Abstract
Clinical trials have shown that hexaminolevulinate (HAL) fluorescence cystoscopy improves the detection of bladder tumors compared with standard white-light cystoscopy, resulting in more efficacious treatment. However, some recent meta-analyses report controversially on recurrence-free rates with this procedure. A systematic review of literature was performed from December 2014 to January 2015 using the PubMed, Embase and Cochrane databases for controlled trials on photodynamic diagnosis (PDD) with HAL. A total of 154 publications were found up to January 2015. Three of the authors separately reviewed the records to evaluate eligibility and methodological quality of clinical trials. A total of 16 publications were considered eligible for analysis. HAL–PDD-guided cystoscopy increased overall tumor detection rate (proportion difference 19%, 95% confidence interval [CI] 0.152–0.236) although the benefit was particularly significant in patients with carcinoma in situ (CIS) lesion (proportion difference 15.7%, 95% CI 0.069–0.245) and was reduced in papillary lesions (Ta proportion difference 5.9%, 95% CI 0.014–0.103 and T1 proportion difference 1.2%, 95% CI 0.033–0.057). Moreover, there were 15% of patients (95% CI 0.098–0.211) with at least one additional tumor seen with PDD. With regard to recurrence rates, the data sample was insufficient for a statistical analysis, although the evaluation of raw data showed a trend in favor of HAL–PDD. This meta-analysis confirms the increased tumor detection rate by HAL–PDD with a most pronounced benefit for CIS lesion.
Introduction
Bladder cancer is the seventh most common cancer in men and in 2015 an estimated 468,351 cases will be diagnosed and 179,753 patients will die of the disease worldwide [Ferlay et al. 2012]. The majority of bladder cancers are nonmuscle invasive (NMIBC), including papillary lesions confined to the urothelium (stage Ta) or invading the lamina propria (stage T1), and carcinoma in situ (CIS; stage Tis) [Hall et al. 2007]. Although fatality is unlikely in NMIBC, the high recurrence rate up to 61% within 1 year and 78% within 5 years means significant morbidity for patients concerned, as does possible progression to muscle invasive disease in up to 17% at 1 year and up to 45% at 5 years [Sylvester et al. 2006]. The probability of recurrence and the unpredictability of progression places a substantial burden on patients and health care resources, as patients require long-term surveillance, additional therapies in the form of intravesical treatments with various agents, and surgery in the case of recurrence [Babjuk et al. 2013], making the lifetime costs of bladder cancer the highest of all malignant diseases [Svatek et al. 2014].
The high recurrence rate associated with NMIBC has been assigned to poor endoscopic detection and, thus, incomplete resection with standard white-light cystoscopy (WLC), especially in the presence of multifocal disease and/or carcinoma in situ [Herr et al. 2008]. As a matter of fact, although cystoscopy remains the gold standard to visualize recurrences, it may miss up to 10–30% of cancer recurrences [Svatek et al. 2005]. Tools to improve visualization of tumors at first trans-urethral resection may result in a more complete resection and thereby reduction of early recurrence rate.
Photo-dynamic diagnosis (PDD) is a method to detect neoplastic lesions by means of fluorescence [Stokes, 1852] that is caused by the interaction of light with specific molecules (fluorophores), which are naturally present in human tissues (endogenous fluorophores), or absorbed by human tissues after external administration (exogenous fluorophores) [Wagnieres et al. 1998].
