Abstract
Background:
This study investigated whether the effectiveness of first-line tyrosine-kinase inhibitors was associated with sites of disease in patients with metastatic renal cell carcinoma (mRCC).
Methods:
A retrospective cohort of consecutive mRCC patients receiving first-line sorafenib (SO) or sunitinib (SU) was analyzed.
Results:
In total, 203 patients received SO and 99 SU. In patients with liver metastasis, SU was associated with a 18% higher risk of time-to-treatment failure (TTF), and a 39% higher risk of death than SO: conversely, patients without liver metastases who received SU showed a 46% decreased risk of TTF and 62% decreased risk of death.
Conclusions:
mRCC patients with liver metastases treated with first-line SO showed a better outcome compared with SU, while mRCC patients without liver metastases treated with first-line SU showed a better outcome compared with SO.
Introduction
Common sites of disease in patients with metastatic renal cell carcinoma (mRCC) include lung (45–60%), bone (30%), lymph nodes (22%), liver (20%), and brain (8%) [Bianchi et al. 2012]. The recent introduction of a number of targeted therapies (TTs) directed at inhibiting either the vascular endothelial growth factor or mammalian target of rapamycin pathways has led to a shift in the treatment of mRCC and has made patient selection extremely important. Prognostic factors, histology, and clinical characteristics are commonly used to guide treatment selection. Nevertheless, all the stratification criteria applied in well-recognized models of prognostic risk factors have only limited utility in predicting response to treatment [Motzer et al. 1999; Heng et al. 2009], and sparse data are available to validate the predictive role of any clinical or biological characteristic [Hahn et al. 2008; Lassau et al. 2010; Jain et al. 2010; Kayani et al. 2011; Rini et al. 2011a; Rini et al. 2011b; Michaelson et al. 2011; Armstrong et al. 2012; Tran et al. 2012].
Although studies performed in the cytokine era suggest that specific metastatic sites have a negative impact on survival [Negrier et al. 2002], and recent data suggest that the presence of bone and liver metastases may worsen the outcome of patients receiving TTs [McKay et al. 2014], there is a paucity of information regarding the potential predictive role of metastatic sites in the era of TTs. In this scenario, the impact of different sites of disease on the outcome of mRCC patients treated with tyrosine-kinase inhibitors (TKIs) is still unclear. It would therefore be interesting to investigate whether the metastatic site might be considered as a predictor of response to treatment.
In this study, we retrospectively investigated whether metastatic sites are associated with time- to-treatment failure (TTF) and overall survival (OS) in mRCC patients treated with first-line sunitinib (SU) or sorafenib (SO).
Patients and methods
A retrospective cohort of consecutive mRCC patients treated with first-line TKIs at the Istituto Nazionale Tumori of Milan between January 2005 and October 2013 was analyzed. All patients receiving upfront TTs with either SU or SO were included. Both agents were administered orally, SU 50 mg once a day for 4 weeks followed by a 2-week resting period, while SO 400 mg was given twice daily continuously. The SU dose was reduced to 37.5 mg or 25 mg and that of SO to 600 mg or 400 mg according to the patient’s pretreatment Eastern Cooperative Oncology Group (ECOG) performance status (PS), or main adverse events during therapy. All patients who were candidates for first-line TKI treatment for metastatic disease were evaluated for the study, including those receiving prior cytokines and those who had not had a nephrectomy. Most of the patients evaluated received a second-line therapy at disease progression. The choice of first-line SO was based mainly on a patient’s characteristics (e.g. prior cytokines, comorbidities, and poor ECOG PS), and availability of the drug at the time of treatment. We collected baseline demographics, clinical and survival data from the patients’ medical records. The study protocol was approved by our Institutional Review Board.
Statistical methods
Baseline data were analyzed by descriptive statistics. TTF was defined as the time from the initiation of first-line TTs to treatment discontinuation for any reason, including objective tumor progression, death, or toxicity, whichever occurred first. Progression of disease was evaluated using the Response Evaluation Criteria in Solid Tumors criteria v.1.1. Patients receiving first-line TTs at the time of the last contact were right censored. OS was defined as the time from the initiation of first-line TTs to death; patients alive or lost to follow up at the time of the last contact were right censored. The last update was performed in December 2013. The Kaplan–Meier method was used to estimate survival functions; the inverse Kaplan–Meier method was used to estimate median follow up, and the Cox regression model was used to estimate hazard ratios (HRs), and to test the statistical interaction between metastatic site and first-line TTs. All statistical tests were two-sided and a p value less than 0.05 was considered statistically significant. Statistical analysis was performed using the SAS software (SAS Institute, Cary, NC, USA) (version 9.2).
