Abstract
Impending and manifest myasthenic crisis (MC) represent life-threatening neurological emergencies that require immediate intervention. Although eculizumab, a terminal complement C5 inhibitor, has established efficacy in generalized myasthenia gravis, most studies have focused on long-term outcomes, and its potential for rapid onset of effect in acute exacerbations has not been well evaluated. We retrospectively analyzed four acetylcholine receptor (AChR) antibody-positive patients (five acute episodes) who received eculizumab as emergency therapy between January 2024 and July 2025. All patients presented with impending or manifest MC; none had received intravenous immunoglobulin, plasma exchange, or neonatal Fc receptor (FcRn) inhibition within the preceding month. According to the Myasthenia Gravis Foundation of America classification, three patients had class IVb and one had class V. Triggers for exacerbation included COVID-19 infection (n = 2), puerperium (n = 1), PD-1 inhibitor exposure (n = 1), and common infection (n = 1). Within 48 h of eculizumab administration, Quantitative Myasthenia Gravis scores improved from 28.2 ± 4.8 to 15.0 ± 4.1, while Myasthenia Gravis Activities of Daily Living scores decreased from 16.8 ± 2.8 to 6.0 ± 4.3. Notable milestones achieved included arterial blood gas improvement within 7.5 h (one patient), nasogastric tube removal within 48 h (three patients), and successful extubation at 17 h (one patient). No treatment-related adverse events were observed. These preliminary findings suggest that eculizumab may be associated with rapid, temporally related clinical improvement within 48 h with a favorable short-term safety profile in AChR antibody-positive patients with impending or manifest MC, particularly those with complement activation triggers. Larger prospective studies with standardized short-term assessment protocols, including frequent neurological evaluations during the first 48 h, are warranted to further validate its efficacy and define its role as an emergency therapy in the acute management of myasthenic exacerbations.
Plain language summary
Myasthenia gravis (MG) is a rare disease in which the body’s immune system interferes with communication between nerves and muscles, leading to weakness in the eyes, face, arms, legs, and sometimes breathing muscles. In its most severe form, called “myasthenic crisis,” patients can lose the ability to breathe on their own and require urgent hospital treatment. Standard emergency treatments such as intravenous immunoglobulin (IVIG) and plasma exchange can be effective, but they may take several days to work, leaving patients dependent on ventilators or feeding tubes for longer. Eculizumab is a medication that blocks part of the immune system known as the complement pathway, which plays a role in damaging the muscle–nerve connection in MG. While eculizumab is approved for generalized MG, most studies have examined its long-term benefits. Much less is known about whether it can act quickly enough to help in emergency situations. In this study, we reviewed four patients with MG (five episodes of severe worsening) treated at our hospital who received eculizumab during an emergency crisis. Triggers included infections, pregnancy recovery, cancer immunotherapy, and COVID-19. Within 48 hours of receiving eculizumab, patients showed meaningful improvement in their muscle strength and daily activities. Some were able to breathe without a ventilator or have feeding tubes removed much sooner than expected. Importantly, no serious side effects were observed in the short term. Our findings suggest that eculizumab may provide rapid improvement in patients experiencing life-threatening MG crises, particularly when the immune system is strongly activated. However, this was a very small study, and more research in larger groups of patients is urgently needed to confirm its safety and effectiveness as an emergency therapy.