Among clinicians, PDD with 5-aminolevulinic acid (ALA) has raised interest because of the preferential accumulation of protoporphyrin IX (PpIX) in neo/dysplastic tissues. This accumulation may be a consequence of dysfunctional heme biosynthesis, precisely a reduction in ferrochelatase enzyme activity, leading to increased cellular uptake of ALA, increased PPIX synthesis and/or reduced PPIX conversion [Peng et al. 1997; Miyake et al. 2009]. However, the low amount of 5-ALA that is internalized in target cells and the low tissue penetration of this drug seems to limit its diagnostic effectiveness and applicability. Since the lipid bilayer of biological membranes is relatively impermeable to charged molecules, the diffusion of intravesical 5-ALA is poor. Thus, more lipophilic 5-ALA derivatives have been explored to enhance bioavailability. Promising results were obtained with hexaminolevulinate acid (HAL, presently commercialized as Hexvix®), an alkyl ester of ALA, which induces a higher PpIX concentration and fluorescence twice as high as compared with 5-ALA [Gaullier et al. 1997]. After penetration into the cell, the ester derivative is hydrolyzed into 5-ALA by nonspecific esterases, leading to the formation of PpIX. As compared with 5-ALA, bladder instillation of esterified derivatives of ALA confer up to 25 times higher fluorescence [Marti et al. 2003].
To date, the use of HAL may be regarded the gold standard for bladder fluorescence cystoscopy, for its diagnostic efficiency with clinically manageable reduced intravesical exposure times of about 1 h [Klem et al. 2006].
Material and methods
Search strategy
A systematic literature search was initially performed from December 2014 to January 2015 using the PubMed, Embase and Cochrane databases for controlled trials on PDD with HAL. The search included only a ‘free-text’ protocol using the keywords: ‘hexaminolevulinate bladder cancer’ or ‘Hexvix’ or ‘hexaminolevulinate’ or ‘photodynamic diagnosis bladder cancer’ across the ‘Title’ and ‘Abstract’ fields. A language limit was used selecting English as the default.
Three of the authors separately reviewed the records to select the studies comparing WLC with blue-light cystoscopy (BLC) using PDD equipment. Studies published only as abstracts and reports from meetings were not included in the review. Discrepancies were resolved by open discussion. Other significant studies cited in the reference lists of the selected papers were evaluated, as were studies published after the systematic search.
Study eligibility
A study was considered eligible to this meta-analysis if it assessed the following: a patient group treated for NMIBC, comparative assessment of transurethral resection of the bladder (TURB) with WLC compared with PDD exclusively with HAL as a photosensitizer, and analysis of tumor detection (evaluated as per lesion or per patient detection) and/or recurrence rates.
Defined end points
The endpoints were: tumor detection rates both at a lesion and patient level, and discrimination between papillary (Ta and T1) and flat lesion (CIS); recurrence rates at 3, 6 and 12 months and false positive detection rates as secondary end point. All data retrieved from the selected studies were recorded in an electronic database.
Statistical analysis
Meta-analysis was carried out by random effects model with proportion differences as outcome variables. Thus, pooled outcome estimates were calculated, accounting for the inter-study variance χ2 [Higgins et al. 2003]. The overall study heterogeneity was assessed using χ2 at p = 5%. Forest plots are provided to show the weight of each study in the overall analysis. The issue of publication bias has been taken into consideration with the asymmetry Begg and Mazumdar adjusted rank correlation test [Egger et al. 1997] accompanied by the corresponding funnel plots.
The statistical analysis was performed using the statistical software STATA version 13.
Limitations of the study
The lack of standard data collection methods among different papers makes it difficult to obtain undisputable conclusions. Some subgroups were too small for statistical analysis.
Results
The literature searches identified 154 publications up to January 2015; reviewers excluded 130 of these on the basis of title or abstracts; 8 were rejected because they did not conform to inclusion criteria. A total of 16 publications were considered eligible for analysis. Among the 16 evaluated papers, seven were unicentric and nine multicentric trials; eight studies were designed as prospective randomized trial and seven as prospective within-patient comparison; one more was an observational comparative controlled trial.
The meta-analysis provided the funnel plot associated to the difference of lesions detected using BLC and WLC (Figure 1). The graph does not involve particular concern about publication bias. The forest plot referred to lesions’ detection displays the observed heterogeneity of the sample of studies which have been selected for the meta-analysis (Figure 2).

