Abstract
Background:
Oral corticosteroids (OCS) are commonly used in the management of autoimmune Myasthenia Gravis (MG), often in high doses for prolonged periods. Exposure to OCS is associated with significant and cumulative adverse effects. There is currently no universal consensus on the approach to OCS use in MG.
Objectives:
To reach a multinational consensus on the appropriate use of OCS in MG, including the role of treatment approaches to minimize dose and support tapering.
Design:
This modified Delphi study established consensus among a panel of 70 general neurologists/neuromuscular specialists from eight countries over two rounds of survey to establish best practice principles regarding the use of OCS in MG.
Methods:
The current literature on OCS use in MG was reviewed. Topics of interest were identified, and a modified Delphi consensus process was created. A steering committee of six experts in MG proposed statements for testing with the wider panel over two rounds of surveys. Consensus was reached if at least 75% agreed or strongly agreed on a 4-point Likert scale.
Results:
Consensus was achieved for all 37 final statements. These statements covered principles of OCS use, including a target for long-term use of ⩽5 mg/day; the role of biologic treatments in minimizing required OCS dose; the need for individualization of approach according to patient factors; and the need for detailed guidance regarding how and when to taper OCS dose.
Conclusion:
These findings reinforce a shift in the management of MG, advocating for the judicious and sparing use of OCS against the backdrop of expanding therapeutic options. As additional evidence-based data emerge, these recommendations should be updated.
Plain language summary
A steering committee of 6 Myasthenia Gravis (MG) experts proposed statements for testing with a wider panel of 70 neurologists/neuromuscular specialists from 8 countries, as part of a modified Delphi process. Consensus was reached for 37 statements covering principles of oral corticosteroid (OCS) use, including a target for long-term use of no more than 5 mg/day; the role of biologic treatments in minimizing required OCS dose and supporting a fast OCS taper (within 4 months); the need for individualization of approach; and the need for greater guidance regarding how and when to taper OCS dose. Key findings include agreement that OCS target dose should be 5 mg/day or lower for most patients with MG, and where OCS use is necessary to maintain symptom control. Alternative treatments with steroid-sparing effects should be used to achieve this target.
Introduction
Autoimmune myasthenia gravis (MG) is a rare neuromuscular disorder caused by the development of autoantibodies targeting the neuromuscular junction and activating the complement system. 1 The severity and extent of muscle weakness vary significantly among individuals with MG, and with symptoms that typically fluctuate in severity over time. 2
The prevalence of MG ranges from 150 to 200 cases per million, and incidence rates have a bimodal distribution in women, with peaks around age 30 and 50. MG incidence below 40 years is more common in women, with a 3:1 ratio compared to men. At age 40–50, incidence is broadly comparable; after age 50, men have a greater incidence at a ratio of 3:2 compared to women. 3
MG that includes muscle weakness outside the distribution of the eyelid and the muscles of eye movement is referred to as generalized MG (gMG), which is highly variable between individuals and ranges from isolated weakness of bulbar muscles to widespread muscle weakness and respiratory failure. 2
MG is currently incurable, but treatments are available to gain initial control of symptoms and to maintain this once the patient’s disease is considered stable. Acetylcholinesterase inhibitors (AChEIs) are often the first treatment to be initiated. If response to AChEIs proves insufficient, immunosuppressive therapies including oral corticosteroids (OCS) and/or nonsteroidal immunosuppressants such as azathioprine and cyclosporine have formed the traditional mainstay of treatment, 4 with high-dose OCS being used often for prolonged durations. 5
Exposure to OCS is associated with systemic side effects, 6 and caution is required in patients with risk factors for pre-existing conditions that may potentially be exacerbated by exposure, such as diabetes, dyslipidemia, cardiovascular disease (CVD), gastrointestinal (GI) disorders, affective disorders, or osteoporosis. 7 Therefore, the established principle of OCS use in gMG is to use as necessary to gain control of symptoms, and then taper the dose to the minimum level required to maintain control.8–10 Once symptom control is achieved, Shelly et al. recommend reducing the OCS dose to below 10 mg prednisone equivalent per day, with an optimal daily dose of 5–7.5 mg per day. 11 In Japan, the daily OCS dose is recommended to be 5 mg/day or lower. 5
Alternate-day corticosteroid dosing is a well-established practice in MG, especially ocular and milder cases, for balancing efficacy with reduced adverse effects.12–14 While this approach has been used in practice for years, Mantegazza et al. 15 suggest that alternate-day dosing is only warranted to manage short-term adverse events related to OCS therapy. While the use of alternate-day dosing regimens can reduce OCS-associated adverse events, it still involves the (potentially chronic) use of OCS at therapeutic levels, and so the risk of side effects remains.
