Abstract
Bladder carcinoma is the most common malignancy of the urinary tract. Approximately 75–85% of patients with bladder cancer present with a disease that is confined to the mucosa (stage Ta, carcinoma in situ) or submucosa (stage T1). These categories are grouped as nonmuscle invasive bladder cancer (NMIBC). Although the management of NMIBC tumours has significantly improved during the past few years, it remains difficult to predict the heterogeneous outcome of such tumours, especially if high-grade NMIBC is present. Transurethral resection is the initial treatment of choice for NMIBC. However, the high rates of recurrence and significant risk of progression in higher-grade tumours mandate additional therapy with intravesical agents. We discuss the role of various intravesical agents currently in use, including the immunomodulating agent bacillus Calmette-Guérin (BCG) and chemotherapeutic agents. We also discuss the current guidelines and the role of these therapeutic agents in the context of higher-grade Ta and T1 tumours. Beyond the epidemiology, this article focuses on the risk factors, classification and diagnosis, the prediction of recurrence and progression in NMIBC, and the treatments advocated for this invasive disease.
Epidemiology
The incidence rate of a cancer is defined as the number of new cases diagnosed per 100,000 people per year. Bladder carcinoma is the most common malignancy of the urinary tract.
Bladder cancer is nearly three times more common in men than in women [Jamal et al. 2005]. The worldwide age standardized incidence rate (ASR) is 10.1 per 100,000 for men and 2.5 per 100,000 for women [Ploeg et al. 2009]. In men, it is the fourth most common cancer after prostate, lung and colorectal cancers, accounting for 6.6% of all cancer cases. In women it is the ninth most common cancer, accounting for 2.4% of all cancers [Jamal et al. 2005]. In Europe, the highest incidence (ASR) has been reported in the western (23.6 in men and 5.4 in women) and southern (27.1 in men and 4.1 in women) regions, followed by northern Europe (16.9 in men and 4.9 in women). The lowest incidence is observed in eastern European countries (14.7 in men and 2.2 in women) [Ferlay et al. 2004].
Approximately 75–85% of patients with bladder cancer present with a disease that is confined to the mucosa [stage Ta, carcinoma in situ (CIS)] or submucosa (stage T1). These categories are grouped as nonmuscle invasive bladder cancer (NMIBC). Of these, approximately 70% present as stage Ta, 20% as T1 and 10% as CIS. Previously, these were categorized as superficial bladder cancers, but following a consensus meeting during the SIU, this name was changed, because ‘superficial’ has a more benign feeling and NMIBC better describes the different potentials of bladder tumours [Soloway, 2007].
Risk factors
Many of the aetiological factors for the development of bladder tumours are known and urologists should be aware of the types of occupational exposure that might be related to urothelial carcinogens. Aromatic amines were the first to be recognized. High-risk groups include workers in the following industries: printing, iron and aluminium processing, industrial painting, gas and tar manufacturing [McCahy et al. 1997; Zeegers et al. 2001; Samanic et al. 2008]. Another prominent risk factor is cigarette smoking, which triples the risk of developing bladder cancer [Bjerregaard et al. 2006; Puente et al. 2006]. Smoking leads to a higher mortality rate from bladder cancer during long-term follow up, even though, in a multivariate analysis, the prognostic effect of smoking was weaker than that of other factors, such as stage, grade, size and multifocality of the tumour [Aveyard et al. 2002]. Former cigarette smokers have a reduced incidence of bladder cancer compared with active smokers. [Augustine et al. 1988]. However, the reduction of this risk down to baseline (age adjusted) takes nearly 20 years after cessation of smoking, a period far longer than for the reduction of risk of cardiovascular disease and lung cancer after smoking has stopped. From a clinical standpoint it is important to realize that not only does smoking increase the risk for developing bladder cancer but also failure to quit smoking once a diagnosis is made predicts a more ominous outcome, even in those diagnosed with noninvasive initial cancers [Fleshner et al. 1999].
Classification
Tumour, node, metastasis classification
The 2002, tumour, node, metastasis (TNM) classification approved by the Union International Contre le Cancer has been widely accepted. This version was updated in 2009, but there were no changes for bladder tumours (Table 1) [Sobin et al. 2009].
Tumour, node, metastasis (TNM) classification 2009.

Histological grading of nonmuscle invasive bladder urothelial carcinomas
In 1998, a new classification of noninvasive urothelial tumours was proposed by the World Health Organization (WHO) and the International Society of Urological Pathology (ISUP) (1998 WHO/ISUP classification) and published by the WHO in 2004 (Table 2) [Epstein et al. 1998; Sauter et al. 2004].
World Health Organization (WHO) grading in 1973 and 2004.

The 2004 WHO classification of the flat lesions includes urothelial hyperplasia, reactive urothelial atypia, and atypia of unknown significance, dysplasia and CIS. Among noninvasive papillary urothelial lesions, the 2004 WHO grading differentiates between papillary urothelial neoplasm of low malignant potential (PUNLMP) and low-grade and high-grade urothelial carcinomas. PUNLMPs are defined as lesions that do not have cytological features of malignancy but show normal urothelial cells in a papillary configuration. Although they have a negligible risk for progression, they are not completely benign and still have a tendency to recur. The intermediate grade (grade 2), which was the subject of controversy in the 1973 WHO classification, has been eliminated [Burger et al. 2008; Pan et al. 2010]. It was shown that the 2004 WHO classification had a better reproducibility than the WHO 1973 classification [Hudson et al. 1995]. The prognostic value of both grading systems (WHO 1973 and 2004) has been confirmed. Attempts to demonstrate better prognostic value of one system over another, however, have yielded controversial results [Burger et al. 2008; Pan et al. 2010; May et al. 2010; Otto et al. 2011]. The majority of clinical trials published to date on TaT1 bladder tumours have been performed using the 1973 WHO classification, and therefore, the guidelines are based on this scheme. Until the prognostic role of WHO 2004 is validated by more prospective trials, both classifications are recommended for use.
Characteristics of stages Ta, T1, Tis
Stage Ta tumours are confined to the urothelium, have a papillary configuration of their exophytic part and do not penetrate from the urothelium into the lamina propria or the detrusor muscle. They are usually of low grade. Although recurrence is common, especially in the setting of multiplicity, progression is rare. Three to eighteen percent of Ta tumours are high grade, with an average of 6.9% [Sylvester et al. 2005a]. The most important factor for progression is grade, not stage.
