Abstract
Immunostimulatory therapies have been a cornerstone of treatment for metastatic renal cell carcinoma (RCC) since the 1990s. However, the use of traditional immunotherapeutic approaches for RCC, such as high-dose interleukin-2 and interferon-α, has been limited by significant systemic toxicities and the need to deliver these therapies at centers of expertise. Furthermore, in spite of the success of these immunostimulatory therapies for some patients with RCC, it is clear that most patients fail to respond to cytokine therapy. More effective immune therapy for RCC has therefore been necessary. The interaction between programmed death-1 (PD-1, present on T cells), and one of its ligands (PD-L1, present on antigen-presenting cells and tumor cells) constitutes an immune checkpoint through which tumors can induce T-cell tolerance and avoid immune destruction. Monoclonal antibodies that disrupt the PD-1/PD-L1 interaction serve as inhibitors of this immune checkpoint, and have demonstrated favorable activity in RCC as monotherapy and in combination with other active agents. This review summarizes the current landscape of anti-PD-1/PD-L1 therapy for RCC, and highlights challenges for the future development of this promising approach.
Introduction
Renal cell carcinomas (RCCs) cause a substantial burden of disease, with an estimated annual incidence of 63,920 new cases and 13,860 deaths in the United States in 2014 [SEER Stat Fact Sheet: Kidney and Renal Pelvis Cancer, 2014]. Several different histologic patterns of RCCs with different biologic characteristics and responses to therapies have been described. These subtypes include clear cell (70–80% of all RCCs), papillary (10–15% of all RCCs), and chromophobe (3–5% of all RCCs).
An enhanced understanding of the pathogenesis of clear cell RCC has led to the development of effective molecularly-targeted therapies. Inhibitors of the vascular endothelial growth factor (VEGF) and mammalian target of rapamycin (mTOR) pathways have demonstrated improvements in objective response rate and progression-free survival in the setting of metastatic RCC when compared with either placebo or interferon alpha [Yang et al. 2003; Motzer et al. 2007; Escudier et al. 2007a, b; Hudes et al. 2007; Motzer et al. 2008; Rini et al. 2011; Motzer et al. 2013]. Despite the successes of VEGF and mTOR-targeted therapies in improving clinical outcomes in metastatic RCC, resistance to tyrosine-kinase inhibitor treatment eventually develops in the vast majority of cases, and long-term disease-free intervals have not been observed with these agents following the discontinuation of treatment. Alternative therapeutic strategies for RCC are therefore necessary.
The observation that some patients with RCC experienced spontaneous regression, possibly due to an innate immune response [Fairlamb, 1981; Vogelzang et al. 1992], led to the exploration of immunotherapeutic strategies in this disease. The most clinically relevant of these treatments have been the immunostimulatory cytokines interferon-α and interleukin-2 (IL-2) [Minasian et al. 1993; Medical Research Council Renal Cancer Collaborators, 1999; Fyfe et al. 1995; Fisher et al. 1988; Negrier et al. 1998; Yang et al. 2003; McDermott et al. 2005; Negrier et al. 2007; Upton et al. 2005]. However, the significant systemic toxicities of high-dose IL-2 limit its use to patients with adequate cardiopulmonary, hepatic, and renal function, and dictate that it can only be administered in centers of expertise. Other immunotherapy strategies have therefore received significant investigational attention.
Checkpoint inhibitors
An enhanced understanding of the precise activators and inhibitors of the immune system has led to therapeutic advances in the treatment of various malignancies. T cells and antigen-presenting cells interact via the T-cell receptor (TCR) on T cells and major histocompatibility complex (MHC)/peptide antigen on antigen-presenting cells (Figure 1). This interaction is complex, and can either lead to T-cell activation against the target antigen or to the opposite effect of immune tolerance or anergy. A variety of costimulatory receptor–ligand interactions dictate which of these opposing outcomes will occur. These interactions form the basis of the ‘two signal hypothesis’ of immune effector cell activation [Bretscher, 1999], in which antigen recognition by the TCR-MHC/peptide complex (signal one) must be accompanied by activation or inhibition from costimulatory molecules (signal two) in order for its downstream effects to occur.

Interaction between the T cell and the tumor cell/antigen-presenting cell.
Among the best characterized of these co-stimulatory molecules is the B7-CD28 super-family [Sharpe and Freeman, 2002]. B7-1 (also known as CD80) and B7-2 (also known as CD86) are present on antigen-presenting cells or tumor cells (Figure 1). B7-1 and B7-2 have dual specificity for the immunostimulatory receptor CD28 and the immunoinhibitory receptor CTLA-4 (cytotoxic T-lymphocyte-associated antigen 4), which are both present on T cells. CTLA-4 functions to dampen the early activity of naïve and memory T cells [Topalian et al. 2012a]. The remarkable clinical success of therapeutic blockade of CTLA-4 with the monoclonal antibody ipilimumab in metastatic melanoma, another immunogenic cancer, was the proof of concept that interruption of an inhibitory signal two at this immune checkpoint can lead to activation of the host immune system against tumor cells, with consequent tumor shrinkage [Phan et al. 2003; Hodi et al. 2010; Robert et al. 2011]. Approximately 20% of patients in these studies experienced prolonged long-term survival, suggesting the power of immune checkpoint blockade in limiting neoplastic growth long after treatment administration.
