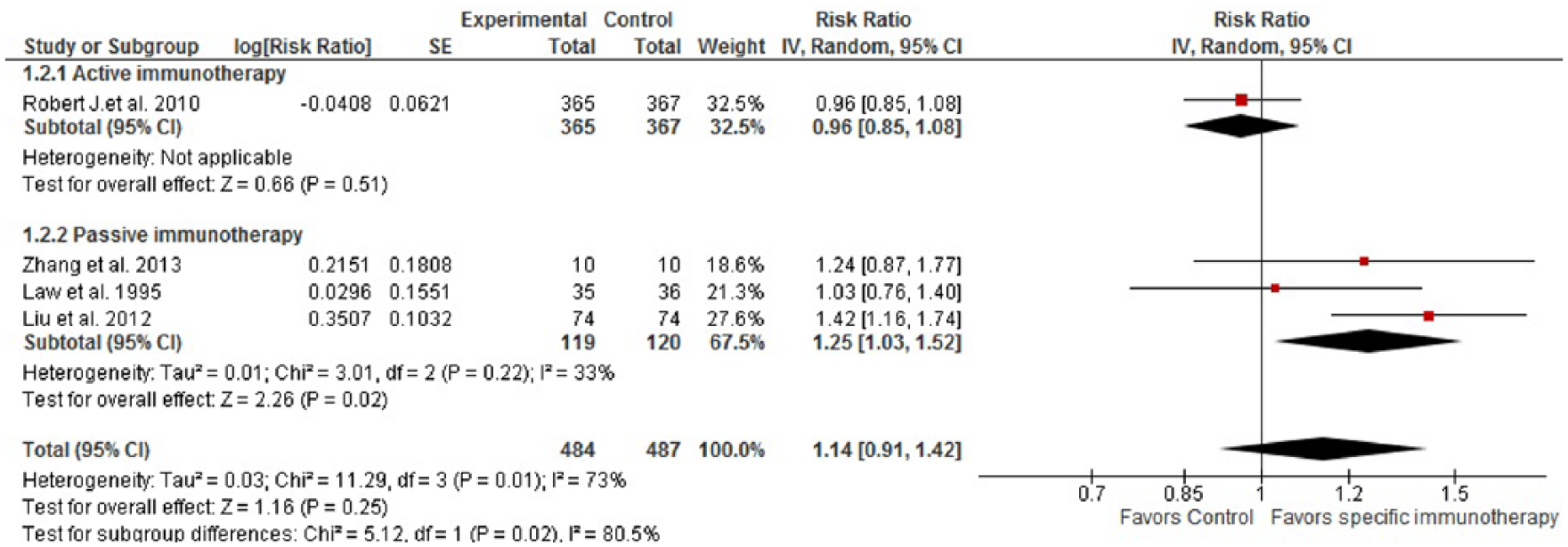
Comparison of tumor response between patients in specific immunotherapy group and control group.
Safety
Overall, 11 studies reported safety parameters in patients undergoing specific immunotherapy. Overall, specific immunotherapies were well tolerated and resulted in less serious adverse events compared with standard treatments (Table 1).
Discussion
In this systematic review, we reviewed and synthesized results of 14 articles reporting 10 controlled clinical trials and 4 studies with historical controls which evaluated the clinical efficacy and safety of specific immunotherapy in 4013 patients with RCC.
Active immunotherapy
Overall, eight different approaches of active immunotherapy including five types of autologous tumor cell vaccines, one peptide-based vaccine, one virus-based vaccine and DC-based vaccines were studied in 3547 patients in nine controlled studies.
Autologous tumor cell vaccines induces a cytotoxic T-lymphocyte (CTL) response toward tumor associated antigens (TAAs) expressed by RCCs themselves [Fishman and Seigne, 2002; Jocham et al. 2004; van Poppel et al. 2009; May et al. 2010]. A number of approaches have been used to enhance the immunogenicity of these vaccines. Reniale® is an autologous tumor cell vaccine prepared by incubation of tumor cell suspension with IFN-γ [Jocham et al. 2004; Doehn et al. 2009; May et al. 2010]. Incubation with IFN-γ leads to increased expression of major histocompatibility complex (MHC) class I and II, intercellular adhesion molecule 1, transporter associated with antigen processing in tumor cell suspensions [Jocham et al. 2004; Doehn et al. 2009; May et al. 2010]. May and colleagues and Jocham and colleagues reported beneficial effect of Reniale® on OS and PFS [Jocham et al. 2004; May et al. 2010].
Another strategy is combining the autologous tumor cell vaccine with a nonspecific component as adjuvant to augment CTL responses [Fishman and Seigne, 2002; Chi et al. 2010]. Tallberg and colleagues treated patients with advanced RCC with an autologous tumor cell vaccine in the form of insolubilized polymer particles mixed with tuberculin or Candida albicans antigen, as adjuvants which resulted in improved OS [Tallberg and Tykkä, 1986]. Pomer and colleagues used irradiated autologous tumor cells infected with Newcastle disease virus (NDV) as an adjuvant which was beneficial in terms of OS [Pomer et al. 1995].
Another approach to enhance the immunogenicity of autologous tumor cell vaccines is gene-modified tumor vaccines [Cayeux et al. 1997; Fishman and Seigne, 2002; Raman and Vaena, 2015]. Pizza and colleagues reported beneficial effect for an allogeneic irradiated RCC cell line, engineered to produce IL-2 (ACHN-IL-2) in combination with autologous metastatic formalin-treated tumor cells [Pizza et al. 2004]. The local IL-2 produced by ACHN-IL-2 can induce inflammatory responses and activate T-cells and natural killer (NK) cells against tumors [Fishman and Seigne, 2002; van Poppel et al. 2009; Chi et al. 2010].