Stage T1 tumours generate from the urothelium but penetrate the basement membrane, which separates the urothelium from the deeper layers. T1 tumours invade into the lamina propria, but not so deep that they reach the detrusor muscle. They are usually papillary; a nodular or sessile appearance suggests deeper invasion. There is significant potential for understaging in patients with nonmuscle invasive tumours, especially for those that appear to be stage T1. Many tumours are found to be more extensive than the transurethral resection (TUR) specimen indicated when patients undergo cystectomy. About one-third of patients believed to have nonmuscle invasive disease at the time of cystectomy were found to actually have muscle invasion, only half of which were organ confined [Stein et al. 2001].
Carcinoma in situ (Tis) is a high-grade (anaplastic) carcinoma confined to the urothelium, but with a flat nonpapillary configuration. Unlike a papillary tumour, Tis appears as reddened and velvety mucosa and is slightly elevated but sometimes not visible. Tis can be local or diffuse. Primary Tis (no previous or concurrent papillary tumours) can be distinguished from secondary Tis (with a history of papillary tumours) and concurrent Tis (in the presence of papillary tumours). It is high grade by definition. Although confined to the urothelium in the same manner as stage Ta, Tis is regarded as a precursor lesion for the development of invasive high-grade cancer. Between 40% and 83% of patients with Tis will develop muscle invasion if untreated, especially if associated with papillary tumours [Althausen et al. 1976]. Some studies have reported worse prognosis in concurrent CIS and T1 tumours compared with primary CIS and in extended CIS [Losa et al. 2000; Griffiths et al. 2002; Solsona et al. 2000]. Various publications have shown that the response to intravesical treatment with Bacillus Calmette-Guérin (BCG) or chemotherapy is an important prognostic factor for subsequent progression and death caused by bladder cancer. Approximately 10–20% of complete responders to BCG instillations will eventually still progress to muscle- invasive disease [Van Gils-Gielen et al. 1995; Hudson et al. 1995; Solsona et al. 2000].
Diagnosis
The most common presenting symptom of bladder cancer is painless macroscopic haematuria, which occurs in about 85% of patients [Varkarakis et al. 1974]. However, haematuria is mostly intermittent so a negative result on one or two specimens has little meaning in ruling out the presence of bladder cancer. The symptom complex of bladder irritability and urinary frequency, urgency and dysuria is the second most common presentation and is usually associated with diffuse CIS or invasive bladder cancer. However, these symptoms almost never occur without (at least) microscopic haematuria. A history of macroscopic haematuria and microscopic haematuria in high-risk patients requires further investigation of the entire urinary tract.
Cystoscopy
All patients suspected of having bladder cancer should have a cystoscopic evaluation. The diagnosis of bladder cancer ultimately depends on cystoscopic examination of the bladder and histological evaluation of the resected tissue. Cystoscopy is initially performed in the office, using flexible instruments. The diagnosis of CIS is made by a combination of cystoscopy, urine cytology and histological evaluation of multiple bladder biopsies [Kurth et al. 1995]. If a bladder tumour has been visualized in earlier imaging studies, diagnostic cystoscopy can be omitted because the patient will undergo a TUR anyhow. A careful description and documentation of the findings is necessary, including the site, size, number and appearance (papillary or solid) of the tumours, as well as a description of mucosal abnormalities. Use of a bladder diagram is recommended.
Urinary cytology – urinary molecular marker tests
Urine cytology is obtained at baseline and to establish the likelihood of high-grade disease. Malignant urothelial cells can be observed on microscopic examination of the urinary sediment or bladder washings. The limitations of microscopic cytology are due to the cytological normal appearance of cells from the well differentiated tumours and because well differentiated cancer cells are more cohesive, they are not readily shed into the urine. Therefore, microscopic cytology is more sensitive in patients with high-grade tumours or CIS. The sensitivity of cytology in CIS detection is over 90%. False-positive cytology may occur in up to 10% of patients and is usually due to urothelial atypia, inflammation, presence of stones in the urinary tract or changes caused by radiation therapy or chemotherapy [Koshikawa et al. 1989]. Cytology should be performed on fresh urine with adequate fixation. Morning urine is not suitable because of the frequent presence of cytolysis.
Molecular urinary markers have not improved the combination of cystoscopy and cytology with relation to detection of bladder tumours. The sensitivity of tests can be improved by using multiple diagnostic marker tests, as suggested by the International Consensus Panel on Bladder Tumour Markers [Lokeshwar et al. 2005], but this will increase costs and it is not clear which tumours are detected and which are still missed. Although it is hoped that by further research these tests can soon make the transition from the laboratory to the clinic, it is essential to evaluate their costs to determine whether they can provide a low-cost and reliable alternative to current cystoscopy methods [Hong and Loughlin, 2008].
Imaging
Renal and bladder ultrasound may be used during initial work-up in patients with haematuria. At the time of initial diagnosis of bladder cancer, computed tomography (CT) urography [or intravenous urogram (IVU)] should be performed only in selected cases (e.g. tumours located in the trigone or high-risk tumours). Especially in muscle-invasive tumours of the bladder and in upper tract tumours, CT urography gives more information than IVU (including status of lymph nodes and neighbouring organs). However, CT urography has the disadvantage of higher radiation exposure compared with IVU. In most centres CT urography has replaced time-consuming IVU investigations.
Therapy
Transurethral resection of bladder tumours
TUR of bladder tumours (TUR-BT) under regional or general anaesthesia is the initial treatment for visible lesions and is performed to remove all visible tumours and to provide specimens for pathological examination to determine stage and grade. Bimanual examination of the bladder should be performed under anaesthesia before prepping and draping unless the tumour is clearly small and noninvasive, and should be repeated after the resection. Fixation or persistence of a palpable mass after resection suggests locally advanced disease. The strategy of resection depends on the size of the lesion. Small tumours (<1 cm) can be resected en bloc; the specimen contains the complete tumour plus a part of the underlying bladder wall. Some experts believe that deep resection is not necessary in small, apparently low-grade lesions with a previous history of TaG1 tumour. Larger tumours should be resected separately in fractions, which include the exophytic part of the tumour, the underlying bladder wall with the detrusor muscle, and the edges of the resection area. The specimens from different fractions must be referred to the pathologist in separate containers to enable a correct diagnosis to be made. Cauterization should be avoided as far as possible during TUR to prevent tissue destruction. Complete and correct TUR-BT is essential to achieve a good prognosis [Brausi et al. 2002]. It has been confirmed that absence of detrusor muscle in the specimen is associated with a significantly higher risk of residual disease and early recurrence [Mariappan et al. 2010].