Targeted therapies, in addition to established nonsteroidal immunosuppressive agents, provide an opportunity to consider how OCS should be optimally used in gMG and how associated side effects may be avoided. 1
Due to a lack of clear evidence, current guidelines offer a general approach but lack detail regarding how the full treatment armamentarium can be employed to achieve treatment goals and minimize potential side effects.8,16 Given these limitations, a modified Delphi approach was agreed to establish an international consensus on the appropriate use of OCS in the management of MG. The outputs are intended to build upon both the evolving evidence base regarding OCS exposure and the development of newer, targeted immune therapies, and to prompt neurologists and neuromuscular specialists to consider how MG can best be managed in their local health system.
Methods
This was a non-interventional modified Delphi consensus (Figure 1). In March 2024, a literature review on the topic of OCS use and tapering was performed using the PubMed and Cochrane databases, along with a general web search using Google (See Figure S1). The findings of the literature search were used to inform the agenda of a steering committee meeting held in March 2024 and to draft an initial set of statements.

Modified Delphi study design.
An international steering committee of MG specialists (all professors or associate professors of neurology/neuromuscular disease) convened in March 2024 to discuss current practice and associated issues regarding OCS use in MG. The steering committee was selected based on published research (including international guidelines) and experience in treating MG.
During the discussion, the steering committee agreed five main domains of focus:
A. Clinical considerations for OCS use in MG
B. Concomitant factors influencing OCS use
C. Dosage, duration, and long-term strategy of OCS use
D. Monitoring and treatment optimization
E. Future recommendations
These themes were discussed further, and consensus statements were agreed upon during the meeting. An electronic survey containing these initial statements was developed and sent to the steering committee, who independently rated the statements as either “accept”, “remove”, or “reword” with suggested changes. This resulted in 39 statements (Table S1) that were developed into a second 4-point Likert survey for distribution to a wider panel in Round 1 of the process.
Recruitment of panel members was based solely on recommendations from the steering committee from their professional peer groups. Panel members were eligible if they were experienced neurologists or neuromuscular specialists with direct experience of the treatment of MG, as determined by the individual steering group members. Those without current MG clinical responsibilities or with industry employment were excluded.
A total of 104 potential respondents were invited to act as panel members. This number was agreed to allow for responder rates of below 100% across two rounds of survey and achieve the target minimum of 50 responses for each round. No financial incentive was offered for participation. As MG is a rare disease, this method was chosen due to the relatively low number of MG specialists among neurologists, and we wished to ensure that responses were informed by a degree of expertise in the field and by geography. The survey presented each statement along with a 4-point Likert scale (“strongly agree,” “tend to agree,” “tend to disagree,” and “strongly disagree”) to allow respondents to indicate their corresponding level of agreement. The survey included an open-text response for panelists to explain any reasons for disagreement.
All feedback was collated and presented to steering committee members ahead of a second steering committee meeting. The survey captured additional demographic data, including country, role (neurologist or neuromuscular specialist), years in role, estimated number of MG patients treated annually, and sex (to ensure representation from both male and female respondents). All responses received were included for the final analysis. The survey was written in English and was not adapted for accessibility.
Stopping criteria for the survey stage (i.e., when no further survey rounds were required) were agreed as a target of 50 responses, two rounds of survey, and a threshold for consensus set at 75% for each statement (a commonly used threshold 17 ).
Completed surveys were analyzed to produce an overall agreement level (i.e., the number of respondents expressing agreement as a percentage of the overall number of respondents for each statement). While the identity of individuals invited to participate in the panel was known to the steering group, the identity of responders was not provided to the steering group to ensure objective analysis of the survey results. The facilitator retained email details of respondents to allow for survey reissue in subsequent rounds.
A briefing of results and any feedback received was sent to the steering committee for review ahead of the second meeting. The steering committee discussed any statements not meeting the threshold for consensus and agreed as a group whether to remove the statements from the final set or modify and reissue the survey for Round 2. For reporting, ACCORD (ACcurate COnsensus Reporting Document) guidelines were adhered to. 18
Results
Note in the following text statements are abbreviated to S (e.g., S1 = Statement 1, S2 = Statement 2 etc.)