Nature and function of PD-1 and PD-L1
Programmed death 1 (PD-1, CD279) is a 288 amino acid type I transmembrane protein encoded by the Pdcd1 gene on chromosome 2 in humans; it functions as a receptor expressed by activated T cells [Keir et al. 2008]. There are two known ligands of PD-1. The first, PD-L1 (also known as B7-H1 or CD274), is a 290 amino acid type I transmembrane protein encoded by the Cd274 gene on chromosome 9 in humans; it is expressed on antigen-presenting cells and tumor cells, and is primarily responsible for the immunosuppressive effects of PD-1 [Sharpe and Freeman, 2002]. The second ligand for PD-1 is known as PD-L2 (also known as B7-DC or CD273); it also functions to inhibit T-cell activation [Latchman et al. 2001; Tseng et al. 2001].
Clinical data have supported the hypothesis that the PD-1/PD-L1 interaction is an important regulator of tumor immune tolerance and tumor growth in RCC. A retrospective analysis of 306 patients who underwent nephrectomy for clear cell RCC at the Mayo Clinic from 1990 to 1994 revealed that patients whose tissue demonstrated expression of PD-L1 (B7-H1) had significantly reduced rates of 5-year cancer-specific survival (41.9%) when compared to those whose tumors did not express PD-L1 (82.9%) [Thompson et al. 2006]. These data suggested that blocking the interaction of PD-L1 (expressed on RCC cells) with PD-1 (expressed on T cells) might reverse tumor-induced immune tolerance and serve as an attractive approach for RCC therapy.
Anti-PD-1 studies in RCC
Phase I
Pre-clinical murine models suggested that interference with the PD-1/PD-L1 interaction results in anti-neoplastic effects against a variety of different tumors [Iwai et al. 2002; Strome et al. 2003]. This led to Brahmer and colleagues’ first-in-human phase I study of MDX-1106/BMS-936558/ONO-4538 (later nivolumab, [Bristol-Myers Squibb; New York, NY, USA]), a fully human monoclonal IgG4 antibody specific for human PD-1, in 39 patients with advanced metastatic melanoma, colorectal cancer, castrate-resistant prostate cancer, non-small cell lung cancer, and RCC [Brahmer et al. 2010]. One patient in this study had clear cell RCC. In this dose-escalation study one patient with colorectal cancer obtained a complete response at the dose of 3 mg/kg. Two patients, the one with RCC and another one with melanoma, experienced partial responses at the 10 mg/kg dose (Table 1).
Single agent anti-PD-1 (nivolumab) studies in RCC.

Abbreviations: n/a, not applicable (only one patient with RCC was enrolled in this study, and therefore objective response rate and stable disease rate could not be calculated); RCC, renal cell carcinoma.
Specific data for immune-related grade 3 or 4 adverse events were not published in this study. The overall rate of grade 3 or 4 adverse events was 5%, 17%, and 13% in the 0.3, 2.0, and 10.0 mg/kg groups, respectively.
The favorable response rate and acceptable toxicity profile of nivolumab in this pilot study led to a multiple-dose trial in 296 patients with different cancers [Topalian et al. 2012b]. This phase I study enrolled patients with advanced melanoma, non-small cell lung cancer, castration-resistant prostate cancer, colorectal cancer, and RCC. A total of 34 of the patients in this study had RCC. They were treated at doses of either 1.0 or 10.0 mg/kg every two weeks for up to 12 cycles, until disease progression or complete response.
In this dose-escalation study no maximum tolerated dose was reached. Only 15 of 296 patients (5%) discontinued treatment because of adverse effects. Grade 3 or 4 adverse effects were present in 14% of patients. Of particular note, drug-induced pneumonitis occurred in 3% of patients and was of grade 3 or 4 severity in three patients (1%). There were three treatment-related deaths (1%) due to pneumonitis in this study.
The efficacy analysis revealed objective responses in 29% (10/34) of patients with RCC. Responses were seen at both doses of nivolumab (1.0 and 10.0 mg/kg). Another nine patients (27%) had stable disease for 24 weeks or more. The median progression-free survival for RCC patients was 7.3 months, with 1-year progression-free survival rate of 35% and 2-year progression-free survival rate of 12%.
Phase II
Based on the favorable efficacy and toxicity data of these two Phase I trials, a randomized, blinded, phase II study of nivolumab in patients with progressive, advanced or metastatic clear-cell RCC began in enrollment in May 2011 and closed to accrual later that year [ClinicalTrials.gov identifier: NCT01354431]. This study evaluated patients who had received at least one prior anti-angiogenic therapy but not more than three previous therapies; patients were randomized to receive doses of 0.3 mg/kg (n = 60), 2.0 mg/kg (n = 54), or 10.0 mg/kg (n = 54) of nivolumab every 3 weeks until disease progression or unacceptable toxicity (Table 1). A total of 168 patients were included in this study, with the primary objective being to evaluate for a dose–response relationship in progression-free survival. The median progression-free survival was 2.7 months in the 0.3 mg/kg group, 4.0 months in the 2 mg/kg group, and 4.2 months in the 10 mg/kg group [Motzer et al. 2014]. The median overall survival was 18.2 months in the 0.3 mg/kg group, 25.5 months in the 2 mg/kg group, and 24.7 months in the 10 mg/kg group. The objective response rates were 20%, 22%, and 20% in the 0.3, 2, and 10 mg/kg groups, respectively, for an overall objective response rate of 21%.