Complete visualization to plan the resection is now facilitated by the initial flexible cystoscopy. In the past it was recommended that a 70° rod lens be used, which allowed maintenance of the anatomic relationships. Bipolar electroresection allows TUR in saline and minimizes the risk of the obturator reflex that can lead to bladder perforation [Shiozawa et al. 2002; Miki et al. 2003]. The use of general anaesthesia with muscle-paralyzing agents or anaesthetic block of the obturator nerve also prevents obturator reflex for tumours located at the lateral wall of the bladder. Resection of diverticular tumours presents a particular risk of bladder wall perforation and accurate staging is difficult to achieve because the underlying detrusor is absent. Invasion beyond the lamina propria immediately involves perivesical fat or stage T3a. Partial or radical cystectomy should therefore be considered for high-grade diverticular tumours.
Complications of transurethral resection of bladder tumours
Intraoperative complications
The major complications of uncontrolled haematuria and clinical bladder perforation occur in less than 5% of cases. Cauterization is used for the bleeding site. The vast majority of perforations are extraperitoneal, but intraperitoneal perforation is possible when resecting tumours at the dome [Collado et al. 2000]. Management of extraperitoneal perforation is usually possible by prolonged urethral catheter drainage. Intraperitoneal perforation is less likely to close spontaneously and often requires open surgical repair. To prevent bladder perforation it is necessary to perform the resection if the bladder is not overdistended.
Postoperative complications
Minor bleeding and irritative symptoms are common side effects in the immediate postoperative period, but sometimes clot retention can occur, especially if an extensive resection was performed. When tumours are located at or near the ureteral orifice, obstruction of the upper tract can occur. Therefore, in these situations a postoperative ultrasound should be performed.
Bladder and prostatic urethral biopsies
Biopsies of any suspicious areas are an important part of a complete evaluation. CIS can present as a velvet-like, reddish area that is indistinguishable from inflammation, or it might not be visible at all. It can be present as an isolated lesion without exophytic tumour or it can accompany TaT1 tumours. When abnormal areas of urothelium are seen targeted ‘cold cup’ biopsies or biopsies with a resection loop should be taken. Biopsies from normal-looking mucosa, so-called random biopsies (R-biopsies), should be performed in patients with positive (high-grade) urinary cytology and in the absence of visible tumour in the bladder. It is recommended that R-biopsies are taken from the trigone, the bladder dome, the right, left, anterior and posterior bladder walls, and the prostate in men. The current consensus is that random biopsies are not indicated in low-risk patients, that is, those with low-grade papillary tumours and negative cytology. The likelihood of detecting CIS, especially in low-risk tumours, is extremely low (<2%) [van der Meijden et al. 1999]. Also, cold cup biopsies from normal-looking mucosa should be performed when the exophytic tumour has a nonpapillary appearance. Material obtained by random or directed biopsies must be sent for pathological assessment in separate containers. In CIS, the coherence and adherence of epithelial cells is decreased, and this feature often results in denuded biopsies when taken by cold cup or a resection loop [Levi et al. 2001]. Involvement of the prostatic urethra and ducts with urothelial cancer in men with NMIBC has been reported [Solsona et al. 1996].
Although the exact risk of prostatic urethra or duct involvement is not known, it seems to be higher if the tumour is located on the trigone or bladder neck, in the presence of bladder CIS and multiple tumours [Matzkin et al. 1999; Mungan et al. 2005]. In these cases and when cytology is positive, with no evidence of tumour in the bladder, or when abnormalities of prostatic urethra are visible, biopsies of the prostatic urethra are recommended. The biopsy is taken from abnormal areas and from the precollicular area (between the 5 and 7 o’clock position) using a resection loop. Also, a prostatic urethral biopsy using the cutting loop may be performed if orthotopic bladder creation is anticipated for high-risk disease [Holzbeierlein et al. 2000].
Fluoresence cystoscopy
Endoscopically, urologists can suspect malignancy based only on the presence of visible changes such as tumours or ‘red spots’. The imperfect sensitivity of cystoscopy potentially explains the high rate of cancer recurrence soon after ‘complete’ removal of all visible tumours.
Photodynamic diagnosis (PDD) is performed using violet light after intravesical instillation of 5-aminolaevulinic acid or hexaminolaevulinic acid. Previous studies have confirmed that fluorescence-guided biopsy and resection are more sensitive than conventional procedures for detection of malignant tumours, particularly for CIS [Jichlinski et al. 2003; Schmidbauer et al. 2004; Hungerhuber et al. 2007].
PDD is most useful for detection of CIS, and therefore, it should be restricted to patients who are suspected of harbouring a high-grade tumour; for example, for biopsy guidance in patients with positive cytology or with a history of high-grade tumour. Because of conflicting data on the reduction in recurrence rate using PDD in papillary tumours, the use of PDD is still questioned in these cases [Stenzl et al. 2010; Witjes et al. 2010]. The additional costs of the equipment and instillation for PDD should be taken into account.
Narrow band imaging (NBI) is a new endoscopic technique that is used to improve the detection of NMIBC. NBI uses light of a narrow bandwidth with centre wavelengths in the blue and green spectrum of light to increase the contrast of the mucosa and small vascular structures with a high resolution, without the use of intravesical instillations. NBI cystoscopy, in addition to white light cystoscopy, improves the detection of NMIBC and might lead to a more complete resection, thereby possibly resulting in fewer early tumour recurrences. However, further validation of the technique is required with comparative studies [Cauberg et al. 2011].