Introduction
Myasthenia gravis (MG) is a prototypical antibody-mediated complement-dependent autoimmune disorder that affects neuromuscular junctions. A 2020 nationwide epidemiological study in China reported an age-standardized mortality rate of 1.86 per million people, with myasthenic crisis (MC) being the predominant cause of death. Notably, 1 15%–20% of patients with MG develop MC, a life-threatening complication, during their disease course. Clinically, it manifests as respiratory failure secondary to respiratory muscle paralysis, frequently necessitating emergent mechanical ventilation (MV). 2
The current evidence-based management of MC remains limited by the paucity of high-quality randomized controlled trials. First-line immunomodulatory therapy for MC still predominantly relies on intravenous immunoglobulin (IVIG) and therapeutic plasma exchange (TPE) as cornerstone rescue therapies.3,4 However, both modalities are constrained by the relatively delayed onset of their therapeutic effects. For instance, a comparative study reported that the TPE group had a mean MV duration of 11.75 days (vs 17.43 days in the IVIG group) and an ICU length of stay of 8.92 days (vs 19.46 days in the IVIG group). 4 Recent studies have identified complement system activation as a pivotal mechanism underlying MC pathogenesis. This concept is directly supported by pathological evidence of extensive postsynaptic membrane destruction at neuromuscular junctions in intercostal muscle biopsy specimens from MC patients. 5
The REGAIN study established the long-term efficacy of C5 complement inhibitors in refractory MG 6 ; however, these agents have historically been employed as rescue therapy only after IVIG/TPE failure. 7 Notably, robust evidence characterizing rapid-onset efficacy during acute-phase management is lacking. Eculizumab, a monoclonal antibody targeting complement C5, exerts its therapeutic effect by specifically inhibiting C5 cleavage, thereby suppressing MAC formation and interrupting key pathological pathways in MG. Pharmacokinetic study shows that serum drug concentrations reach steady state after 4 weeks of continuous administration, 8 with sustained potent complement inhibitory activity, as evidenced by >90% of patients achieving complete C5 inhibition. Monitoring data confirmed high-level terminal complement pathway suppression in most patients at both trough and peak concentrations. Although in vitro studies have indicated rapid biological effects within hours of administration, 8 clinical observations suggest that symptomatic improvement typically requires prolonged drug accumulation. Recent clinical trials have demonstrated that eculizumab treatment leads to rapid symptomatic improvement in patients with MG, as evidenced by favorable extubation success rates in mechanically ventilated patients.9,10 However, robust evidence supporting the use of this agent as a first-line emergency therapy during the acute phase is currently lacking, with only limited case reports available in the literature. 11 Existing clinical evidence has primarily focused on its application as a salvage treatment for patients who have failed conventional immunosuppressive therapies. 7
This study retrospectively evaluated four patients with anti-acetylcholine receptor (AChR) antibody-positive generalized myasthenia gravis (gMG) who received eculizumab during MC or an impending crisis. The focus was on its rapid therapeutic effects and safety profile in acute exacerbations.
Methods
Study participants
This retrospective study enrolled patients with anti-AChR antibody-positive gMG who received eculizumab as first-line emergency therapy for impending or manifested myasthenia crisis at Shenzhen People’s Hospital between January 1, 2024, and July 31, 2025. During this period, eculizumab was the sole C5 complement inhibitor accessible in China, as ravulizumab was not yet available. All participants met the international diagnostic criteria for an MG crisis (requiring MV) or an impending crisis (defined as imminent progression to crisis within days to weeks), 12 as confirmed by three senior neurologists. The inclusion criteria were mandatory AChR antibody positivity, completion of standardized assessments, meningococcal prophylaxis, and a baseline laboratory workup. The key exclusion criteria were a history of IVIG, TPE, or FcRn inhibition within the preceding month.
Study design and data collection
Using a retrospective observational design, we comprehensively documented the following: (1) demographic characteristics, (2) disease features, (3) comorbidities, and (4) history of prior treatment. Disease duration was defined as the interval from the initial onset of gMG symptoms to the current crisis. Using the time of eculizumab administration as the reference point, pre- and posttreatment outcomes were systematically evaluated, including (1) the Myasthenia Gravis Foundation of America (MGFA) Quantitative Myasthenia Gravis (QMG) score; (2) the Myasthenia Gravis Activities of Daily Living (MG-ADL) score; and (3) objective physiological parameters (time to oxygenation improvement, MV weaning time, and nasogastric tube removal time).