Amongst all patients who had an objective response (n = 35), 54% had responses lasting for at least 12 months. No dose-response was noted for progression-free survival or objective response rate; however, longer median overall survival was obtained at the 2.0 and 10.0 mg/kg doses.
Grade 3 or 4 treatment-related adverse events were present in 5% of patients at the 0.3 mg/kg dose, 17% of patients at the 2 mg/kg dose, and 13% of patients at the 10 mg/kg dose. Side effects led to the discontinuation of treatment in 2%, 11%, and 7% of patients in the 0.3 mg/kg group, 2 mg/kg group, and 10 mg/kg groups, respectively. No instances of grade 3 or 4 pneumonitis were observed.
This study indicated that nivolumab was well-tolerated from a safety perspective. Its favorable response rate of 21% and encouraging overall survival across all doses demonstrated that further study in a randomized, phase III trial was warranted. However, no dose–response relationship was established in this study; the response rate was constant across increasing doses of nivolumab. Furthermore, the decrease in objective response rate between the phase I study (29%) and this phase II study (21%) suggests that nivolumab’s clinical effectiveness may be limited to a minority of patients, and that combination approaches should be explored.
Phase III
The CheckMate 025 study is a phase III randomized trial of nivolumab versus everolimus in patients with advanced RCC [ClinicalTrials.gov identifier: NCT01668784]. Patients who received one or two prior anti-angiogenic therapies for advanced RCC (but not more than three total previous therapies) were randomized to receive nivolumab at 3 mg/kg every 2 weeks versus everolimus until disease progression or unacceptable side effects (Table 4). The primary endpoint of this study was overall survival. In July 2015, the independent Data Monitoring Committee concluded that the study had met its primary endpoint. While full results remain forthcoming, this study will likely establish nivolumab as a new standard of care for previously treated patients with metastatic kidney cancer [Bristol-Myers Squibb, 2015].
Anti-PD-L1 studies
Phase I
Blockade of one of PD-1’s ligands, PD-L1, was simultaneously investigated as a possible therapeutic target for a variety of malignancies. BMS-936559 (also known as MDX-1105) is a fully human IgG4 monoclonal antibody directed against PD-L1. Brahmer and colleagues reported a phase I trial of BMS-936559 in 207 patients with advanced non-small cell lung cancer, melanoma, ovarian cancer, colorectal cancer, pancreatic cancer, gastric cancer, breast cancer, or RCC who had tumor progression after at least one previous therapy for advanced or metastatic disease [Brahmer et al. 2012]. Of this group, 17 patients had RCC (Table 2).
Single agent anti-PD-L1 studies in RCC.

Objective responses were demonstrated in 2 of the 17 patients with RCC (12%). The response durations for these two patients were 4 and 17 months. An additional 7 patients with RCC (41%) had stable disease for at least 24 weeks. Immune-related adverse events were present in 81 patients (39%). Grade 3 or 4 side effects that were attributable to potential immune causes were present in 10 patients (5%). Because of the relatively low response rate and high percentage of immune-related side effects, BMS-936559 will not be pursued for further development for RCC.
More recently, atezolizumab (formerly MPDL3280A, Genentech; South San Francisco, CA, USA), a human IgG1 monoclonal antibody directed against PD-L1, has been evaluated in a first-in-human phase I trial of patients with advanced, incurable cancer [Herbst et al. 2014]. Atezolizumab inhibits the interaction of PD-L1 with both PD-1 and B7.1, and also has an engineered Fc domain that is designed to inhibit antibody-dependent cell-mediated cytotoxicity, thereby preventing cytotoxicity against activated T-cells that express PD-L1 and further augmenting upregulation of the immune antitumor response. In this trial 277 patients with advanced solid tumors were administered atezolizumab in a dose-escalating fashion every 3 weeks (Table 2). These included 69 patients with RCC [McDermott et al. 2014]. Of note, this trial permitted enrollment of both clear cell RCC (n = 62 evaluable in the efficacy analysis) and non-clear cell (n = 7 evaluable in the efficacy analysis) RCC histologies.
A maximum tolerated dose was not reached in this study. Analysis of toxicity revealed that grade 3 or 4 treatment-related adverse events were present in 35 of 277 patients (13%). Grade 3 or 4 adverse events that were related to immune phenomena were presents in three subjects (1%); no cases of grade 3 or greater pneumonitis were observed. Of the cohort of patients with RCC, grade 3 treatment-related adverse events occurred in 11 patients (16%), and no grade 4 adverse events have been observed.
The overall objective response rate for the entire cohort of patients with RCC was 15% (Table 2). One complete response was noted. The 24-week progression-free survival rate was 51%, and the median duration of response was 54 weeks (range 2.7+ to 68.1+ weeks). Objective responses were noted both in patients without PD-L1 immunohistochemical positivity in tumor-infiltrating immune cells (objective response rate 10%) and in patients with PD-L1 immunohistochemical positivity (defined as 1+, 2+, or 3+ staining) in tumor infiltrating immune cells (objective response rate 20%). Intriguingly, higher response rates were noted in the subgroup analysis of MSKCC poor-risk [Motzer et al. 1999] patients with PD-L1 positivity by immunohistochemistry (objective responses in 4 of 7 patients, 57%).