Second resection
There is a significant risk of residual tumour after the initial TUR of TaT1 tumours [Brausi et al. 2002; Miladi et al. 2003]. Repeat TUR-BT is usually appropriate in the evaluation of T1 tumours because a repeat TUR can demonstrate worse prognostic findings in up to 25% of specimens [Schwaibold et al. 2000]. A second TUR should be considered when the initial resection is incomplete; for example, when multiple and/or large tumours are present, or when the pathologist has reported that the specimen contains no muscle tissue (TaG1 excluded). Furthermore, a second TUR should be performed when a high-grade or T1 tumour has been detected at initial TUR [Kulkami et al. 2010]. Previous studies have demonstrated that a second TUR can increase recurrence-free survival [Grimm et al. 2003; Lopez-Beltran et al. 2004; Divrik et al. 2006].
There is no consensus about the strategy and timing of a second TUR. Most authors recommend resection 2–6 weeks after the initial TUR. The procedure should include resection of the primary tumour site.
Recurrence progression
TaT1 tumours
Patients with a TaT1 bladder tumour are divided into low-risk, intermediate-risk and high-risk groups [Millán-Rodríguez et al. 2000a, 2000b]. When using these risk groups, however, no distinction is usually drawn between the risk of recurrence and progression. Although prognostic factors indicate a high risk for recurrence, the risk of progression might still be low, and other tumours might have a high risk of recurrence and progression. To predict separately the short-term and long-term risks of recurrence and progression in individual patients, the European Organization for Research and Treatment of Cancer (EORTC)-Genitourinary (GU) Group developed a scoring system and risk tables (Tables 3–5) based on the six most significant clinical and pathological factors [Sylvester et al. 2006]:
Weighting used to calculate recurrence and progression scores.
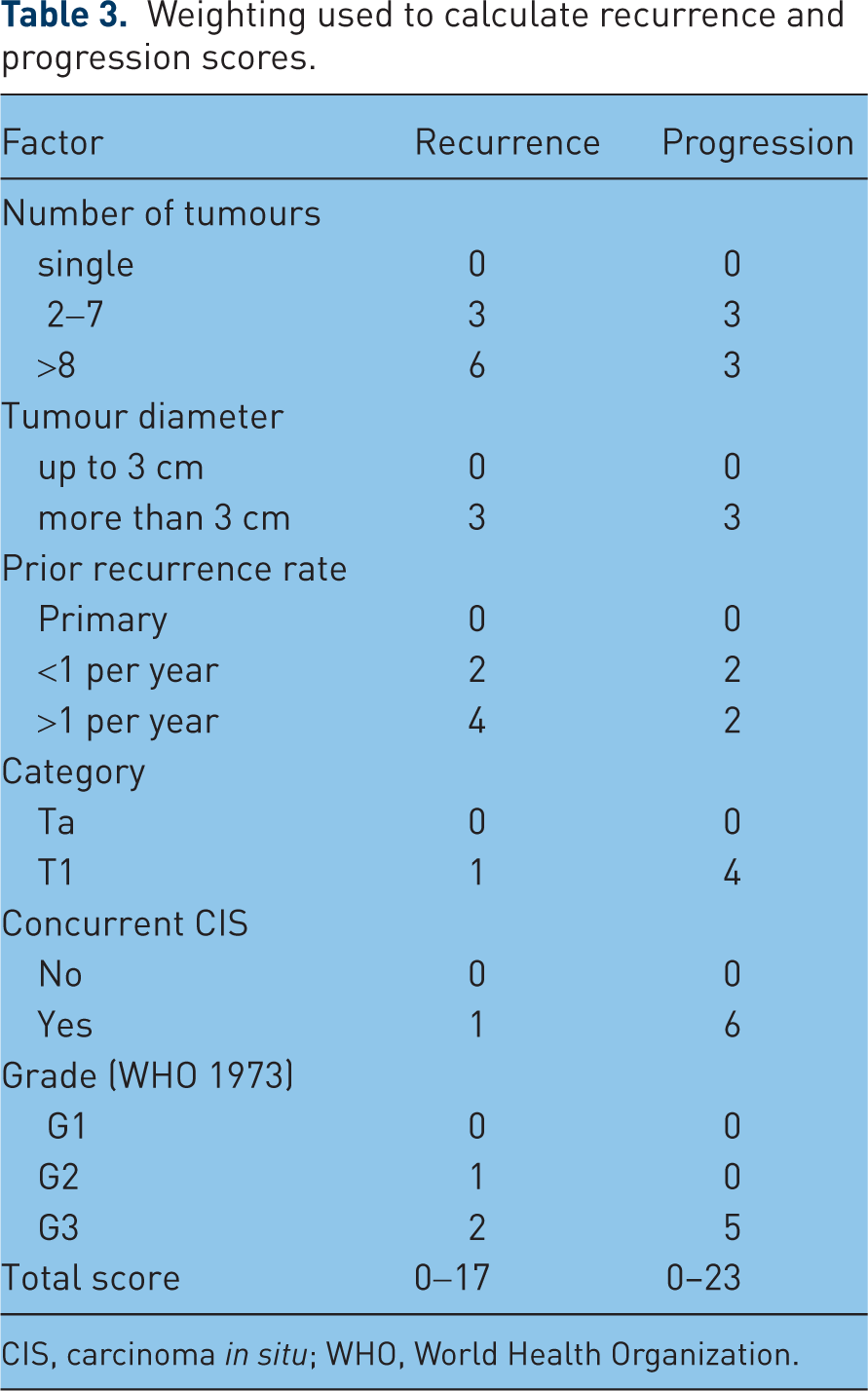
CIS, carcinoma in situ; WHO, World Health Organization.
Probability of recurrence according to total score.
