Abstract
Background:
Urethral strictures or fistulas are common complications after phalloplasty. Neourethral defects pose a difficult reconstructive challenge using standard techniques as there is generally insufficient ventral tissue to support a graft urethroplasty. We report our experience with local fasciocutaneous flaps for support of ventrally-placed buccal mucosal grafts (BMGs) in phalloplasty.
Methods:
A retrospective review of patients who underwent phalloplasty and subsequently required revision urethroplasty using BMGs between 2011 and 2015 was completed. Techniques, complications, additional procedures, and outcomes were examined.
Results:
A total of three patients previously underwent phalloplasty with sensate radial forearm free flaps (RFFFs): two female-to-male (FTM) gender reassignment, and one oncologic penectomy. Mean age at revision urethroplasty was 41 years (range 31–47). Indications for surgery were: one meatal stenosis, four urethral strictures (mean length 3.6 ± 2.9 cm), and two urethrocutaneous fistulas. The urethral anastomosis at the base of the neophallus was the predominant location for complications: 3/4 strictures, and 2/2 fistulas. Medial thigh (2) or scrotal (1) fasciocutaneous flaps were used to support the BMG for urethroplasty. One stricture recurrence at 3 years required single-stage ventral BMG urethroplasty supported by a gracilis musculocutaneous flap. All patients were able to void from standing at mean follow up of 8.7 months (range 6–13). A total of two patients (66%) subsequently had successful placement of a penile prosthesis.
Conclusions:
Our early results indicate that local or regional fasciocutaneous flaps enable ventral placement of BMGs for revision urethroplasty after phalloplasty.
Introduction
Phalloplasty, or the creation of a neophallus, can be undertaken for congenital anomalies such as aphallia or micropenis, but more often, is performed following penile trauma, oncologic penectomy, or during gender-confirming surgery for female-to-male (FTM) transgender patients. These procedures are technically difficult and require consideration of patient goals including satisfactory appearance, tactile and erogenous sensation, ability to engage in penetrative sexual intercourse and voiding in a standing position [Wroblewski et al. 2013].
To create an anatomically acceptable sensate neophallus, the most common techniques utilize innervated microvascular free tissue transfer, usually from the forearm [Gilbert et al. 1995; Kim et al. 2009; Wroblewski et al. 2013; Morrison et al. 2015; Morrison, 2016]. Free flap phalloplasty has been described as a one-stage, two-stage or multi-staged procedure [Meyer et al. 1986; Sohn et al. 1991; Gilbert et al. 1995; Fang et al. 1999; Kim et al. 2009]. A tube-in-tube design from a fasciocutaneous free flap is generally used to create the neourethra in conjunction with the neophallus. Prelaminating a free flap with mucosa prior to harvest can also create a neourethra [Chang and Hwang, 1984; Schaff and Papadopulos, 2009]. Once the patient is able to void independently and there has been interval development of sensation, an inflatable or malleable penile prosthesis can be inserted to allow for penetrative sexual intercourse [Pariser et al. 2015b].
Regardless of differences in timing or technique, urethral complications including fistulas and strictures are common. Urethral fistulas or strictures have been reported in up to 58% of phalloplasties, but generally occur in <30% [Rohrmann and Jakse, 2003; Morrison, 2016]. Conservative measures including local wound care and serial urethral dilatation usually are insufficient and surgical repair is often required [Levine and Elterman, 1998]. Reconstructing these defects can be challenging, and a satisfactory result may not be achievable using standard urologic techniques. Due to insufficient ventral tissue to support a graft (generally buccal mucosa) on the neophalluses, alternative reconstructive techniques are necessary to enable adequate repair. Here we present a novel approach to neophallus revision urethroplasty using fasciocutaneous flap reinforcement of buccal mucosal grafts (BMGs) and report our early results.
