Abstract
Background:
Patients with atrial fibrillation (AF) who survive spontaneous intracerebral hemorrhage (ICH) face competing risks of thromboembolism and recurrent bleeding.
Objectives:
To evaluate the safety and efficacy of initiating oral anticoagulants versus avoiding anticoagulation in adults with AF after spontaneous ICH.
Design:
Systematic review and meta-analysis of randomized-controlled clinical trials (RCTs).
Data sources and methods:
We searched MEDLINE, Scopus, and ClinicalTrials.gov up to August 28, 2025, for eligible RCTs randomizing adults with AF and prior spontaneous ICH to start oral anticoagulation versus avoid anticoagulation. Efficacy outcomes included the occurrence of new ischemic stroke (primary) and ischemic major adverse cardiovascular events (MACE; secondary). Safety outcomes included recurrent ICH (primary), hemorrhagic-MACE, all-cause mortality at follow-up, and cardiovascular death (secondary). Risk ratios (RRs) with 95% confidence intervals (CIs) were pooled using random-effects meta-analysis.
Results:
Six RCTs were included, comprising 403 patients in the anticoagulation group and 395 in the avoid-anticoagulation group. Anticoagulants reduced the rates of new ischemic stroke (RR = 0.20; 95% CI: 0.06–0.72; I2 = 60%; number needed to treat = 9) and ischemic-MACE (RR = 0.41; 95% CI: 0.23–0.75; I2 = 32%). Anticoagulants were associated with higher rates of recurrent ICH (RR = 3.14; 95% CI: 1.41–7.01; I2 = 0%; number needed to harm = 19) and hemorrhagic-MACE (RR = 2.35; 95% CI: 1.32–4.21; I2 = 1%). All-cause mortality at 90 days (RR = 1.06; 95% CI: 0.69–1.64; I2 = 28%) and cardiovascular death (RR = 0.98; 95% CI: 0.34–2.87; I2 = 63%) did not differ between the two groups. Leave-one-out sensitivity analyses supported the overall direction of effects, with some attenuation when individual trials were omitted.
Conclusion:
In AF survivors of spontaneous ICH, restarting oral anticoagulation lowers ischemic events but raises risks of recurrent ICH and major bleeding, without a clear early mortality difference. Potential benefits may outweigh risks in selected patients within a multidisciplinary framework. Adequately powered RCTs are needed to refine agent choice, timing, and patient selection.
Trial registration:
PROSPERO CRD420251135299 (registered August 27, 2025).
Keywords
Introduction
Patients with spontaneous intracerebral hemorrhage (ICH) face a substantial risk for major adverse cardiovascular events (MACE), including ischemic stroke, systemic embolism, and myocardial infarction. 1 Among these individuals, those with atrial fibrillation (AF) face an especially elevated risk of ischemic complications, a risk that often exceeds predictions based on conventional scoring systems such as CHA2DS2-VASc. 2 Oral anticoagulation has been shown to reduce the risk of ischemic stroke in patients with AF by nearly two-thirds compared with no therapy. 3 However, this benefit comes at the expense of an increased risk of bleeding, while patients with prior ICH have historically been excluded from pivotal randomized-controlled clinical trials (RCTs) evaluating anticoagulation in AF, leaving significant uncertainty regarding the net clinical benefit of restarting anticoagulation in this population. 4
The recent European Stroke Organisation (ESO) guidelines underscore the uncertainty about the net benefit of long-term oral anticoagulation in reducing the risk of MACE and death or dependence compared with the avoidance of anticoagulants in patients with AF and spontaneous ICH. Moreover, the 2022 American Heart Association/American Stroke Association (AHA/ASA) recommendations state that the resumption of oral anticoagulation may be considered approximately 7–8 weeks after the index hemorrhage, following individualized assessment of thromboembolic and hemorrhagic risks to optimize the balance of benefits and risks.5,6
Recent RCTs and observational studies have provided new evidence regarding the balance of ischemic and hemorrhagic risks in patients with AF and a history of ICH, and they suggest that initiating or resuming anticoagulation after ICH may lower the risk of ischemic events and improve functional outcomes but may concurrently increase the risk of recurrent ICH and other major bleeding events.7–9 However, their results do not provide definitive guidance on the optimal management of these patients, leaving considerable uncertainty in clinical decision-making.
Meta-analyses and pooled analyses of earlier trials suggested potential benefits of anticoagulation in reducing ischemic events.10,11 However, existing meta-analyses leave key questions unresolved, particularly regarding patient selection, timing of anticoagulation resumption, and the balance of ischemic benefit against hemorrhagic risk. Importantly, the recent availability of data from additional RCTs offers an opportunity to re-evaluate the evidence base and better inform clinical decision-making for this high-risk population.7,12
In this context, we conducted a systematic review and meta-analysis of RCTs comparing initiation of oral anticoagulants versus avoidance of anticoagulation in patients with AF and a history of spontaneous ICH. We aimed to synthesize the current evidence to evaluate the safety and efficacy of oral anticoagulation, thereby providing updated and clinically relevant insights to guide management decisions and future guideline recommendations.
Materials and methods
Standard protocol approvals, registrations, and patient consents
This systematic review and meta-analysis was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO; registration number CRD420251135299, August 27, 2025). The study is reported in accordance with the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 13 Owing to its design as a systematic review and meta-analysis, institutional ethics approval and informed consent were not required.
Data sources, searches, and study selection
A systematic search of the literature was conducted based on the patient, intervention, comparison, and outcome model 14 to identify available RCTs enrolling adult patients with spontaneous ICH and AF (P: population) initiating oral anticoagulants (I: intervention) versus avoiding anticoagulants with or without placebo (C: control) and investigating the outcomes of interest as outlined below (O: outcome). The anticoagulants in the intervention group included any direct oral anticoagulant (DOAC, i.e., apixaban, rivaroxaban, dabigatran, or edoxaban) or vitamin K antagonists (VKA). Patients in the control group received standard care for patients with AF and ICH that could include no antithrombotic therapy or administration of antiplatelets, excluding anticoagulants or other experimental agents.
Ongoing trials, observational cohort studies, non-controlled studies, case series, and case reports were excluded. Commentaries, editorials, and narrative reviews were also excluded.
The literature search was performed independently by four reviewers (L.P., N.M.P., A.T., and E.Ba.). The electronic databases MEDLINE, Scopus, and ClinicalTrials.gov were searched, employing search strings that included the terms “intracerebral hemorrhage,” “atrial fibrillation,” and “anticoagulants.” The complete search algorithm is available in the Supplemental Material. No language or other restrictions were applied. Our search spanned from the inception of each database to August 28, 2025. To ensure completeness, reference lists of eligible studies and abstracts from international conferences were also screened. All identified records were independently evaluated by four reviewers (L.P., N.M.P., A.T., and E.Ba.), with discrepancies resolved through discussion with a fifth tie-breaking reviewer (G.T.).
Quality control, bias assessment, and data extraction
Risk of bias among eligible studies was independently evaluated by four reviewers (L.P., N.M.P., M.C., and G.P.), employing the Cochrane Collaboration Risk-Of-Bias 2 tool (RoB 2) for RCTs. 15 Any disagreements were settled by consensus after discussion with a fifth author (G.T.).
