Abstract
Background:
The treatment of juvenile myoclonic epilepsy (JME) is limited, with most patients requiring long-term medication and over half experiencing seizure recurrence upon drug withdrawal. As a third-generation antiseizure medication, brivaracetam (BRV) has emerged as a promising therapeutic option. Its efficacy has been investigated in focal epilepsies and genetic generalized epilepsies (GGEs), with promising results.
Objectives:
This study aims to evaluate the safety and preliminary efficacy of BRV as an off-label initial monotherapy in patients with newly diagnosed JME, with a specific focus on the control of myoclonic seizures.
Design:
A prospective, single-center, and observational study.
Methods:
This study prospectively enrolled drug-naïve patients with JME. All participants received BRV monotherapy. Clinical data were collected at baseline and after a 6-month follow-up period, including demographic characteristics, electroencephalography (EEG), cranial magnetic resonance imaging (MRI), and comprehensive neuropsychological assessments. Changes in seizure frequency, cognitive function, levels of anxiety and depression, sleep quality, and quality of life from baseline to the 6-month follow-up were analyzed and compared.
Results:
A total of 19 patients were included with a mean age of 20.26 ± 6.88 years (median: 18, interquartile range: 8), and a male-to-female ratio of 12:7. The average age of onset was 14.58 ± 3.42 years, and the average duration of epilepsy prior to BRV treatment was 5.71 ± 7.40 years (median: 2, interquartile range: 5). The mean frequency of myoclonic seizures at baseline was 38.79 ± 45.60 times per month (median: 10, interquartile range: 86). Eighteen patients (94.73%) experienced both generalized tonic-clonic seizures (GTCS) and myoclonic seizures, one patient only experienced myoclonic seizures, The MRI findings were negative in all patients (100%). The EEG of all patients at baseline was abnormal, revealing 3–5.5 Hz generalized spike-and-wave or polyspike-and-wave discharges. At the 6-month evaluation, all patients achieved seizure-free status (p < 0.001), neuropsychological assessments also demonstrated significant improvement, including Montreal Cognitive Assessment (MoCA; p < 0.001), Hamilton Anxiety Scale (HAMA; p < 0.001), Hamilton Depression Scale (HAMD; p < 0.001), Pittsburgh Sleep Quality Index (PSQI; p < 0.001), and Quality of Life in Epilepsy-31 (QOLIE-31; p < 0.001). Only one patient complained of poor sleep after BRV administration.
Conclusion:
This study suggests that BRV may offer promising efficacy, specifically in controlling myoclonic seizures and favorable tolerability as an off-label initial monotherapy for JME patients. While the evaluation of efficacy against GTCS requires longer follow-up, our findings support the potential of BRV as a therapeutic option for JME. Further randomized controlled trials are warranted to validate these observations.
Plain language summary
The treatment of juvenile myoclonic epilepsy (JME) is limited, with most patients requiring long-term medication and over half experiencing seizure recurrence upon drug withdrawal. Brivaracetam (BRV) has shown promising therapeutic potential in focal epilepsies and genetic generalized epilepsies (GGEs).
This study evaluated the efficacy and safety of BRV as the initial monotherapy in patients with JME in clinical practice.
A total of nineteen patients were included. Following treatment with BRV, all patients achieved seizure-free status (p<0.001), neuropsychological assessments also demonstrated significant improvement, including Montreal Cognitive Assessment (MoCA) (p<0.001), Hamilton Anxiety Scale (HAMA) (p<0.001), Hamilton Depression Scale (HAMD) (p<0.001), Pittsburgh Sleep Quality Index (PSQI) (p<0.001), and Quality of Life in Epilepsy-31 (QOLIE-31) (p<0.001). Only one patient complained of poor sleep.
This study suggests that BRV may offer promising efficacy and tolerability as an initial monotherapy for JME, supporting its potential as a first-line treatment option. Further randomized controlled trials involving larger cohorts and extended follow-up periods are required to validate these findings.