Begg’s funnel plot for publication bias with the 95% confidence limits. Sample symmetry graphical assessment for the studies comparing the proportion of lesions detected using different lights.
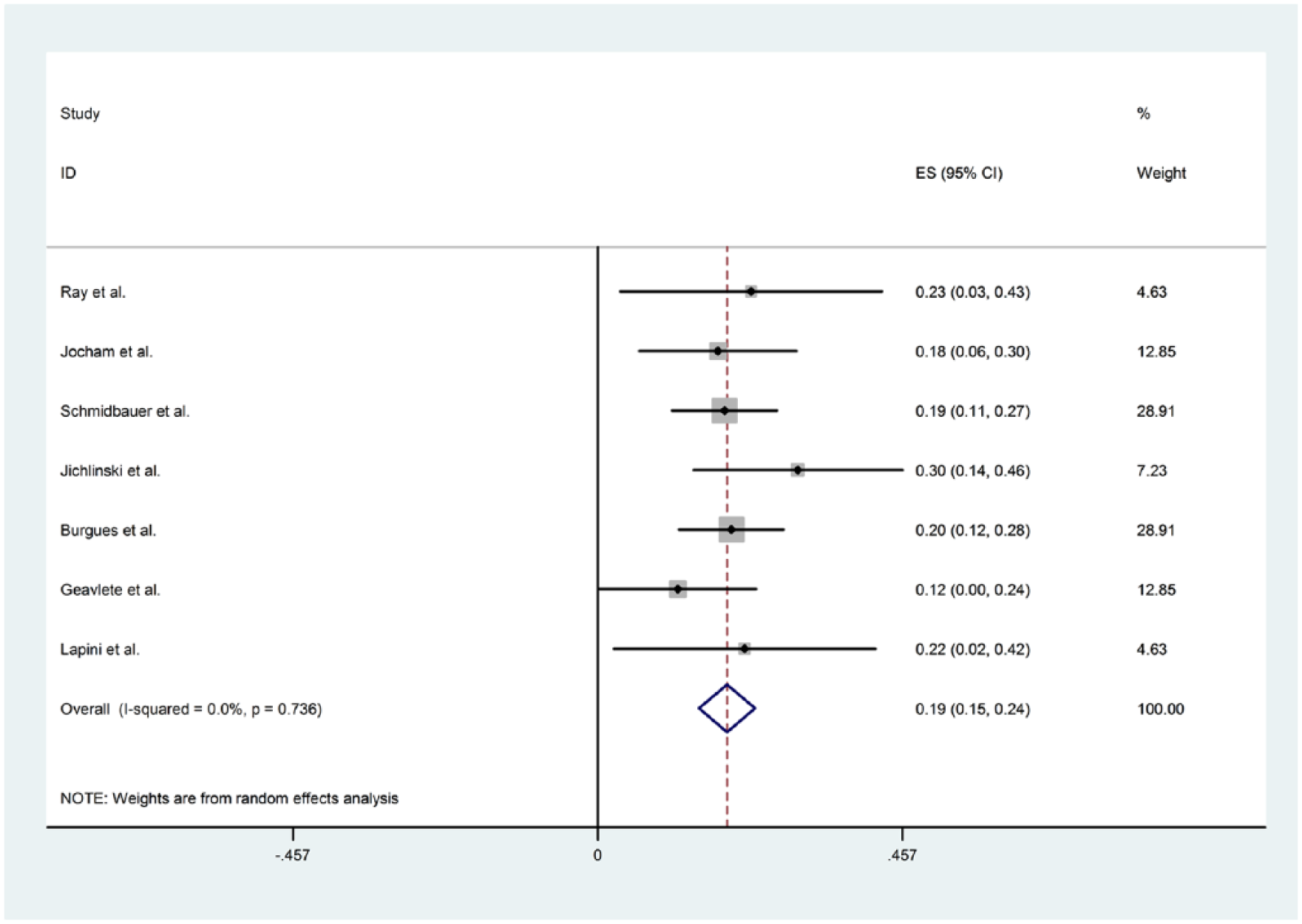
Forest plot for the graphical examination of the heterogeneity of the studies used to compare lesions’ detection using blue light and white light. The dashed line refers to the difference in the proportion of lesions detected using different lights.