Data extraction was performed in structured reports, including study name, country, recruitment period, intervention and comparison characteristics, the number of included patients, their baseline characteristics, including age, sex, CHA2DS2-VASc score, hematoma volume, and location of ICH, and the prespecified outcomes of interest.
Outcomes
The primary efficacy outcome of interest was the occurrence of a new ischemic stroke. The secondary efficacy outcome was the incidence of ischemic MACE, which was the composite of ischemic stroke, systemic arterial embolism, pulmonary embolism, or myocardial infarction.
The primary safety outcome was ICH recurrence. Secondary safety outcomes included the incidence of hemorrhagic MACE (comprising spontaneous ICH or major extracranial hemorrhage), all-cause mortality at follow-up, and cardiovascular death as defined in each study.
Statistical analysis
For the pairwise meta-analysis, risk ratios (RR) with corresponding 95% confidence intervals (CIs) were calculated for dichotomous outcomes, that is, the comparison of outcome events among patients receiving anticoagulants versus patients avoiding them. For every outcome of interest, pooled proportions with 95% CIs were estimated for both arms following application of the variance stabilizing double arcsine transformation. Furthermore, the absolute risk difference (ARD) with 95% CIs was calculated for the primary efficacy and the primary safety outcomes using the formula: ARD = [(RR × rate in no-AC group) − rate in AC group]. For the primary efficacy outcome, the number needed to treat (NNT) was calculated using the formula: NNT =
The threshold for statistical significance for the above analyses was set at a two-sided p-value of <0.05. Heterogeneity was assessed with the I2 and Cochran’s Q test. For the qualitative interpretation of heterogeneity, I2 values <25%, between 25% and 50%, and >50% were considered to represent low, moderate, and significant heterogeneity, respectively. The significance level for the Q statistic was set at <0.1. Small-study effects, as a proxy for publication bias, were assessed when at least four studies were included in the analysis of the outcomes of interest, using both funnel plot inspection and Egger’s linear regression test. 19 Prediction intervals (PI) were also calculated for all outcomes of interest. We conducted a prespecified leave-one-out sensitivity analysis to assess the robustness of the pooled estimates by sequentially excluding each study from the meta-analysis. In addition, we generated Baujat plots for each outcome to assess the contribution of individual studies to overall heterogeneity and pooled effect estimates. 20
The above statistical analyses were performed using the R software version 4.4.2 (package: meta). 21
Data availability statement
All data generated or analyzed in this study are included in this article and its Supplemental Information Files.
Results
Literature search and included studies
The systematic database search yielded a total of 103, 113, and 113 records from the MEDLINE, SCOPUS, and ClinicalTrials.gov databases, respectively (Figure 1). After excluding duplicates and initial screening, we retrieved the full text of 11 records that were considered potentially eligible for inclusion. After reading the full-text articles, seven were further excluded (Supplemental eTable 1). Furthermore, after searching the reference lists of published articles and international conference abstracts, two additional records were considered potentially eligible, and they were finally included. Finally, a total of six eligible RCTs met the inclusion criteria for this systematic review and meta-analysis (Table 1).7,12,22–25 Regarding the ELDERCARE trial, which enrolled high-risk elderly population with AF, we restricted inclusion to the subgroup of participants with a history of ICH for the present meta-analysis. 23 Data for this subgroup were obtained from the COCROACH individual participant data meta-analysis, which provided subgroup-level outcome data for participants with prior ICH. 11 STATICH consisted of two parallel randomized trials: one evaluating the initiation of antiplatelet therapy in patients with vascular disease and another evaluating oral anticoagulant therapy in patients with AF. 12 For this meta-analysis, we included only the anticoagulant arm of STATICH, as it specifically addresses the population and intervention of interest, that is, patients with AF who survived ICH and were randomized to resume oral anticoagulation or avoid anticoagulation. For STATICH, data were extracted from the presentation of the study at the European Stroke Organisation Conference 2025. In all included RCTs, the control arm permitted the use of antiplatelet agents at the investigators’ discretion, except for ELDERCARE-AF, in which participants in the control group received a placebo. Regarding follow-up duration, although it varied across trials, all studies required at least 6 months of follow-up, and the median follow-up period was broadly similar, ranging from approximately 1.2 to 2 years, supporting reasonable comparability of outcome capture across studies. This systematic review and meta-analysis included a total of 798 patients with a history of spontaneous ICH. Participants were assigned to two treatment strategies: initiation of oral anticoagulants (n = 403; mean age 78.5 years; 37% female; mean CHA2DS2-VASc score 4.2; 40% with lobar ICH; mean volume of ICH 7.4 mL; mean time from ICH to enrollment 91 days) or avoidance of anticoagulation (n = 395; mean age 78.2 years; 37% female; mean CHA2DS2-VASc score 4.1; 35% with lobar ICH; mean volume of ICH 6.4 ml; mean time from ICH to enrollment 96 days). Full baseline characteristics of the included trials, including raw counts by study arm and sub-study, are summarized in Table 1 and presented in the Supplemental Material (Supplemental eFigures 1–6). There were no significant differences in baseline characteristics between the two groups.
Baseline characteristics of the included studies.
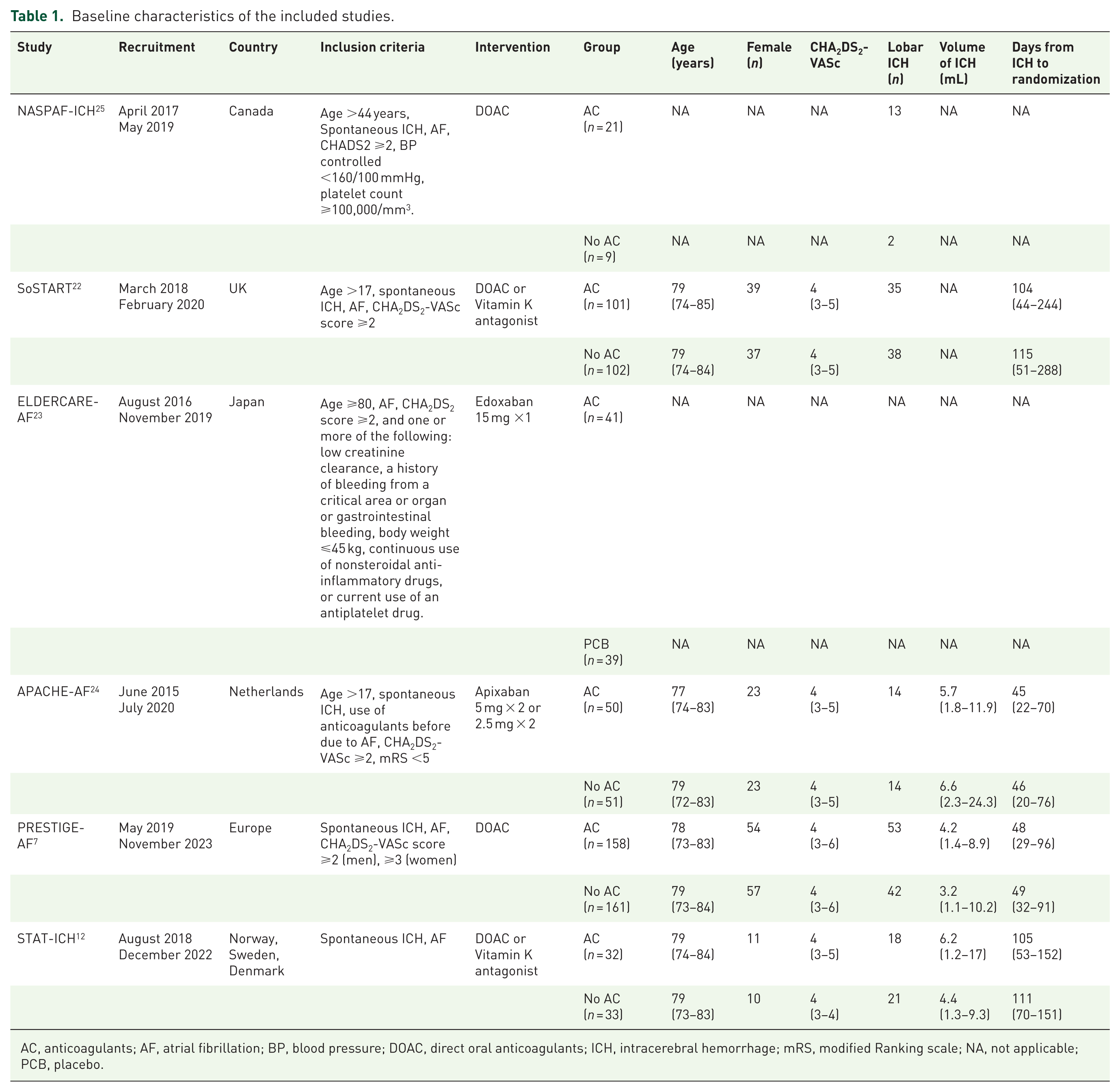
AC, anticoagulants; AF, atrial fibrillation; BP, blood pressure; DOAC, direct oral anticoagulants; ICH, intracerebral hemorrhage; mRS, modified Ranking scale; NA, not applicable; PCB, placebo.