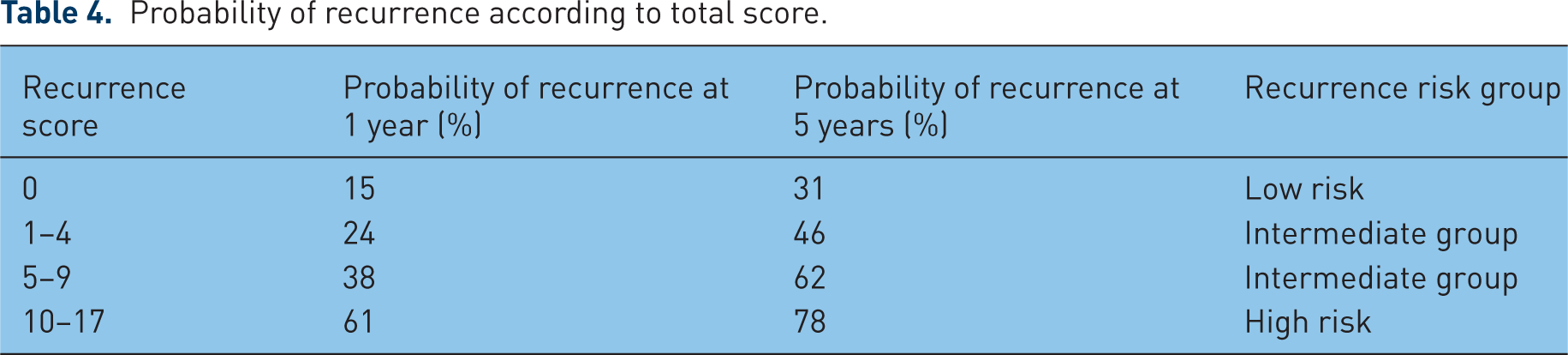
Probability of progression according to total score.

tumour size,
prior recurrence rate,
T-category,
presence of concurrent CIS,
tumour grade,
number of tumours.
Carcinoma in situ
Patients with CIS are always categorized in the high-risk group. Without any treatment, approximately 54% of patients with CIS progress to muscle-invasive disease [Lamm et al. 1992]. Unfortunately, there are no reliable prognostic factors that can be used to predict the course of the disease and specify the most dangerous cases. Various publications have shown that the response to intravesical treatment with BCG or chemotherapy is an important prognostic factor for subsequent progression and death caused by bladder cancer [Van Gils-Gielen et al. 1995; Hudson and Herr, 1995; Solsona et al. 2000; Chade et al. 2010]. Approximately 10–20% of patients who have a complete response eventually progress to muscle-invasive disease compared with 66% of those who do not respond [Van Gils-Gielen et al. 1995; Hudson and Herr, 1995; Solsona et al. 2000].
Adjuvant treatment
Perioperative intravesical therapy
It is believed that tumour cell implantation immediately after resection is responsible for early recurrences and this has been used to explain the observation that initial tumours are most commonly found on the trigone and lower side walls of the bladder, whereas recurrences are often located near the dome [Heney et al. 1981]. Thus the assumption is that intravesical chemotherapy can kill these cells before implantation. A high variability in the 3-month recurrence rate has been demonstrated, indicating that TUR is probably incomplete in a large number of cases due to overlooked tumours or recurrences in a high percentage of patients [Brausi et al. 2002]. Therefore, adjuvant therapy should be considered in all patients and resection techniques improved. It needs to be shown that an improvement in resection technique (a complete resection of all tumours) is perhaps more important than adjuvant instillations. The choice of therapy may be considered on an individual basis according to what risk is acceptable for the patient and the urologist.
One immediate instillation of chemotherapy within 6–24 h of TUR was shown to significantly reduce recurrence rate compared with TUR alone [Sylvester et al. 2004]. Further studies are required, however, to determine the definitive role of immediate chemotherapy before BCG or further chemotherapy instillations in intermediate- and high-risk groups. There is no single chemotherapeutic drug that is superior with regard to efficacy. Mitomycin C, epirubicin and doxorubicin have all shown a beneficial effect [Sylvester et al. 2004]. So, an early immediate instillation is indicated in tumours at low risk of progression (single, primary, papillary lesions) as the only intravesical treatment and in those presumably at intermediate risk, for which a single instillation is considered as the initial stage of further intravesical therapy. In tumours that are presumably at high risk of recurrence/progression (solid lesions, positive urinary cytology), an immediate instillation is an option because it can have a positive impact on recurrence rate through prevention of tumour cell implantation. However, whether this immediate instillation in combination with a course of further adjuvant treatment improves the outcome compared with a course of adjuvant treatment alone remains to be determined.
In case a bladder perforation is suspected after the TUR-BT or if significant haematuria is present, the immediate postoperative chemotherapy instillation should be omitted.
Additional adjuvant intravesical chemotherapy
For patients at low risk of tumour recurrence (and without a suspected bladder wall perforation), a single immediate instillation of a chemotherapeutic agent is considered the standard treatment. No further treatment should be given in these patients before subsequent recurrence. For other patients, however, a single immediate instillation remains an incomplete treatment because the likelihood of recurrence and/or progression is considerable. The effect of the immediate instillation of chemotherapy occurs during the first and second year [Solsona et al. 1999; Hinotsu et al. 1999]. The choice between further chemotherapy and immunotherapy largely depends on the risk that needs to be reduced: recurrence or progression. A combined analysis of the EORTC-GU Group and Medical Research Council data, comparing intravesical chemotherapy with TUR alone, demonstrated that chemotherapy prevents recurrence but not progression [Pawinski et al. 1996]. The ideal duration and intensity of the instillation schedule remains undefined [Sylvester et al. 2008]. The available data do not support any treatment longer than 1 year. If chemotherapy is given, the drug should be used at its optimal pH and the concentration of the drug maintained during instillation by reducing fluid intake. One of the instillation schedules used in an adjuvant setting is weekly mitomycin C 40 mg for 6 weeks, and in the case of negative cystoscopy, monthly instillations for 1 year.
Intravesical BCG immunotherapy
Intravesical immunotherapy results in a massive local immune response characterized by induced expression of cytokines in the urine and bladder wall and by an influx of granulocytes and mononuclear cells. BCG is an attenuated mycobacterium developed as a vaccine for tuberculosis. The vaccine is reconstituted with 50 ml of saline and should be administered through a urethral catheter under gravity drainage soon thereafter because aggregation occurs [Ratliff et al. 1994]. The exact antitumour action of BCG has not been elucidated until now, but several groups have demonstrated that an increase in T helper 1 urinary cytokines (e.g. gamma, interluekin-2) following BCG instillations predicts an improved outcome [de Reijke et al. 1996; Ratliff et al. 1986; Haaff et al. 1986].