Round 1
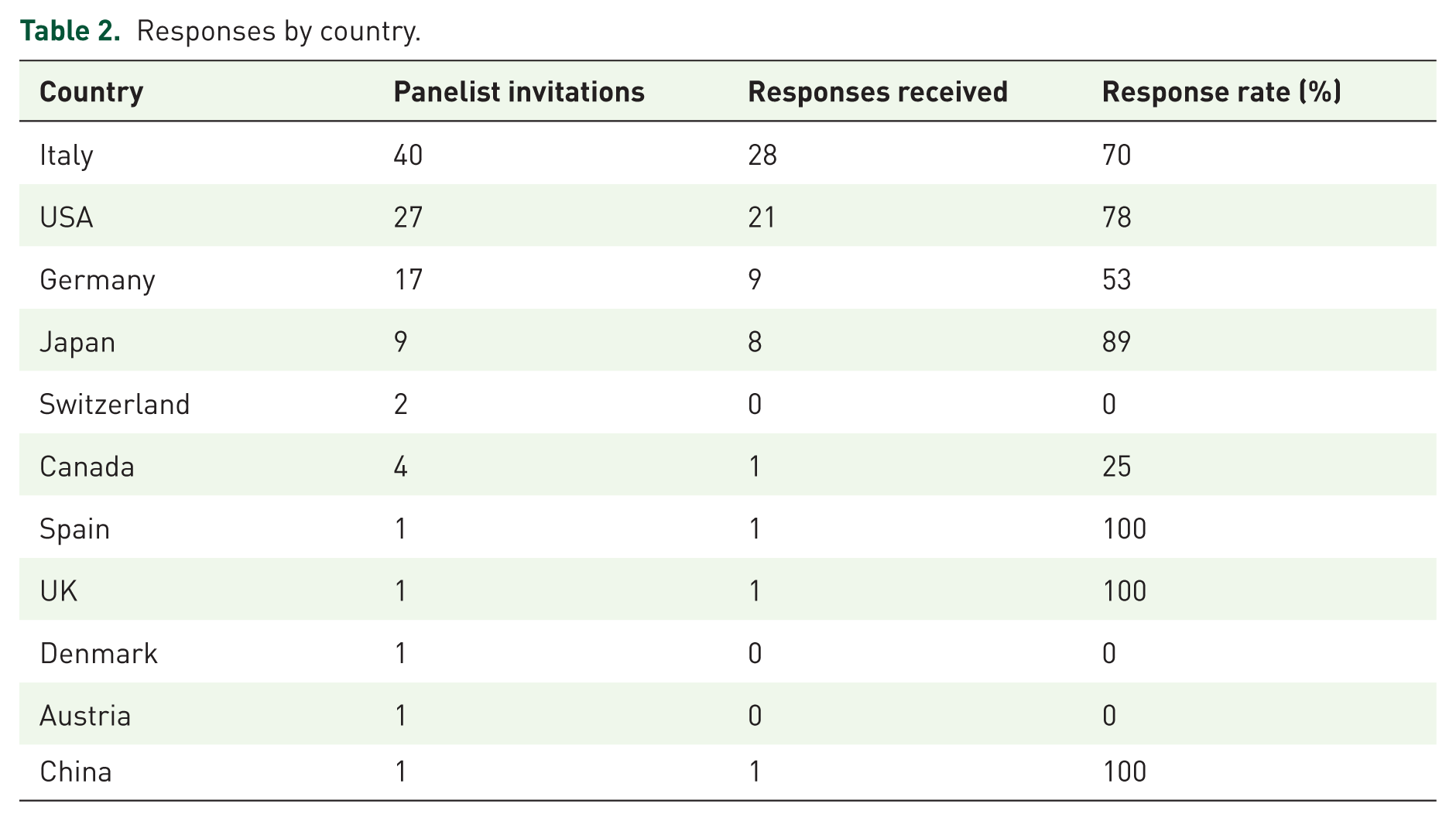
In total, 70 completed surveys were received, representing a response rate for the first round of 68%. Respondent role, time in role, and country are shown in Table 1. Twenty-two respondents identified as female, 45 as male, and one preferred not to disclose. Responders were based in Italy (n = 28), USA (n = 21), Germany (n = 9), Japan (n = 8), Canada, China, Spain, and the UK (all n = 1; Table 2).