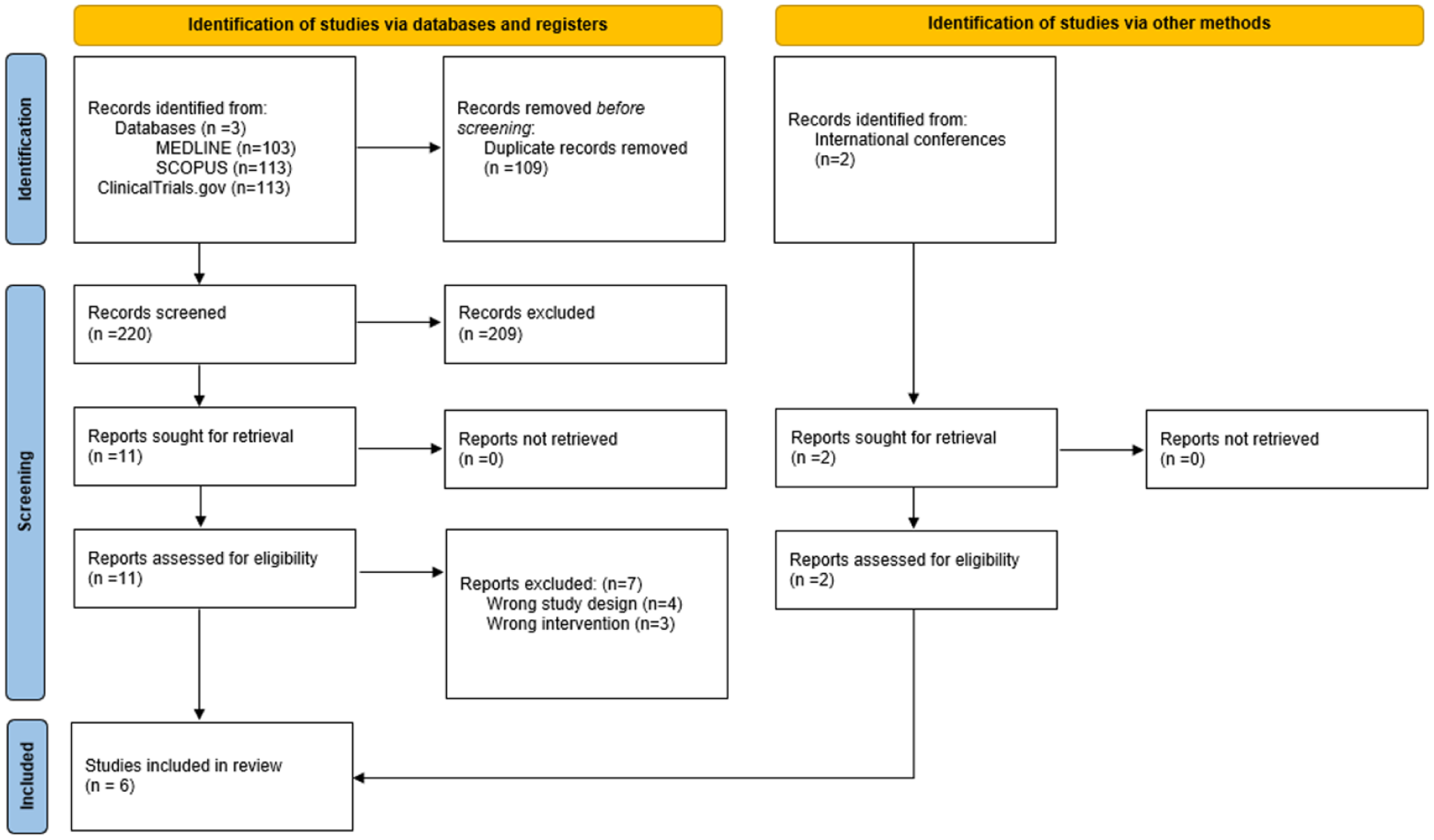
Flow chart of the systematic review.
Quality control of included studies
The risk of bias assessment for the included RCTs is summarized in Supplemental eFigure 7. All studies employed an appropriate randomization process, resulting in a low risk of bias for that domain. The ELDERCARE trial used a placebo in the control group and did not present deviations from the intended intervention, leading to a low risk of bias in this domain. 23 The remaining trials presented some deviations from the intended intervention, resulting in some concerns for this domain. Regarding missing outcome data, the PRESTIGE-AF and ELDERCARE trials had a proportion of patients who discontinued or withdrew consent, leading to some concerns of risk of bias in that domain.7,23 The remaining trials did not report any missing outcome data. All studies had a low risk of bias in the measurement of outcomes, as outcome assessors were blinded to treatment allocation. In addition, the risk of bias due to selective reporting was judged to be low across all included studies.
Quantitative analyses
An overview of the analyses for primary and secondary outcomes is presented in Table 2.
Overview of analyses for the outcomes of interest.

CI, confidence interval; MACE, major adverse cardiovascular event; NNH, number needed to harm; NNT, number needed to treat; RR, risk ratio.