This long-term follow-up data indicate that atezolizumab is tolerable from a safety perspective but has a modest objective response rate (15%) in advanced RCC. This limited efficacy as a single agent in unselected RCC patients suggests that further study of atezolizumab in RCC should be based on combination therapy with other immune-checkpoint or angiogenesis inhibitors.
PD-1 and PD-L1-directed therapy in combination
The above-noted objective response rates and favorable toxicity profiles of anti-PD-1 and anti-PD-L1 therapies in early phase clinical trials have led to interest in combining these agents with other immunomodulatory molecules and anti-angiogenesis therapies, in an effort to augment the extent and duration of responses for patients with metastatic RCC.
Combinations of anti-PD-1/PD-L1 and angiogenesis inhibitors
The immunomostimulatory effects of anti-angiogenesis therapies have been described by multiple investigators. These effects include increased trafficking of T cells into tumors [Manning et al. 2007, Shrimali et al. 2010], reduction of immunosuppressive cytokines, and reduction in immunosuppressive Treg cells [Kusmartsev et al. 2008; Roland et al. 2009]. Anti-angiogenic therapies may therefore serve as primers for subsequent or concurrent immunostimulatory therapies. This provides the rationale for combination of anti-VEGF treatment and anti-PD-1/PD-L1 therapy for metastatic RCC (Tables 3 and 4).
Anti-PD-1/PD-L1 in combination with angiogenesis inhibition or other immune checkpoint inhibition in RCC.
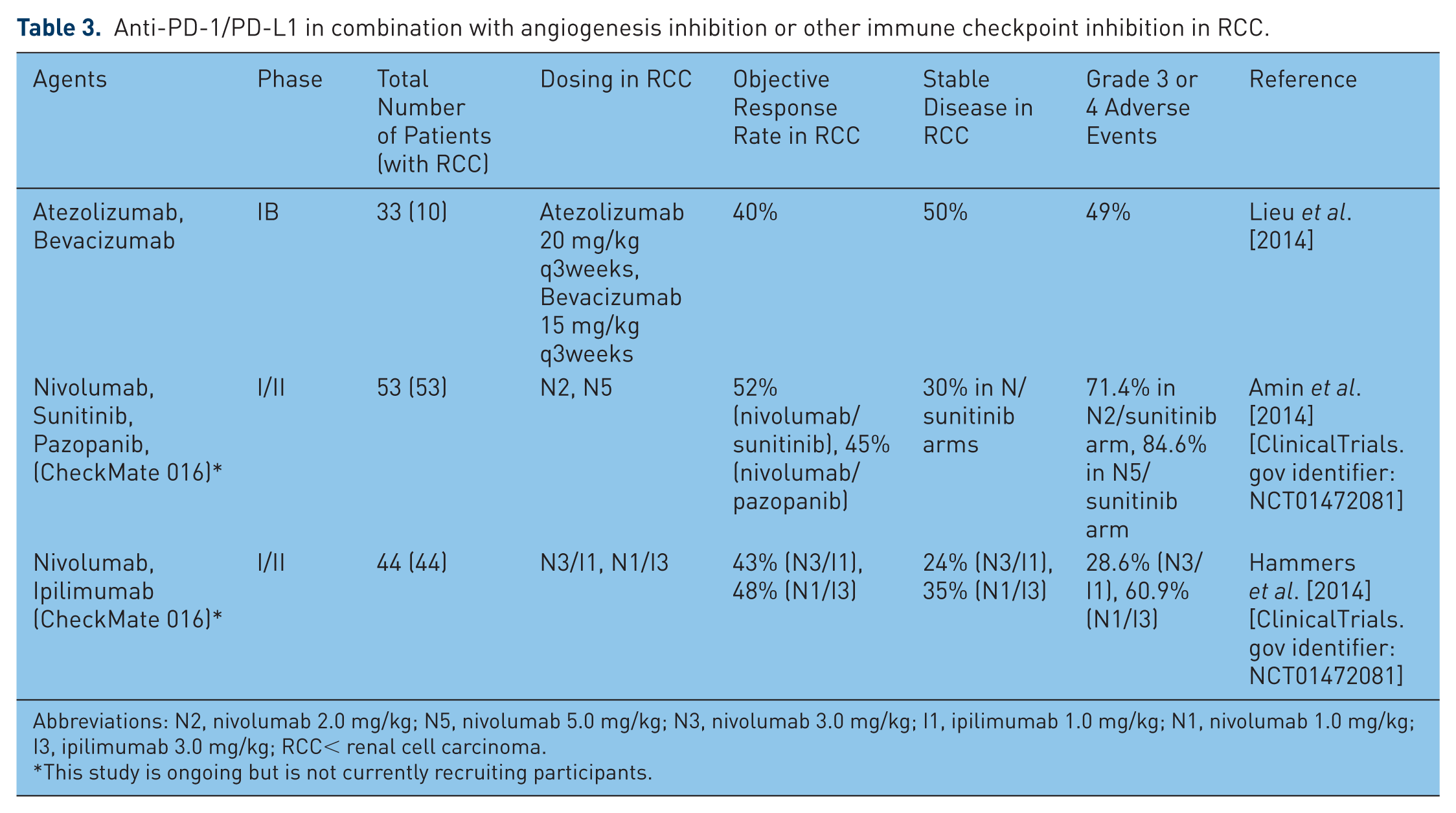
Abbreviations: N2, nivolumab 2.0 mg/kg; N5, nivolumab 5.0 mg/kg; N3, nivolumab 3.0 mg/kg; I1, ipilimumab 1.0 mg/kg; N1, nivolumab 1.0 mg/kg; I3, ipilimumab 3.0 mg/kg; RCC< renal cell carcinoma.