Four meta-analyses have confirmed that BCG after TUR is superior to TUR alone or TUR and chemotherapy for the prevention of recurrence of NMIBC in patients with Ta and T1 tumours [Shelley et al. 2001; Han et al. 2006], at high risk of tumour recurrence [Shelley et al. 2004] and intermediate- or high-risk status [Böhle et al. 2003]. Also, there are studies which have confirmed the superiority of BCG over the combination of epirubicin and interferon [Duchek et al. 2010], mitomycin C [Järvinen et al. 2009] or epirubicin [Sylvester et al. 2010] alone for prevention of tumour recurrence, in intermediate- and high-risk tumours. Although BCG is a very effective treatment, there is a consensus that not all patients with NMIBC should be treated with BCG due to the risk of toxicity. Ultimately, the choice of treatment depends on the patient’s risk of recurrence and progression (see Tables 4 and 5). The use of BCG does not alter the natural course of tumours at low risk of recurrence (see Tables 4 and 5), and could be considered to be overtreatment for this patient category. In patients with tumours at high risk of progression, for whom cystectomy is not an option, BCG including at least 1 year of maintenance is indicated.
In patients at intermediate or high risk of recurrence and intermediate risk of progression, BCG with 1 year of maintenance is more effective than chemotherapy for prevention of recurrence; however, it has more side effects than chemotherapy. For this reason BCG with maintenance and intravesical chemotherapy both remain an option. The efficacy of BCG after TUR-BT for high-risk papillary disease has been demonstrated in several series of T1 lesions, with recurrence rates of 16–40% and progression rates of 4.4–40%, a substantial improvement compared with TUR- BT alone [Cookson and Sarosdy, 1992; Pansadoro et al. 1995; Herr, 1997; Jimenez-Cruz et al. 1997; Gohji et al. 1999; Hurle et al. 1999].
For optimal efficacy, BCG must be given in a maintenance schedule, consisting of weekly instillations for 6 weeks and three weekly instillations at month 3, 6 and 12, if cystoscopy and cytology are negative [Böhle et al. 2003; Malmström et al. 2009; Sylvester et al. 2010]. If a recurrence is found at the 3-month cystoscopy, again 6 weeks of BCG instillations can be given followed by the maintenance schedule as described above. In the EORTC-GU Group meta-analysis, only patients who received maintenance BCG benefited. Induction BCG instillations are classically given according to the empirical 6-weekly induction schedule that was introduced by Morales in 1976 [Morales et al. 1976]. The optimal number of induction instillations and the optimal frequency and duration of maintenance instillations remain unknown [Zlotta et al. 2000]. Treatments usually begin 2–4 weeks after the TUR-BT, allowing time for the re-epitheliazation to minimize the potential for intravasation of live bacteria [Lamm, 1992a, 1992b]. In the event of a traumatic catheterization, macroscopic haematuria and symptomatic urinary tract infection, the treatment should be delayed for several days.
BCG toxicity
Serious side effects are encountered in less than 5% of patients and can be effectively treated in virtually all cases [van de Meijden et al. 2003]. The side effects can be divided into local (e.g. voiding complaints, haematuria) and systemic side effects (e.g. fever, hepatitis, pneumonitis, allergic reactions). Major complications can appear after systemic absorption of the drug. Thus, BCG should not be administered during the first 2 weeks after TUR-BT in patients with macroscopic haematuria, symptomatic urinary tract infection or after traumatic catheterization. BCG should not be used in immunocompromised patients (immunosuppression, HIV) [Lamm et al. 1992]. Although these are the recommendations, personal experience has revealed no major side effects in patients undergoing renal transplantation. Before applying intravesical BCG therapy the urologist should be aware of how to recognize and treat BCG-induced complications.
Morbidity secondary to intravesical BCG may present both locally and systemically. Most patients suffer a self-limited irritative voiding syndrome. Often there are not unified criteria for the management of BCG side effects. Irritative voiding symptoms are among the most frequent symptoms, generally self-limited, but if these persist (>48 h) the urologist will have to treat them depending on their intensity and duration with symptomatic therapy (e.g. anticholinergics). Macroscopic haematuria is not infrequent and diminishes with an expectant approach and water intake, however a urinary tract infection or residual tumour should be excluded. A febrile syndrome, if present, is usually self-limited to the first 24–48 h and below 38.5°C without general status affectation. The majority of local and systemic side effects are seen during the induction and the first half year of maintenance. During further maintenance BCG toxicity does not increase and instillations are generally well tolerated [Van der Meijden et al. 2003]. Recognition of risk factors, particularly traumatic catheterization or concurrent cystitis, that result in systemic BCG absorption, as well as the prompt and appropriate treatment of early side effects should significantly decrease the incidence of severe toxicity [Lamm et al. 1992].
Patients who develop mild symptoms of cystitis following BCG instillation, even if accompanied by low-grade fever, usually do not require specific therapy other than analgesics. As noted above, symptoms usually resolve within 48 h.
Acute fever higher than 39.0°C may occasionally develop but does not necessarily signify BCG infection. In this setting it may be impossible to distinguish an infectious from a noninfectious event. Such patients are best seen, evaluated and sometimes hospitalized for observation. Empiric therapy with a fluoroquinolone antibiotic should be considered until the etiology of the fever is established. A fluoroquinolone will treat the majority of non-BCG bacterial urinary tract infections and has reasonable antimycobacterial activity.
The cardinal sign of BCG infection is a relapsing fever with drenching night sweats persisting beyond 48 h. For acute symptoms or those that persist beyond 48 h, antituberculous therapy should be prescribed. Knowing when to administer and when to withhold BCG will prevent most complications, but even when all precautions are taken, some complications will occur. The initial step in the treatment of infectious complications is the use of isoniazid. Routine prophylactic isoniazid should not be given because animal studies have confirmed that immune stimulation, and presumably antitumour activity, can be inhibited by isoniazid prophylaxis. However, when cystitis persists for more than 2 days or is so severe that it does not respond to symptomatic treatment, isoniazid 300 mg daily is used to control the symptoms, prevent progressive infection and avoid the overgrowth of BCG, which can result in excessive organisms and suppression of the immune response. If symptoms progress despite isoniazid treatment or do not begin to abate within 1–2 weeks, rifampicin 600 mg daily is added. Rifampicin is given from the beginning in patients with potentially severe extravesical BCG infection such as pneumonitis, hepatitis or nephritis. In patients with symptoms such as fever, malaise or bladder irritation that respond within a few days, it is generally necessary to continue antituberculous antibiotics for only 2 weeks [Lamm, 1992a, 1992b; Rodriguez et al. 2009]. Patients with extravesical infection and those who do not respond promptly to treatment are treated for 3 months, and those with severe or deep-seated infection are treated for 6 months. Patients with disseminated infection should also be hospitalized and treated with antituberculous agents with or without glucocorticoids. Patients who develop infectious complications severe enough to require antituberculous therapy should generally not receive further BCG bladder instillations.
