Abstract
Background:
For treatment of relapsing-remitting multiple sclerosis (RRMS), a broad range of disease-modifying therapies (DMT) is available. However, few comparative effectiveness studies between different drugs have been performed.
Objectives:
This study aimed to compare the efficacy and treatment continuation of natalizumab and ocrelizumab in a real-world cohort of patients with relapsing-remitting multiple sclerosis (RRMS) from two German university hospitals.
Methods:
We performed a retrospective analysis of RRMS patients who initiated treatment with natalizumab or ocrelizumab between January 2016 and April 2019 at the German university hospitals of Mainz and Düsseldorf. Bayesian propensity score matching was conducted to correct for differences in baseline characteristics. Our primary outcome was no evidence of disease activity [NEDA-3: no relapses, no confirmed disability progression, and no magnetic resonance imaging (MRI) activity] and its subcomponents. Secondary outcomes included measurement of neurofilament light chain (NfL) in serum, analysis of premature discontinuation, and evidence of rebound activity in patients switching from natalizumab to ocrelizumab.
Results:
We identified 63 patients starting treatment with natalizumab and 76 patients starting with ocrelizumab. Binary logistic regression showed that treatment with natalizumab or a higher number of relapses in the previous year were independently associated with a higher risk for relapses. Patients receiving natalizumab had a higher probability of premature discontinuation of therapy (p = 0.002). After propensity score matching of the two treatment arms, 55 patients remained per group. NEDA-3 after 30 months of follow-up was reached by 53.1% in the ocrelizumab group and 36.1% in the natalizumab group (p = 0.177). Ocrelizumab was superior to natalizumab concerning the occurrence of relapses in log-rank test (p = 0.019). NfL levels in serum were low under both treatments. Patients who switched from natalizumab to ocrelizumab showed no increased rebound activity.
Discussion:
This study provides class IV evidence that treatment of RRMS patients with ocrelizumab and natalizumab show comparable effectiveness in combined endpoints, while ocrelizumab might be more effective in preventing the occurrence of relapses.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system. 1 For treatment of relapsing-remitting multiple sclerosis (RRMS), a broad range of disease-modifying therapies (DMTs) is available. By targeting different aspects of autoimmune inflammation, these therapies reduce relapse rates and disability progression. 2 Although a certain hierarchy of therapies is acknowledged in clinical practice, comparative effectiveness studies between different drugs are scarce, and thus, the ability to provide evidence-based advice to patients is limited.3,4
Natalizumab is a humanized neutralizing monoclonal antibody directed against α4 integrin that blocks the transmigration of lymphocytes across the blood–brain barrier. 5 It was approved for treatment of RRMS after showing beneficial effects in two large phase III trials. In the AFFIRM study, 67% of patients treated with natalizumab remained relapse-free after 2 years compared with 41% in the placebo group, and both clinical progression and magnetic resonance imaging (MRI) activity were significantly decreased upon treatment with natalizumab. 6 The SENTINEL trial showed superior efficacy of a combination therapy of natalizumab and interferon beta-1a compared with interferon beta-1a alone. 7
A rare but often fatal adverse event in natalizumab-treated patients is progressive multifocal leukoencephalopathy (PML) caused by John Cunningham virus (JCV). Therefore, PML risk stratifications are continuously improved based mainly on JCV index values, prior immunosuppressive therapy, and length of natalizumab treatment.8–10 Moreover, extended interval dosing (EID) of natalizumab is an increasingly established alternative for patients at risk. 11 In this context, a recent phase IIIb trial showed a higher number of new or enlarging T2 lesions, but a similar proportion of patients with new MRI lesions, relapses, or confirmed disability worsening when receiving natalizumab once every 6 weeks compared with once every 4 weeks. 12 Finally, patients with a positive anti-JCV antibody status are usually switched to another DMT depending on yearly benefit–risk re-evaluation according to individualized prediction models, 8 although this may be a challenging task as cessation of natalizumab treatment often results in severe rebound activity. 13 Nevertheless, treatment with natalizumab is well established and effective, also in the long term. 14
Another highly effective DMT is ocrelizumab, a recombinant humanized monoclonal antibody against CD20-positive B cells. Two phase III trials in RRMS, OPERA I and II, revealed that 47.9% and 47.5%, respectively, of patients treated with ocrelizumab had no evidence of disease activity (NEDA) by 96 weeks, compared with 29.2% and 25.1%, respectively, in the interferon beta-1a group. 15 NEDA is a concept of combined disease-relevant outcomes. The most commonly applied form used in both clinical trials and real-world studies is NEDA-3, defined as no relapses, no confirmed disability progression, and no active MRI lesions (both new or enlarged T2 lesions and contrast-enhancing lesions). 16 Relevant long-term side effects of ocrelizumab are occurrence of hypogammaglobulinemia and increased risk of infections, comparable with other B cell–depleting drugs.17–19 For example, treatment with ocrelizumab was associated with a more severe disease course of SARS-CoV-2 in a large international cohort. 20
Both natalizumab and ocrelizumab are high-efficacy DMT for RRMS (and in the case of ocrelizumab also for primary progressive MS), and are commonly used to treat patients with severe disease activity. Although they can be administered as a first-line treatment (‘induction’), most patients are initially treated with low-risk platform agents and then switched to high-efficacy DMT in case of disease activity (‘treatment escalation’). Alternatively, for example, in case of side effects, patients may change to a drug of comparable efficacy, but different mode of action (so-called ‘lateral switch’). Therefore, real-world patient cohorts differ from cohorts in the phase III trials, in which therapy–naïve patients or those switching from injectable substances are preferably included.6,15 In contrast to trial cohorts, real-world studies represent heterogeneous patient populations concerning factors such as age, Expanded Disability Status Scale (EDSS) score, and comorbidities. Moreover, as both study designs and control groups differ between the trials for natalizumab and ocrelizumab, it is not possible to draw any direct conclusions on comparative effectiveness. Consequently, a comprehensive risk–benefit assessment critically depends on the evaluation of data collected by comparative observational studies done in the real-world setting. In this study, we therefore wanted to compare the efficacy and treatment continuation of natalizumab and ocrelizumab in a real-world RRMS cohort from two German university hospitals. To this aim, we compared NEDA-3 and its subcomponents as well as neurofilament light chain (NfL) levels under both treatments, and analyzed reasons for premature discontinuation (PD) and rebound activity after natalizumab cessation.