Statistical analysis
Data were presented as mean ± SD for continuous variables. Statistical analysis was performed using GraphPad Prism 10.0 software (GraphPad Software, LLC, San Diego, CA, USA).
Results
Clinical characteristics
This study enrolled four patients with MG who met the inclusion criteria: one male (25%) and three female (75%). All patients were laboratory-confirmed to be AChR antibody-positive and presented with MC or an impending crisis. Three patients (75%) were classified as MGFA class IVb and one (25%) as MGFA class V. During the study period, five crisis or impending crisis events were recorded, including one patient who experienced two distinct crisis episodes. Table 1 summarizes the baseline clinical characteristics of the patients.
Baseline characteristics and pretreatment status of AChR antibody-positive gMG patients with myasthenic crisis or impending crisis prior to eculizumab therapy.
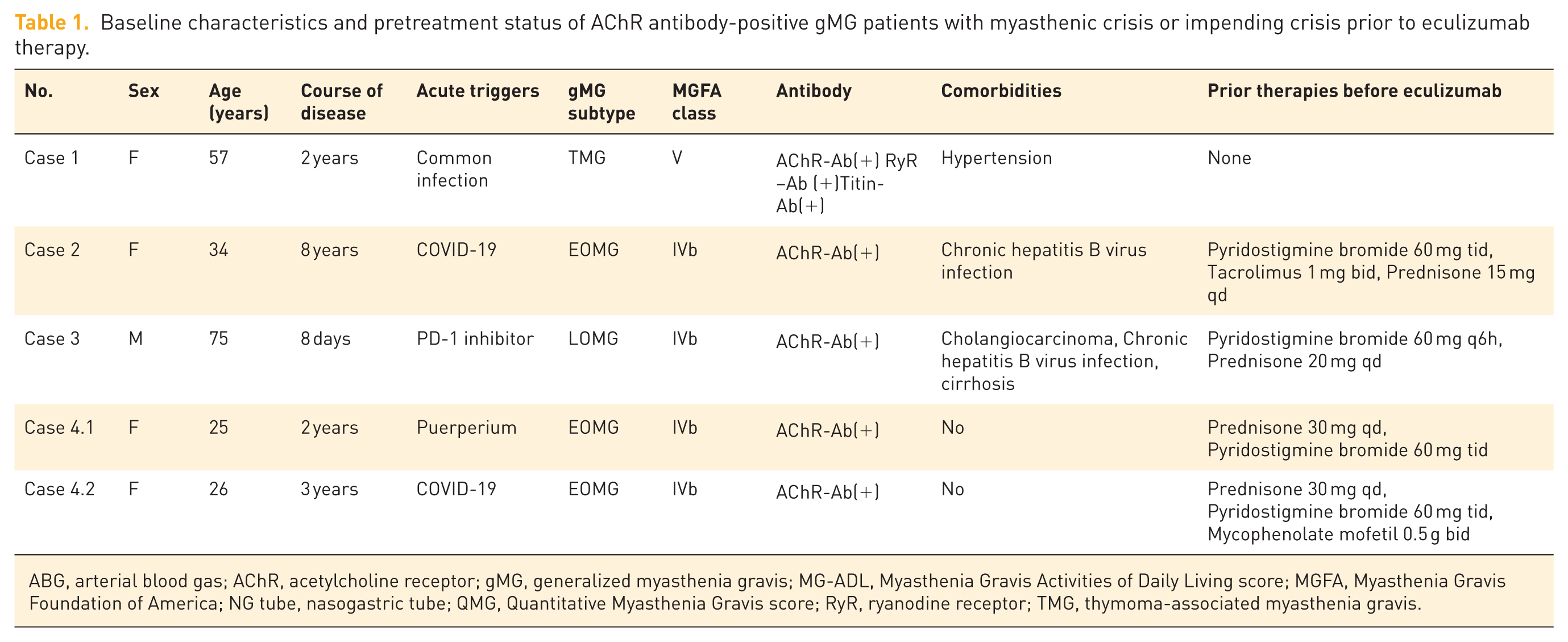
ABG, arterial blood gas; AChR, acetylcholine receptor; gMG, generalized myasthenia gravis; MG-ADL, Myasthenia Gravis Activities of Daily Living score; MGFA, Myasthenia Gravis Foundation of America; NG tube, nasogastric tube; QMG, Quantitative Myasthenia Gravis score; RyR, ryanodine receptor; TMG, thymoma-associated myasthenia gravis.