Results
Patient population
In total, 309 mRCC patients received either first-line SO (n = 206) or SU (n = 103). Eight patients were excluded from the analysis because of the contemporary absence of TTF and OS data; therefore a total of 301 (97.4%) patients were analyzed (SO, n = 202; SU, n = 99). Most patients (87%) had received a prior nephrectomy and 37% had previously received cytokines. The majority of patients (63%) had more than one metastatic site.
Metastatic lesions to the lung were present in 237 (66%) patients; 136 (38%) patients presented with metastases to the lymph nodes, 100 (28%) patients had bone metastases, 63 (18%) patients had metastases to the liver, 16 (5%) had metastases to the brain, and 125 (35%) patients had lesions to other sites. The two groups differed in terms of PS (ECOG PS = 2 in 14% of SO-treated patients versus 5% in the SU population), metastatic sites (lung: 71% versus 54%; lymph nodes: 44% versus 28%), number of concomitant metastatic sites (= 3 in 24% versus 10%), and prior cytokines (54% versus 7%). Table 1 summarizes demographics, disease and treatment characteristics in the overall population and in the two subgroups.
Demographic, pathological, and previous treatment characteristics.

Adrenal, soft tissue, pleural, or kidney.
Treatment effectiveness
After a median follow up of 64.1 months (range: 10.0–93.2 months) 194 out of 203 (96%) patients in the SO group reached the TTF event; after a median follow up of 27.0 months (range: 1.0–60.1 months) 58 out of 99 (59%) patients in the SU group reached the TTF event. Median TTF for the SO group was 9.0 months (95% confidence interval [CI]: 7.1–12.0), while median TTF for the SU group was 16 months (95% CI: 12.0–21.0). At