Methods
Study design
This was an independent retrospective cohort study including patients from two German university hospitals (Mainz and Düsseldorf). Clinical and MRI data were collected prospectively by each MS center at routine clinic visits according to the national treatment guidelines. Data were then collected retrospectively by chart review for this study. There was no interference with medical care received by the included patients.
Study population
We included patients who (a) had a diagnosis of RRMS and (b) initiated treatment with natalizumab or ocrelizumab between January 2016 and April 2019 at the university hospitals of Mainz and Düsseldorf. Owing to the real-world setting, patients were diagnosed according to the McDonald criteria applicable at the time of diagnosis; however, all patients fulfilled the 2017 McDonald criteria. 21 Follow-up data were available until 18–30 months after start of treatment except for patients with PD. We excluded data from patients who were lost to follow-up due to continuing treatment in another center. Patients starting treatment with natalizumab were excluded if they had previously received natalizumab at any time point. During the first 24 months of treatment, all patients treated with natalizumab received infusions in a 4-week interval. After month 24, some patients were switched to EID in order to reduce PML risk. 11 These patients were not excluded from further analysis as this was considered representative of real-world experience. Patients starting treatment with ocrelizumab were excluded if they were switching from another B cell–depleting therapy such as rituximab. Importantly, patients with progressive forms of MS receiving ocrelizumab were not included in the study as we considered comparison to natalizumab not to be relevant for this group. A flow chart of the study population is shown in Supplementary Figure 1. Sample sizes were sufficient for the conclusions achieved in the study as shown by the Bayesian posterior distribution analyses of effect sizes (Supplementary Figure 2).
Data harmonization
Both centers used harmonized patient care guidelines as published in the quality manual of treatment recommendations by the German Competence Network Multiple Sclerosis (KKNMS). 22 Shared worksheets were used in both centers for collection of data. All data were checked for consistency by the first author.
Outcome measurements
Our primary outcome was the proportion of patients with NEDA-3 [no evidence of disease activity, defined as no relapses, no confirmed disability progression, and no active MRI lesions (both new or enlarged T2 lesions and contrast-enhancing lesions)]. 16 Each of these subcomponents was also analyzed separately.
A relapse was defined as a monophasic clinical episode with new patient-reported symptoms and objective findings typical of MS, developing acutely or subacutely, with a duration of at least 24 h, with or without recovery, and in the absence of fever or infection. 21 Disease progression was defined as an increase in EDSS score of 1.5 points from a baseline score of 0, of 1.0 point from a baseline score between 1.0 and 5.0, or of 0.5 points from a baseline score greater than 5.0, confirmed after 3 months.16,23
Concerning MRI activity, we analyzed both new and enlarged hyperintense lesions on T2-weighted images and presence of gadolinium-enhancing lesions on T1-weighted images, and further analyzed these parameters as a combinatory endpoint. MRI was performed on 3 Tesla magnetic resonance (MR) scanners. Scans of all MS patients were performed in the context of the clinical routine, and all MR images were analyzed by a neuroradiologist with regard to new or enlarging lesions as well as to contrast-enhancing lesions. Therefore, a T1-weighted sequence, a T1-weighted fluid-attenuated inversion recovery (FLAIR) sequence, and contrast-enhanced T1-weighted images were obtained for the detection of radiological disease activity. Baseline MRI scans were performed not more than 3 months before or 1 month after treatment start. For each patient, the same scanner was used during follow-up if possible, although this could not be achieved in all cases in clinical practice.
As a secondary outcome, we included measurement of NfL in serum (sNfL) as a marker for axonal damage that has been shown in recent years to correlate with treatment response in MS.24,25 This analysis was only available for a subgroup of patients.
Neurofilament measurements
NfL was included as a secondary outcome if available. Follow-up values under treatment were assessed at the earliest time point available, but not before 3 months after treatment start. In total, follow-up values were available for 55 patients (ocrelizumab n = 23, natalizumab n = 30) after a mean time to follow-up of 9.9 months. For 30 patients (ocrelizumab n = 17, natalizumab n = 13), both baseline and follow-up values were available (mean time to follow-up = 8.8 months). NfL values were assessed from the patient’s sera using a standardized protocol described in detail previously. 25 NfL levels were determined in duplicates by single molecule array with an SiMoA HD-1 (Quanterix, Billerica, MA, USA) using the Nf-Light Advantage Kits (Quanterix) according to the manufacturer’s instructions. All coefficients of variation (CVs) of the two replicates were below 20%, resulting in a mean intra-assay CV of 6.8%. Low and high controls, consisting of recombinant human NfL antigen, were included in each sample run to monitor plate-to-plate variation (low: mean = 3.0 pg/ml, inter-assay CV = 4.0%; high: mean = 132.1 pg/ml, inter-assay CV = 6.9%). The NfL measurements were performed blinded and without information on clinical data.