Clinical presentation
Case 1: Thymoma-associated myasthenia gravis with myasthenic crisis, rapidly bridged to thymectomy).
A 57-year-old female patient presented with a 2-year history of recurrent fluctuating limb weakness, which had aggravated over the preceding 2 months, with the development of dysphagia and ptosis. She was admitted emergently due to fever and respiratory failure; arterial blood gas (ABG) analysis revealed a PCO2 level >130 mmHg. Her consciousness was restored after emergency endotracheal intubation. Physical examination revealed diplopia, mild ptosis, and Medical Research Council (MRC) grade 4 muscle strength in all limbs. Serological antibody testing using a cell-based assay was positive for AChR (titer 1:320), ryanodine receptor (RyR; titer 1:320), and titin (titer 1:320) antibodies. Chest computed tomography (CT) revealed a thymoma. The diagnosis was confirmed as thymoma-associated MG (TMG), classified as MGFA class V, with MG-ADL and QMG scores of 17 and 30, respectively.
Immunosuppressive therapy was initiated immediately, consisting of prednisone (30 mg once daily) and mycophenolate mofetil (MMF; 0.5 g twice daily), combined with symptomatic treatment with pyridostigmine bromide (60 mg three times daily). Plasma-derived products required for IVIG (IVIG preparations) and PLEX (replacement fluids such as albumin or fresh frozen plasma) were not immediately available and required allocation. In addition, the patient’s family refused to reestablish invasive vascular access, precluding the timely initiation of PLEX. Informed consent was obtained from the patient’s family to facilitate rapid weaning from MV, and the patient received an infusion of eculizumab (900 mg) with concurrent prophylactic penicillin. The patient showed rapid crisis reversal, as evidenced by successful weaning from the ventilator 17 h postinfusion. At the 48-h follow-up, the QMG score decreased to 12 and the MG-ADL score decreased to 3. The nasogastric tube was removed on day 5. By day 6, the QMG score had further improved to 9, and the MG-ADL score was 0. A second eculizumab infusion was administered on day 7 along with meningococcal vaccination.
After clinical stabilization, the patient underwent a thymectomy on day 12 and experienced a smooth intraoperative course and good postoperative recovery. Histopathological examination confirmed type B3 thymoma, and the patient was subsequently transferred to the Radiation Oncology Department for adjuvant treatment. Eculizumab treatment was administered a total of four times, once weekly, twice before thymoma surgery, and twice after surgery. Once the condition stabilized, the patient declined further long-term eculizumab infusion. Monthly follow-up visits were conducted for 9 months, during which the ADL score consistently remained at 0. The long-term medication regimen consisted of MMF (0.5 g) twice daily, pyridostigmine (60 mg) three times daily, and gradual steroid tapering.
Case 2: Highly active, early-onset AChR-positive generalized myasthenia gravis with recurrent crises and bulbar sequelae.
A 34-year-old female was diagnosed with AChR antibody-positive gMG 8 years ago and initially presented with diplopia, limb weakness, and dysphagia. Initial treatment with pyridostigmine bromide and prednisone resulted in residual limb fatigue. Seven years ago, an infection-triggered disease exacerbation led to an impending crisis characterized by worsened dysphagia. The patient was treated with steroid pulse therapy and azathioprine (later discontinued due to hepatotoxicity), resulting in residual tongue atrophy. The patient was maintained on regular low-dose prednisone and pyridostigmine. Five years prior, another infection precipitated respiratory failure that required intubation. Despite treatment with IVIG and immunosuppressive therapy (including prednisone and tacrolimus), the patient experienced recurrent relapses, requiring multiple IVIG rescue courses.