Introduction
Juvenile myoclonic epilepsy (JME) is a common type of idiopathic generalized epilepsy (IGE), affecting about 9.3% of all patients with epilepsy. 1 The disease typically presents between the ages of 8 and 40 years, with a clear female predominance, and approximately 15% of JME cases evolve from childhood absence epilepsy (CAE). 2 The main clinical symptoms are myoclonic seizures, generalized tonic-clonic seizures (GTCS), and sometimes absence seizures. 3 Interictal electroencephalogram (EEG) typically shows 3–5.5 Hz generalized spike-and-wave (SW) or polyspike-and-wave (PSW) discharges. 4
Currently, anti-seizure medications (ASMs) remain the primary treatment for JME and are effective in approximately 65% of patients. 5 Valproic acid (VPA) has long been considered a first-line treatment for JME. However, its long-term administration is associated with severe adverse effects (AEs), particularly in female patients, such as weight gain, menstrual disorder, polycystic ovary syndrome, hair loss, hepatic damage, and tremor. 6 Furthermore, evidence indicates that VPA can cross the placental barrier and exhibit obvious teratogenicity. 7 Levetiracetam (LEV) has been shown to exhibit comparable efficacy to VPA for the treatment of JME.8–11 Nevertheless, Mbizvo et al. 12 found that the incidence of behavioral side effects in children receiving LEV treatment can be as high as 23%, thereby restricting its application. Furthermore, recent meta-analysis revealed that 78% of JME patients experienced recurrence of seizures after the discontinuation of ASMs. 5 Most patients require lifelong treatment, imposing a substantial burden on both their families and society. Therefore, effective and well-tolerated drugs for JME are urgently needed.
Brivaracetam (BRV) is a third-generation of ASMs, which was initially approved in Europe in 2016 as an adjunctive therapy or monotherapy for focal epilepsy in patients aged 16 and older. In 2024, it was approved in China for the treatment of patients with partial seizures (older than 16 years old). Similar to LEV, BRV acts by binding to the synaptic vesicle protein 2A (SV2A), thereby inhibiting neurotransmitter release. 13 However, BRV differs from LEV in other pharmacological profiles. Firstly, BRV does not inhibit α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors and high-voltage calcium channels, lowering the incidence of irritability and aggression 14 ; Secondly, BRV has 15–30 times higher affinity to SV2A. 15 Besides, BRV enters the brain at least sevenfold faster than LEV. 16 It has been shown to be effective in the treatment of focal epilepsy, 17 and studies have also reported its efficacy in the treatment of generalized epilepsy.18,19 Current evidence for BRV in JME is limited to small case series, or investigations of its adjunctive therapy. Prospective studies evaluating BRV as an initial monotherapy for newly diagnosed JME patients are still lacking.
This study prospectively enrolled a cohort of patients with JME to evaluate the safety and preliminary efficacy of off-label BRV monotherapy.
Methods
Study design
This was a prospective, observational, single-center study conducted at Beijing Tiantan Hospital. A total of 19 patients diagnosed with JME were enrolled between January 2025 and May 2025. Clinical data were collected at enrollment and at the 6-month follow-up to assess the efficacy and safety of BRV, as well as its effects on cognition, psychology, sleep, and quality of life.
This study received ethical approval from the Medical Ethics Committee of Beijing Tiantan Hospital, Capital Medical University. All patients or their legal representatives provided written informed consent.
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, as recommended by the EQUATOR Network.
Patients
The inclusion criteria were as follows: (1) aged 12–60 years with a definitive diagnosis of JME according to established diagnostic criteria. 4 (2) No prior history of ASMs use. (3) Written informed consent from the patients or their caregivers. The exclusion criteria included: (1) severe hepatic or renal impairment. (2) Pre-existing severe psychiatric disorders (such as psychosis, severe depression). (3) A history of vagus nerve stimulation therapy or other ASMs use. (4) Female patients who were pregnant, breastfeeding, or planning to become pregnant during the study period. (5) Inability to complete data collection or follow-up. Withdrawal criteria included patients with poor medication adherence, defined as actual oral dosage less than 80% of the prescribed dosage.
Treatment
All patients received off-label BRV monotherapy, administered orally twice daily with weight-based dosing. For patients weighing ⩾50 kg, BRV was initiated at 50 mg twice daily. For those weighing <50 kg, the initial dose was 1 mg/kg twice daily. The dosage was then individually adjusted based on the frequency of seizures and clinical response.
Data acquisition and evaluation of treatment efficacy
Clinical data were collected, including gender, age, age of onset, type and frequency of seizures, EEG, cranial magnetic resonance imaging (MRI), treatment, and prognosis. The baseline seizure frequency of myoclonic seizures was determined based on the patient’s clinical history for one or several months preceding enrollment. It should be noted that most patients sought medical attention due to a recent exacerbation of myoclonic seizures and GTCS. All patients underwent 12-h continuous video-EEG (VEEG) monitoring before and after treatment. Electrodes were placed according to the international 10–20 system. The 12-h VEEG recording included both wakefulness and natural sleep. Standard activation procedures, including hyperventilation, intermittent photic stimulation, and eye opening/closing tests, were performed for all patients during the recording. All patients were assessed with the following scales at both baseline and the 6-month follow-up: Montreal Cognitive Assessment (MoCA), Hamilton Anxiety Scale (HAMA), Hamilton Depression Scale (HAMD), Pittsburgh Sleep Quality Index (PSQI), Quality of Life in Epilepsy-31 (QOLIE-31), and Activities of Daily Living. Then a comparison was performed between baseline and follow-up regarding seizure frequency and scale scores.