Statistical analysis
Information collected at treatment start with natalizumab or ocrelizumab (hereafter termed baseline) for all patients included age, sex, EDSS score, 26 years since diagnosis, years since first symptoms, number of relapses in previous year, number of previous DMT, last previous DMT, reason for DMT switch, and presence of gadolinium enhancement on baseline MRI.
Baseline characteristics of the two treatment groups were compared by Fisher’s exact test and Mann–Whitney U test as appropriate. Odds ratios were calculated using logistic regression (dependent variable: occurrence of relapse; covariates: EDSS at baseline, number of relapses in previous year, number of previous DMTs, years since first manifestation, years since diagnosis, age at baseline and treatment with natalizumab).
In order to correct for non-randomization and eliminate the potential influence of confounding factors such as age and sex for the analyzed groups, we performed a Bayesian propensity score matching of the groups. Propensity score computation was performed as described elsewhere. 27 In brief, for propensity score matching, we used the open-source Bayesian spatial propensity score matching (BSPM) toolbox (https://sejdemyr.github.io/r-tutorials/statistics/tutorial8.html) with RStudio (1.1.456). The first step in BSPM is to estimate the propensity score through a logistic regression. Bayesian networks are generative models, and to generate the joint probability distribution of the regression model, the generative process is stated below. By Bayesian theorem, the posterior distribution of the network is the product of the likelihood and the prior. In many cases, the exact posterior distribution cannot be solved analytically, but it can be approximated with stochastic methods. In this study, we used the NO-U-Turn Sampler (NUTS) in the Hamiltonian Monte Carlo algorithm. We set up a NUTS sampler with a single chain, 3000 samples, and 200 warm-up samples were discarded. After inferring the propensity score from the Bayesian logistic regression model, the second step of BPSM is to match pairs based on their propensity score distances. The objective of the matching is to form balanced groups, that is, minimizing the propensity score distance. In this study, for the matching we used the Caliper matching, which is computationally cheap and intuitive. 28 After matching, we performed the log-rank testing.
Data were analyzed with IBM SPSS Statistics for Windows, Version 28.0 (IBM Corp. Released 2021, Armonk, NY, USA) and GraphPad Prism software (version 9.3.0 for Windows, GraphPad Software, San Diego, CA, USA). The statistical tests used are indicated in the figure or table legends. p values < 0.05 were considered significant.
Results
Analysis of total patient cohort
Patient characteristics
Between January 2016 and April 2019, 155 patients met inclusion criteria at the two centers. A total of 63 patients starting with natalizumab and 76 patients starting with ocrelizumab could be included in further analyses (see Supplementary Figure 1). Patients receiving ocrelizumab were older, had longer disease duration, a higher EDSS at baseline, and a higher number of previous DMTs than patients starting with natalizumab. Detailed baseline characteristics are shown in Table 1. A total of 27.6% of patients starting with ocrelizumab were switching directly from natalizumab, while 42.1% of ocrelizumab patients had previously been treated with natalizumab, directly before switching or at any time point in the past. There were no patients switching from B cell–depleting therapy in the natalizumab group.