Five days before the current admission, she developed a fever, followed by head drop, ptosis, dysphagia / difficulty in mastication, and respiratory distress. Laboratory tests confirmed COVID-19 and revealed elevated inflammatory markers, including C-reactive protein (89.46 mg/L), procalcitonin (0.097 ng/mL), interleukin-6 (46.73 pg/mL), and white blood cell count (12.90 × 109/L). Neurological examination revealed mild ptosis but intact extraocular movements, dysarthria accompanied by three-grooved tongue atrophy, dysphagia necessitating nasogastric tube placement, and proximal weakness (MRC grade 3 in the upper limbs and grade 4 in the lower limbs). She was classified as MGFA class IVb and had MG-ADL and QMG scores of 19 and 28, respectively.
The patient had refractory AChR-positive MG and an incomplete previous response to IVIG. Long-term eculizumab therapy was planned prior to admission; therefore, eculizumab was initiated directly during the acute phase according to a pre-established treatment plan. Her baseline regimen, pyridostigmine bromide (60 mg three times daily), tacrolimus (1 mg twice daily), and prednisone (15 mg once daily) was supplemented with an infusion of eculizumab (900 mg) and COVID-19 antiviral therapy, with concurrent prophylactic penicillin. Significant improvements in swallowing and respiratory function occurred within 24 h, permitting nasogastric tube removal 41 h postinfusion. The QMG score improved to 15 and the MG-ADL score to 4 at the 48-h follow-up, and by day 6, the scores had further improved to QMG 9 and MG-ADL 3. Long-term regular eculizumab therapy was continued, initially weekly and then every 2 weeks from the 5th week along with pyridostigmine, tacrolimus, and prednisone. Monthly follow-up showed an ADL score of 1 over 10 months.
Case 3: PD-1 inhibitor-induced generalized myasthenia gravis in impending crisis. 11
A 75-year-old male with PD-1 inhibitor (pembrolizumab)-induced MG with impending MC received emergency eculizumab treatment and showed rapid improvement in respiratory and myasthenic symptoms. Detailed case information is available in a previously published article. 11 During 11 months of follow-up, he received biweekly intravenous infusions of 1200 mg eculizumab, with pyridostigmine maintained at 60 mg every 8 h. Tumor therapy with capecitabine was continued. The patient remains stable with minimal symptoms and no evidence of tumor recurrence, with good treatment tolerance and no adverse events during the follow-up period.
Case 4: Early-onset myasthenia gravis with recurrent impending crises and prominent bulbar involvement.
A 25-year-old woman initially presented 2 years prior with diplopia, dysarthria, and dysphagia requiring nasogastric tube placement. Laboratory testing revealed markedly elevated AChR antibodies and low-frequency repetitive nerve stimulation confirmed a decremental response. Chest CT revealed remnants of thymic involution. She was diagnosed with AChR antibody-positive gMG during an impending crisis. IVIG therapy led to clinical improvement (nasogastric tube removed on day 5), followed by maintenance prednisone (30 mg/day) and pyridostigmine bromide (60 mg three times daily).