Assessment of BRV safety
The safety of BRV was evaluated by monitoring the incidence and frequency of AEs such as mood disturbances, somnolence, and behavioral changes, as well as tracking changes in laboratory parameters throughout the study.
Statistical analysis
SPSS 24.0 software (IBM Corp., Armonk, N.Y., USA) for Windows was used for statistical analysis. The normality of data distribution of continuous variables was tested by the Shapiro–Wilk test. Continuous variables were shown as the mean ± standard deviation or the median (interquartile range (IQR)), and categorical variables were shown as frequencies (proportions). Group comparisons were performed using the paired t test or the Wilcoxon signed-rank test. A p-value < 0.05 (two-sided) was regarded as statistically significant.
Results
Clinical characteristics
Detailed demographic and clinical features of patients at baseline and at the 6-month follow-up were summarized in Table 1. A total of 19 JME patients with a mean age of 20.26 ± 6.88 years (median: 18, IQR: 8) were enrolled, and 7 patients were female (36.84%). The average age of onset was 14.58 ± 3.42 years. And the average duration of epilepsy prior to BRV treatment was 5.71 ± 7.40 years (median: 2, IQR: 5). Notably, the variation in disease duration reflects a subset of patients with delayed diagnosis, characterized by historically infrequent seizures that recently exacerbated, thereby prompting medical consultation. In total, 18 patients experienced both GTCS and myoclonic seizures (94.73%), 1 patient only experienced myoclonic seizures, and absence seizures were not observed in any patient. The MRI findings were negative in all patients (100%). The EEG of all patients at baseline was abnormal, revealing 3–5.5 Hz generalized SW or PSW discharges. All 19 enrolled patients completed the 6-month follow-up period and data collection process. After 6 months of BRV treatment, 14 patients (73.68%) exhibited an absence of epileptiform discharges during the 12-h VEEG monitoring, and the other 5 patients showed only rare discharges with prominent improvement.
Clinical characterization of patients with JME.
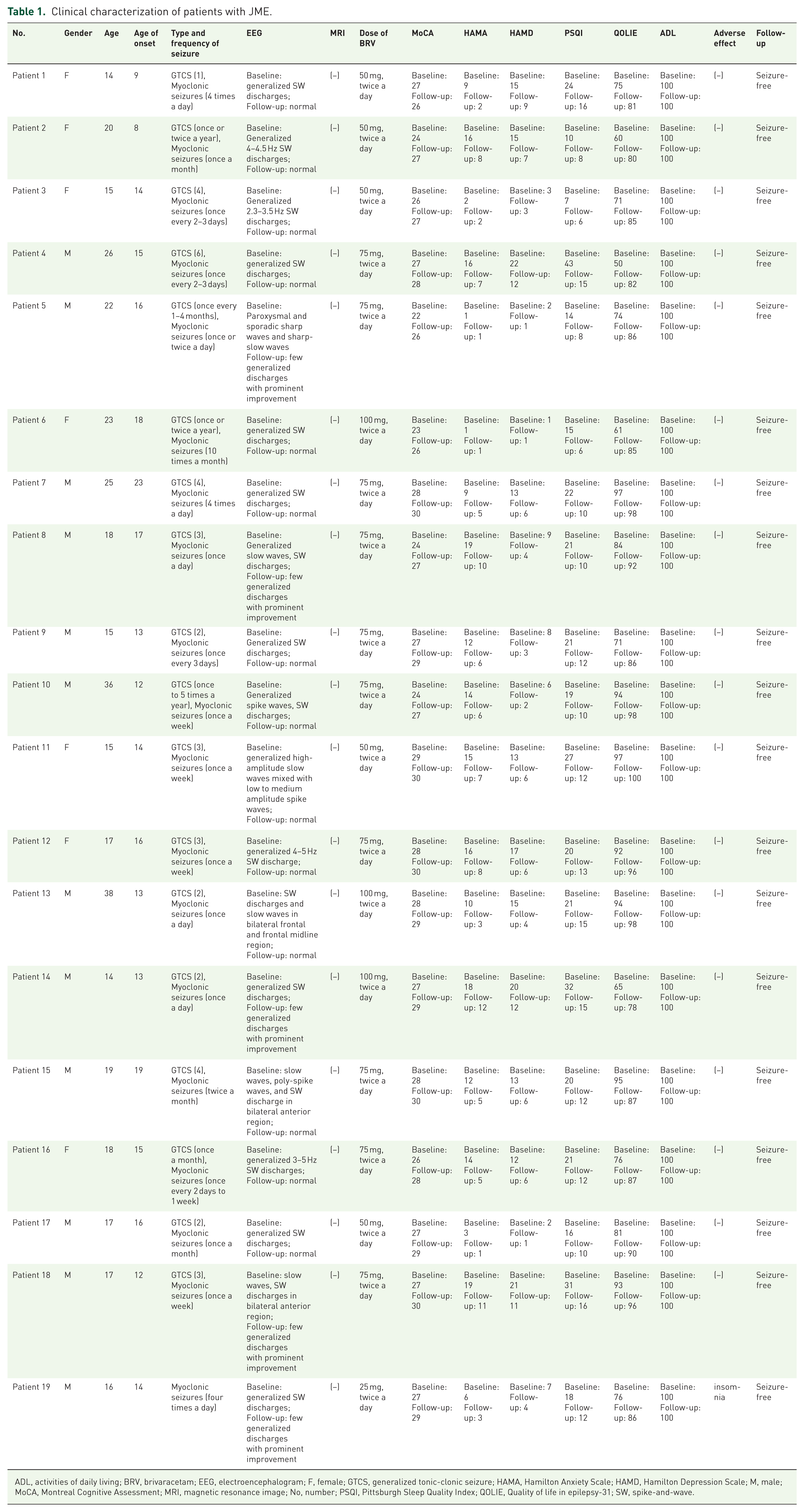