Baseline characteristics of the total cohort.
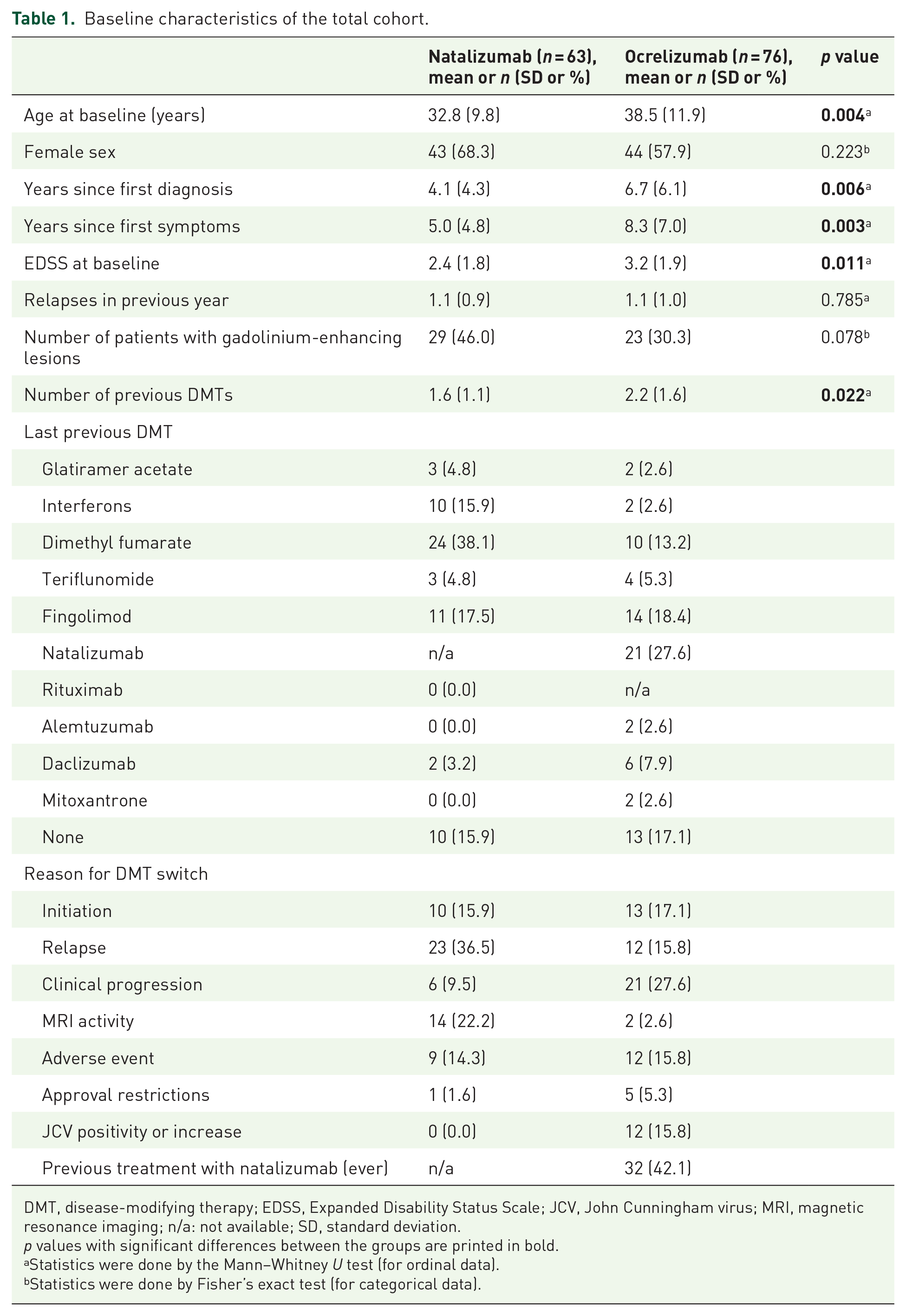
DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; JCV, John Cunningham virus; MRI, magnetic resonance imaging; n/a: not available; SD, standard deviation.
p values with significant differences between the groups are printed in bold.
Statistics were done by the Mann–Whitney U test (for ordinal data).
Statistics were done by Fisher’s exact test (for categorical data).
Relapse activity in total patient cohort
In order to assess the risk for relapses under treatment, we first performed a binary logistic regression in the total patient cohort. Factors independently associated with a higher risk for relapses were treatment with natalizumab [odds ratio (OR) = 3.53, 95% confidence interval (CI) = 1.40–8.88, p = 0.007] and a higher number of relapses in the previous year (OR = 1.83, 95% CI = 1.09–3.09, p = 0.022) (Figure 1(a)). We then repeated the analysis after exclusion of all relapses occurring during the first 4 weeks of treatment with natalizumab or ocrelizumab, as these might occur before onset of action after treatment initiation. Both treatment with natalizumab and the number of relapses in the previous year remained significantly associated with a higher risk for relapses (OR = 2.98, 95% CI = 1.13–7.84, p = 0.027 and OR = 1.80, 95% CI = 1.06–3.04, p = 0.029, respectively) (Figure 1(b)). Previously naïve patients and patients switching from another DMT had a comparable risk for relapses in the total patient cohort and in both treatment groups analyzed separately (Supplementary Table 1).

Odds ratios for the risk for relapses in the total patient cohort. Statistics were performed using binary logistic regression. (a) Odds ratios including all relapses occurring after start of treatment. (b) Odds ratios after exclusion of relapses that occurred during the first 4 weeks after start of treatment as these might be considered carry-over disease activity. In both approaches, the number of relapses in the previous year and treatment with natalizumab significantly increased the risk for relapses.
PD
PD was defined as discontinuation before completion of 24 months of therapy, as this treatment duration is usually the goal when switching to a new treatment. There was a significantly higher frequency of PD in the natalizumab group (30.2%) than in the ocrelizumab group (9.2%, p = 0.002) (Table 2). In the natalizumab treatment group, PD was most often due to JCV seroconversion or increase in antibody index (36.8% of PD), followed by pregnancy and treatment failure (21.1% each). The main reasons for PD in the ocrelizumab treatment group included treatment failure (42.9% of PD) followed by conversion to secondary progressive multiple sclerosis (SPMS) (28.6% of PD). Accordingly, analysis of patients with PD showed a higher proportion of relapses compared with those continuing on the respective treatment (Supplementary Table 2).
Premature discontinuation.