Methods
Patients
Our institutional review board approved this study (S15-00615). A retrospective review identified patients who previously had phalloplasty then subsequently underwent revision urethroplasty using BMG at New York University Langone Medical Center, NY, USA between May 2011 and August 2015. Both electronic medical records and office charts were reviewed, and data was collected on patient demographics, techniques, complications, requirement of additional procedures, and outcomes. Patients who had phalloplasty performed by our surgical team and those referred to our institution for revision procedures were included. Patients were excluded if they underwent a separate procedure for revision urethroplasty or were not deemed candidates for revision urethroplasty. Conservative techniques for management were initially trialed prior to surgical consideration. For the context of this work, reported complications were Clavien grade III, or those requiring surgical intervention, or higher [Dindo et al. 2004].
Statistics and follow up
Data collected were not sufficient for univariate or multivariate statistical analysis, so all data are presented as descriptive. Data from follow-up appointments were aggregated and included in the descriptive statistics of the patients.
Operative technique
Prior to manipulation of the neophallus, a suprapubic catheter was placed for urinary diversion in all patients. A handheld Doppler was then used to locate the vascular pedicle of the neophallus and trace its course within the flap to ensure preservation. Prior incisions at the base of the penis were used for access and extended cranially into the medial and native urethral regions. Once the urethra was identified, urethroscopy assessed for stricture formation. If the stricture was distal and the length was 1.5 cm or less, it was then evaluated for possible Heineke–Mikulicz repair [Lumen et al. 2010]. However, all cases ultimately required formal urethral excision with BMG urethroplasty (Figure 1a). BMGs were harvested in the standard fashion, as previously described [Zhao et al. 2015]. The oral mucosa donor site was either closed primarily or, more commonly, closure was augmented with an acellular dermal matrix (ADM). Depending on the neourethral defect, either a partial or completely circumferential BMG was used. For circumferential urethral defects, a dorsal graft was inset to create the urethral plate (Figure 1b). Following, a ventral graft was sutured to the lateral edges of the dorsal graft, and secured to a ventrally-placed fasciocutaneous flap from either the scrotum or medial thigh (Figure 1c). Besides supporting the buccal graft for urethroplasty, the fasciocutaneous flap served to fill dead space, and allowed for closure without undue tension (Figure 1d).