the time of analysis, 160 out of 204 (78%) patients in the SO group and 31 out of 97 (32%) in the SU group had died. Median OS in the SO group was 19.9 months (95% CI: 16.0–25.1), while median OS for the SU group was not yet defined. Survival function estimates are plotted in Figure 1.
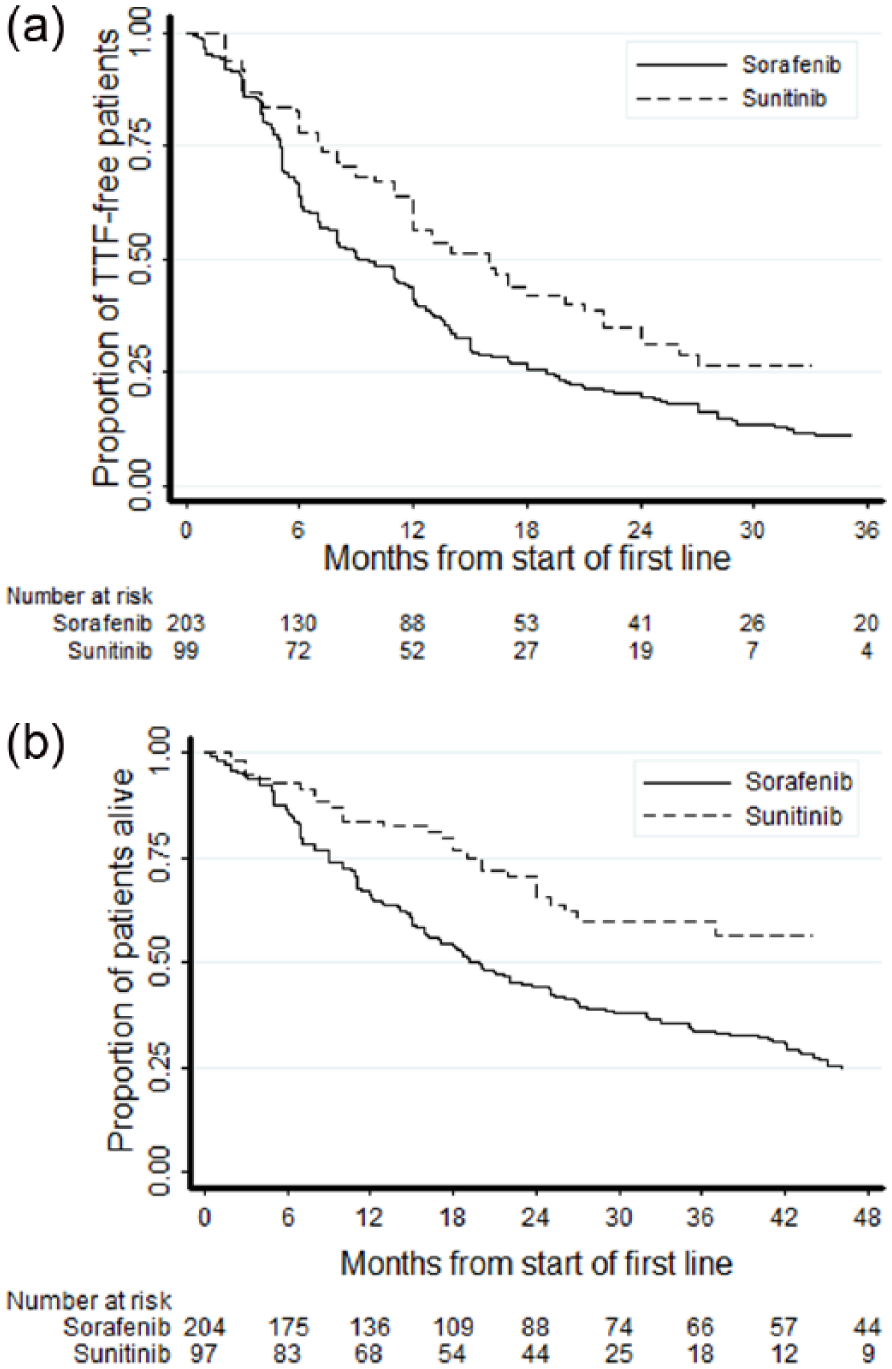
Kaplan–Meier plots of time-to-treatment failure (a) and overall survival (b) in patients treated with first-line sunitinib and first-line sorafenib. TTF, time-to-treatment failure.
Interaction between treatment outcomes and metastatic sites
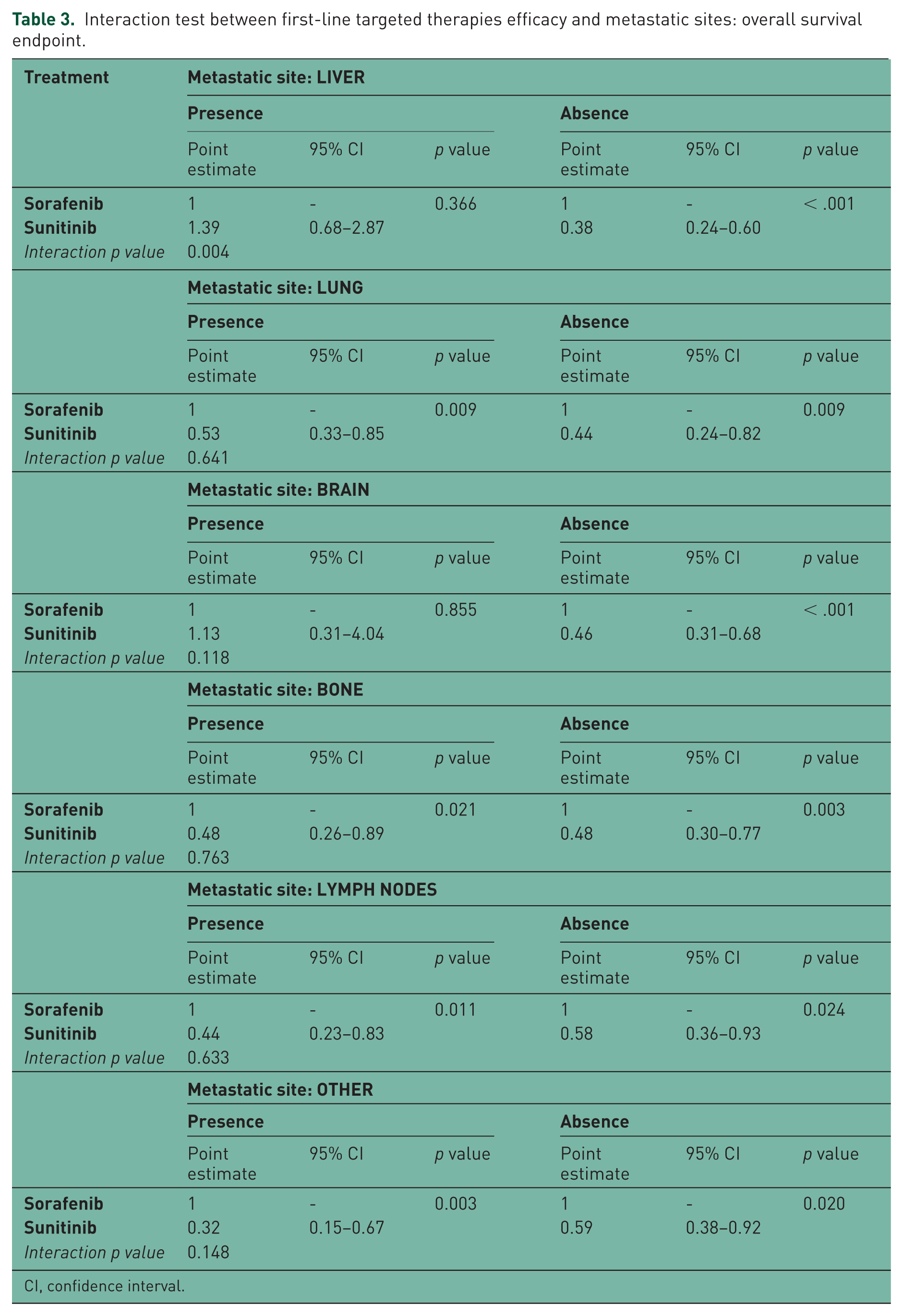
Results on testing the interaction between first-line TTs efficacy and metastatic sites are shown in Tables 2 and 3. A significant interaction between first-line treatment and metastatic sites was disclosed for the liver site (TTF: p = 0.034; OS: p = 0.004): SU was associated with an 18% higher risk, compared with SO (HR: 1.18; 95% CI: 0.63–2.22), of TTF, while patients without liver metastases who received SU showed a 46% (HR: 0.54; 95% CI: 0.39–0.75) decreased risk compared with SO (Figure 2). SU was associated with a 39% higher risk of death compared with SO (HR: 1.39; 95% CI: 0.68–2.87) in patients with liver metastases, while patients without liver metastases treated with SU showed a 62% decreased risk of death compared with SO (HR: 0.38; 95% CI: 0.24–0.60; p = 0.004) (Figure 2). The predictive role of liver metastases was uncertain when introducing the Motzer score, Fuhrman grade, and number of metastatic sites into a multivariate model (TTF: p = 0.308; OS: p = 0.077) (see Table 4).
Interaction test between first-line targeted therapies efficacy and metastatic sites: time-to-treatment failure endpoint.