ADL, activities of daily living; BRV, brivaracetam; EEG, electroencephalogram; F, female; GTCS, generalized tonic-clonic seizure; HAMA, Hamilton Anxiety Scale; HAMD, Hamilton Depression Scale; M, male; MoCA, Montreal Cognitive Assessment; MRI, magnetic resonance image; No, number; PSQI, Pittsburgh Sleep Quality Index; QOLIE, Quality of life in epilepsy-31; SW, spike-and-wave.
Efficacy and safety analysis
The mean frequency of myoclonic seizures at baseline was 38.79 ± 45.60 times per month (median: 10, IQR: 86). At the 6-month evaluation, all patients had achieved seizure-free status (p < 0.001, Figure 1(A)). Cognitive function was evaluated with the MoCA scores, revealing significant improvement after the treatment with BRV (p < 0.001, Figure 1(B)). Symptoms of anxiety and depression, assessed by HAMA and HAMD, also demonstrated notable improvement (p < 0.001, Figure 1(C) and (D)). Moreover, both PSQI and QOLIE-31 scores exhibited substantial improvements at follow-up compared to baseline (p < 0.001, Figure 1(E) and (F)). Notably, there were no reports of BRV discontinuation due to AEs. Only one patient complained of poor sleep after BRV administration. No other AEs were observed.

Distribution of (A) myoclonic seizure frequency, (B) MoCA, (C) HAMA, (D) HAMD, (E) PSQI, and (F) QOLIE-31 scores at admission and at follow-up in JME patients.
Discussion
JME is generally considered a lifelong disease that requires reasonable and long-term medication to control seizures. Recent evidence has indicated that the seizure-free rates of JME patients after initiating ASMs treatment were 64.8%, 29.5%, and 14.6% at 1, 3, and 5 years, respectively. 20 This highlights the common occurrence of drug resistance in JME patients, underscoring the importance of selecting the appropriate ASMs. The first-line treatment for JME is VPA, but it is not appropriate for women of reproductive age due to the obvious AEs and teratogenicity. 21 LEV has comparable efficacy to VPA, but psychiatric and behavioral side effects restrict its application. 22 Lamotrigine (LTG) is well-tolerated but less effective than VPA and LEV. 23 Furthermore, some ASMs may aggravate seizures, such as oxcarbazepine, carbamazepine, and gabapentin. 24 Therefore, it is of significant clinical value to identify new ASMs with both high efficacy and good tolerability.