All selected papers and their descriptive baseline characteristics are shown in Table 1, while overall results of meta-analysis are reported in Table 2.
Descriptive baseline characteristics of selected paper.

BL, blue light; WL, white light; D, detection; R, recurrence.
Meta-analysis summary results.

BL, blue light; WL, white light; Δ, difference; PD, proportion difference.
Tumor detection
The evaluation of tumor detection rates between different studies is difficult. This is due mainly to the lack of standard data collection methods: some studies reported only detection at patient level or at lesion level, some other have reported the overall detection or additional detection with BLC only.
We have subdivided results on tumor detection in two main groups: patient detection and lesion detection series. Additional subgroups were defined as: overall additional tumor detection rate with BLC and additional detection rates according to their histopathological finding.
The majority of the evaluated papers have shown the superiority of HAL–PDD-guided cystoscopy over WLC alone in tumor detection. Tables 3 and 4 show comparison of tumor detection rates between BLC and WLC in the selected trials.
Differential of patient detection rate for BL versus WL cystoscopy (at least one more lesion detected).
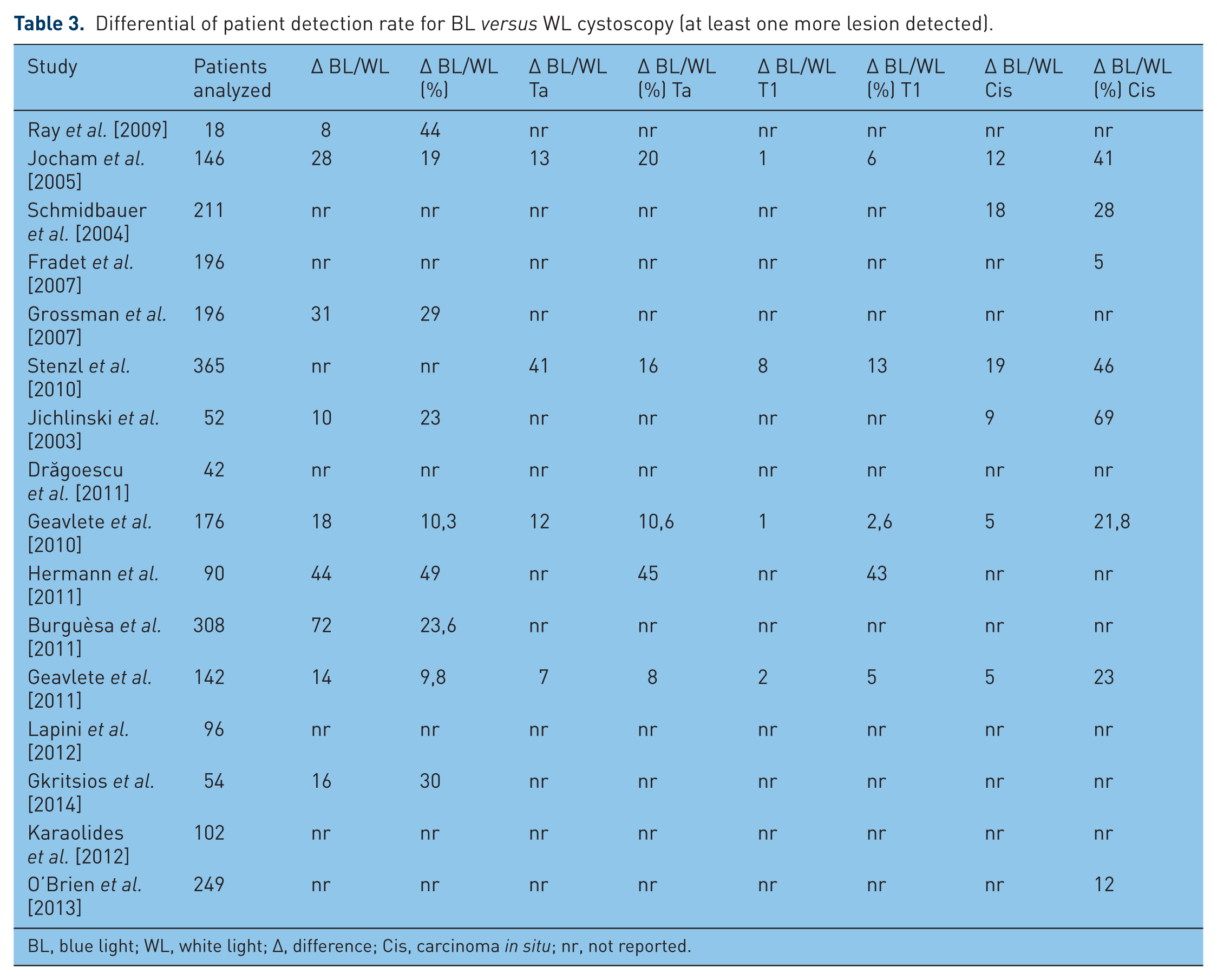
BL, blue light; WL, white light; Δ, difference; Cis, carcinoma in situ; nr, not reported.
Differential of lesion detection rate with BL and WL cystoscopy.

BL, blue light; WL, white light; Δ, difference; Cis, carcinoma in situ; nr, not reported.
Patient detection rates