Regarding the primary efficacy outcome, patients receiving anticoagulants had significantly lower rates of new ischemic stroke compared to those avoiding anticoagulants (RR = 0.20; 95% CI: 0.06–0.72; p = 0.01; PI: 0.01–6.40; 5 studies; I2 = 60%; p for Cochran’s Q = 0.04; Figure 2). The ARD was −0.11 (95% CI: −0.13 to −0.04). The NNT was 9 (95% CI: 8–26). In the exploratory subgroup analysis comparing trials using DOACs only versus those allowing DOACs or VKAs, no significant difference was observed between subgroups for the primary efficacy outcome (p for subgroup differences = 0.84; Supplemental eFigure 8). Regarding timing from the index ICH to randomization, the exploratory subgroup analysis demonstrated a statistically significant benefit among patients randomized >8 weeks after ICH (p for subgroup difference <0.01; Supplemental eFigure 9). Concerning secondary efficacy outcomes, the rates of ischemic MACE were significantly lower for patients receiving anticoagulants compared to those avoiding them (RR = 0.41; 95% CI: 0.23–0.75; p = 0.004; PI: 0.11–1.59; 5 studies; I2 = 32%; p for Cochran’s Q = 0.21; Figure 3).
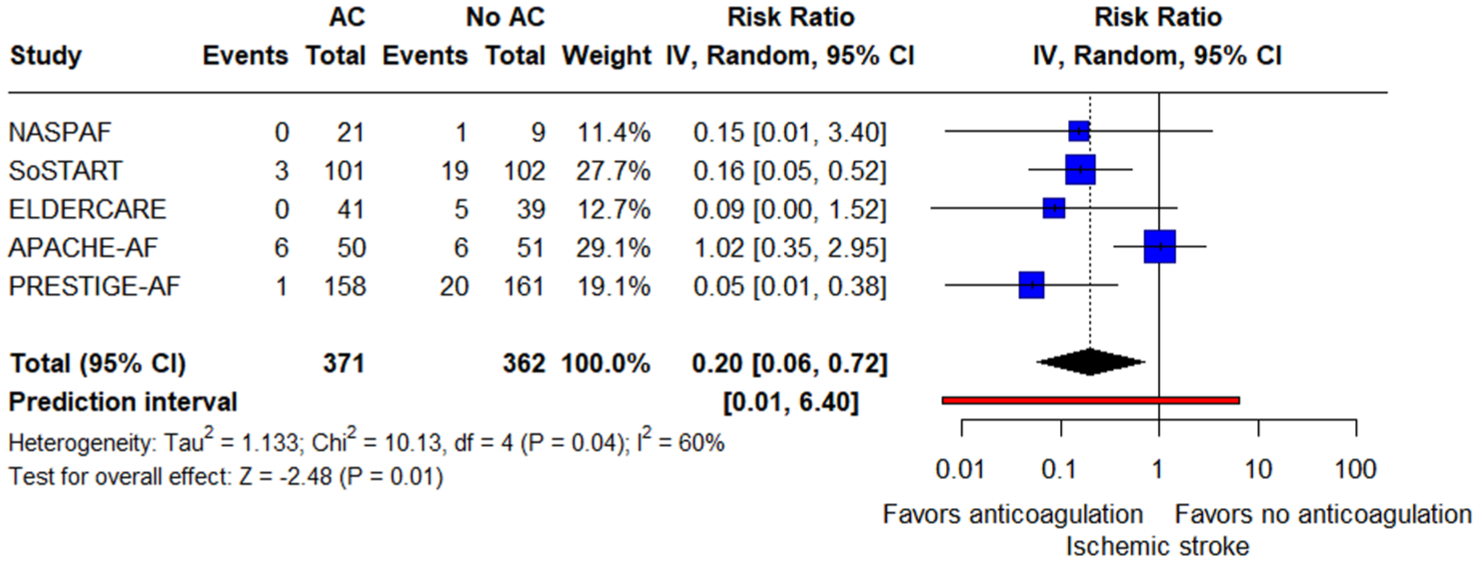
Forest plot presenting the risk ratio of new ischemic stroke among patients treated with oral AC versus patients treated without oral AC (no AC).
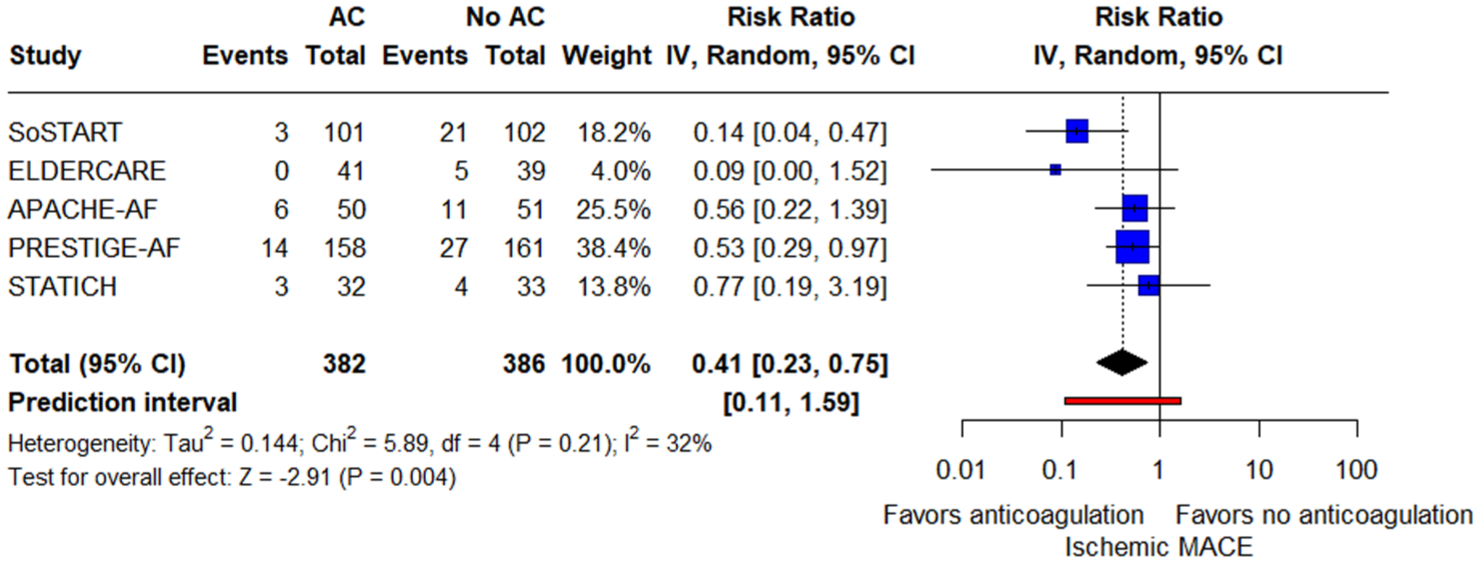
Forest plot presenting the risk ratio of ischemic major adverse cardiovascular events among patients treated with oral AC versus patients treated without oral AC (no AC).
Regarding the primary safety outcome, the rate of new ICH was significantly higher among patients who started with anticoagulants (RR = 3.14; 95% CI: 1.41–7.01; p = 0.005; PI: 0.85–11.57; 5 studies; I2 = 0%; p for Cochran’s Q = 0.45; Figure 4). The ARD was 0.05 (95% CI: 0.01–0.13). The NNH was 19 (95% CI: 8–109). In the exploratory subgroup analysis comparing trials using DOACs only versus those allowing DOACs or VKAs, no significant difference was observed between subgroups for the primary safety outcome (p for subgroup differences = 0.13; Supplemental eFigure 10). Similarly, no significant subgroup difference was observed regarding time from ICH to randomization for the primary safety outcome (p for subgroup differences = 0.81; Supplemental eFigure 11). Finally, there were no significant differences between lobar and non-lobar index ICH in the exploratory subgroup analysis stratified by ICH location (p for subgroup differences = 0.87; Supplemental eFigure 12).