CI, confidence interval.
Interaction test between first-line targeted therapies efficacy and metastatic sites: overall survival endpoint.
CI, confidence interval.

Kaplan–Meier plots of time-to-treatment failure and overall survival by site of metastasis in patients with (a, c) or without (b, d) liver metastases. TTF, time-to-treatment failure.
Interaction test between first-line targeted therapies efficacy and metastatic sites for time-to-treatment failure (a) and overall survival (b) endpoints; the Motzer prognostic score, the Fuhrman grade, and the number of metastatic sites were introduced in the Cox regression model.

Discussion
This retrospective study aimed to determine the predictive role of different metastatic sites of disease in mRCC patients treated with upfront TTs. The unfavorable prognostic role of some sites of disease such as bone and liver is well recognized [Motzer et al. 2007], and may be due to a more aggressive disease and a lower efficacy of anticancer treatments.
Several studies have demonstrated that first-line SU is associated with a marked activity in terms of both overall disease control rate and time to tumor progression in mRCC treatment-naïve patients. Specifically the efficacy of SU was demonstrated in a randomized phase III trial in all Memorial Sloan–Kettering Cancer Center prognostic groups [Motzer et al. 2007], and also in a retrospective analysis of an unselected population of patients treated in a clinical practice setting [Gore et al. 2009]. Recently, the randomized, phase III SWITCH trial showed no superiority of the sequence SO followed by SU versus the opposite sequence in terms of overall median progression-free survival [Michel et al. 2014].