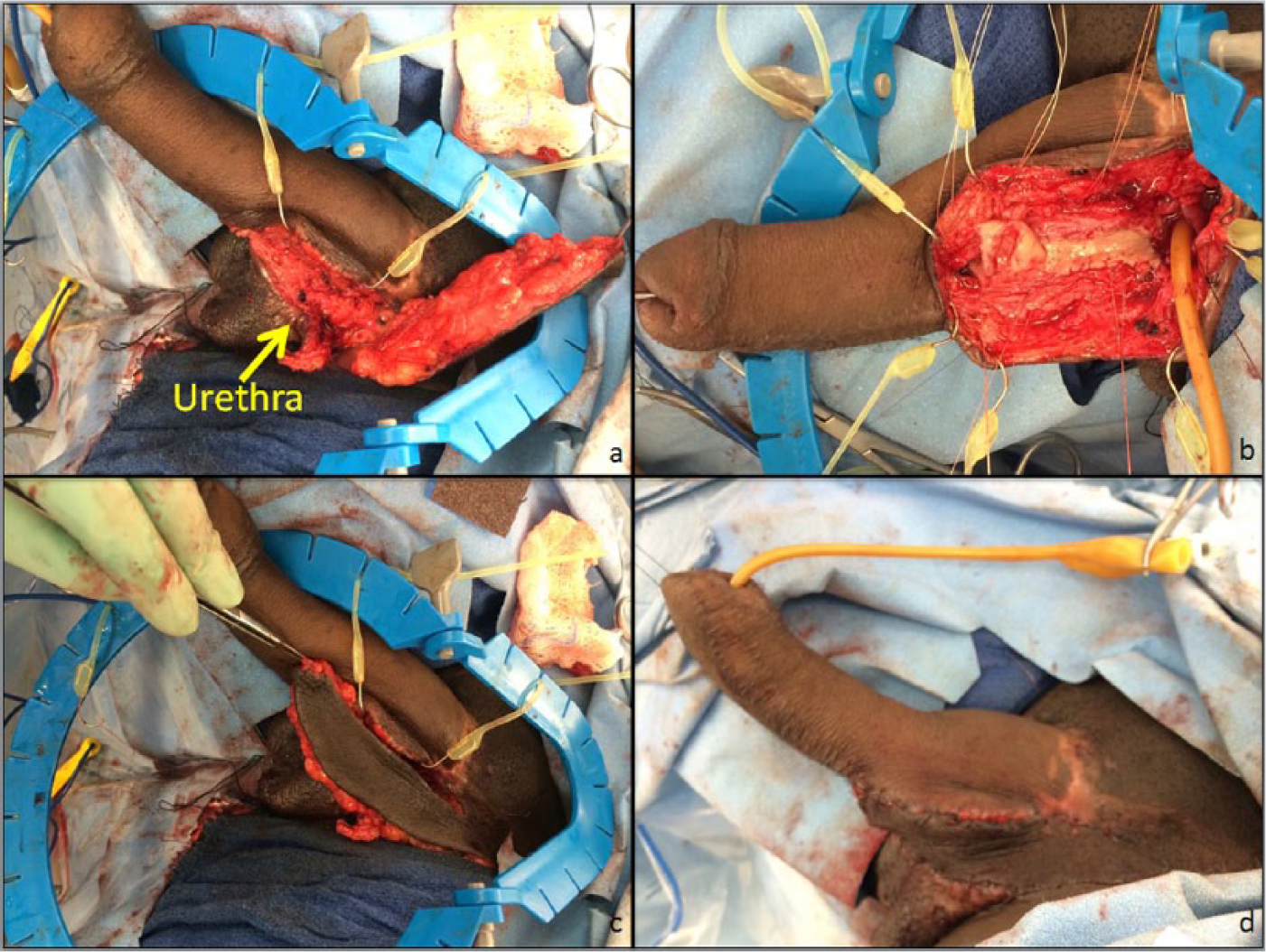
Depending on the neourethral defect (Figure 1a), either a partial or completely circumferential BMG was used. For circumferential urethral defects, a dorsal graft was inset to create the urethral plate (Figure 1b). Following, a ventral graft was sutured to the lateral edges of the dorsal graft, and secured to a ventrally-placed fasciocutaneous flap from either the scrotum or medial thigh (Figure 1c). Besides supporting the buccal graft for urethroplasty, the fasciocutaneous flap served to fill dead space, and allowed for closure without undue tension (Figure 1d).
For the urethrocutaneous fistula present at the base in patient three, a rotational scrotal fasciocutaneous flap was designed as it provided adequately vascularized tissue for interposition after standard fistula repair. In the remaining two patients, fasciocutaneous medial thigh propeller flaps were designed. Using a freestyle technique, Doppler signals were localized in the medial thigh overlying the adductor muscles and marked out appropriately. A propeller flap (flap base of 4–5 cm) was then designed to cover the defect with its point of rotation centered over the perforator signal. Starting from the distal end of the flap, dissection was taken down through the subcutaneous tissue to the level of the fascia. The distal flap was elevated suprafascially, and then transitioned to a subfascial plane in the middle aspect of the flap and continued in this plane up to the flap base. Following elevation, the Doppler signal was again reassessed prior to and after rotation of the flap. The bridging medial skin between the flap base and the defect was opened and local soft tissue flaps were elevated to allow for flap inset over the urethral repair. The medial thigh donor site was closed primarily.
Results
Overall, three patients with a history of phalloplasty who subsequently underwent revision BMG urethroplasty with fasciocutaneous flap reinforcement were identified (Table 1). All three cases involved sensate radial forearm free flaps (RFFFs). A total of two of the three patients were FTM transsexuals, and had their initial operation for gender dysphoria at an outside institution. The third patient had primary reconstruction with our team following penectomy for penile squamous cell carcinoma.
Outcomes of revisional urethroplasty using fasciocutaneous flap reinforcement of BMGs.