BRV is a new ASM characterized by high affinity to SV2A. It demonstrates a superior profile in selectivity, affinity, and brain penetrability compared to LEV. 25 Previous studies have shown that BRV is effective in patients with focal epilepsy.17,26–28 In a randomized controlled trial involving Asian adult patients with focal epilepsy, the 50 and 200 mg daily dose of BRV demonstrated 50% responder rates of 41.1% and 49.3%, respectively. 29 There is also evidence for the efficacy of BRV in genetic generalized epilepsies (GGEs). A study by Strzelczyk et al. 30 included 61 patients with refractory GGEs who had previously failed 2–3 ASMs. It reported 82% retention rate at 3 months and 69% at 5 months, with 36% responder rate and 25% seizure-free rate at 3 months. In a study by Fonseca et al. 31 involving 37 patients with GGEs who had experienced inadequate efficacy or AEs from other ASMs, BRV therapy resulted in a 6-month retention rate of 81.1% and a seizure-free rate of 62.2%. In addition, a prominent reduction in interictal epileptiform discharges was observed in 52% of patients. These studies provide support for the use of BRV in a wide range of epilepsy types.
Currently, data on BRV for the treatment of JME are still limited. This study represents an initial small prospective, single-center observational evaluation of BRV as off-label monotherapy in patients with JME. Our findings demonstrated a 100% seizure-free rate with respect to myoclonic seizures, with 73.68% of patients showing complete normalization of their 24-h VEEG at the 6-month follow-up. These results showed higher efficacy outcomes than those reported in the existing studies on BRV treatment for JME. Strzelczyk et al. 30 retrospectively reported the positive response among 16 patients with refractory JME, revealing a responder rate of 60%, with a seizure-free rate of 40%. Stephen and Brodie 18 prospectively reported that 4 out of 16 (25%) patients with uncontrolled JME achieved seizure-free following 6 months of BRV therapy. The differences in these study results may be related to the selection of patient populations. Specifically, our study primarily enrolled previously untreated JME patients, while the other two studies mainly focused on refractory cases. Furthermore, Navarro 32 described a pregnant woman with drug-resistant JME, whose seizure frequency decreased from once every day to once every month after the treatment of BRV, and she later delivered a healthy newborn. This provides additional evidence supporting the application of BRV in specific patient populations.
In addition to effective control of myoclonic seizures, our study also observed significant improvements in cognitive function, anxiety and depression levels, sleep quality, and overall quality of life after BRV treatment. These changes may be associated with the effective seizure control and favorable tolerability of BRV. BRV was well-tolerated with a relatively low incidence of AEs, which is consistent with previous studies. Lattanzi et al. 17 reported that 7.5% of patients experienced somnolence, 3.4% experienced headache, and 3.1% experienced dizziness. More importantly, BRV has shown potential in improving behavioral AEs associated with LEV. The study by Hirsch et al. 33 revealed that 28 out of 49 patients (57.1%) who switched directly from LEV to BRV due to psychiatric side effects experienced improvement in their psychiatric symptoms. Toledo et al. 34 also demonstrated that BRV does not increase anger levels and can improve LEV-related behavioral AEs. In our cohort, only 1 patient reported poor sleep; improvements in HAMA and HAMD scores were observed in 17 patients (89.47%), and no behavioral AEs were reported. The absence of AEs may be attributed to the small sample size and short follow-up period.
This study has several limitations. First, the small sample size and gender imbalance may limit the statistical power and generalization of our findings. Second, the follow-up period in this study was relatively short. Given the low baseline frequency of GTCS, this duration was insufficient to definitively assess the efficacy of BRV against this specific seizure type. Further studies with extended follow-up periods are needed to confirm the long-term efficacy and safety of BRV in JME patients. Third, although the self-controlled design enables the evaluation of individual responses, the lack of a LEV-monotherapy control group limits comparative interpretation. Fourth, we utilized standardized adult-validated scales (e.g., QOLIE-31, HAMA) to ensure longitudinal data consistency as adolescents transition into adulthood. However, we acknowledge this may overlook specific psychological aspects better captured by adolescent-specific scales like CHEQOL-25, which should be considered in future studies. Future studies should involve larger, multicenter, prospective randomized controlled trials with extended follow-up periods to better assess the efficacy and safety of BRV.
Conclusion
In conclusion, this study may suggest the therapeutic potential of BRV as an initial monotherapy for patients with JME, specifically for the control of myoclonic seizures. Our findings might indicate a promising responder and seizure-free rate, as well as good tolerability. However, further randomized controlled trials involving larger cohorts and extended follow-up periods are required to validate these findings.
Supplemental Material
sj-docx-1-tan-10.1177_17562864261440217 – Supplemental material for Brivaracetam monotherapy in juvenile myoclonic epilepsy: a safety and efficacy evaluation
Supplemental material, sj-docx-1-tan-10.1177_17562864261440217 for Brivaracetam monotherapy in juvenile myoclonic epilepsy: a safety and efficacy evaluation by Jing-Wen Zuo, Ying-Yue Dai, Wen-Jing Liu, Jia-Ying Zhang, Xiao-Qiu Shao, Qun Wang and Rui-Juan Lv in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