This study is ongoing but is not currently recruiting participants.
Currently enrolling anti-PD-1/PD-L1 trials in RCC.
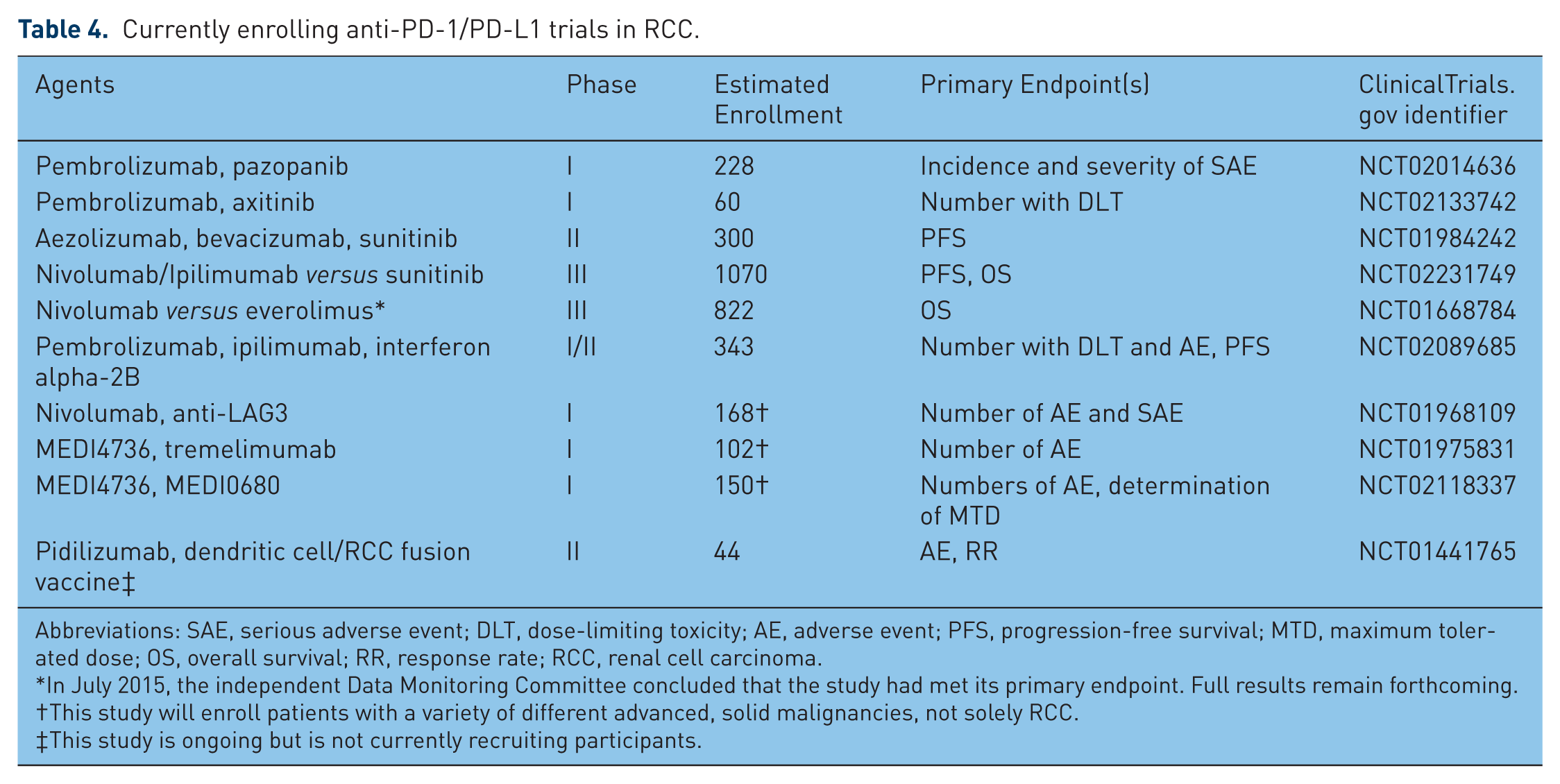
Abbreviations: SAE, serious adverse event; DLT, dose-limiting toxicity; AE, adverse event; PFS, progression-free survival; MTD, maximum tolerated dose; OS, overall survival; RR, response rate; RCC, renal cell carcinoma.
In July 2015, the independent Data Monitoring Committee concluded that the study had met its primary endpoint. Full results remain forthcoming.
This study will enroll patients with a variety of different advanced, solid malignancies, not solely RCC.
This study is ongoing but is not currently recruiting participants.
Data from a recent phase 1B study of PD-L1 blockade with atezolizumab in combination bevacizumab were recently presented [Lieu et al. 2014]. Ten patients with metastatic clear-cell RCC were enrolled in this trial, in which patients received atezolizumab at 20 mg/kg every 3 weeks and bevacizumab at 15 mg/kg every 3 weeks in a dose-escalating fashion. Four of the 10 patients (40%) demonstrated an objective response, and another five patients demonstrated stable disease (50%). Objective responses in this small cohort of patients appeared to be unrelated to PD-L1 tumor positivity, with responses noted in two patients with 1+ immunohistochemical staining and in one patient with no immunohistochemical staining for PD-L1. Of the entire study population (n = 35, including patients with RCC as well as colorectal cancer and other solid tumors), 49% experienced a grade 3 or 4 adverse event; however, only one of these adverse events was attributed to atezolizumab (one case of neutropenia), and no grade 4 adverse events or deaths were attributed to atezolizumab. This small study suggests that the combination of anti-PD-L1 therapy and VEGF inhibitor therapy may have clinical efficacy in metastatic clear-cell RCC.