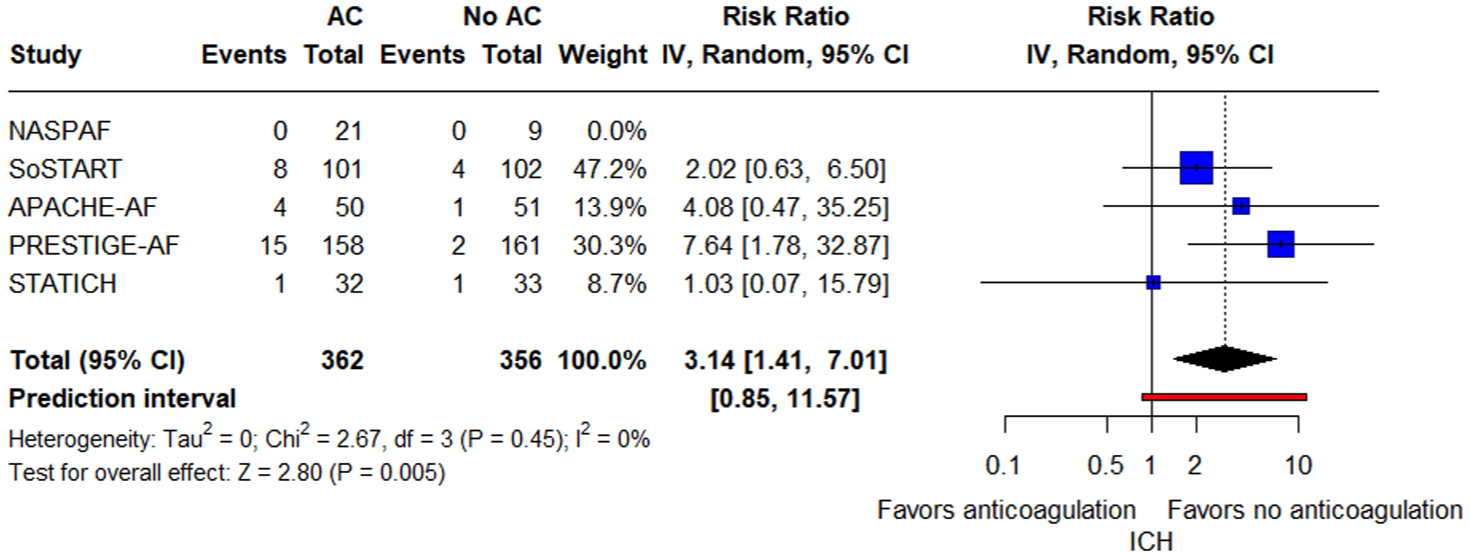
Forest plot presenting the risk ratio of new intracranial hemorrhage among patients treated with oral AC versus patients treated without oral AC (no AC).
For secondary safety outcomes, the rates of hemorrhagic MACE were significantly higher in the group of patients who started anticoagulants (RR = 2.35; 95% CI: 1.32–4.21; p = 0.004; PI: 1.07–5.20; 6 studies; I2 = 1%; p for Cochran’s Q = 0.41; Figure 5). Regarding all-cause mortality at follow-up, the rates did not differ between the two groups (RR = 1.06; 95% CI: 0.69–1.64; p = 0.79; PI: 0.42–2.68; 6 studies; I2 = 28%; p for Cochran’s Q = 0.23; Figure 6). Similarly, there was no significant difference in cardiovascular death between the two groups (RR = 0.98; 95% CI: 0.34–2.87; p = 0.976; PI: 0.02–52.62; 4 studies; I2 = 63%; p for Cochran’s Q = 0.07; Figure 7).

Forest plot presenting the risk ratio of hemorrhagic major adverse cardiovascular events among patients treated with oral AC versus patients treated without oral AC (no AC).
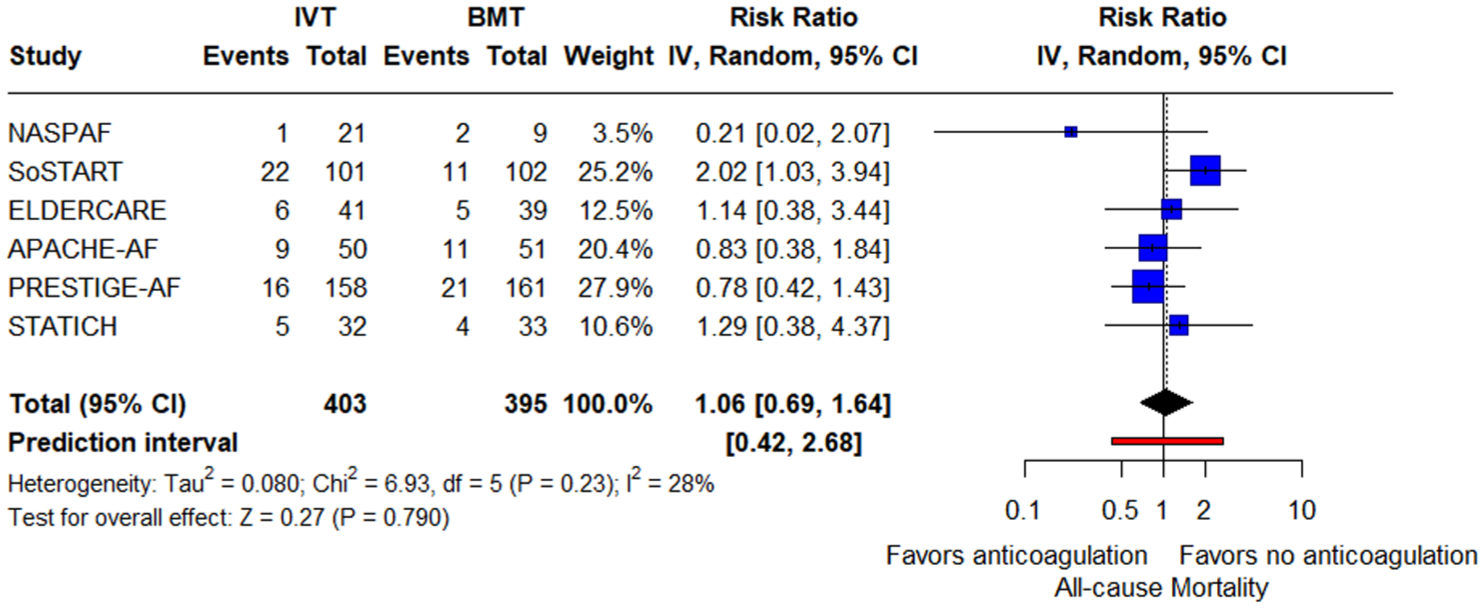
Forest plot presenting the risk ratio of all-cause mortality at follow-up among patients treated with oral AC versus patients treated without oral AC (no AC).

Forest plot presenting the risk ratio of cardiovascular death among patients treated with oral AC versus patients treated without oral AC (no AC).