Our data evaluated the interaction between treatment effectiveness of TKIs and the metastatic sites. The 97 patients receiving first-line SU showed a remarkable treatment duration of 16 months, while patients treated with SO had a median treatment duration of 9 months. It should be noted that, in the present study, the patients population was treated in a clinical practice setting and the efficacy assessment was therefore performed in a less rigorous fashion than in controlled trials. This may justify the longer median duration of first-line treatment observed in our study with both TKIs compared with those reported in the pivotal SU and SO trials [Motzer et al. 2006; Escudier et al. 2007]. In our daily clinical practice we do not suggest to change therapy too early in patients with an indolent disease showing a very slow progression along with clinical benefit. Moreover, we usually continue the same treatment in the case of mixed radiological response, and prefer to manage the adverse events with proper supportive care and dose modifications rather than discontinue the treatment if not strictly necessary.
Our study may suggest that sites of disease be considered as potential predictive factors of effectiveness for SU and SO used as first-line therapy. The greater effectiveness of SU over SO in the presence of lung, lymph node, brain, and bone metastatic involvement was somehow expected. Moreover, in patients without liver metastases SU showed a 46% decreased risk of progression and a 62% decreased risk of death compared with SO. Altogether these data were in keeping with previous studies that indirectly suggested a greater efficacy of SU in terms of overall disease control and time to tumor progression [Motzer et al. 2006; Escudier et al. 2009]. Interestingly, a recent study by Kakizoe and colleagues showed that the response at 1 month after TKI treatment initiation, evaluated by the decreased ratio of 18F-2-fluoro-2-deoxyglucose using positron emission tomography/computed tomography in mRCC patients, did not differ between metastatic sites [Kakizoe et al. 2014], although the timing of response assessment and the use of a nonstandard technique for the evaluation of response should be taken into account.
The lower efficacy reported with SU compared with SO in the presence of liver metastases was unexpected. The 18% increased risk of treatment interruption and the 40% higher risk of death reported with SU should be considered with caution due to some limitation of our study: first of all, its retrospective nature and limited sample size; secondly, the two subgroups of patients receiving SU and SO as first-line treatment were not well balanced due to a greater number of unfavorable prognostic features in the SO subgroup. Moreover, only 3 out of 16 patients who received SU showed liver-only metastases, while the remaining 13 cases reported concomitant metastases to different sites including lung, brain, lymph nodes, or bone. On the other hand, patients receiving SO treatment represented a selected population from the past that was pretreated with cytokines in 54% of cases, before SU or SO was available. When looking at the OS data it should be taken into account that mOS in both SU and SO groups was evaluated from the time of TKI commencement rather than from the time of diagnosis of metastatic disease. Nevertheless while in the SU group TKI was commenced at the time of diagnosis of metastatic disease, in the SO group the prior treatment with cytokines reported in most cases might have affected prognosis and outcome.
Taking all these limitations into account, our study suggests that, in presence of liver metastases, SO might be more effective than SU. Our study should not be considered as conclusive in terms of the identification of liver involvement as a predictive factor of response to SO, and the hypothesis that liver metastases could have a predictive role for the effectiveness of first-line TKIs should be further investigated in a larger controlled study. However data from a study on hepatocellular carcinoma (HCC), where SO turned out to be the only agent able to slow down disease progression [Llovet et al. 2008], suggest that a potential organotropism of this agent for the liver cannot be excluded for mRCC patients. Liver disease may have intricate roles in drug pharmacokinetics, biotransformation, and clearance. SO metabolism is significantly altered in the liver tumor tissue of patients with HCC. Sometimes, alterations increase the bioavailability of the drug resulting in an increased cumulative SO dose [Chu et al. 2008]. Nevertheless the mechanisms underlying the possible relationship between altered SO metabolism in HCC tissue and liver metastases from RCC, as well as similarities between the clinical benefit of SO in HCC and liver metastases from RCC patients, still remain unknown and should be further investigated.
In conclusion, our analysis suggests that the site of disease may be considered as a potential predictive factor which could help, to some extent, treatment selection; further research is needed to confirm or discard these preliminary findings.
Footnotes
Acknowledgements
Editorial assistance for the preparation of this manuscript was provided by Luca Giacomelli, PhD; this assistance was supported by internal funds.
Conflict of interest statement
GP was an advisor for Bayer, Bristol-Myers Squibb, GlaxoSmithKline, Janssen, and Novartis, and received honoraria from Astellas Pharma and Pfizer. The other authors declare no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