ADM, acellular dermal matrix; BMG, buccal mucosal graft; FTM, female-to-male; RFFF, radial forearm free flap.
The mean age at initial phalloplasty was 41 years (range: 31–47 years). In all three patients, the initial complication requiring intervention occurred at the base of the neophallus. Both FTM transsexual patients were complicated by urethral stricture involving the base while the reconstructive patient in our series presented with a urethrocutaneous fistula. The indications for revision urethroplasty in these patients included a single meatal stenosis, four urethral strictures (mean length 3.6 ± 2.9 cm), and two urethrocutaneous fistulas. The neophallic base was the predominant complication location: 75% (3/4) of strictures and 100% (2/2) of fistulas (Table 1).
The choice of fasciocutaneous flap design varied slightly in our study population. Specifically, two patients had medial thigh propeller fasciocutaneous flaps that ranged from 14–20 cm in length and 4–5 cm in width., and one patient had a much smaller local fasciocutaneous flap designed from the scrotal tissue (2 cm × 1 cm). The latter ultimately recurred at the site of repair 37 months later and was repaired with buccal mucosal flap reinforced with a gracilis musculocutaneous flap. To date, all three patients in this series are able to void while standing with a mean follow up of 8.7 months (range: 6–13 months) after revision urethroplasty. A total of two patients (66%) subsequently underwent penile prosthesis placement without complication (Table 1).
BMGs were harvested for the primary phalloplasty in all three patients. Our group revisited this donor site a total of five times for the three patients in this series. Overall, three of the four buccal mucosal harvests used ADM to close the donor site (75%). No oral donor site morbidity was noted.
Discussion
Recalcitrant strictures, and subsequent urethrocutaneous fistulas often complicate total phallic reconstruction. This is particularly true at the anastomotic site of the native urethra and the neourethra at the base of the neophallus. Initial stricture rate described in 1994 by Fang and colleagues were as low as 12.5% in a series of 56 patients [Fang et al. 1994]. However, multiple subsequent studies have rates as high as 58%, but generally <30%, for neourethral strictures and urethrocutaneous fistulas [Rohrmann and Jakse, 2003; Morrison, 2016]. Given this difficult and frequent clinical problem, recent efforts have aimed to determine the optimal salvage techniques in revisional urethroplasty following total phallic reconstruction.
It is postulated that neourethral strictures result from a relative ischemia of the urethral tissue due to the recipient bed for graft tissue relying on subsequent angiogenesis, and the high tendency for urethral grafts to kink due to gravitational forces on the neophallus [Levine and Elterman, 1998]. Pariser and colleagues described their experience with patients requiring revisional urethroplasty following phalloplasty. In their study, a 50% success (nonrecurrence) rate was determined using BMG alone with a median follow up of 9.5 months [Pariser et al. 2015a].
To reduce the risk of recurrent urethral strictures or urethrocutaneous fistulas, we believe the transfer of additional well-vascularized tissue is imperative to reinforce BMG urethroplasty. Improved outcomes were reported with the use of periurethral flaps to support ventrally-placed buccal mucosa grafts in urethroplasty of the native urethra [Cordon et al. 2014]. However, since a neophallus lacks periurethral dartos or spongiosum, an alternative source of tissue must be identified. Muscle flaps could be used to achieve this goal, but these flaps are bulky, can result in a functional donor site defect, and often require additional coverage with skin grafting at the recipient site. Fasciocutaneous flaps, on the other hand, provide thin pliable tissue with excellent vascularity, minimal donor site defect, and generally do not require skin grafting.