The pooled proportions per arm for each outcome of interest are presented in Supplemental eTable 2 in the Supplemental Material.
Regarding the leave-one-out sensitivity analyses for the primary efficacy outcome, pooled RRs ranged between 0.12 and 0.29 with I2 = 0%–70%; the effect remained directionally consistent but lost nominal significance when omitting SoSTART (RR = 0.20; 95% CI: 0.03–1.17) or PRESTIGE-AF (RR = 0.29; 95% CI: 0.08–1.06). Excluding APACHE-AF eliminated between-study heterogeneity (RR = 0.12; 95% CI: 0.05–0.29; I2 = 0%). For ischemic MACE, results were robust across all omissions (RR: 0.34–0.53; I2 = 0%–46%), with statistical significance retained. For recurrent ICH, the association persisted across most leaves (RR: 3.02–3.67; I2 = 0%–23%) but became non-significant when omitting PRESTIGE-AF (RR = 3.14, 95% CI: 0.82–5.59). For hemorrhagic MACE, estimates ranged RR = 1.68–2.61; I2 = 0%–20% and lost significance only when PRESTIGE-AF was excluded (RR = 1.68, 95% CI: 0.81–3.46). All-cause mortality remained non-significant throughout (RR = 0.85–1.19; I2 = 0%–42%), as did cardiovascular death (RR = 0.59–1.57; I2 = 0%–81%), with omission of SoSTART reducing heterogeneity (I2 = 0%) but not altering conclusions. Sensitivity analyses are presented in Supplemental eFigures 13–18. To complement these analyses, we generated Baujat plots to visualize the contribution of individual trials to overall heterogeneity and pooled effects, yielding similar results (Supplemental eFigures 19–24). Regarding the assessment of publication bias, funnel plots were broadly symmetric for all-cause mortality, recurrent ICH, and ischemic MACE. For ischemic stroke, visual inspection suggested asymmetry driven by APACHE-AF, although Egger’s test was not significant (p = 0.27). Hemorrhagic MACE showed significant asymmetry (p = 0.02), indicating possible small-study effects or publication bias (Supplemental eFigures 25–29). A trim-and-fill analysis was conducted and imputed two hypothetical studies and produced an adjusted pooled RR of 2.64 (95% CI, 1.33–5.26), which was nearly identical to the unadjusted estimate, indicating that the observed asymmetry had minimal influence on the overall result (Supplemental eFigure 30).
Discussion
In this systematic review and meta-analysis of 6 RCTs comprising 798 patients with a history of spontaneous ICH and indication for anticoagulants due to AF, we found that oral anticoagulants were associated with a significantly lower likelihood of occurrence of new ischemic stroke (NNT = 9) and ischemic MACE. Oral anticoagulants were linked to an increased risk of a new ICH (NNH = 19), and hemorrhagic MACE, but they were not associated with a significantly higher risk of cardiovascular death or all-cause mortality at follow-up. Overall, the results suggest competing risks: a significant reduction in ischemic outcomes with a concomitant increase in hemorrhagic events, with no demonstrable early mortality difference.
Regarding efficacy outcomes, resuming oral anticoagulation in AF patients with prior spontaneous ICH produced a large relative reduction in ischemic events, including new ischemic stroke (RR 0.20; 95% CI: 0.06–0.72; NNT = 9) and ischemic MACE (RR 0.41; 95% CI: 0.23–0.75). This effect translates into a meaningful absolute benefit for this population in the prevention of stroke. Although early mortality was unchanged, the prevention of ischemic events is likely to confer substantial functional gains. Accordingly, in patients whose hemorrhagic risk is not prohibitive, these efficacy data support considering anticoagulation as part of individualized, shared decision-making.
Regarding safety outcomes, anticoagulation increased recurrent ICH (RR 3.14; 95% CI 1.41–7.01; NNH 19) and hemorrhagic MACE (RR 2.35; 95% CI 1.32–4.21), without excess cardiovascular death or 3-month all-cause mortality. On point estimates, the NNH (19) is roughly double the NNT (9), implying that benefit events may be about twice as frequent as serious harms over comparable horizons. This suggests a net clinical benefit on average, while acknowledging wide CIs and the need to weigh outcome severity in patient-centered decisions. These outcomes also differ in clinical weight and impact: ischemic strokes after anticoagulation withdrawal vary in severity but could often be disabling or fatal, whereas recurrent ICH, although less frequent, can have devastating consequences for affected patients. Thus, numerical comparisons of NNT and NNH should be interpreted in light of the differing functional and prognostic burden of these events. Our exploratory subgroup analysis detected no statistically significant difference by index ICH location (lobar vs non-lobar), but this inference is limited by data available from only two trials.7,24
Across outcomes, between-trial heterogeneity ranged from minimal to substantial (Table 2). Effects on ischemic stroke were the most variable (moderate–substantial heterogeneity with a wide PI), the ischemic composite was more consistent (modest heterogeneity), and bleeding outcomes showed little statistical heterogeneity overall, despite wide PIs driven by few events. Mortality estimates were relatively consistent, whereas cardiovascular death was notably heterogeneous. Leave-one-out analyses suggested that the ischemic stroke signal attenuated when omitting SoSTART or PRESTIGE-AF, and the significance for recurrent ICH and hemorrhagic MACE depended in part on PRESTIGE-AF; ischemic MACE was robust across omissions. The disproportionate influence of PRESTIGE-AF and SoSTART likely reflects their larger sample sizes, broader inclusion criteria, and differences in restart timing and anticoagulant selection. PRESTIGE-AF enrolled a multicenter European cohort with earlier restart and exclusive DOAC use, whereas in SoSTART, 97 of 101 participants in the anticoagulation arm received a DOAC and 4 received a VKA, reflecting the predominance of modern agents. These design and pharmacological differences plausibly explain their greater statistical influence, whereas smaller pilot trials contributed minimally to overall variability. Baujat plots confirmed that APACHE-AF contributed most to overall heterogeneity, whereas PRESTIGE-AF and SoSTART exerted the greatest influence on pooled efficacy and safety estimates. Quantitatively, exclusion of these high-impact trials shifted pooled RRs for ischemic stroke from 0.20 (95% CI 0.06–0.72) to 0.20 (0.03–1.17) and 0.29 (0.08–1.06), respectively, and for hemorrhagic MACE from 2.35 (1.32–4.21) to 1.68 (0.81–3.46). Visual inspection and Egger’s testing suggested possible small-study effects for hemorrhagic MACE, while asymmetry for ischemic stroke was not statistically supported. Likely contributors include variation in eligibility (e.g., lobar vs deep ICH), timing of restart, agent/dose (DOAC vs VKAs; low-dose regimens), allowance of antiplatelets, and follow-up durations. In addition, although Egger’s test indicated possible funnel plot asymmetry for hemorrhagic MACE, this finding is based on a small number of trials and should be interpreted cautiously. The trim-and-fill method imputed two hypothetical studies but did not materially alter the pooled estimate, suggesting that the observed asymmetry likely reflects small-study variability rather than publication bias. Given the limited number of studies and sparse events, such analyses remain exploratory and unstable. We also acknowledge that the trim-and-fill should be interpreted cautiously, given the limited number of studies. Collectively, these sources of between-study variability and imprecision caution against overgeneralization and underscore the need for larger, adequately powered RCTs to provide definitive estimates and to refine strategies for anticoagulation after ICH.