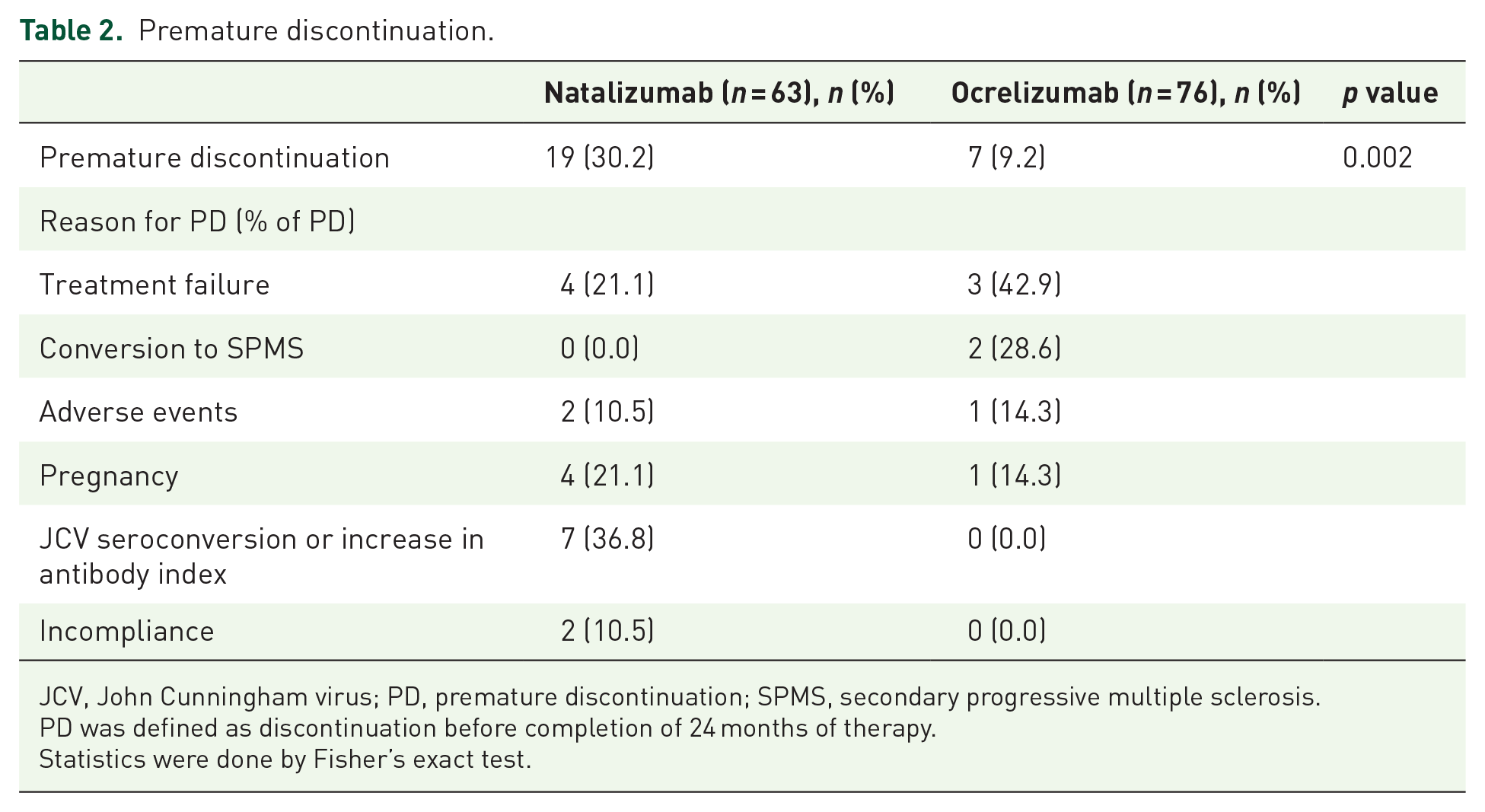
JCV, John Cunningham virus; PD, premature discontinuation; SPMS, secondary progressive multiple sclerosis.
PD was defined as discontinuation before completion of 24 months of therapy.
Statistics were done by Fisher’s exact test.
Subgroup analyses
Disease activity in matched patient cohort
To analyze disease activity in more detail, we next performed propensity score matching of the two treatment groups in order to adjust for differences in baseline characteristics (Table 3 and Supplementary Figure 3). Patients successfully matched were then compared in a Kaplan–Meier failure curve by log-rank test. In line with the analysis in the whole patient cohort, relapses occurring during the first 4 weeks of treatment were excluded. This analysis confirmed a higher risk for relapses in the group treated with natalizumab compared with ocrelizumab (p = 0.019) (Figure 2(a)). Relapses occurring in the natalizumab group were not due to EID, as patients on EID after month 24 showed a comparable relapse risk to patients on standard interval dosing (data not shown). There was no significant difference between the groups concerning confirmed disease progression or MRI activity, defined as a combinatory endpoint of new or enlarged T2 lesions and contrast-enhancing lesions, although there was a trend toward more MRI activity in the natalizumab-treated group (Figure 2(b) and (c)).
Baseline characteristics with adjusted values after propensity score matching.
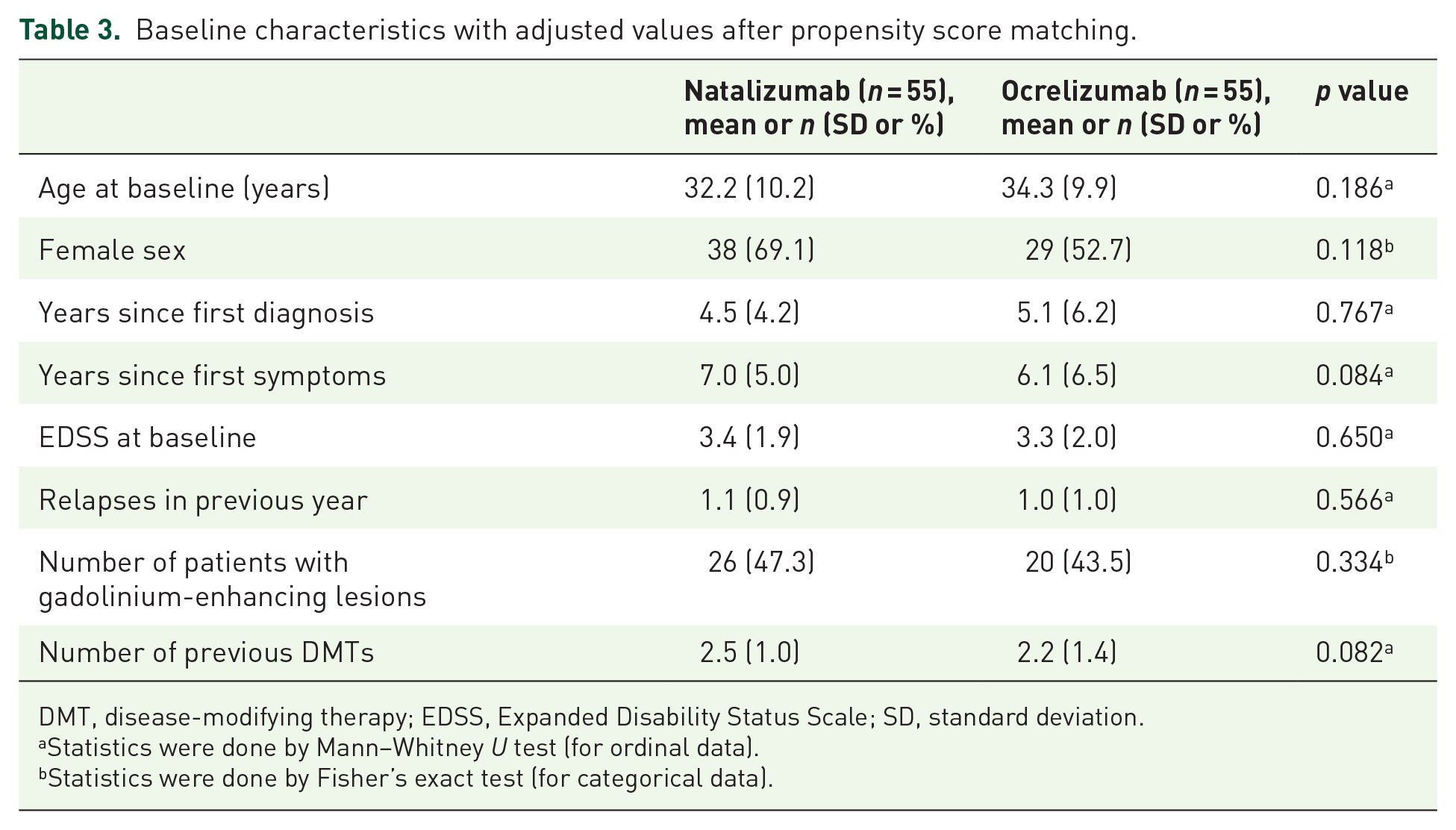
DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; SD, standard deviation.
Statistics were done by Mann–Whitney U test (for ordinal data).
Statistics were done by Fisher’s exact test (for categorical data).