Among the 16 evaluated papers, nine reported data on patient tumor detection as shown in Table 3. In 15% (95% confidence interval [CI] 0.098–0.211) of patients at least one additional tumor was identified only by BLC (overall data on Ta, T1 or CIS). This benefit was observed in patients with Ta tumors (in 4% of patients, 95% CI 0.020–0.066) and in patients with CIS/flat lesion (5.9%, 95% CI 0.021–0.096). Data on T1 tumor detection rates were insufficient to obtain a statistically significant estimated pool.
The benefit of BLC was particularly significant in patients with Tis lesion and was reduced in papillary lesions.
Lesion detection rate
Lesion detection rates of selected studies are reported in Table 4.
A total of 2313 lesions were detected in the patients evaluated (lesion detected by BLC and/or WLC).
Overall additional lesion detection rate was 19% (95% CI 0.152–0.236, Ta, T1 and Tis lesions).
Six studies reported additional Ta tumor detection rates with BLC, ranging from 9% to 42% of the total Ta tumors detected [Jocham et al. 2005; Schmidbauer et al. 2004; Grossman et al. 2007; Burguèsa et al. 2011; Geavlete et al. 2011; Lapini et al. 2012], with a proportion difference of 5.9% (95% CI 0.014–0.103) of additional lesion detection rates with BLC. The same studies reported additional T1 tumor detection rates ranging from 4.9% to 9% of the total T1 tumors detected [Schmidbauer et al. 2004; Fradet et al. 2007; Burguèsa et al. 2011; Geavlete et al. 2011; Lapini et al. 2012], only Jocham and colleagues showed no differences in T1 tumors detection rates [Jocham et al. 2005]. Proportion difference for T1 tumors was 1.2% (95% CI −0.033 to −0.057).
The detection of Tis lesions was reported in six studies [Jocham et al. 2005; Schmidbauer et al. 2004; Fradet et al. 2007; Burguèsa et al. 2011; Geavlete et al. 2011; Lapini et al. 2012] showing greater additional detection rates with BLC, ranging from 24% to 94% of the total Tis lesions detected. Overall additional Tis lesions detection rate was 15.7% (95% CI 0.069–0.245).