To our knowledge, this is the first report describing the use of fasciocutaneous flaps to reinforce traditional BMG urethroplasty in neophallus patients. For two patients in our study, we reinforced BMGs with medial thigh fasciocutaneous propeller flaps, which allowed us to bring in additional healthy tissue with a second distinct vascular supply to help support graft survival. Moreover, this flap is thin and pliable which is preferable on the shaft of the neophallus. Both patients who underwent BMG reinforcement with fasciocutaneous perforators flaps are able to void while standing with a mean follow up of 10 months. For our third patient, a smaller fasciocutaneous flap was used based off of native scrotal tissue. Over 6 years later, this patient had a stricture recurrence, and the decision was made to revise the urethra with a BMG and a medial thigh fasciocutaneous flap. However, due to inadequate perforators, we had to use a gracilis musculocutaneous flap instead to reinforce the graft. At a 6-month follow up, this patient is able to void while standing and reports improvement in urine stream.
It has been our experience that a significant number of phalloplasties are based more inferiorly than the origin of the natural male penis. In the transsexual population this is most likely related to the more inferior/posterior location of the clitoris and vagina in relation to the anterior pubic bone compared with the penis and testicles. This limits the ability to utilize ventral based flaps for coverage in revisional urethroplasty BMG placement. When fasciocutaneous flaps are used for BMG graft support they can offer unforeseen benefits to phallic outcomes including improved projection, increased length, and elevation of the neophallus superiorly into a more natural male position.
The literature continues to support BMG as an ideal graft for urethral reconstruction [Markiewicz et al. 2007; Andrich and Mundy, 2008]. In addition, BMG has been described in the literature for use in the primary neophallus creation and for secondary revisions following fistulae or strictures [Levine and Elterman, 1998; Rohrmann and Jakse, 2003; Pariser et al. 2015a]. One study described outcomes of nine urethroplasties using split thickness skin grafts, full thickness skin grafts, vaginal mucosa, transitional epithelium from the bladder, and BMGs [Levine and Elterman, 1998]. There were no complications in the patients who received BMGs at mean follow up of 16 months but the remaining six patients who underwent other techniques had either failure leading to perineostomy (two cases of split thickness skin grafts) or restricture requiring additional procedures (three cases) [Levine and Elterman, 1998]. Thus, the authors concluded BMGs should be the first-line tissue for urethroplasty. Our cohort demonstrated similar results. In fact, we harvested BMGs for all urethral reconstructions despite the fact that all our patients had prior BMG grafts in their primary phalloplasty.
Oral donor site morbidity has been well documented. A randomized study by Wong and colleagues comparing BMG donor site primary closure and allowing healing by secondary intention found greater earlier postoperative pain with primary closure [Wong et al. 2014]. A separate randomized study showed evidence for less pain, earlier return to full diet, and earlier return to full mouth opening when the donor site was not closed [Rourke et al. 2012]. For the patients in our study, BMGs were harvested multiple times and thus it was decided to use an ADM to replace donor site mucosa in the majority of harvests. No patients in our study developed intraoral complications. Our experience was supported by Akyuz and colleagues where the majority of their patients had more urologic pain than oral pain, and over 95% of their patients stated that they could tolerate BMG harvest again [Akyuz et al. 2014].
Limitations of this study are related to generalizability and the small sample size of our cohort. Due to the positive outcomes that our patients experienced following placement of fasciocutaneous flaps over BMG grafts for revisional urethroplasty, it is felt that the data point to overall improved outcomes. Of course larger clinical studies are needed before these results can be generalized to all patients requiring revisional urethroplasty after phalloplasty.
Our study offers strong support for the use of adjunctive fasciocutaneous flaps to reinforce BMG urethroplasties. Despite our relatively small case series, our results have been promising with revision rates and complications lower than previous reports. When anatomically possible, we suggest that reconstructive surgeons should incorporate additional healthy tissue to help support BMG urethroplasty in complex urethral reconstructions. However, this is a retrospective review of a single surgical team’s experience in a small number of patients. A larger sample size and longer follow up will help elucidate the ultimate utility of this technique.
Conclusion
Urethral strictures and fistulas are frequent and challenging complications of total phalloplasty, often requiring surgical intervention due to the limited success of conservative management. Our study demonstrates successful neophallus revision urethroplasty using fasciocutaneous flap reinforcement of BMGs. Our early results indicate that this novel technique may be a viable option for revision urethroplasty reconstruction in patients with a neophallus.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