Case 4.1: One year prior, the patient developed another impending crisis after a cesarean section, characterized by generalized weakness and tachypnea, which resolved very slowly after IVIG administration (20 g/day for 5 days). She was maintained on a regular prednisone and pyridostigmine regimen. Forty days after discharge, she experienced a third exacerbation characterized by severe dysphagia and limb weakness (rendering her nonambulatory). Pulmonary function tests revealed a forced vital capacity (FVC) of 540 mL, consistent with MGFA class IVb, and MG-ADL and QMG scores of 18 and 32, respectively. The patient had a suboptimal response to previous IVIG therapy and declined further IVIG or invasive plasmapheresis. After a thorough consultation, she opted for treatment with a novel targeted biological agent. Rescue therapy with eculizumab (900 mg) and concurrent prophylactic penicillin yielded rapid improvement. Within 24 h, MG-ADL and QMG scores decreased to 12 and 22, respectively, and the FVC increased to 1904 mL. At 48 h postinfusion, the nasogastric tube was removed, and by day 4, the FVC had further improved to 3035 mL (with QMG and MG-ADL scores of 14 and 4, respectively). The patient declined thymectomy for personal reasons and was discharged on MMF (0.5 g) twice daily; however, long-term eculizumab maintenance therapy was discontinued owing to financial constraints.
Case 4.2: One month prior, the patient experienced acute exacerbation following sleep deprivation and COVID-19 infection, exhibiting dyspnea and dysphagia that required nasogastric tube reinsertion. Upon admission, she met the criteria for MGFA class IVb (MG-ADL 18, QMG 31), and 76 mL of respiratory secretions was suctioned within the first hour. The patient had a well-documented history of robust clinical response to eculizumab. Given the rapid deterioration in the current exacerbation and the urgent need for a fast-acting therapy, eculizumab was initiated immediately during the acute phase. Emergency administration of eculizumab (900 mg) with concurrent prophylactic penicillin, combined with neostigmine (1 mg intramuscularly) and COVID-19 antiviral therapy, resulted in the elimination of secretion accumulation within 35 min. After 24 h, her clinical status had markedly improved (MG-ADL and QMG scores of 2 and 12, respectively), permitting nasogastric tube removal. The patient received eculizumab treatment twice but discontinued the therapy after achieving disease stability for financial reasons. The long-term medication regimen consisted of MMF (0.5 g) twice daily, pyridostigmine (60 mg) three times daily, and gradual steroid tapering. Monthly follow-up visits were conducted with an ADL score of 0 at the 4-month follow-up.
The clinical course of all patients is shown in Figure 1, and MG-ADL and QMG scores after eculizumab treatment are presented in Figure 2.

Clinical courses.

Changes in MG-ADL and QMG scores before and after eculizumab infusion (within 7.5–48 h).
Clinical outcomes and functional assessments
All four enrolled MG patients (five acute exacerbation episodes) demonstrated rapid clinical improvement and favorable safety profiles after eculizumab treatment. The patient in the MC group (Case 1) achieved ventilator weaning 17 h postinfusion and nasogastric tube removal on posttreatment day 5. Nasogastric tube removal was achieved within 48 h postinfusion in three of the four impending crisis events (three patients), with a mean duration of 2.5 ± 1.7 days. ABG parameters improved within 7.5 h in one impending crisis (Case 3). Quantitative assessments revealed marked reductions from baseline in both QMG scores (28.2 ± 4.8 to 15.0 ± 4.1) and MG-ADL scores (16.8 ± 2.8 to 6.0 ± 4.3). The initial response was observed within 7.5–48 h (mean, 30.3 ± 17.5 h; see Figure 2).