PIs were calculated to reflect the expected range of true effects in a new, comparable population. The wide PI observed for ischemic stroke (0.01–6.40) indicates that, while the average pooled estimate favors anticoagulation, the true effect could vary considerably across settings. This variability reflects both statistical uncertainty and real-world heterogeneity in trial populations, restart timing, and clinical practices. Consequently, although the overall direction of evidence supports anticoagulation in appropriately selected patients, the findings should be applied cautiously at the site level, with recognition that individual patient characteristics and management patterns may meaningfully influence the balance of risks and benefits. By contrast, the narrower PIs for ischemic MACE (0.11–1.59) and hemorrhagic MACE (1.07–5.20) suggest more consistent associations across trials. For recurrent ICH, the PI (0.85–11.57) remained wide, underscoring residual uncertainty in bleeding risk estimation. The PIs for all-cause mortality (0.42–2.68) and cardiovascular death (0.02–52.62) encompass both potential benefit and harm, aligning with the non-significant pooled effects for these outcomes. Collectively, these intervals emphasize that while pooled RRs provide a summary estimate, true effects may vary substantially between clinical settings, reinforcing the importance of individualized, context-specific decision-making when applying these results.
Several large ongoing trials are positioned to clarify agent choice, timing, and net clinical benefit after ICH. ENRICH-AF (NCT03950076) is an event-driven, multinational study evaluating edoxaban in AF survivors of ICH, initiating therapy ⩾14 days after the index event; its primary endpoint is time to first ischemic stroke, hemorrhagic stroke, or systemic embolism. ASPIRE (NCT03907046) is a randomized, double-blind, phase III U.S. trial comparing apixaban with aspirin in patients with recent ICH and non-valvular AF, with planned 12–36-month follow-up. A3-ICH (NCT03243175) will further test apixaban with left atrial appendage closure (LAAO) or usual care without anticoagulants and has a projected population of 300 patients with AF and prior history of ICH. Together, these trials should deliver more precise estimates of both thromboembolic benefit and hemorrhagic risk, enable stratified analyses by hemorrhage subtype and timing of restart, and help define practical strategies like agent selection, dose, and initiation window for anticoagulation after ICH. More adequately powered RCTs of this nature remain essential to provide definitive guidance.
Our findings are consistent with the ESO 2025 guideline stance that the net long-term benefit remains uncertain and that enrollment in RCTs is encouraged, and they align with AHA/ASA 2022 recommendations that resumption at 7–8 weeks may be reasonable after individualized assessment.5,6 In addition, our results are in line with the latest meta-analysis by D’Anna et al., 10 which likewise found lower ischemic stroke risk with anticoagulation alongside higher recurrent ICH risk, and no clear differences in all-cause or cardiovascular mortality. Compared with that study (4 RCTs, n = 653), our analysis integrates a broader and more contemporary evidence base by including two additional randomized datasets not incorporated previously—the ICH subgroup of ELDERCARE-AF 23 and the oral anticoagulation arm of STATICH 12 —resulting in eight eligible RCTs and nearly 800 participants. We also applied harmonized composite outcomes (ischemic and hemorrhagic MACE) to enhance clinical interpretability, calculated absolute measures of effect (ARD, NNT/NNH), and used PIs and prespecified leave-one-out sensitivity analyses to assess between-study variability and robustness. These differences in eligibility criteria, outcome definitions, and analytic framework likely explain modest divergences in effect estimates compared with D’Anna et al. while providing a more comprehensive and up-to-date synthesis of randomized evidence. Taken together, our synthesis supports guideline-consistent, patient-centered decision-making while underscoring the need for ongoing adequately powered RCTs to refine definitive strategies for agent selection, dose, and timing after ICH.
Decisions regarding anticoagulation after ICH should be grounded in rigorous phenotyping and structured risk appraisal. In ENRICH-AF, a safety review mandated withdrawal of participants with lobar ICH or non-aneurysmal convexity subarachnoid hemorrhage (cSAH) because of an excess risk of recurrent intracranial bleeding, underscoring the vulnerability of these phenotypes. 26 Patients with probable cerebral amyloid angiopathy (CAA) which is typically characterized by lobar hemorrhage, cortical superficial siderosis, cSAH, and a high burden of lobar cerebral microbleeds carry a particularly high risk of rebleeding and, in general, should not receive oral anticoagulation.26,27 Additional features that weigh against resumption include a very heavy microbleed burden (e.g., ⩾10 cerebral microbleeds (CMB) in CAA-unrelated ICH or ⩾5 lobar CMBs in CAA-related disease), uncontrolled hypertension, and chronic alcohol misuse, according to the ESO guidelines. 5 Risk scores can inform but must not dictate decisions: CHA2DS2-VASc quantifies thromboembolic risk, whereas HAS-BLED provides a bleeding framework but has important limitations (development primarily in VKA-treated cohorts, modest predictive performance, and omission of key neuroimaging markers).28,29 Accordingly, patient selection should integrate clinical variables like age, blood pressure control, comorbidities, ICH mechanism and location, and MRI biomarkers (microbleeds, superficial siderosis, cSAH) within a multidisciplinary evaluation, favoring anticoagulation chiefly in non-CAA, deep hypertensive ICH survivors with well-controlled risk factors and low microbleed burden, or high risk of thromboembolic events (mechanical valve or LVAD) and generally avoiding it in CAA phenotypes.5,6
The timing of anticoagulation resumption after ICH should be individualized and contingent on radiographic stability of the hematoma, strict blood-pressure control, and the presumed ICH mechanism.30,31 Current guidance supports a cautious delay, with the AHA/ASA suggesting consideration at 7–8 weeks in carefully selected patients. 6 In our exploratory subgroup analysis, timing of anticoagulation reinitiation appeared to influence ischemic risk reduction. Trials resuming therapy beyond 8 weeks after ICH showed a significant reduction in ischemic stroke, whereas earlier restart (⩽8 weeks) was not associated with benefit. However, these results derive from a few studies and should be interpreted with caution. No significant timing effect was observed for recurrent ICH. The findings suggest that later restart may favor efficacy without clearly increasing hemorrhagic risk, although adequately powered RCTs are needed to confirm this hypothesis.
Regarding agent and dose, DOACs are generally preferred over VKAs due to markedly lower intracranial bleeding rates in AF populations, better clinical and neuroimaging outcomes, and predominant DOAC use in contemporary RCTs.6,32–34 In our exploratory subgroup analysis, comparing studies in which patients received only DOACs versus those in which both DOACs and VKAs could be used, no significant differences were observed between the subgroups for either efficacy or safety outcomes. Notably, in SoSTART, only four participants received VKAs, and STATICH did not report the exact number of patients treated with VKAs. These findings are therefore limited by the small and uncertain exposure to VKAs across trials. Overall, the available evidence suggests that DOACs represent the preferred option for patients requiring anticoagulation after ICH. VKAs should be reserved for indications such as mechanical valves or moderate–severe rheumatic mitral stenosis. 6 Dosing should follow on-label criteria rather than empirical under-dosing, which compromises efficacy without clear safety gain.35,36
From a clinical perspective, our findings suggest that resuming oral anticoagulation after ICH may provide net benefit in appropriately selected patients with AF, primarily through the prevention of ischemic stroke and other thromboembolic events. However, the decision to restart should be individualized, taking into account the underlying ICH phenotype (lobar vs non-lobar), baseline hemorrhagic risk, comorbidities, and the patient’s competing ischemic risk profile. In particular, patients with a high thromboembolic burden (e.g., elevated CHA2DS2-VASc score) may derive greater absolute benefit, whereas those with markers of high hemorrhagic vulnerability may warrant a more conservative approach. Multidisciplinary evaluation integrating neurology, cardiology, and hematology perspectives remains essential to guide timing and agent selection.