Kaplan–Meier failure curves for clinical and radiological outcomes. (a) Risk of relapses. (b) Risk of disease progression. (c) Risk of MRI activity, shown as combined endpoint of new or enlarged T2 lesions and contrast-enhancing lesions. The y-axis shows the percentage of patients from the propensity score–matched cohort, the x-axis gives the time in months. Data for the natalizumab treatment group are shown by the purple dashed line; data for the ocrelizumab treatment group are shown by the black line. The number of patients from whom data were available at each time point is given in the tables below each graph. Statistics were done by the log-rank test.
Combined analysis of clinical and radiological outcomes revealed that 53.1% of patients treated with ocrelizumab and 36.1% of patients treated with natalizumab achieved the NEDA-3 endpoint (no relapses, no confirmed disability progression, and no MRI activity) after 30 months (p = 0.177) (Figure 3). Cox regression analysis of combined disease activity confirmed comparable effectiveness of both treatment groups in the matched cohort [hazard ratio (HR) = 0.78, 95% CI = 0.44–1.39, p = 0.393], with similar results obtained in the total patient cohort (Supplementary Table 3).

Kaplan–Meier survival curve for NEDA-3 outcome. The y-axis shows the percentage of patients from the propensity score–matched cohort, the x-axis gives the time in months. Data for the natalizumab treatment group are shown by the purple dashed line, and data for the ocrelizumab treatment group are shown by the black line. The number of patients from whom data were available at each time point is given in the table below. Statistics were done by the log-rank test.
NfL measurement
sNfL was measured by SiMoA in a subcohort of patients at baseline and under treatment in order to further analyze subclinical disease activity. Owing to the limited sample size, no matching between the groups was performed. Treatment with ocrelizumab significantly reduced NfL levels (Figure 4(a)). NfL levels under treatment were low and not significantly different between the ocrelizumab and the natalizumab group (Figure 4(b)).