Discussion
MG, an autoimmune disorder of the neuromuscular junction, is characterized by AChR antibody positivity in approximately 80% of patients. These pathogenic antibodies impair AChR function through three principal mechanisms: competitive blockade of acetylcholine-binding sites; antibody-mediated internalization, and degradation of AChRs; and complement system activation, which leads to postsynaptic membrane damage. Clinical observations reveal that approximately 20% of patients with MG present with MC as their initial manifestation. 13 Notably, patients with impending crises often progress to crisis within days to weeks. 12 As the most critical complication of MG, the current standard treatments for MC are TPE and IVIG. 12 Although their overall efficacy is comparable, 14 the 2016 International Consensus Guidance from the Myasthenia Gravis Foundation of America suggests that expert experience favors TPE because of its potentially faster onset of action and higher response rates. 12 Clinical decision-making requires careful consideration of the following: TPE’s rapid action but associated risks of hemodynamic instability and catheter-related complications versus IVIG’s delayed efficacy (typically 4–5 days) but a more favorable safety profile. Additional factors include comorbidities (e.g., TPE is contraindicated in sepsis, whereas IVIG should be used with caution in hypercoagulable states or renal insufficiency) and healthcare resource accessibility. 12
Therapeutic rationale for employing eculizumab or efgartigimod during impending and manifest MC is established on clinical evidence from real-world studies: both agents significantly reduce clinical scores within 1 week of treatment initiation. 15 Recent advances in understanding MG pathogenesis and challenges related to blood product availability have highlighted emerging targeted biologics (e.g., the C5 inhibitor eculizumab, FcRn antagonist efgartigimod and rozanolixizumab, and long-acting C5 inhibitor ravulizumab) for MG management.16–18 Eculizumab has demonstrated significant efficacy not only in the long-term management of chronic-phase MG 6 but has also shown promising results in exploratory studies of MC treatment.19–21 Similarly, the use of efgartigimod has been reported in case reports on MC management. 22 Current evidence demonstrates that precise immunomodulation is pivotal for improving MG outcomes.15,16,23–25
Recent clinical evidence has demonstrated the rapid therapeutic onset of complement C5 inhibitor eculizumab in MG management. The pivotal REGAIN study established its clinical efficacy within 4 weeks of treatment, with most patients achieving an optimal response by week 12. 6 Subsequent investigations revealed a further shortened median time to clinical effect of 7 days, 20 with case reports documenting clinical responses as early as day 2. 19 This provides critical evidence for its acute-phase application. In our case series, despite potential confounders (e.g., concurrent pyridostigmine bromide in case 1 due to the initial diagnosis, neostigmine administration in case 4 due to dysphagia, and an inability to swallow oral medications), all subjects exhibited marked clinical improvement within 48 h. These findings suggest that this approach may be a viable option for first-line treatment of patients with impending or early-stage MC, particularly in urgent clinical situations where rapid symptom control is essential. Although further studies are needed to confirm its efficacy, these results provide preliminary evidence supporting its potential role in such scenarios.
The onset of acute MG exacerbation is closely associated with aberrant complement system activation triggered by diverse factors. Complement activation may be directly induced via pathogen-associated molecular patterns or indirectly mediated through immune complex-driven classical pathways. 26 Recent studies have demonstrated that the SARS-CoV-2 spike (S) protein specifically activates the alternative complement pathway, 27 whereas its nucleocapsid (N) protein initiates the lectin pathway by binding to the mannose-binding lectin-associated serine protease 2.28,29 Furthermore, nonneutralizing IgG antibodies (particularly those targeting the N protein) generated during early infection may form virus-antibody complexes, thereby excessively activating the classical complement pathway and exacerbating inflammatory cascades and tissue injury. Seasonal coronavirus cross-reactive antibodies may also have contributed to this process. 30 Beyond infectious triggers, systemic complement activation is frequently observed in cancer patients receiving immune checkpoint inhibitors (ICIs), 31 whereas postpartum MG worsening typically occurs within months of delivery. 12 Among the five acute exacerbation episodes analyzed in this study, the triggers included SARS-CoV-2 infection (n = 2), puerperium (n = 1), PD-1 inhibitor therapy (n = 1), and common infections (n = 1). Notably, the pathological features of these rapidly progressive episodes may predominantly align with a complement-mediated direct tissue injury mechanism, which could explain the rapid therapeutic response to eculizumab.