Patients with AF suffered spontaneous ICH should first receive a structured non-pharmacologic secondary-prevention program including tight blood-pressure control, avoidance of heavy alcohol use, smoking cessation, weight management and treatment of sleep apnea, mitigation of fall risk, and minimization of unnecessary antithrombotics and nonsteroidal anti-inflammatory drugs, since these measures reduce recurrent ICH and overall vascular risk and are endorsed by contemporary ESO and AHA/ASA guidance.5,6 When the hemorrhagic risk is prohibitive and long-term oral anticoagulation is unsuitable, LAAO is the principal non-pharmacologic option for cardioembolic stroke prevention.37–39 Contemporary guidance from ESO recommendations questions the certainty of benefit in AF patients with prior ICH history, endorses considering LAAO when long-term oral anticoagulation is unsuitable, and encourages enrollment in RCTs. 5 Randomized evidence comparing LAAO with oral anticoagulation in this population remains limited; phase III trials are ongoing or have been recently completed, including A3-ICH (NCT03243175) and CLEARANCE (NCT04298723), while CLOSURE-AF (NCT03463317) has finished recruitment and STROKECLOSE (NCT02830152) is suspended. LAAO should be embedded within comprehensive secondary prevention that addresses modifiable determinants of hemorrhage and vascular risk.5,6,40 Collectively, these non-pharmacologic measures complement LAAO and may meaningfully reduce recurrent ICH and overall vascular events when long-term anticoagulation is not pursued.
Future trials should be adequately powered and pragmatic, enroll across the full spectrum of ICH phenotypes rather than excluding the highest-risk groups, and prespecify stratification by ICH location and CAA biomarkers; compare DOACs against both anticoagulation avoidance and LAAO; rigorously test timing (earlier vs later restart) and dosing strategies; standardize core outcomes that include functional status, health-related quality of life, and long-term mortality; mandate central imaging with harmonized definitions; and incorporate adaptive or event-driven designs with transparent safety monitoring.41,42
Our present meta-analysis has several methodological strengths. It synthesizes randomized evidence only, minimizing confounding and selection bias inherent to observational designs and providing the most reliable estimates available for AF patients after spontaneous ICH. To our knowledge, this is the only meta-analysis to date that includes and synthesizes the latest, complete set of randomized data, incorporating all contemporary trials on resuming oral anticoagulation after spontaneous ICH in patients with AF. Clinically interpretable metrics were emphasized through the calculation of NNT/NNH and pooled proportions, as well as prespecified leave-one-out analyses for every outcome to assess stability. We also reported 95% PIs to reflect the range of true effects expected in a new, comparable population.
Despite its strengths, there are also important limitations in this study to consider. This is a study-level meta-analysis, and the synthesis is constrained by the small number of trials and modest sample size, resulting in imprecision, wide PIs, and limited power for the outcomes. Trial designs varied with respect to eligibility criteria, timing of restart, agent and dose, concomitant antiplatelet use, and follow-up duration, contributing to between-study heterogeneity. Follow-up duration varied across the included trials, ranging from approximately 6 to 24 months. Such heterogeneity in observation windows may have influenced event capture and comparability between studies and should be considered when interpreting pooled estimates. Inference for certain endpoints was sensitive to single trials in leave-one-out analyses, and small-study effects were suggested for hemorrhagic MACE. Another limitation of our study is the lack of standardized functional outcomes across trials, such as 90-day modified Rankin Scale, longer-term health-related quality of life, or cognitive measures. This constraint limits adjudication of net clinical benefit, especially given the neutral early mortality signal, and shifts interpretation toward event counts rather than functional independence and quality of life. Subgroup analyses based on anticoagulant type, timing from the index ICH to randomization, and index ICH location were exploratory and derived from a limited number of studies. Results should be interpreted with caution, and these analyses serve primarily as hypothesis-generating evidence. Finally, generalizability is constrained by trial selection and protocol amendments; therefore, applicability to younger populations, varied health systems, or higher-risk phenotypes remains uncertain.
Conclusion
This meta-analysis of randomized data indicates that, in survivors of spontaneous ICH with AF, oral anticoagulation reduces the risk of ischemic stroke but increases the risk of recurrent ICH and major hemorrhage, with no clear early mortality difference. Importantly, the NNT was approximately half the NNH, suggesting that, on average, the potential benefits may outweigh the risks in carefully selected patients. However, residual uncertainty remains due to heterogeneity, wide PIs, and sensitivity to individual trials. This underscores the need for a patient-centered strategy that balances stroke prevention against the potential for serious bleeding, guided by rigorous phenotyping and multidisciplinary shared decision-making. Future randomized trials and individual-patient data meta-analysis are essential to optimize timing, agent and dosing selection, and to refine non-pharmacologic alternatives for this vulnerable population.
Supplemental Material
sj-docx-2-tan-10.1177_17562864251406065 – Supplemental material for Safety and efficacy of oral anticoagulation in patients with intracranial hemorrhage and atrial fibrillation: a systematic review and meta-analysis of randomized controlled clinical trials
Supplemental material, sj-docx-2-tan-10.1177_17562864251406065 for Safety and efficacy of oral anticoagulation in patients with intracranial hemorrhage and atrial fibrillation: a systematic review and meta-analysis of randomized controlled clinical trials by Nikolaos M. Papageorgiou, Lina Palaiodimou, Aikaterini Theodorou, Eleni Bakola, Maria Chondrogianni, Georgia Papagiannopoulou, Apostolos Safouris, Eleni Anagnou, Panagiota-Eleni Tsalouchidou, Effrosyni Koutsouraki, Theodore Karapanayiotides, Efstathios Boviatsis, Christos Krogias, Sotirios Giannopoulos, Diana Aguiar de Sousa, Mira Katan, Thorsten Steiner and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-1-tan-10.1177_17562864251406065 – Supplemental material for Safety and efficacy of oral anticoagulation in patients with intracranial hemorrhage and atrial fibrillation: a systematic review and meta-analysis of randomized controlled clinical trials
Supplemental material, sj-pdf-1-tan-10.1177_17562864251406065 for Safety and efficacy of oral anticoagulation in patients with intracranial hemorrhage and atrial fibrillation: a systematic review and meta-analysis of randomized controlled clinical trials by Nikolaos M. Papageorgiou, Lina Palaiodimou, Aikaterini Theodorou, Eleni Bakola, Maria Chondrogianni, Georgia Papagiannopoulou, Apostolos Safouris, Eleni Anagnou, Panagiota-Eleni Tsalouchidou, Effrosyni Koutsouraki, Theodore Karapanayiotides, Efstathios Boviatsis, Christos Krogias, Sotirios Giannopoulos, Diana Aguiar de Sousa, Mira Katan, Thorsten Steiner and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