Recurrence rates
Only seven papers have reported data on recurrence rates in patients treated with BLC and WLC. Available recurrence rates are summarized in Table 5.
Recurrence rate for BL and WL cystoscopy.

BL, blue light; WL, white light; Δ, difference; nr, not reported.
Recurrence was used to evaluate if an improvement in tumor detection could reduce recurrence rates up to 12 months. We have considered the recurrence rate as the number of patients with a recurrence at 3, 6 and 12 months divided by the total number of patients analyzed.
With this definition we have obtained a mean overall recurrence rate of 28.9% and 44.2% for BLC and WLC, respectively. In addition, we have found a recurrence rate of 10.2% versus 18.1% at 3 months, a recurrence rate of 10.5% versus 22.3% at 6 months and 14.8% versus 32.3% at 12 months for BLC and WLC. Unfortunately, the data sample was insufficient for a statistical analysis, however, the evaluation of raw data showed a trend in favor of BLC.
False positive detection rates
The false positive detection rate is the number of suspicious lesions that had negative histology divided by the total number of areas biopsied with each technique.
A T test on the pooled percentage values, weighted on the number of lesions detected in each study of the meta analysis, of the white light false positive and blue light false positive was statistically significant (p < 0.05), getting pooled percentage estimates of 15.65 (SD = 7.06) for the white light and 22.35 (SD = 9.24) for the blue light.
We found a large variation in the false positive detection rate among centers for BLC and WLC (range 7–86% and 3–81%, respectively) as shown in Table 6. This may be explained by a learning curve with the fluorescence technique.
Comparison of false positive detection rates between WL and BL cystoscopy.

BL, blue light; WL, white light.
Discussion
Since the first report on PDD by Kriegmair and colleagues in 1994 [Kriegmair et al. 1994], many expectations with regard to improved diagnosis of bladder tumors have been met. Most of the initial reports on the clinical use of PDD in NMIBC showed that the tumor detection rate was increased at about 20% by this new method, and that recurrence, i.e. persister, rates in most cases were reduced on follow-up cystoscopy in the same amount [Dindyal et al. 2008; Mowatt et al. 2011]. It was also concluded from these findings that a consistent reduction of bladder tumor recurrences would help to significantly reduce the costs for bladder cancer treatment [Dindyal et al. 2008].
After two decades of PDD, reevaluation of its efficacy in the therapy of NMIBC can shed light on the present position of fluorescence endoscopy in global treatment concepts. The results of the present meta-analysis confirm the increased tumor detection rate by BLC in about 20%. The most pronounced benefit was demonstrable for CIS, with a superior detection rate ranging from 24% to 94% compared with WLC. This has been reported by other investigators before [Isfoss, 2011; Kausch et al. 2010]. The benefit for papillary lesions was also demonstrable, but minor compared with CIS: it was 9–42% for pTa tumors and 0–9% for pT1 tumors. These findings seem to be in close line with clinical experience: while flat lesions are difficult to detect or discriminate from benign morphologic changes of the urothelium, exophytic tumors are endoscopically more identifiable, except when they are very tiny or multilocular. In these cases, when single small lesions may be missed, PDD is a valuable tool for diagnosis and prevention of tumor persistence.
Although not statistically significant, the improved tumor detection rate with BLC was well reflected by a decrease of tumor recurrences at 3, 6 and 12 months in the present meta-analysis. The difference was 8%, 12% and 17.5%, respectively. This may be the most relevant finding, since it definitely reduces the number of consecutive operations and the surgery-related morbidity for patients and the costs for the health care system.
While expert panels recommend the use of PDD for bladder tumor resection as well in the initial as in the follow-up situation [Malmström et al. 2012; Witjes et al. 2010], this method has not been integrated in the EAU guidelines yet [Babjuk et al. 2013]. The main reason, besides single studies that failed to demonstrate a significant benefit in recurrence rates with BLC, may be that tumor progression rates have not shown any reduction compared with standard WLC resection. While pTa tumors rarely progress to muscle-invasive disease, and pT1 tumors are also detectable with WLC in most cases, there is a broad consensus that CIS with its potential for stage progression is a domain of BLC at present. Thus, it is surprising, that progression rates have not been markedly improved in the publications on BLC hitherto.