Respondent role, time in role, and country.

Responses by country.
Results from Round 1 demonstrated agreement (⩾75%) for 35 statements, of which 25 (64%) achieved ⩾90% agreement. Four statements (10%) failed to achieve consensus. The steering committee agreed to modify and reissue two of the statements for retesting with the panel and remove two from the statement set.
Round 2
The updated statements were reissued to respondents from Round 1 (n = 70) along with an overview of the results from Round 1. At the end of the second round, 50 responses were received from the panel (general neurologists = 20, neuromuscular specialists = 30). The two revised statements (S8, S10) achieved consensus. Over the two rounds, all statements in the final set (n = 37) achieved consensus (Table 3, Figure 2 and Figure S2). The response rate for Round 2 was 71%.
Overall agreement scores, Round 1 (n = 70) and Round 2 (n = 50) combined (all numbers rounded to the nearest percentage).

IVIg, plasma exchange, methylprednisolone pulse therapy, or a combination of these.

Overall agreement scores, Round 1 (n = 70) and Round 2 (n = 50) combined.
Based on the final statement set the following principles are proposed:
Principle 1: A treatment goal for gMG is minimal symptom expression (MSE), or full symptom-free remission* with OCS dose ⩽5 mg/day
To mitigate the well-documented adverse effects of OCS, 6 the consensus underscores that MSE (defined as MG activities of daily living profile score 0 or 1 19 ) or remission is the desired therapeutic target, achievable with the lowest effective dose and shortest treatment duration. A more proactive approach to reducing reliance on OCS should incorporate frequent patient reassessment to adjust dosage in light of disease control milestones and adverse effect profiles. This aligns with the consensus that patient outcomes can be optimized by combining OCS sparingly with other therapies, leveraging tailored regimens to improve quality of life while minimizing harm (S18, 96%; S22, 99%; *equivalent to Pharmacological Remission as defined by Jaretzki et al. 20 ).
Principle 2: OCS should be used at the lowest therapeutic dose
OCS have a vital role in achieving rapid disease control in acute exacerbations,21–23 but to avoid the pitfalls of long-term high-dose OCS use, clinicians should plan for use in the short term and at the lowest therapeutic dose (S2, 93%; S21, 91%). Patients should be informed of the risks of prolonged OCS use and the importance of tapering once therapeutic goals are met (S28, 99%).
Principle 3: Rapid-onset immune therapies – Whether used as short-term bridging options or as chronic treatments – Can support earlier tapering of OCS
Combining OCS with rapid-onset therapies should become the standard to achieve swift disease control and taper steroid exposure safely. Notably, respondents agreed that immune therapies with a fast onset of action, including intravenous immunoglobulin (IVIg), therapeutic plasma exchange (PLEX), methylprednisolone pulse therapy, complement inhibitors, and FcRn inhibitors, enable clinicians to initiate tapering more rapidly 24 and thereby reducing cumulative OCS dose (S9, 96%; S10, 100%).
Principle 4: OCS exposure is associated with adverse effects that add to the patient burden and long-term costs of care
Clinicians are urged to incorporate not only clinical but also economic and quality-of-life metrics when considering OCS use. Considering the long-term costs associated with OCS,25,26 including managing adverse effects and diminished patient quality of life, strengthens the argument for early OCS tapering (S4, 91%; S19, 89%).
Principle 5: Use of OCS should be based on careful consideration of all patient factors and only when the benefits clearly outweigh the risks
Given the heterogeneous nature of gMG, individualized therapy plans must weigh the risks and benefits of OCS in light of comorbid conditions. Multidisciplinary collaboration (e.g., with endocrinologists, cardiologists, and pulmonologists) should form the backbone of this principle, ensuring comorbidities are adequately managed to facilitate safe OCS use (S14, 100%; S16, 94%).
Principle 6: A plan for tapering OCS dose should be developed before initiation
An individualized tapering strategy, initiated as soon as MSE is achieved, should be a cornerstone of gMG management. Protocols should emphasize close monitoring for signs of adrenal insufficiency or symptom recurrence, with rapid intervention protocols in place (S32, 100%).