A larger phase II study of the combination of atezolizumab and bevacizumab for patients with previously untreated metastatic RCC is currently enrolling patients (Table 4 [ClinicalTrials.gov identifier: NCT01984242]). A total of 300 patients will be randomized between the following three arms, until progression of disease: (1) atezolizumab at the fixed dose of 1200 mg every 3 weeks for eight cycles, (2) atezolizumab at 1200 mg every 3 weeks for eight cycles and bevacizumab at 15 mg/kg every 3 weeks, and (3) sunitinib at 50 mg per day for 4 weeks of 6 week cycles. This trial will allow comparison of combination anti-PD-L1/anti-VEGF therapy with anti-PD-L1 monotherapy and anti-VEGF monotherapy.
Combination trials of anti-PD-1 therapy and anti-VEGF therapy that are ongoing but are not currently recruiting participants include the CheckMate 016 trial, a phase I study of nivolumab in combination with sunitinib, pazopanib, or ipilimumab in patients with metastatic clear-cell RCC (Table 3, [ClinicalTrials.gov identifier: NCT01472081]). The tyrosine-kinase inhibitor arms of this study included dose-escalation cohorts with nivolumab/sunitinib and nivolumab/pazopanib at two different doses of nivolumab (2.0 mg/kg every 3 weeks and 5.0 mg/kg every 3 weeks, N2 and N5 arms, respectively), in combination with sunitinib 50 mg daily for 4 out of every 6 weeks or pazopanib 800 mg daily in a continuous fashion.
Initial data from this trial indicated that the N5/sunitinib arm has no dose-limiting toxicity; a maximum tolerated dose was not reached for seven patients treated at each dose. The nivolumab/sunitinib arm was therefore expanded to include 19 more treatment-naïve patients with metastatic RCC at the N5 dose [Amin et al. 2014]. However, the nivolumab/pazopanib arm demonstrated four dose-limiting toxicities out of 20 patients who were treated at the N2 dose (three patients with elevated AST/ALT and one patient with fatigue). This translated to a 25% rate of discontinuation due to treatment-related adverse events, and led to closure of the nivolumab/pazopanib half of the study.
Grade 3 or 4 adverse events were noted in 71.4% of patients in the N2/sunitinib arm and in 84.6% of patients in N5/sunitinib arm; these included fatigue (11.5%), diarrhea (11.5%), and increased hepatic transaminases (elevated alanine aminotransferase in 18.2%, elevated aspartate aminotransferase in 9.1%). In addition, grade 3 or 4 renal toxicity was observed at an unexpectedly increased rate in the nivolumab/sunitinib arms, with one patient each (3%) experiencing increased creatinine, acute renal failure, and autoimmune nephritis. Grade 3 pneumonitis occurred in one patient in these arms.
The objective response rate for the total combination of 33 patients in both of the nivolumab/sunitinib arms was 52% (one complete response and 16 partial responses), as defined by RECIST 1.1 criteria, with median duration of response of 54 weeks (range 18.1 to 80+) and median progression-free survival 48.9 weeks. An additional 10 patients (30%) experienced stable disease. In contrast, the objective response rate for the 20 patients in the nivolumab/pazopanib arm was 45% (no complete responses and nine partial responses), with median response duration 30 weeks and median progression-free survival 31.4 weeks. Of note, PD-L1 expression on tumors was measured retrospectively using the Dako immunohistochemistry assay for PD-L1. Using the definition of PD-L1 positivity as greater than 1% tumor membrane staining, objective responses were noted in 45.5% of PD-L1 positive patients in the nivolumab/sunitinib group, and in 42.4% of PD-L1 negative patients in this group. Using the more stringent definition of PD-L1 positivity of 5% tumor membrane staining to indicate PD-L1 positivity, objective responses were noted in 15.2% of patients with PD-L1 positive tumors and in 72.7% of patients with PD-L1 negative tumors.
This small study demonstrated a higher objective response rate with the combination of nivolumab/sunitinib than with historical studies of either agent alone (21% and 31%, respectively) in the treatment of metastatic RCC [Motzer et al. 2014, 2007]. Furthermore, many of the patients who did respond to this combination therapy have had ongoing responses (10 of 17 patients, 59%), with a median duration of response of 54 weeks (range 18.1–80+ weeks). As noted previously, the nivolumab/pazopanib arm of this trial was discontinued because of early dose-limiting toxicities, and similar rates of grade 3 or 4 transaminase elevations in the nivolumab/sunitinib arm (elevated alanine aminotransferase in 18.2%, elevated aspartate aminotransferase in 9.1%) have discouraged further development of this combination therapy as well.
Several other combinations of PD-1/PD-L1 blockade and anti-angiogenesis inhibitors are currently undergoing clinical trial investigation (Table 4). This includes the phase I study of pazopanib and/or pembrolizumab (also known as MK-3475, [Merck, Whitehouse Station, NJ, USA]), a humanized IgG4 monoclonal antibody against PD-1, in treatment-naïve patients with metastatic RCC [ClinicalTrials.gov identifier: NCT02014636], as well as a phase I study of MK-3475 and axitinib [ClinicalTrials.gov identifier: NCT02133742].