Wood and colleagues investigated vitespen (HSPPC-96) which is a heat-shock protein (glycoprotein 96) peptide complex derived from autologous tumors [Wood et al. 2008; van Poppel et al. 2009]. This peptide-based vaccine acts as an individualized vaccine by capturing the antigenic fingerprint of a patient’s specific cancer and induces MHC class I-restricted immune responses [Doehn and Jocham, 2008; Yang, 2008; Wood and Mulders, 2009].
Amato and colleagues treated patients with clear cell RCC with a virus-based vaccine, a modified vaccinia virus Ankara vector engineered to express the oncofetal antigen 5T4 (MVA-5T4; TroVax®) [Amato et al. 2010]. The 5T4 antigen is expressed at high levels on the cell surface of both primary and metastatic tumor cells in RCC which makes it a potential target for both T-cell and antibody-mediated effector responses [Amato et al. 2010; Kim et al. 2010; Harrop et al. 2012; Zhang et al. 2012].
DCs as the most potent antigen-presenting cells can present TAAs effectively to both CD4+ and CD8+ T-cells and consequently induce both CTL and humoral responses against malignant cells [Fishman and Seigne, 2002; van Poppel et al. 2009; Zhan et al. 2012; Isaak et al. 2015; Raman and Vaena, 2015]. However, Isaak and colleagues reported that DCs pulsed with either human telomerase reverse transcriptase peptide or allogeneic or autologous tumor lysate were not beneficial in treatment of patients with metastatic RCC (mRCC) [Isaak et al. 2015].
In our systematic review we detected a trend in improved OS and PFS for active immunotherapies, but these effects were not statistically significant. However, Reniale®, ACHN-IL-2 admixed with autologous metastatic formalin-treated tumor cells, autologous tumor cell vaccine (insolubilized polymer particles) and NDV virus-infected autologous tumor cells had a significant beneficial effect on OS. It is of note that DC-based vaccines had a significant negative effect on OS. Efficacy of active immunotherapy, especially DC-based vaccines relies on activation and interaction of various elements of the immune system and requires a healthy immune system to achieve therapeutic efficacy [Stadler, 2000; Fishman and Seigne, 2002; van Poppel et al. 2009; George et al. 2011; Raman and Vaena, 2015]. On the other hand, patients with RCC are frequently characterized by a state of ‘immune dysfunction’ due to the immunosuppressive effects of RCC [Fishman and Seigne, 2002; Amato et al. 2010; Chi et al. 2010; George et al. 2011].
Passive immunotherapy
Overall, three passive immunotherapies including autologous CIK cells, ALT and LAK cells were studied in 329 patients in four controlled studies.
Liu and colleagues and Zhang and colleagues both reported improved OS for patients treated with CIK cells [Liu et al. 2012; Zhang et al. 2013]. CIK cells, a novel and heterogeneous population of immune effector cells, are activated T-cells with NK cell properties with MHC unrestricted cytotoxicity toward malignant cells [Liu et al. 2012; Zhan et al. 2012; Zhang et al. 2013; Zhao et al. 2015].
In ALT, autologous lymphocytes get depleted of suppressor cells and then become activated in an autologous lymphokine mixture and are transfused back to the patient [Osband et al. 1990; George et al. 2011; Bedke et al. 2014; Zang et al. 2014]. Osband and colleagues reported that ALT had positive outcome on OS of patients with mRCC [Osband et al. 1990].
Law and colleagues investigated LAK cells in the treatment of patients with mRCC [Law et al. 1995]. LAK cells are a heterogeneous population of cells consisting primarily of NK cells and T-cells. NK cells within LAK are more cytotoxic against malignant cells especially NK-resistant tumor cells [West et al. 2011; Galluzzi et al. 2012; Bedke et al. 2014].
Our systematic review showed that overall passive immunotherapy has a significant beneficial effect on OS but not on PFS.
Unlike active immunotherapeutic approaches, passive immunotherapies directly provide antitumor components for the immune system and therefore, their success depends on the amount of immune components administered to the body. Consequently, dose and pharmacokinetics are important factors in the success of passive immunotherapies. It can be speculated that in some of the studies of passive immunotherapies that yielded negative results, dosage and frequency of administration of passive immunotherapies might have been inappropriate.
Limitations
This meta-analysis had some limitations. First, there was considerable clinical heterogeneity in designs, interventions and populations of studies included in this systematic review. To overcome this limitation, we rated quality of studies and employed subgroup meta-analysis and a random effects model. Second, some outcomes in some studies were not reported properly so we could not include them in our meta-analysis. We tried to address this in the quality rating of studies under selective reporting domains. Third, the risk of bias was high in some studies and in several papers and enough information was not provided to allow judgment of bias in particular domains. Since the number of included studies was too few for reported outcomes (<10), we could not assess publication bias using a funnel plot.
Conclusion
In conclusion, results of this systematic review based on the available literature suggest that some specific immunotherapies such as Reniale®, ACHN-IL-2, NDV virus-infected autologous tumor cells, ALT and CIK treatment can improve clinical outcomes of patients with RCC. Our result suggests that further larger studies with higher power are required to evaluate long-term as well as short-term outcomes of specific immunotherapy in patients with RCC. In addition, the advent of other immunotherapies such as checkpoint inhibitors has opened new windows for the treatment of RCC. Therefore, further systematic reviews are needed to evaluate clinical efficacy of nonspecific immunotherapies such as check-point inhibitors to evaluate their potential value for treatment of RCC.
Footnotes
Acknowledgements
This study was approved by the research ethics committee of Tehran University of Medical Sciences, Iran.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