Principle 7: Tapering can be considered as soon as an improvement over baseline is noted with an aim to achieve minimum OCS dose within 4 months
Leveraging the effectiveness of biologic therapies can support faster tapering regimens, 24 thereby reducing the cumulative OCS burden while maintaining disease control (S24, 99%; S25, 89%, S26, 97%).
Discussion
This study sought to establish consensus on the appropriate use of OCS in gMG. Both the steering committee and Delphi panel included individuals with significant expertise and seniority in the management of gMG, and this project was able to establish agreement on the principles of OCS use in gMG using a robust Delphi method with two rounds of survey informed by feedback.
The speed of onset associated with OCS provides an important treatment option, which is acknowledged in international consensus guidelines.8,27 The efficacy of OCS has been established in retrospective studies and a limited number of randomized controlled trials.28,29 However, these were very early studies, and the quality of evidence is considered low by the standards of today. 21 Consequently, the long-term efficacy of high-dose OCS is under scrutiny.30,31 The evidence surrounding the risks of long-term OCS exposure is growing, both in the associated increase in health risk,6,32 and the impact on the patient experience. 33 Some studies suggest that even short-term (<4 weeks) or low-dose exposure can alter the function of the hypothalamic-pituitary-adrenal axis.9,34 The evidence base, therefore, supports minimizing OCS use in terms of both length of exposure and dose.5,35 Although OCS treatment is inexpensive, analysis of predictors of high healthcare costs among gMG patients in the US identified “number of days with corticosteroids” prior to index exacerbation as an important predictor of high costs. 36 This suggests that the longer a patient is exposed to OCS, the greater the costs associated with managing their health.
Japanese guidelines (2022) 5 state that the first goal of treatment is the achievement of minimal manifestations with a dose of oral prednisolone of ⩽5 mg per day (MM-5 mg). Low-dose corticosteroids combined with calcineurin inhibitors are administered from the outset with fast-acting therapies (PLEX, IVIg, methylprednisolone pulse therapy, or a combination of these) to improve any residual symptoms. While not specifically addressed in all international MG guidelines, methylprednisolone pulse therapy has been reported in observational studies and case series as a potential adjunct to induce rapid improvement in selected patients with gMG, often as part of a combined regimen.37,38
German guidelines (2023) 39 recommend that OCS should be used “over the shortest possible period of time” with fast-acting therapies used according to severity, while international guidelines (2016 16 and 2020 8 ) place less emphasis on OCS dose reduction. These differences may go some way to explaining the disparity between the US and Italy (43% and 57%, respectively), and Japan and Germany (100% and 89%, respectively) regarding statement 10 during Round 1. Had the respondent groups been equal in number, then this statement may well have achieved consensus agreement overall, demonstrating a specific limitation in this study.
Once the patient has achieved the desired symptom control, tapering the OCS down to the minimum required to maintain response is recommended.8,10,40 Sharshar et al. 41 found that a shorter tapering regimen can be used to achieve minimal manifestation status at 12 months which is sustained at 15 months. In addition, several sources recommend reducing daily OCS dose in greater steps initially, followed by smaller increments when the daily dose is below 15 mg.7,9,34,40 This Delphi panel agreed that in practice, tapering could be achieved rapidly (<4 months) with the IVIg, PLEX, methylprednisolone pulse therapy, complement inhibitors, and FcRn inhibitors.24,41,42
Myasthenic crisis (MC) is a life-threatening manifestation of MG, which is usually treated with PLEX or IVIg and subsequent increase in OCS dosage where indicated.5,39,43 Respondents agreed with the use of OCS doses above 10 mg/day to avoid deterioration of symptoms and emerging MC for those unresponsive or unsuitable for nonsteroidal immunotherapies (S11, 91%). This supports the use of OCS as a last resort when other treatments are contraindicated or not tolerated, or have not shown efficacy. If an individual does require the chronic use of OCS, then an alternate-day dosing schedule is an option that can reduce the risk of OCS-related side effects while maintaining control of symptoms,13–15 but ultimately, some risk will remain.