Combinations of anti-PD-1/PD-L1 and other checkpoint inhibitors
In contrast to other immune checkpoint pathways such as CTLA-4/B7, which interferes solely with the initiation of primary immune responses, the PD-1/PD-L1 pathway appears to be critical for the interruption of both the initiation and the maintenance of the immune response [Fife et al. 2009]. It has therefore been theorized that dual blockade of the PD-1/PD-L1 axis along with other immune checkpoints may augment the immune up-regulation of any single-agent checkpoint blockade alone.
As noted above, the CheckMate 016 trial is a phase I study of nivolumab (N) in combination with sunitinib, pazopanib, or ipilimumab (I) in patients with metastatic clear-cell RCC [ClinicalTrials.gov identifier: NCT01472081]. The nivolumab/ipilimumab data from this study were presented at the American Society of Clinical Oncology annual meeting in June 2014 [Hammers et al. 2014]. In this study patients with metastatic RCC who had favorable or intermediate prognosis based on the MSKCC score were randomized to receive either nivolumab 3 mg/kg and ipilimumab 1 mg/kg (n = 21, N3/I1 arm) or nivolumab 1 mg/kg and ipilimumab 3 mg/kg (n = 23, N1/I3 arm) every 3 weeks for 4 doses, followed by nivolumab 3 mg/kg every two weeks until progression or toxicity. The objective response rate in this trial was 43% (nine patients) in the N3/I1 arm and 48% (11 patients) in the N1/I3 arm.
The durability of response is difficult to estimate at present; however, seven of nine patients (77.8%) in the N3/I1 had ongoing responses, and nine of the 11 patients (81.8%) in the N1/I3 arm had ongoing responses at the time of that data presentation. Adverse events of any grade were noted in 76.2% of patients in the N3/I1 arm and in 100% of patients in the N1/I3 arm. Grade 3 or 4 adverse events occurred in 28.6% of patients (n = 6) in the N3/I arm, and in 60.9% (n = 14) of patients in the N1/I3 arm. These consisted of elevated lipase (14.3% in the N3/I1 arm, 26.1% in the N1/I3 arm), elevated ALT (0% in the N3/I1 arm, 26.1% in the N1/I3 arm), diarrhea (4.8% in the N3/I1 arm and 13.0% in the N1/I3 arm), and elevated amylase (4.8% in the N3/I1 arm and 4.3% in the N1/I3 arm). Treatment was discontinued because of an adverse event in 2 patients (9.5%) in the N3/I1 arm, and in 6 patients (26.1%) in the N1/I3 arm.
These data demonstrate that the combination of nivolumab and ipilimumab has a tolerable safety profile and encouraging efficacy in the treatment of metastatic RCC. Expansion cohorts at the N3/I1 and N1/I3 doses were subsequently enrolled, and another cohort of nivolumab 3 mg/kg and ipilimumab 3 mg/kg (N3/I3) was added to the study. Accrual for this trial has been completed.
Several other combinations of PD-1/PD-L1 inhibitors and other immune checkpoint blockers are currently under investigation (Table 4). These include combinations of pembrolizumab (MK-3475) and ipilimumab [ClinicalTrials.gov identifier: NCT02089685], nivolumab and an anti-LAG3 monoclonal antibody [ClinicalTrials.gov identifier: NCT01968109], the anti-PD-L1 agent MEDI4736 and the CTLA-4 inhibitor tremelimumab [ClinicalTrials.gov identifier: NCT01975831], and the PD-1 inhibitor MEDI0680 with MEDI4736 ([ClinicalTrials.gov identifier: NCT02118337], Table 4). All of these are phase I studies whose goals broadly include determining dose-limiting toxicities and treatment-related adverse events of combination PD-1/PD-L1 blockade with other immune checkpoint inhibitors.
In addition, a phase III trial of nivolumab and ipilimumab versus sunitinib in previously untreated patients with metastatic RCC (CheckMate 214) was opened to accrual in October 2014 [ClinicalTrials.gov identifier: NCT02231749]. This trial aims to randomize 1070 patients with advanced or metastatic RCC to one of two study arms: (1) nivolumab 3 mg/kg and ipilimumab 1 mg/kg every 3 weeks for four doses, followed by nivolumab 3 mg/kg every 2 weeks until disease progression or unacceptable toxicity, or (2) sunitinib 50 mg orally daily for 4 out of 6 weeks continuously until disease progression or unacceptable toxicity. The primary outcome measures of this study are progression-free survival and overall survival. It is expected to be completed in September 2019.
Combinations of anti-PD-1/PD-L1 and other immunomodulators
PD-1/PD-L1 blocking agents have also been combined with other immunostimulatory therapies, in an effort to further augment the immune antitumor response in RCC. This includes a phase II study of the PD-1 inhibitor CT-011 (also known as pidilizumab [CureTech LTD, Yavne, Israel]) in combination with a dendritic cell/RCC tumor cell fusion autologous vaccine [ClinicalTrials.gov identifier: NCT01441765]. This study is currently ongoing.