What is the future of PDD? Throughout the last years, novel imaging technologies have been introduced in bladder cancer diagnosis [Lerner et al. 2015]: narrow-band imaging (NBI), optical coherence tomography (OCT) and confocal laser endoscopy (CLE) have the potential to detect bladder tumors beyond the borders of macroscopic visibility. A meta-analysis for NBI showed improved tumor detection rates similar to PDD at about 20% [Li et al. 2013]. Data on improvement and accuracy of tumor detection by OCT and CLE are still lacking [Liu et al. 2012]. These technologies may be used alone or in combination with PDD [Gladkova et al. 2013]. However, they will have to prove equivalence or superiority to PDD, not only in tumor detection rates, but also in recurrence and, possibly, progression rates, as well as cost efficiency, before they may be accepted for clinical routine.
From a patient’s and urologist’s perspective, there is no argument not to use the best available tools to visualize bladder tumors during resection, as no surgeon would perform surgery without his glasses. Even if the benefit of PDD is restricted to only part of the patients, cost efficacy in the course of disease is a compelling argument for the use of fluorescence endoscopy for bladder tumor resection.
Several new photosensitizing agents and imaging technologies have been developed for improved visibility and detectability of bladder cancer.
Hypericin is a photosensitizer with promising applications in photodynamic diagnosis for bladder cancer, and can be used with the same imaging system that is used for the porphyrin-related substrates [Vandepitte et al. 2011].
Technologic improvements may further enhance our ability to detect and stage bladder tumors and distinguish benign from malignant disease [Lerner et al. 2015; Zlatev et al. 2015]: NBI is a macroscopic imaging modality that improves WLC by providing increased contrast between normal and abnormal tissue on the basis of neovascularity. NBI cystoscopy has been shown to improve detection rates of bladder tumors [Cauberg et al. 2010], and is associated with lower recurrence rates and longer recurrence-free survival (RFS) time [Herr et al. 2011] than conventional WLC. A single-surgeon randomized study comparing 2-year RFS of patients with NMIBC using NBI or WLC showed that NBI cystoscopy improves completeness of bladder tumor resection, which reduces the frequency of early and late tumor recurrences [Herr, 2015].
OCT, a real-time and high-resolution imaging technology, delineates subsurface microarchitecture information from bladder lesions. It has the ability to discriminate between benign or malignant lesions [Manyak et al. 2005] and noninvasive and invasive cancers [Goh et al. 2008].
Molecular imaging associates optical imaging technologies with cancer-specific molecular agents to improve the specificity of disease detection [Lerner et al. 2015; Zlatev et al. 2015]. CLE, as OCT, allows in situ tissue characterization with high resolution [Sonn et al. 2009].
The combination of these macroscopic and microscopic visualization techniques has the ability for significant improvement of bladder tumor detection and in situ histological characterization [Lerner et al. 2015].
Additional imaging techniques that currently under development, such as endoluminal high-frequency ultrasound [Yuan et al. 2008], time- and spectral-resolved two-photon imaging [Cicchi et al. 2010] and coherent anti-Stokes Raman scattering microscopy [Gao et al. 2011], might also become procedures for bladder tumor detection, diagnosis and staging in future.
To improve the results in NMIBC primary objectives are to have more accurate diagnostics and more effective therapies. In a more favorable economic conditions, such as the one before the financial collapse of 2008, it should make available to every department of urology the best diagnostic technology (i.e. fluorescence cystoscopy) and the best therapeutic technologies (i.e. intravesical electro-osmotic mitomycin [Di Stasi et al. 1999, 2006, 2011]).
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