Prophylactic ofloxacin decreased the incidence of moderate to severe adverse events associated with BCG intravesical therapy. Compliance with induction and maintenance therapy may be improved by adjuvant ofloxacin therapy. However, long-term comparative studies with other preventive strategies must be carried out to confirm these findings [Colombel et al. 2006].
Several attempts to decrease BCG-induced toxicity have been published. The Spanish group found that dose reduction of BCG resulted in less toxicity with similar efficacy in low- and intermediate-risk patients. However, in high-risk patients the full dose is still advised [Martinez-Pineiro, 2002]. Another strategy could be to reduce the number of instillations (two instead of six during the induction weeks) because the immune reaction was shown to be more favourable in mice [de Boer et al. 2005].
Carcinoma in situ
If concurrent CIS is found in association with muscle-invasive bladder cancer, therapy is determined according to the invasive tumour. The detection of CIS with TaT1 tumours increases the risk of recurrence and progression of TaT1 tumours [Sylvester et al. 2006; Fernadez-Gomez et al. 2009] and further treatment is mandatory. CIS cannot be cured by an endoscopic procedure only. Histological diagnosis of CIS must be followed by further treatment, either intravesical BCG instillations or radical cystectomy. No consensus exists about whether conservative therapy (intravesical BCG instillations) or aggressive therapy (cystectomy) should be done, especially in combination with concurrent high-grade papillary tumours. There is a lack of randomized trials comparing instillation therapy and early cystectomy as immediate primary treatment. Tumour-specific survival rates after early cystectomy for CIS are excellent, but as many as 40–50% of patients might be overtreated [Van der Meijden et al. 2005].
The American Urological Association Guidelines and the European Association of Urology (EAU) guidelines supported BCG as the preferred initial treatment option for CIS [Smith et al. 1999; Sylvester et al. 2005b].
Treatment of failure following intravesical therapy
Patients with high-grade Ta, T1 or CIS are at high risk for recurrence and, more importantly, progression. Thus, both the American Urological Association and the EAU recommend initial intravesical treatment with BCG followed by maintenance therapy for a minimum of 1 year. The complete response rate to BCG therapy in patients with high-risk NMIBC can be as high as 80%. However, most patients with high-risk disease suffer from recurrence. Treatment with BCG is considered to have failed in the following situations:
When a muscle-invasive tumour is detected during follow up.
When a high-grade, nonmuscle invasive tumour is present at both 3- and 6-month follow up. In patients with a tumour present at 3 months, an additional BCG course can achieve a complete response in over 50% of cases, both in patients with papillary tumours and CIS [Herr et al. 2003], but with increasing risk of progression [Gallagher et al. 2008; Lerner et al. 2009].
Any worsening of the disease under BCG treatment, such as a higher number of recurrences, higher T-stage or higher grade, or the appearance of CIS, in spite of an initial response.
In patients whose condition fails to respond to conservative treatment with noninvasive recurrences and who refuse surgical therapy or are not suitable candidates for surgery, the treatment options become even more complicated. Patients with nonmuscle invasive recurrence of urothelial bladder carcinoma after intravesical chemotherapy can benefit from BCG instillations [Huncharek et al. 2004].
However, if this treatment fails again, the treatment options are limited and include an alternative immunotherapy regimen, low-dose BCG plus interferon-alpha [Gallagher et al. 2008], chemotherapy with intravesical gemcitabine [Mohanty et al. 2008; Di Lorenzo et al. 2010] or docetaxel [Barlow et al. 2009]. Device-assisted therapy with electromotive mitomycin C given sequentially with BCG might not only reduce the recurrence rate but also reduce progression and disease-specific mortality, although currently there is no trial in a specific population with ‘BCG failure’ [Yates and Roupret, 2010].
Another bladder-preserving treatment is chemotherapeutic instillations combined with hyperthermia [Witjes et al., 2009; Colombo et al. 2011]. It was shown that in patients with primary CIS whose condition failed to respond to BCG-CIS treatment with chemotherapy using the Synergo® system (Synergo ® Medical Enterprises, Amstelveen,The Netherlands), complete responses of around 50% after 2 years could be achieved. However, more data are needed. To achieve a more homogeneous heating of the bladder we have started a trial with external hyperthermia in combination with mitomycin instillations. The results of this approach are awaited.
To date, however, further research is necessary for all conservative secondary treatment options to determine which might be the most efficacious. All conservative treatments should be considered investigational. Currently, cystectomy remains the standard of care for high-risk patients whose condition has failed to respond to BCG therapy [Lightfoot et al. 2011].
Urine markers have been studied extensively to help diagnose bladder cancer and thereby decrease the need for cystoscopy. However, no marker is available at present that can sufficiently warrant this. Several urinary markers have higher but still insufficient sensitivity compared with cytology. To identify an optimal marker that can delay cystoscopy in the diagnosis of bladder cancer, large prospective and standardized studies are needed [Tilki et al. 2011]. The same is true for prognostic markers, which could help in selecting patients who need aggressive therapy from the beginning to prevent delay of effective treatment and increasing the risk to the patient.
The role of early cystectomy
Many cases of high-grade NMIBC will progress to invasion and with the risk of cancer death. Although the initial response rate to BCG therapy in patients with CIS can be above 80%, patients whose condition fails to respond have a 50% chance of disease progression and a higher likelihood of disease-specific mortality [Catalona et al. 1987; Nadler et al. 1994]. Therefore, some experts consider it reasonable to propose immediate cystectomy to patients with NMIBC who are at high risk of progression. According to the risk tables of the EORTC (Tables 3–5) these patients have multiple recurrent high-grade tumours, high-grade T1 tumours or high-grade tumours with concurrent CIS. In these patients the following treatment options should be discussed: immediate cystectomy and conservative treatment with BCG instillations. Patients should be informed about the benefits and risks of both approaches.