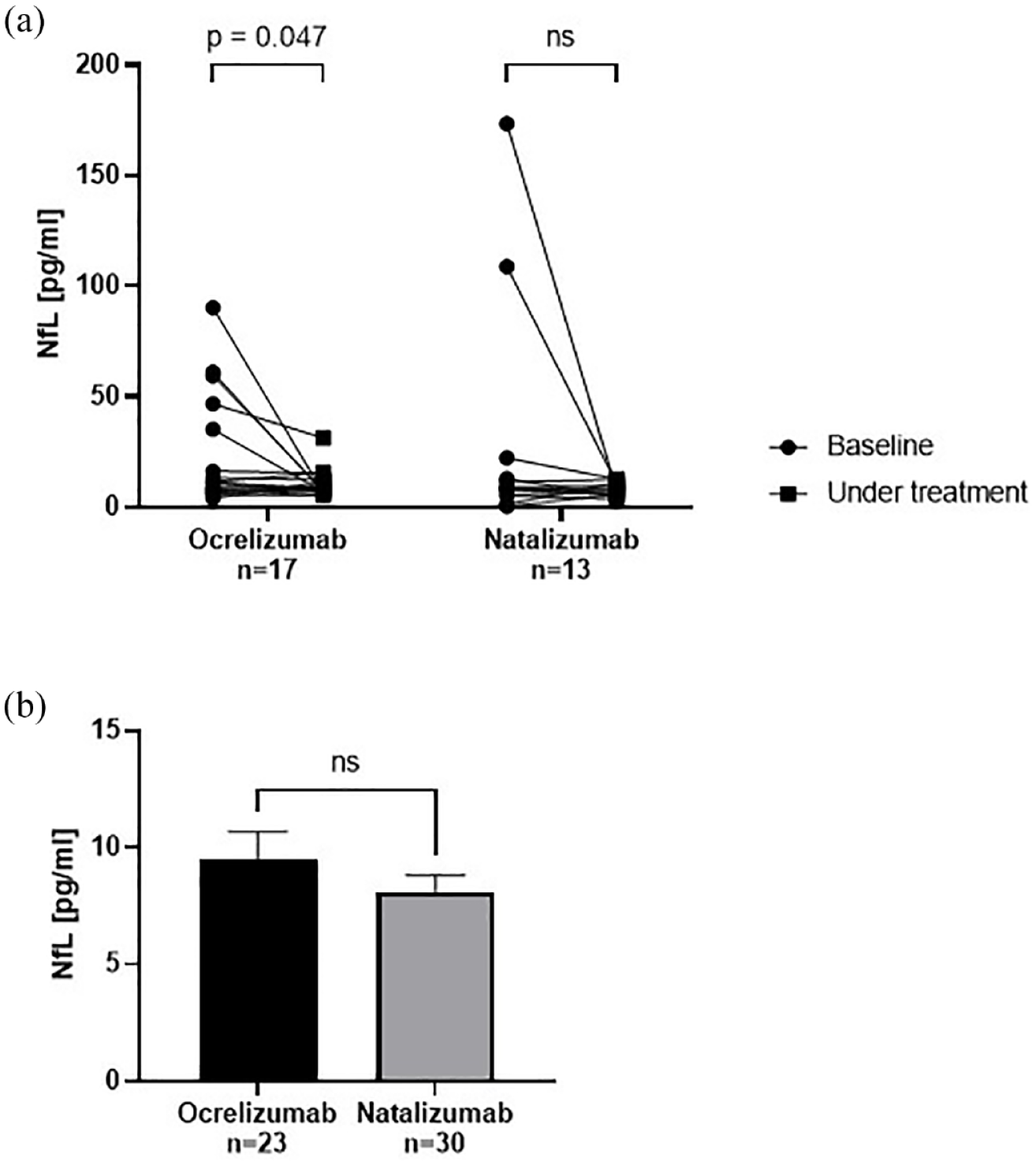
Analysis of neurofilament light chain in serum. (a) NfL levels were analyzed in a subcohort of patients before and under treatment with ocrelizumab or natalizumab. Ocrelizumab treatment significantly decreased NfL levels compared with baseline. Statistics were done by multiple paired t tests. (b) NfL levels under treatment were not significantly different between the ocrelizumab and the natalizumab group. Data are shown as mean value ± SEM. Statistics were done by the Mann–Whitney test.
Rebound activity after natalizumab cessation
Of the total group treated with ocrelizumab, 27.6% of patients directly switched from natalizumab. Disease activity data for these direct switchers were compared with the rest of the ocrelizumab-treated group. The occurrence of relapses, confirmed disability progression, MRI activity, and the proportion of patients with NEDA-3 were comparable between both groups (Table 4). Thus, there was no evidence for enhanced rebound activity after natalizumab cessation for patients switching to ocrelizumab.
No increased rebound activity in patients switching from natalizumab to ocrelizumab.

MRI, magnetic resonance imaging; NEDA, no evidence of disease activity.
Data for up to 24 months of follow-up were included.
Statistics were done by Fisher’s exact test.
Discussion
In this study, we analyzed comparative effectiveness and treatment continuation of two highly effective DMT, natalizumab and ocrelizumab, in a real-world RRMS cohort. Overall, there was no significant difference between the proportions of patients reaching the combined endpoint NEDA-3. Treatment with ocrelizumab was associated with a lower risk for relapses than treatment with natalizumab in our cohort. Furthermore, patients treated with natalizumab had a higher risk for PD of therapy. For patients switching from natalizumab, our data support the evidence for reduced rebound activity after switching to a B cell–depleting therapy.
In order to compare the effectiveness of different DMT, there have been attempts to infer combined endpoints such as NEDA-3 from different phase III trials. 29 Furthermore, several recent network meta-analyses calculated rankings for all available DMT, setting natalizumab and ocrelizumab at comparable levels of treatment efficacy.30–32 Although these comparisons are helpful for establishing therapeutic hierarchies, one of their major limitations lies in the artificial conceptualization of the underlying studies. For example, the AFFIRM trial for natalizumab excluded patients with a relapse within 50 days before the administration of the first dose and patients treated with interferon beta, glatiramer acetate, cyclosporine, azathioprine, methotrexate, or intravenous immune globulin within the previous 6 months or treated with interferon beta, glatiramer acetate, or both for more than 6 months in total. 6 In other words, the trial preferentially included patients who had been stable during at least 50 days without having received basic therapy. This population clearly does not reflect clinical routine, in which especially patients suffering from disease activity despite basic treatment are escalated to natalizumab. In the OPERA trials for ocrelizumab, more than 70% of patients included were therapy naïve, and almost no patients had previously received natalizumab, 15 while in our real-world cohort, only 17.1% received ocrelizumab as first-line treatment and 42.1% had, at some point, been previously treated with natalizumab, with 27.6% being direct switchers. In sum, these exemplary points highlight the importance of real-world data in comparative analysis of treatment efficacy. Nevertheless, the groups analyzed in this study also had relatively low EDSS and a proportion of patients (15.9% natalizumab and 17.1% ocrelizumab) without pretreatment, which may have affected the results.