Blockade of the PD-1/PD-L1 axis is also being investigated in combination with traditional cytokine-based immunotherapy. The Keynote-029 trial is a phase I/II trial designed to examine safety and tolerability of the anti-PD-1 monoclonal antibody MK-3475 and either the cytokine interferon alpha-2b or ipilimumab in advanced melanoma and RCC [ClinicalTrials.gov identifier: NCT02089685]. This trial is currently enrolling participants, and is estimated to be completed in January 2018 (Table 4).
Unanswered questions and future directions
In spite of the efficacy and favorable toxicity profile of immune checkpoint inhibitors in metastatic RCC, many unanswered questions remain. These include, among others, better defining the role of PD-L1 positivity as a biomarker for response to anti-PD-1/PD-L1 therapy, developing better predictive models for response to immune checkpoint blockade, determining how to approach immune checkpoint blockade in patients with non-clear cell RCC, and assessing the durability of response to immune checkpoint blockade after discontinuation of anti-PD-1/PD-L1 agents.
The role of PD-L1 positivity
The precise role of PD-L1 positivity in predicting response to anti-PD-1/anti-PD-L1 targeted therapy in metastatic RCC remains to be defined. Correlative data from several clinical trials has suggested that PD-L1-positive RCC tumors may be more responsive to checkpoint blockade; however, this finding has been inconsistent, and it is clear that responses have also been seen in PD-L1-negative tumors. Moreover, the very definition of a cut-off for PD-L1 ‘positivity’ (e.g. immunohistochemical staining of 1% or 5%) has varied among different trials. Further study is necessary not only to standardize the definition of PD-L1 positivity, but also to elucidate its role as a possible predictive factor.
PD-1/PD-L1 inhibition in non-clear cell RCC
PD-L1 status may have an important role in RCCs of non-clear cell histology. Up to 80% of RCCs have clear cell histology. Previous studies have indicated that clear cell RCC may respond better to immunotherapy than other RCC histologic variants, such as papillary or chromophobe; for this reason patients with non-clear cell histologies have largely been excluded from the above-mentioned clinical trials [Chowdhury et al. 2011]. Recently, the PD-L1 status of 101 patients with non-clear cell RCC was assessed and was correlated with time to recurrence and overall survival [Choueiri et al. 2014]. In this study, PD-L1 positivity was defined and quantified in two different ways: (1) as ⩾5% tumor cell membrane staining, and (2) as a percentage of PD-L1-positive tumor-infiltrating mononuclear cells (TIMC), with 0% = 0, <5% = 1, and ⩾5% = 2. The majority of these patients were stage I (53%), and few had metastatic disease (23%). Only 10.9% were PD-L1-positive by the criteria of PD-L1 expression in tumor cell membrane; however, 56.4% were PD-L1-positive by a TIMC PD-L1 score of >0. PD-L1 expression in the tumor cell membrane correlated with higher Fuhrman Grade and TNM stage.
After an overall median follow-up of 5 years, patients with PD-L1-positive tumor cells had a significantly increased risk of death (hazard ratio = 6.41, p < 0.001) compared with those with PD-L1-negative tumor cells. In addition, a statistically nonsignificant association was observed between positive PD-L1 expression in TIMC and decreased overall survival (hazard ratio = 2.49, p = 0.08). Both PD-L1-positive tumor cells and PD-L1-positive TIMC were associated with shorter time to recurrence. This study suggests that PD-L1-positivity is a negative prognostic factor in non-clear cell RCC, and that blockade of the PD-1/PD-L1 axis may prove beneficial in future studies of non-clear cell RCC.
Developing predictive models
Beyond tumor PD-L1 status, it is possible that the most accurate predictive models for the effectiveness of PD-1/PD-L1 blockade may incorporate multiple factors, including clinical characteristics such as MSKCC risk score, IDMC criteria [Heng et al. 2009], degree of immune infiltrate of the tumor, tumor mutational signature, and gene expression characteristics. Further study is necessary in order to integrate these variables into the most useful predictive model, in order to assure the most appropriate patient selection for immune checkpoint inhibitor therapy in metastatic RCC.
Demonstrating durable benefit after cessation of immune checkpoint blockade
Although objective response rates of PD-1/PD-L1 blockade in RCC are encouraging, it remains to be seen whether or not responses to anti-PD-1/PD-L1 therapy will be durable, thereby leading to persistent disease control after the discontinuation of immunotherapy. Such durability of responses has been noted with other immunotherapies such as high-dose IL-2 in RCC and melanoma and ipilimumab in metastatic melanoma.
Conclusion
Targeted therapy against the PD-1/PD-L1 immune checkpoint pathway has produced encouraging objective response rates in patients with metastatic RCC. While full results remain forthcoming, nivolumab has reportedly met its primary endpoint of demonstrating an overall survival benefit when compared to everolimus for patients with previously-treated metastatic RCC in the randomized, Phase III, CheckMate 025 study, and will likely establish a new standard of care for this population [Bristol-Myers Squibb, 2015]. Combinations of anti-PD-1/PD-L1 and other agents active against RCC, such as angiogenesis inhibitors, cytokines, vaccines, and other immune checkpoint inhibitors, have demonstrated higher response rates than single-agent therapy alone, albeit at the expense of higher toxicities. Defining the safest combination of these agents and developing predictive algorithms to enhance selection of those patients most likely to benefit from checkpoint inhibitor therapy will be crucial to the success of these agents in the future. Furthermore, the improved objective response rates of these combination therapies must be confirmed in prospective, randomized trials.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