Adrenal insufficiency (AI) may occur during steroid use, during a taper, and up to 4 years after OCS cessation. Adrenal function can be assessed by morning cortisol levels and/or adrenocorticotropic hormone (ACTH) stimulation test. 44 In practice, AI may be misattributed to myasthenic deterioration and lead HCPs to cease tapering. Instead, HCPs should continue to taper safely while being mindful of endocrinology guidance around adrenal testing or following a slower tapering regimen to avoid adrenal crisis – a life-threatening emergency requiring prompt recognition and treatment. 44
As with all studies, some limitations have been identified. While the expert panel was international, the majority of the collaborators were from Italy, the USA, Germany, and Japan, with a small number from the UK, China, Spain, and Canada. As a consequence of using the steering groups’ professional network to identify panel members, respondents were almost entirely based in Italy, the USA, Germany, and Japan. These are all high-income countries where high-cost therapies are routinely used in clinical practice, and there was a lack of representation from low- and middle-income countries (LMICs) where practice may be more strongly dictated by the level of access that neurologists have to specific treatments. Consequently, we might expect clinicians in LMICs to rely more heavily on therapies with a low acquisition cost that are freely available for use, such as OCS. It should therefore be acknowledged that the generalizability of these findings may be limited globally, but the principle of minimizing OCS use where possible still applies, and all prescribers should consider how this can be applied in their daily practice.
In addition, there was a limitation regarding representation by country: due to the relatively low numbers of panelists, the overall level of consensus is greatly influenced by practice in countries with the greatest representation (e.g., USA and Italy), and if there were equal representation on the panel for all included countries, there may have been a difference in the levels of consensus achieved.
Conclusion
The consensus findings reinforce a shift in the management of gMG, including a reframing of the role of OCS. Advocating for the judicious and sparing use of OCS against the backdrop of expanding therapeutic options. By attempting to quantify and prioritizing the lowest effective dose and shortest feasible duration of OCS, the consensus aligns with the principle of shared decision-making and active partnership between patients and HCPs in a safety-first approach that acknowledges the cumulative risks associated with steroid therapy.
As in other therapy areas, OCS should not be considered a key component for long-term treatment in gMG. Furthermore, the emphasis on integrating steroid-sparing and fast-onset therapies into treatment regimens reflects the broader evolution of gMG care. These targeted treatments not only enhance clinical efficacy but also reduce long-term adverse outcomes, aligning with therapeutic goals of disease remission and improved quality of life.
This consensus serves as a call-to-action for neurologists and neuromuscular specialists to consider their approach to OCS in gMG management. Incorporating these principles into clinical practice may improve treatment outcomes, reduce patient burden, and inform future guidelines. Ongoing education, research, and collaboration among clinicians are vital to ensure that advances in therapeutic strategies continue to translate into tangible benefits for gMG patients globally.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251398375 – Supplemental material for Appropriate use of steroids for patients with generalized Myasthenia Gravis: an international Delphi study
Supplemental material, sj-docx-1-tan-10.1177_17562864251398375 for Appropriate use of steroids for patients with generalized Myasthenia Gravis: an international Delphi study by Heinz Wiendl, Hiroyuki Murai, Pushpa Narayanaswami, Francesco Saccà, Nicholas J. Silvestri and James F. Howard in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors wish to thank the following individuals for their participation and feedback in the Delphi panel:
Prof. Jeffrey Allen: University of Minnesota, Minneapolis, MN, USA. Prof. Anthony A. Amato: Brigham and Women’s Hospital/Harvard Medical School, USA. Prof. Alvino Bisecco: Department of Advanced Medical and Surgical Sciences – University of Campania “Luigi Vanvitelli”, Naples, Italy. Dr. Silvia Bonanno: Neuroimmunology and Neuromuscular Disorders Unit, Fondazione IRCCS Istituto Neurologico Carlo Besta, Milan, Italy. Dr. Marco Capobianco: Neurology Department, S. Croce and Carle Hospital, Cuneo, Italy. Prof. Andrea Corse: University of North Carolina, Chapel Hill, NC, USA. Dr. Elena Cortés-Vicente: Neuromuscular Diseases Unit, Hospital de la Santa Creu i Sant Pau, Barcelona, Spain. Dr. Nunzia Cuomo: University of Naples Federico II, Italy. Prof. Emanuele D’Amico: Department of Medical and Surgical Sciences, University of Foggia, Foggia, Italy. Dr. Claudia dell’Aquila: UOC Neurologia Stroke Unit di Venere Hospital, Bari, Italy. Prof. Mazen Dimachkie: The University of Kansas Medical Center, USA. Dr. B. Jane Distad: University of Washington, USA. Dr. Vincenzo Di Stefano: Dept of Biomedicine, Neuroscience and Advanced Diagnostics (Bi.N.D.), University of Palermo, 90127 Palermo, Italy. Dr. Miriam L Freime: The Ohio State University, USA. Dr. Maria Pia Giannoccaro: IRCCS Istituto delle Scienze Neurologiche di Bologna, Italy and Dipartimento di Scienze Biomediche e Neuromotorie, Università di Bologna, Italy. Prof. Namita A. Goyal: UC Irvine, USA. Prof. Neelam Goyal: Stanford University Hospital. Palo Alto, Ca, USA. Prof. Kelly Graham Gwathmey: Virginia Commonwealth University, USA. Prof. Francesco Habetswallner: Cardarelli Hospital, Clinical Neurophisiology Department, Naples, Italy. Dr. Tim Hagenacker: Dep. of Neurology, University Medicine Essen, Germany. Dr. Francesca Lapenna: Neurophysiopathology Unit, Policlinico Hospital, Bari, Italy. Dr. Yuebing Li: Neuromuscular Center, Department of Neurology, Cleveland Clinic, USA. Dr. Lorenzo Maggi: Neuroimmunology and Neuromuscular Diseases Unit, Fondazione IRCCS Istituto Neurologico Carlo Besta, Milan, Italy. Prof. Andreas Meisel: Charité Universitätsmedizin Berlin, 10098 Berlin, Germany, Department of Neurology with Experimental Neurology, Germany. Dr. Nico Melzer MD: Department of Neurology, Medical Faculty and University Hospital, Heinrich-Heine-University Düsseldorf, Germany. Prof. Arthur Melms: Hertie-Institute for Clinical Brain Research, University of Tübingen, Tübingen, Germany. Prof. Srikanth Muppidi: Stanford Neuroscience Health Center, Palo Alto, CA, USA. Prof. Jacqueline Palace: Nuffield Department of Clinical Neurology and Neurology Department, Oxford University Hospitals Trust, UK. Dr. Chiara Pane: Department of Neuroscience, Reproductive Science and Dentistry, University “Federico II”, Naples, Italy. Prof. Mamatha Pasnoor: The University of Kansas Medical Center, USA. Dr. Marc Pawlitzki: Department of Neurology, Medical Faculty, University Hospital Düsseldorf, Düsseldorf, Germany. Prof. Stefano C. Previtali: Vita-Salute San Raffaele University, Italy. Dr. Dario Ricciardi: UOC Clinical Neurophysiology – AORN Cardarelli, Naples, Italy. Prof. Carmelo Rodolico: Department of Clinical and Experimental Medicine, University of Messina, Italy. Dr. Alessio Sarnataro: University of Naples Federico II, Italy. Prof. Dr. Jörn Peter Sieb: Neurologische Klinik, HELIOS Hanseklinikum Stralsund, Stralsund, Germany. Prof. Dr Benedikt Schoser: Friedrich-Baur-Institute, Department of Neurology, LMU clinics, Germany. Prof. Gordon Smith: Virginia Commonwealth University, Richmond, USA. Dr. Masanori P. Takahashi: Osaka University Graduate School of Medicine, Japan. Prof. Dr. Christiane Schneider-Gold: Department of Neurology, St. Josef Hospital, Ruhr-University Bochum, Gudrunstr. 56, 44791 Bochum, Germany. Dr. Manlio Sgarzi: ASST Papa Giovanni XXIII Bergamo Neuroscience department, Italy. Prof. Takamichi Sugimoto: Department of Clinical Neuroscience and Therapeutics, Hiroshima University, Hiroshima, Japan. Dr. Shigeaki Suzuki: Keio University School of Medicine, Tokyo, Japan. Dr. Kimiaki Utsugisawa: Hanamaki General Hospital, Japan. Dr. Akiyuki Uzawa: Department of Neurology, Graduate School of Medicine, Chiba University, Japan. Dr. Claudia Vinciguerra: Neurology Unit, Department of Medicine, Surgery and Dentistry, University Hospital of Salerno, Salerno, Italy. Dr. Mitsuru Watanabe: Department of Neurology, Kyushu University Hospital, Fukuoka, Japan. Prof. Dr. Gil I. Wolfe: Jacobs School of Medicine and Biomedical Sciences, Univ. at Buffalo/SUNY, Buffalo, NY, USA. Prof. Chongbo Zhao: Department of Neurology, Huashan Hospital, Fudan University, China
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