Real-world studies have gained importance in studying MS treatment responses during the last years. For example, several studies have focused on long-term efficacy and safety of natalizumab.14,33,34 Furthermore, real-world studies have compared effectiveness of natalizumab with other DMT such as fingolimod.35–37 To the best of our knowledge, this study is the first direct comparison between natalizumab and ocrelizumab treatment in a real-world RRMS cohort. Interesting insights, however, might be gained from studies including rituximab as another B cell–depleting antibody. In a recent comparative effectiveness study on several DMT using clinic-based registry data and linked electronic health records, natalizumab was associated with a higher 1-year and 2-year relapse rate compared with rituximab, with relapses being defined as both clinical and radiological events. 38 Similar trends concerning natalizumab and rituximab were observed in other retrospective studies.39,40 These results fit well with our data on relapse risk.
Besides their clinical characteristics, analysis of contemporary patient cohorts allows integration of new paraclinical markers in order to gain a comprehensive picture of treatment response. sNfL is an emerging marker for disease activity reflecting axonal damage, and although it is not specific for MS, it has been shown to correlate with relapse rate and treatment response on a group level.24,25 As it allows for detection of subclinical disease activity, it might provide a helpful additional tool to guide treatment decisions in the near future. In our subgroup analysis, serum levels of NfL were successfully suppressed by both natalizumab and ocrelizumab treatment. Although reduction of NfL levels compared to baseline was significant only for ocrelizumab, these results should be interpreted with caution as the subgroups were small and therefore precluded matching for covariates.
Apart from therapeutic effectiveness, adherence of patients to treatment regimens is of vital importance for clinical success. Our results show that PD of therapy is significantly more frequent in patients treated with natalizumab compared with ocrelizumab. The main reason for PD in the natalizumab group was an increase in risk for PML as indicated by JCV seroconversion or increase in antibody index. It is important to note that PML cases also occur under treatment with B cell–depleting drugs. 41 Most of the cases, however, reported in MS patients under ocrelizumab so far were considered carry-over PML after natalizumab treatment, 42 and there is no established risk stratification for ocrelizumab such as anti-JCV antibody index. Further reasons for PD more frequent in the natalizumab group included incompliance, which is possibly due to the infusion interval of 4 weeks compared to 6 months for ocrelizumab, and pregnancy. The latter might be due to the fact that available (albeit limited) data point toward safety of exposure to natalizumab during the first trimester; therefore, the drug might have been given preferentially to women with high disease activity and sub-acute family planning until conception. 43 Thus, reasons for PD might sometimes even reflect advantages of specific DMT.
In the ocrelizumab group, PD was less frequent in general and rather due to treatment failure or conversion to SPMS than to adverse events. The observation period in this study, however, might be too short to register long-term side effects such as hypogammaglobulinemia or infections. 44
In most cases, discontinuation actually implies switching to another therapy. In case of natalizumab, cessation of treatment might lead to severe rebound activity, especially in patients with high disease activity and a high level of disability prior to natalizumab treatment.45,46 Several retrospective studies suggest that switching to a B cell–depleting therapy is superior to other DMT.47,48 In fact, a retrospective multicenter study showed relapses in only 8% of patients (95% CI = 0–19%) switching from natalizumab to ocrelizumab, compared with a risk of reemerging disease activity of 45% (95% CI = 0.41–0.49) after natalizumab cessation known from the literature.48,49 Our data are in line with this hypothesis, as patients switching from natalizumab to ocrelizumab displayed a comparable stability of disease as patients initiating with ocrelizumab or switching from other drugs.
In sum, this study provides class IV evidence for comparative effectiveness of natalizumab and ocrelizumab in the treatment of RRMS. Limitations of this study include the retrospective, non-randomized design and the limited cohort size and length of follow-up. Furthermore, differences in the patient cohorts assigned to a certain treatment might have influenced the results despite matching procedures. For example, a large proportion of patients in the ocrelizumab group (42.1%) had been treated with natalizumab at any time point in the past, while there were no ‘reverse switchers’ (i.e. switching from ocrelizumab to natalizumab). Patients receiving natalizumab were more often treated with interferon beta or dimethyl fumarate before switching, and the occurrence of relapses was a more frequent reason for switching. Moreover, PML risk stratification influenced choice of treatment.
Nevertheless, we think that real-world comparative analyses improve our abilities to provide individualized advice to patients. For example, patients with high relapse rates might benefit from early treatment with ocrelizumab. Further prospective randomized trials are needed in order to confirm these results and to guide both patients and clinicians through the broadening choice of DMTs.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221142924 – Supplemental material for Comparative effectiveness of natalizumab versus ocrelizumab in multiple sclerosis: a real-world propensity score–matched study
Supplemental material, sj-docx-1-tan-10.1177_17562864221142924 for Comparative effectiveness of natalizumab versus ocrelizumab in multiple sclerosis: a real-world propensity score–matched study by Katrin Pape, Leoni Rolfes, Falk Steffen, Muthuraman Muthuraman, Melanie Korsen, Sven G. Meuth, Frauke Zipp and Stefan Bittner in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank Dr Cheryl Ernest for proofreading the manuscript and Theresa Wingert for technical assistance. The authors also thank all patients who were part of this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