Delay of cystectomy in these patients might lead to decreased disease-specific survival [Raj et al. 2007]. In patients in whom cystectomy is performed at the time of pathological high-risk nonmuscle-invasive disease, the 5-year disease-free survival exceeds 80% [Stein et al. 2001; Madersbacher et al. 2003; Hautmann et al. 2006; Shariat et al. 2006a, 2006b, 2006c; Ghoneim et al. 2008].
Follow up
Cystoscopy is the hallmark of surveillance
The EAU recommendations for follow up in patients after TUR of NMIBC are [Leblanc et al. 1999; Zeeger et al. 2000; Oge et al. 2000; Holmang et al. 2001; Fujii et al. 2003; Borhan et al. 2003; Soloway et al. 2003; Gofrit et al. 2006; Sylvester et al. 2006]:
Patients with TaT1 tumours at low risk of recurrence and progression (Tables 3–5) should have a cystoscopy at 3 months. If negative, the following cystoscopy is advised 9 months later, and then yearly for 5 years.
Patients with TaT1 tumours at high risk of progression and those with CIS should have a cystoscopy and urinary cytology at 3 months. If negative, the following cystoscopy and cytology should be repeated every 3 months for a period of 2 years, and every 6 months thereafter until 5 years, and then yearly. Yearly imaging of the upper tract is recommended.
Patients with TaT1 tumours at intermediate risk of progression (about one-third of all patients) should have an in-between follow-up scheme using cystoscopy and cytology, which is adapted according to personal and subjective factors.
During follow up in patients with positive cytology and no visible tumour in the bladder, R-biopsies or biopsies with PDD (if equipment is available) and investigation of extravesical locations (CT urography, prostatic urethra biopsy) are recommended.
A number of tumour markers have shown the ability to improve upon the sensitivity of cytology, but specificity is lower for most. Because of these facts, there are no markers that are accurate enough to replace cystoscopy or cytology in the follow up of NMIBC. Change in lifestyle habits can be advised and the most important are an increase in fluids, smoking cessation and a low-fat diet.
New imaging developments
Optical coherence tomography
Optical coherence tomography (OCT) is a novel, real-time endoscopic imaging modality that permits delineation of microarchitectural features of bladder lesions. It may provide an extension of conventional cystoscopy by allowing noninvasive examination of bladder tissue at microscopic resolution (10–20 µm) [Goh et al. 2008]. It is not possible to examine the whole bladder using OCT, but it could be helpful to decrease the high number of false-positive biopsies if photodynamic diagnosis is used because OCT can provide the urologist with direct information on the architecture of the suspect area. Since this technique looks at the microarchitecture of the lesions in the bladder, it could also be used to grade the tumours in a more objective manner because the grading systems are known to be subject to a high inter- and intra-observer variability. Based on OCT-measured optical attenuation [µ(t)], the grade of bladder urothelial carcinoma could be assessed in real time [Cauberg et al. 2010].
Raman molecular imaging
Raman molecular imaging (RMI) is an optical technology that combines the molecular chemical analysis of Raman spectroscopy with high-definition digital microscopic visualization. This approach also permits visualization of the physical architecture and molecular environment of cells in the urine. In 1995, Feld and colleagues first recorded Raman spectra from the bladder. They showed that bladder cancer has a greater nucleic acid content and lower lipid content than normal bladder urothelium [Feld et al. 1995]. Urothelium, lamina propria and muscle layers could be clearly distinguished based on Raman spectra. Lamina propria spectra were dominated by signal contributions of collagen and the smooth muscle layer showed strong signal contributions of actin. The urothelium had a relatively strong lipid signal contribution. This technology can be applied in vivo by thin, flexible fibre optic catheters for analysis of the molecular composition of the normal and pathological bladder without the need for biopsies [de Jong et al. 2002]. A three-group algorithm constructed by Raman spectroscopy differentiated normal bladder, cystitis and urothelial carcinoma/CIS with over 90% sensitivity and specificity. In addition, this could accurately characterize urothelial carcinoma into low (G1/G2) or high (G3) grade and superficial (pTa) or invasive (pT1/pT2) stage. It has the potential to provide immediate pathological diagnoses during TUR-BT [Crow et al. 2004].
Draga and colleagues studied the feasibility of Raman spectroscopy for the diagnosis of bladder cancer in vivo. They demonstrated that high- volume Raman spectroscopy could be used in vivo as an objective clinical tool for real-time staging of bladder cancer invasion [Draga et al. 2010a, 2010b].
New drugs
Apaziquone
Apaziquone is a promising drug for intravesical use in patients with NMIBC [Witjes and Kolli, 2008]. The complete response of a marker lesion in 67% of patients was followed by a recurrence-free rate of 56.5% at 1-year follow up and 49.5% at 2-year follow up. These long-term results are good in comparison with the results of other ablative studies [Hendricksen et al. 2009]. Early recurrences after treatment with apaziquone are infrequent and the interval to recurrence is significantly longer compared with the historical recurrence rates for these patients. Larger prospective randomized trials are warranted to confirm these results [Jain et al. 2009]. Local side effects were comparable to side effects due to other chemotherapy instillations [van der Heijden et al. 2006].
Gemcitabine
Gemcitabine seems especially promising for the treatment of intermediate-risk NMIBC. Gemcitabine has a molecular weight of 299 D, lower than that of commonly used intravesical chemotherapeutic agents such as mitomycin C (389 D) and doxorubicin (589 D). This may enable gemcitabine to penetrate the bladder mucosa with beneficial effects in the treatment of early invasive bladder cancer (T1 disease). At the same time the molecular weight is high enough to prevent significant systemic absorption in an intact bladder [Contero and Frea, 2006]. The safety of gemcitabine is tested in different instillation schemes, drug concentrations and administered volumes. Its safety profile is excellent, with good tolerability and minimal toxicity up to 2000 mg/50 ml for 2 h instillations. In comparison with other drugs, the ablative efficacy of gemcitabine is good. The first studies on prophylactic efficacy in intermediate-risk, high-risk and BCG-refractory patients are promising but limited by the small number of patients studied. Exploring phase II and comparative randomized phase III studies should provide additional information on gemcitabine and its benefit to clinical practice [Hendricksen and Witjes, 2007].
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for- profit sectors.
All authors state that they have no conflict of interest.