In the management of acute exacerbations of MG, the speed of onset is one of the most critical determinants in therapeutic decision-making, whereas treatment cost remains a significant and unavoidable consideration. The two most widely used acute-phase interventions, IVIG and TPE, impose substantial economic burdens. 32 For instance, in the Chinese market, a single course of IVIG costs approximately ¥20,000–40,000, whereas a single course of TPE costs ¥20,000–50,000. In contrast, a single dose of eculizumab (900 mg) was approximately ¥7554. It is important to note that our observations regarding the potential cost-effectiveness of eculizumab were strongly influenced by medical settings in China. In the acute crisis scenarios addressed in our study, patients typically required only 1–2 doses of eculizumab to achieve rapid and stable improvement. When costs are calculated based on this real-world dosing pattern, the total expenditure may in certain cases be lower than that of a full course of IVIG or TPE. However, we also explicitly acknowledge that drug pricing, reimbursement policies, and resource allocation vary substantially across regions. Therefore, this economic inference may not be generalizable to other healthcare systems. More importantly, we emphasize the potential advantage of eculizumab in terms of its rapid onset of action, which may be clinically meaningful for the management of severe MG exacerbations or situations requiring prompt stabilization. This approach provides clinicians with additional therapeutic options in selected circumstances.
Eculizumab has multifaceted clinical benefits in the treatment of myasthenic crises. First, mechanistically, this agent selectively inhibits complement C5, thereby precisely blocking MAC formation and effectively mitigating neuromuscular junction injury. This targeted mechanism differs fundamentally from that of conventional therapies (e.g., IVIG or TPE), which act through nonspecific immune modulation or circulating antibody clearance. Second, eculizumab exhibited superior efficacy kinetics. Clinical observations indicate significant symptom improvement in some patients within 48 h, which is a critical advantage for critically ill patients requiring rapid ventilator weaning to avoid prolonged intubation-associated complications. Third, eculizumab maintains its efficacy against diverse MC triggers including infections, perioperative stress, pregnancy/postpartum states, and PD-1 inhibitor-induced exacerbations. Fourth, eculizumab can induce therapeutic responses even in patients in whom conventional salvage therapies have failed, offering a novel option for refractory cases. 20 Notably, most reports in the literature describe eculizumab as a rescue treatment after failure of IVIG and/or PLEX, and observed improvements may partly reflect effects of prior treatment. 33 In contrast, our patients were treated with eculizumab from the outset, thereby avoiding interference from traditional rescue medications. Finally, by selectively targeting the terminal complement pathway while preserving proximal complement functions, including microbial opsonization, immune response initiation, and immune complex clearance, eculizumab maintains potential utility in patients with concurrent infections (except encapsulated bacteria such as Neisseria meningitidis and Streptococcus species). 8
This investigation has several important limitations that warrant consideration. First, due to the limited sample size and retrospective study design, potential selection bias and confounding factors may have compromised the reliability of our findings. Second, regarding efficacy assessment, the current study lacked standardized evaluation time points. Future studies should incorporate more frequent assessments (e.g., every 12 h) within the first 48 h postadministration to quantify the clinical response time of eculizumab more precisely. Third, inclusion of ICU patients and those with concurrent infections introduces confounding, as adjunctive interventions were often necessary. This concomitant therapy complexity hinders accurate assessment of the independent therapeutic efficacy of eculizumab. Future multicenter, large-scale randomized controlled trials employing stricter efficacy assessment criteria and standardized treatment protocols are warranted to validate the independent therapeutic effects of this agent.
Conclusion
Our case series provides preliminary observations suggesting that eculizumab may provide rapid clinical improvement within 48 h with a favorable short-term safety profile in AChR antibody-positive patients presenting with impending or manifest MC, particularly in patients with suspected complement activation triggers. However, its retrospective design, small sample size, and concomitant therapies precluded causal inference. Therefore, these findings should be regarded as hypothesis-generating only. Future multicenter randomized controlled trials should prioritize direct efficacy comparisons with conventional therapies (IVIG/PE) and the establishment of standardized short-term assessment protocols (e.g., neurological function assessments every 12 h) to further validate its rapid therapeutic efficacy and safety, and to clarify its role as an emergency therapy in acute myasthenic exacerbations.
